Significance
Here we demonstrate that bisphenol A (BPA) exposure during a time point analogous to the second trimester in humans has real and measurable effects on brain development and behavior. Furthermore, our study is the first, to our knowledge, to show that bisphenol S, a replacement used in BPA-free products, equally affects neurodevelopment. These findings suggest that BPA-free products are not necessarily safe and support a societal push to remove all structurally similar bisphenol analogues and other compounds with endocrine-disruptive activity from consumer goods. Our data here, combined with over a dozen physiological and behavioral human studies that begin to point to the prenatal period as a BPA window of vulnerability, suggest that pregnant mothers limit exposure to plastics and receipts.
Keywords: endocrine disruption, androgen receptor, aromatase, hyperactivity
Abstract
Bisphenol A (BPA), a ubiquitous endocrine disruptor that is present in many household products, has been linked to obesity, cancer, and, most relevant here, childhood neurological disorders such as anxiety and hyperactivity. However, how BPA exposure translates into these neurodevelopmental disorders remains poorly understood. Here, we used zebrafish to link BPA mechanistically to disease etiology. Strikingly, treatment of embryonic zebrafish with very low-dose BPA (0.0068 μM, 1,000-fold lower than the accepted human daily exposure) and bisphenol S (BPS), a common analog used in BPA-free products, resulted in 180% and 240% increases, respectively, in neuronal birth (neurogenesis) within the hypothalamus, a highly conserved brain region involved in hyperactivity. Furthermore, restricted BPA/BPS exposure specifically during the neurogenic window caused later hyperactive behaviors in zebrafish larvae. Unexpectedly, we show that BPA-mediated precocious neurogenesis and the concomitant behavioral phenotype were not dependent on predicted estrogen receptors but relied on androgen receptor-mediated up-regulation of aromatase. Although human epidemiological results are still emerging, an association between high maternal urinary BPA during gestation and hyperactivity and other behavioral disturbances in the child has been suggested. Our studies here provide mechanistic support that the neurogenic period indeed may be a window of vulnerability and uncovers previously unexplored avenues of research into how endocrine disruptors might perturb early brain development. Furthermore, our results show that BPA-free products are not necessarily safer and support the removal of all bisphenols from consumer merchandise.
In humans and rodent models, gestational exposure to bisphenol A (BPA) has been associated with increased risk of developing social (e.g., aggression), psychiatric (e.g., depression), and behavioral (e.g., hyperactivity) challenges later in life (1–7). BPA is a compound used in the production of diverse consumer products, ranging from baby bottles to thermal paper used for credit card receipts (8–10). Even though adults can experience adverse health following continued exposure to BPA, the fetal brain is especially vulnerable because of an immature xenobiotic-metabolizing system and blood–brain barrier (11, 12). Exactly how BPA exposure in utero translates into neurodevelopmental disorders later in life is only beginning to be explored (13–15).
Despite a wide body of research illustrating adverse effects of BPA, controversy exists around the true effects of low-dose exposure, as is most often the case in humans. In accordance with standardized toxicological testing procedures, government agencies in the United States (the US Environmental Protection Agency, USEPA), Canada (Health Canada), and Europe (the European Food Safety Authority, EFSA) have established tolerable daily intake levels, ranging from 25–50 μg BPA⋅kg body weight−1⋅d−1 (16–18). Given these restrictions and societal pressure, manufacturers seeking BPA alternatives have turned to primarily bisphenol S (BPS) to produce “BPA-free” products (19). Indeed, a strong negative correlation between BPA and BPS levels exists, whereby thermal paper that contained high quantities of BPA (milligrams per gram) demonstrated low quantities of BPS (nanograms per gram), and vice versa, suggesting that BPS is the primary replacement for BPA in thermal receipts (20). A recent examination of urine samples in the United States and Asia confirmed previous work showing that 93% of people had detectable levels of BPA but surprisingly showed that 81% had detectable levels of BPS, illustrating the wide-spread use of this poorly known bisphenol analog in consumer products (21).
The physiological effects of BPA on adults are well documented, but the mode of BPA action on the developing brain has yet to be defined clearly, especially in the hypothalamus, which plays a known role in neuroendocrine disorders that are on the rise, including obesity and precocious puberty, as well as anxiety and hyperactivity (1, 2, 4, 10, 22). BPA is commonly thought to exert its effects by acting as a weak estrogen receptor (ER) agonist (8), although antagonism at androgen receptors (ARs) and thyroid receptors (ThRs) has been shown also (23). Proliferating cells in the developing hypothalamus express ERs and ARs (24, 25), and a role for sex steroids (e.g., estrogen, testosterone) in regulating neurogenesis is emerging (26–29). Although results are mixed (30, 31), neurobehavioral studies in humans suggest that the prenatal period could be a window of BPA vulnerability (1–4, 7). Given that BPA is a known endocrine disruptor and that steroid hormones increasingly are being shown to play a role in cell differentiation, our objective here was to determine whether BPA-mediated behavioral phenotypes were the consequence of altered neurogenesis, a developmental process that occurs during the second trimester of gestation. Further, we also studied whether BPS, a common replacement for BPA, likewise causes precocious neurogenesis and concomitant hyperactive behaviors.
Results
BPA Exposure During Hypothalamic Neurogenesis Induces Hyperactivity.
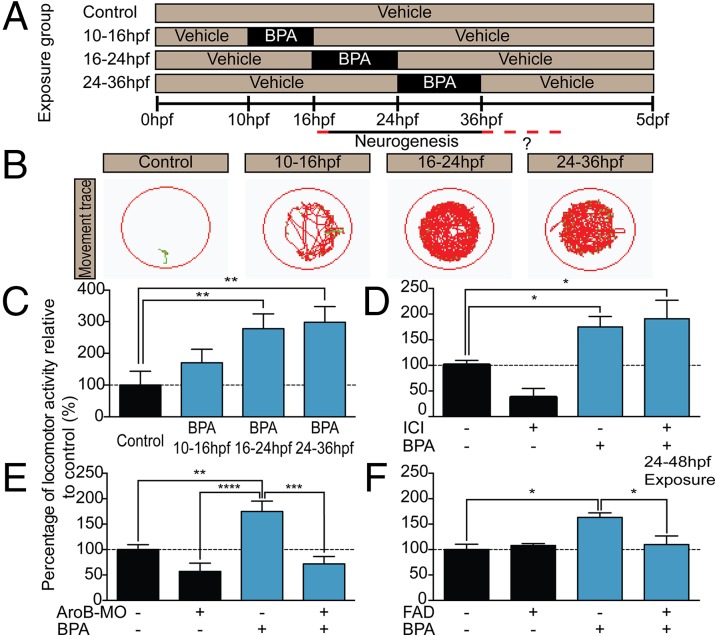
First, we sought to recapitulate the human data using the neurodevelopmentally similar zebrafish as a model and asked whether acute BPA exposure during a time point analogous to gestation would cause behavioral changes in larvae. Specifically, zebrafish embryos were exposed to BPA just before (10–16 h post fertilization; hpf), at the onset of (16–24 hpf), and at the peak of (24–36 hpf) hypothalamic neurogenesis. BPA was washed out after each restricted time point, and zebrafish were assayed at the larval stage, 5 d post fertilization (dpf), for changes in locomotor activity (Fig. 1A; see SI Materials and Methods for further dosing information). Low-dose BPA exposure at 0.1 μM just before neurogenesis (10–16 hpf) had no effect on locomotion but resulted in 2.8- and 2.9-fold increases in locomotor activity when exposure occurred during the early (16–24 hpf) and late (24–36 hpf) neurogenic periods, respectively (Fig. 1 B and C). Precedents in zebrafish demonstrate that this type of locomotor activity (i.e., hyperactivity burst) can be an indicator of anxiety-like behavior (32, 33). We also found that chronic BPA exposure (across 0–5 dpf) resulted in an inverted U-shaped hyperactivity dose–response curve (Fig. S1A), consistent with previous findings (32). Given that physiological responses to endocrine disruptors are known to be biphasic (10), with elevated responses at nanomolar concentrations, we next asked whether a lower dose of BPA also would cause hyperactive behavior. Additionally, because BPA is thought to act via ERs, we sought to determine whether ER inhibition alleviated BPA effects on hyperactivity. Indeed, the same BPA dose found in a local water body (Oldman River, Alberta, Canada) (34) (0.0068 μM, considered a very low dose) (Fig. 1D) and a more moderate BPA dose (1 μM) (Fig. S1B) caused hyperactivity that, unexpectedly, was not blocked by the broad ER ligand ICI 182,780 (hereafter, “ICI”) (Figs. 1D and 2C). This failure to antagonize BPA effects indicated that BPA might not act via classic nuclear (i.e., ERα, ERβ) or membrane-bound (i.e., mER, GPR30) ERs. This same ICI dose was sufficient to antagonize ER activity, because it blocked vtg1 expression in gonad and liver tissues (Fig. S2).
Fig. 1.
BPA exposure induces hyperactive behaviors in larval zebrafish during the window of hypothalamic neurogenesis. (A) Treatment paradigm for BPA exposure. (B) Representative locomotor activity scribes with fish (green) and movement (red) shown. Locomotion in 5-dpf controls and groups exposed to 0.1 μM BPA from 10–16 hpf is compared with locomotion in groups exposed to 0.1 μM BPA from 16–24 hpf and 24–36 hpf. (C) Quantified locomotor activity for 5-dpf zebrafish treated with 0.1 μM BPA from 10–16 hpf, 16–24 hpf, and 24–36 hpf. (D) Locomotor activity in 5-dpf zebrafish coexposed from 0–5 dpf to 0.0068 μM BPA + 1 μM ICI. (E) Locomotor activity in 5-dpf AroB morphants exposed from 0–5 dpf to 0.0068 μM BPA. (F) Locomotor activity in 5-dpf zebrafish coexposed from 24–48 hpf to 1 μM BPA + FAD. (D and E) BPA exposure in AroB morphants and BPA+ICI treatment were run in the same experiment. Results have been separated for simplicity, and controls are shown twice. Data in C–F are shown as mean ± SEM; *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001 [one-way ANOVA, Tukey’s Honestly Significant Difference test (Tukey's HSD)]; n = 5–10 fish per group.
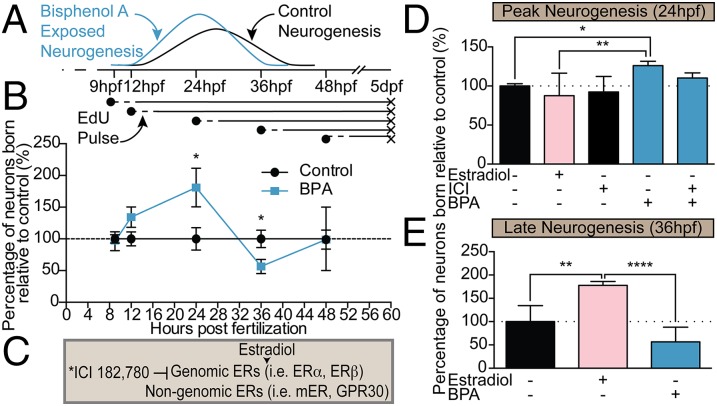
Fig. 2.
Precocious neurogenesis in the hypothalamus following BPA exposure is independent of ER signaling. (A) Cartoon of the experimental design and sample neurogenic curves for BPA-exposed (blue) and control (black) groups. (B) Quantification of neurons born at each time point in BPA (0.0068 μM) relative to vehicle treatment (set to 100%). Neuronal birth was identified by EdU and α-HuC colabeling in BPA-treated and control coronal sections through the hypothalamus. Data are shown as mean ± SEM; *P < 0.05 (Student’s t test); n = 3–11 fish per group. (C) Representation of ICI interaction with ERs. *ICI 182,780 is an antagonist to genomic ERs and membrane-bound ER (mER) but is an agonist toward GPR30. (D) Neuronal birth in 5-dpf zebrafish exposed to 0.0068 μM estradiol or 0.0068 μM BPA or coexposed to 0.0068 μM BPA + 1 μM ICI and pulsed with EdU at 24 hpf. (E) Neuronal birth in 5-dpf zebrafish exposed to 0.0068 μM 17β-Estradiol or 0.0068 μM BPA and pulsed with EdU at late neurogenesis (36 hpf). Data in D and E are shown as mean ± SEM; *P < 0.05, **P < 0.01, ****P < 0.0001 (ANOVA, Tukey’s HSD); n = 3–11.
These data were puzzling, because BPA generally is considered to act via ERs. We therefore looked to other components of the estrogen synthesis pathway and asked whether aromatase B (AroB), the key enzyme for local estradiol synthesis, which is expressed in hypothalamic progenitor cells, mediates the effects of BPA. First, we relied on transient knockdown of AroB with targeted morpholinos (MOs) (Fig. S3) and observed a complete block of the BPA-mediated increase in locomotor activity at both very low (0.0068 μM) (Fig. 1E) and moderate (1 μM) (Fig. S1C) BPA doses. Second, we used the selective aromatase inhibitor fadrozole (FAD) (1 μM) to determine whether AroB catalytic activity was required for BPA-mediated behavioral changes. Indeed, coexposure to very-low-dose BPA (0.0068 μM) + FAD (1 μM) (Fig. 1F) and moderate-dose BPA (1 μM) + FAD (1 μM) (Fig. S1D) during the hypothalamic neurogenic window (24–48 hpf) lowered BPA-induced hyperactivity nearly to control levels, suggesting that AroB enzymatic activity indeed is required.
BPA Causes Precocious Neurogenesis in the Hypothalamus.
Next, we sought to test our hypothesis that these behavioral changes are the result of altered neurogenesis in the hypothalamus. Zebrafish hypothalamic progenitors undergo neuronal differentiation between 18–36 hpf (35), and neural development is complete by 48 hpf. By 5 dpf, zebrafish emerge as prey-seeking larva. To assess quantitatively the timing of neuronal birth in embryonic zebrafish exposed from 0–5 dpf to very-low-dose (0.0068 μM) BPA or vehicle control, we performed birthdating experiments by pulse-labeling embryos with 5-ethynyl-2′-deoxyuridine (EdU) at specific time points before (9 and 12 hpf), during (24 and 36 hpf), and after (48 hpf) the neurogenic window (Figs. 2A and 3). We then assessed 5-dpf zebrafish for the number of neurons born (i.e., that exited the cell cycle) at each time point of EdU exposure (which therefore retained high levels of the thymidine analog) and that also were colabeled with the early neuronal marker HuC/D (Fig. 3). The number of newly born neurons in the hypothalamus of BPA-treated embryos increased significantly, to 180% relative to controls (set at 100%), at 24 hpf (Figs. 2B and 3 and Fig. S4A) and decreased to 60% of control levels by 36 hpf (Figs. 2 and 3 and Fig. S4B), consistent with the notion that precocious neurogenesis at 24 hpf exhausted a portion of the progenitor pool normally reserved for 36 hpf. In contrast, hypothalamic neurogenesis was not significantly altered at 9, 12, or 48 hpf—time points outside the window of neurogenesis (Fig. 2B). Moreover, neuronal birth in several other embryonic brain regions was examined, including the thalamus, tectum, and hindbrain, and no significant changes in neurogenesis were detected; thalamic data are shown in Fig. 4. In addition, a moderate (1 μM) BPA dose did not increase neurogenesis statistically in zebrafish pulsed with EdU at 24 hpf (Fig. S4A) or decrease neurogenesis in zebrafish pulsed with EdU at 36 hpf (Fig. S4B). Taken together these data suggest that the effect of BPA exposure is brain region-specific and nonlinear, with nanomolar concentrations perhaps having greater potency in inducing neurogenesis than micromolar concentrations.
Fig. 3.
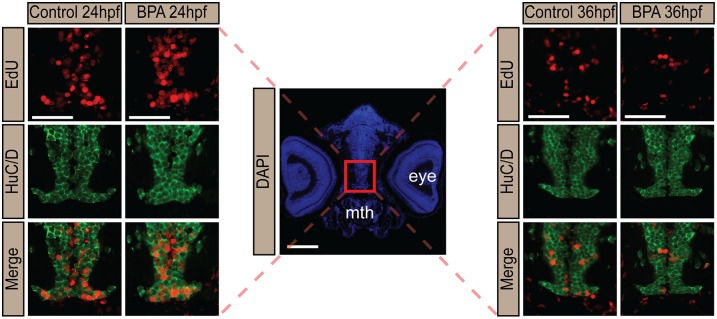
BPA exposure induces precocious neuronal birth in the hypothalamus. Representative immunohistochemistry images of rostral hypothalamus in larval zebrafish exposed to 0.0068 μM BPA at 0–5 dpf. EdU (red), α-HuC (green), and merged hypothalamic sections are shown. The red box in the DAPI (blue)-stained image marks the hypothalamus. Mth, mouth. (Scale bar: 50 μm.)
Fig. 4.
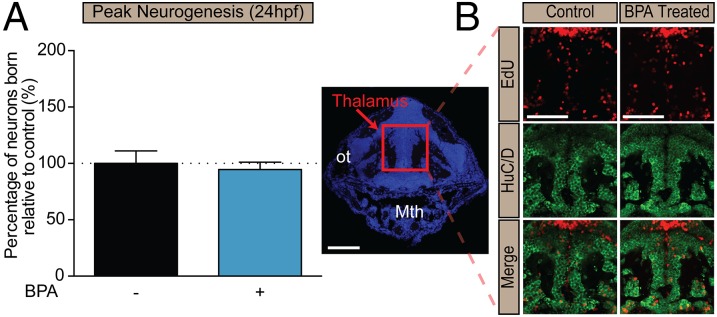
BPA exposure does not cause precocious neurogenesis in other brain regions. (A) Quantification of neurons born at 24 hpf in the thalamus of larvae exposed to 0.0068 μM BPA at 0–5 dpf. Data are shown as mean ± SEM; P > 0.05 (Student’s t test); n = 3 or 4 fish. (B) Representative immunohistochemistry images of the thalamus of a larval zebrafish exposed to 0.0068 μM BPA at 0–5 dpf and pulsed with EdU at 24 hpf. Mth, mouth; ot, optic tract. (Scale bar: 50 μm.)
Given that BPA is widely considered to function as a weak estrogen agonist (23) [the BPA EC50 is 10,000-fold lower than that of endogenous estrogen (36)] and that estrogen has been shown to regulate neurogenesis (26, 28), we examined whether the same low dose (0.0068 μM) of estrogen (17β-estradiol) likewise caused precocious hypothalamic neurogenesis. Strikingly, treatment with 0.0068 μM 17β-estradiol had no effect on hypothalamic neuronal birth at 24 hpf (Fig. 2D) but significantly induced neurogenesis at 36 hpf (Fig. 2E), perhaps unmasking an unappreciated temporal competence window in hypothalamic development for BPA- and 17β-estradiol–sensitive periods. Consistent with our behavioral data, coexposure with 0.0068 μM BPA + 1 μM ICI failed to reduce significantly the number of neurons born at 24 hpf (Fig. 2D), suggesting that BPA functions in a nonestrogenic manner.
Mechanistic Evaluation of BPA-Induced Precocious Neurogenesis.
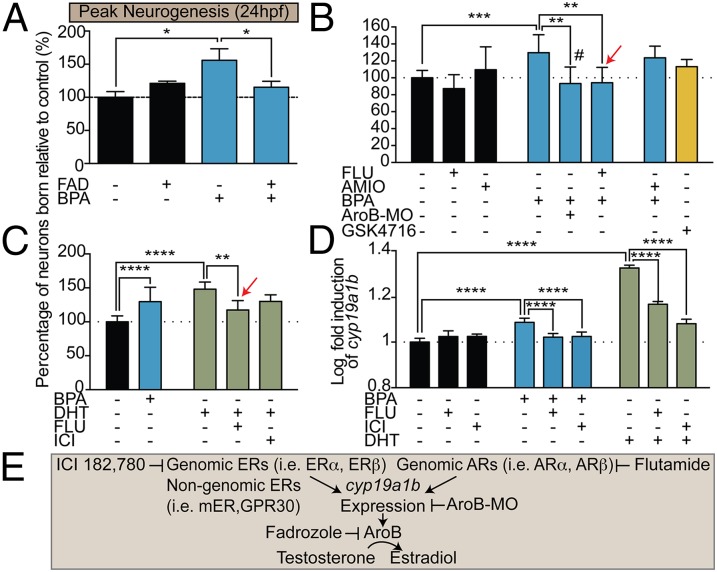
To determine whether AroB also plays a role in BPA-mediated changes in hypothalamic neurogenesis, we coexposed developing embryos to very-low-dose (0.0068 μM) BPA + 1 μM FAD. Strikingly, coexposure to BPA + FAD significantly reduced the number of neurons born at 24 hpf, suggesting that BPA-induced precocious hypothalamic neurogenesis also is dependent on AroB (Fig. 5 A and E). Moreover, increased hypothalamic neurogenesis was attenuated in AroB morphants exposed to very-low-dose BPA (Fig. 5 B and E). BPA has been shown previously to bind a wide range of other receptors, including ThRs, ARs, and estrogen-related receptors (ERRs) (23, 37). To identify the receptor upstream of AroB activation, we exposed zebrafish to the following treatment paradigms: (i) BPA (0.0068 μM) and the AR antagonist flutamide (6.17 μM) (38); (ii) BPA (0.0068 μM) and the ThRα/ThRβ transcriptional repressor amiodarone (50 nM) (39); or (iii) the ERR GSK4716 agonist alone (0.1 μM; no broad antagonist is commercially available) (32). Coexposure to BPA + flutamide significantly attenuated neurogenesis by 40% at 24 hpf, suggesting that AR activation is at least partially required to induce precocious neurogenesis (Fig. 5 B and E). In contrast, cotreatment with BPA + amiodarone did not reduce significantly the number of neurons born, and, conversely, treatment with GSK4716 did not increase neurogenesis (Fig. 5B).
Fig. 5.
BPA-induced precocious neurogenesis is mediated via ARs and aromatase. (A) Quantification of neuronal birth in 5-dpf zebrafish coexposed from 0–5 dpf to 1 μM FAD + 0.0068 μM BPA and pulsed with EdU at 24 hpf. (B) Neuronal birth in 5-dpf zebrafish exposed from 0–5 dpf to 0.1 μM GSK4716 alone or coexposed to BPA + 50 nM amiodarone (AMIO) or to BPA + 6.17 μM flutamide (FLU). The red arrow indicates exposure at 8–48 hpf. The hash mark (#) indicates the AroB morphant (AroB-MO) exposed to BPA. (C) Neuronal birth in 5-dpf zebrafish exposed from 0–5 dpf to 0.0068 μM BPA or 1 μM DHT or coexposed to 1 μM DHT from 0–5 dpf and to 6.17 μM FLU (red arrow) for 8–48 hpf or to 1 μM ICI from 0–5 dpf. (D) Log-transformed relative AroB (cyp19a1b) expression at 48 hpf in zebrafish exposed to 0.0068 μM BPA or 1 μM DHT or coexposed to BPA or 1 μM DHT + 6.17 μM FLU or 1 μM ICI at 8–48 hpf. Data in A–D are shown as mean ± SEM; *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001 (ANOVA, Tukey’s HSD); n = 3–13. (E) Diagram illustrating targets of various pharmacological agents and AroB MO.
Because BPA is commonly thought to act as an AR antagonist and not an AR agonist (23), we reasoned that if BPA indeed activates ARs either directly or indirectly, exposure to the endogenous AR agonist should phenocopy BPA-mediated precocious neurogenesis. Therefore we treated zebrafish embryos with dihydrotestosterone (DHT), a nonaromatizable androgen that is unable to be converted by AroB to estradiol and which binds AR with greater affinity than testosterone (40). Zebrafish exposed to 1 μM DHT (41) displayed a significant increase in the number of early hypothalamic neurons born (160%), whereas cotreatment of embryos with DHT + flutamide (6.17 μM) significantly attenuated neuronal birth (Fig. 5C). In addition, coexposure of DHT (1 μM) + ICI (1 μM) failed to block DHT-mediated increased neurogenesis, further suggesting that very-low-dose BPA at this time point acts through ARs and not ERs to promote hypothalamic neurogenesis.
This unexpected finding led us to question how AR activation links to AroB catalytic activity. Evidence supporting AR-mediated regulation of AroB activity in the hypothalamus was shown previously using androgen-insensitive testicular feminized male rats (42). Moreover, studies in rat and nonhuman primates show that androgen treatment up-regulates aromatase expression and catalytic activity within the nonhuman primate and rat hypothalamus (43). In zebrafish, androgens have been shown to increase AroB (cyp19a1b) expression, although this up-regulation was attributed to the conversion of androgens to estrogenic metabolites, which in turn activate ERs that bind to AroB regulatory elements (41). However, the AroB promoter does contain androgen response elements (AREs) (44). Thus, to characterize the link between BPA-mediated precocious neurogenesis and ARs and AroB catalytic activity, we confirmed that AroB (cyp19a1b) expression was induced in larval zebrafish exposed to 0.0068 μM BPA (45). In addition, we explored whether cotreatment with BPA (0.0068 μM) and the AR antagonist flutamide (6.17 μM) affected AroB (cyp19a1b) expression. Using quantitative reverse transcriptase PCR (qRT-PCR), we found that AroB (cyp19a1b) transcripts at 48 hpf were elevated in zebrafish treated from 8–48 hpf with both BPA (0.0068 μM) and DHT (1 μM) (Fig. 5D). Significantly, coexposure to BPA + flutamide (6.17 μM) as well as DHT (1 μM) + flutamide (6.17 μM) reduced cyp19a1b expression when these compounds were administered across the neurogenic window (8–48 hpf), showing that BPA can induce transcription of AroB (cyp19a1b) via ARs specifically during this developmental period. Interestingly, coexposure of BPA (0.0068 μM) and ICI (1 μM) as well as DHT (1 μM) and ICI (1 μM) also significantly decreased AroB (cyp19a1b) transcript levels (Fig. 5D). Thus, because antagonism of ERs had no effect on BPA- and DHT-mediated precocious neurogenesis (Figs. 2D and 5C), we propose that ER-driven AroB transcription is uncoupled from the neurogenic phenotype of BPA-exposed zebrafish. Combined, these data support the hypothesis that BPA might act via agonism of ARs to increase AroB transcription to drive precocious neurogenesis.
BPS Exposure Alters Brain Development and Behavior.
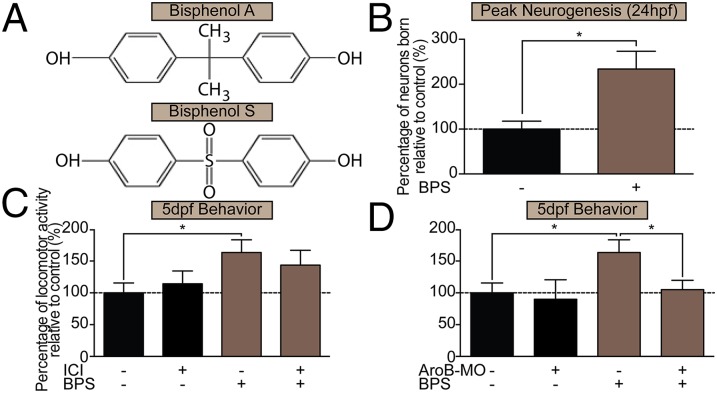
Because of the societal push to rid consumer products of BPA, manufacturers have replaced BPA with structurally similar monomer compounds, particularly BPS (Fig. 6A) (19). Because BPS also displays endocrine-disruptive activity (46), we examined whether BPS had effects similar to those of BPA on precocious hypothalamic neurogenesis and pursuant hyperactive behaviors. We exposed embryonic zebrafish to BPS at the same very low BPA dose used previously, 0.0068 μM. Strikingly, this BPS exposure resulted in a 240% increase in neuronal birth in the rostral hypothalamus at 24 hpf (Fig. 6B) and a significant (160%) increase in the same locomotor bursting activity, which was reduced by transient knockdown of AroB (Fig. 6D) but not by treatment with 1 μM ICI (Fig. 6C). Together, these data imply that both BPS and BPA influence hypothalamic development and may act through a similar AroB-mediated mechanism.
Fig. 6.
BPS exposure increases neurogenesis and hyperactive behavior via a mechanism similar to that of BPA. (A) Chemical structures of BPA and BPS. (B) Neuronal birth in 5-dpf zebrafish exposed from 0–5 dpf to 0.0068 μM BPS. Data are shown as mean ± SEM; *P < 0.05 (Student’s t test); n = 6. (C) Locomotor activity in fish coexposed from 0–5 dpf to 0.0068 μM BPS + 1 μM ICI. (D) Locomotor activity in 5-dpf controls and AroB morphants exposed from 0–5 dpf to 0.0068 μM BPS. Data in C and D are shown as mean ± SEM; *P < 0.05 (ANOVA, Tukey’s HSD); n = 3–9. (C and D) BPS exposure in AroB morphants and BPS+ICI treatment were run in same experiment. Results have been separated for simplicity, and controls are shown twice.
Discussion
Our study is the first, to our knowledge, to show that BPA/BPS can alter the normal developmental timing of this critical neuroendocrine center, the consequences of which can lead to early synaptogenesis and improper fine-tuning of the brain later in development. In addition, we are the first, to our knowledge, to link BPA to AR and AroB signaling during hypothalamic development. Finally, our study provides further evidence for the hypothesis that the prenatal period is indeed a window of vulnerability to BPA/BPS in humans and suggests that pregnant woman should be particularly mindful of their use of plastics and the handling of thermal receipt papers throughout gestation, especially during the second trimester.
Region-specific perturbations in neurogenesis have been linked to altered synaptic connectivity (47), which can lead to downstream effects on proper pruning and manifest in behavioral deficits later in life. Exactly how the timing of neurogenesis becomes altered is an active area of research. For example, proliferating cells in the developing cortex, hippocampus, olfactory bulb, and hypothalamus all express ERs (48) and ARs (25, 49), and a clear role for sex steroids (e.g., estrogen, testosterone) in regulating neurogenesis in vivo (26, 28) and in vitro (50, 51) is emerging, especially in the adult brain (27, 29, 52). In embryonic and adult zebrafish, ERs and AroB are coexpressed in hypothalamic radial glia and are thought to control differentiation and migrational changes that occur in response to estrogen exposure (29, 53). These findings have led to the notion that AroB-expressing radial glia are plastic and initiate adult neurogenesis in response to estrogenic challenges as a way to maintain homeostatic physiologies (52). In the developing mammalian brain, AroB is broadly expressed in radial glia throughout the CNS and becomes restricted to only a small subset of neurogenic niches by adulthood, the most studied of which is the hippocampus (26, 43). Indeed, blocking ERs halfway through the mouse neurogenic window (at embryonic day15) results in decreased proliferation in the developing neocortex (26), illustrating the sex steroid sensitivity of progenitors in mammals. Within the adult zebrafish hypothalamus, antagonism of ERs also decreases cell proliferation (29), demonstrating conserved roles of ER signaling among teleosts and mammalian progenitors. Interestingly, AroB inhibition in the adult zebrafish forebrain was shown to have no significant effect on progenitor cell proliferation or neuronal differentiation (29), implying differences in progenitor biology in embryonic zebrafish versus adults. Furthermore, we propose that we have uncovered a temporal competency window for BPA effects on hypothalamic neurogenesis that is separate from estrogen signaling. This phenotype could be caused by changes in the expression of all three zebrafish ERs (ERα, ERβ1, and ERβ2), which increases dramatically between 24 and 48 hpf (53). Given that the peak of BPA susceptibility occurs at 24 hpf, and we show a role for ARs in BPA-mediated precocious neurogenesis, it will be interesting to see whether AR expression increases earlier than ER expression in the developing hypothalamus. Considered together, the known influence of sex steroids on adult neurogenesis and the evolving role for these hormones in regulating neural development point to a need to understand fully how endocrine-disrupting chemicals can influence this process in utero.
Evidence for androgen pathway-mediated increases in AroB transcription is conflicting in both mammalian and lower vertebrates. For example, up-regulation of AroB transcripts following androgen treatment has been shown in mammals, ricefield eel, and in teleosts such as zebrafish and channel catfish; direct AR regulation of AroB has been demonstrated in ricefield eel, but in teleosts increased AroB expression is attributed to signaling by estrogenic metabolites and not to ARs directly (41, 43, 54–56). In rodents (mice and rats), however, results of AR-mediated changes in AroB expression are mixed, with reports that androgens increase, decrease, or have no effect on AroB transcription (57–60). Interestingly, AREs have been identified in the promoter of AroB (cyp19a1b) in teleosts and humans, suggesting that AR may modulate AroB directly; however, AREs have not been identified in murine animals, perhaps suggesting that there is an indirect mechanism of AroB modulation in these species (43, 44). Here, through the use of an AR antagonist, we show that AR pathway activation also can increase AroB expression in teleosts and starts to build a connection between BPA and AR activation, which then modulates AroB. Because BPA conventionally is thought to act as an AR antagonist, our finding that BPA indeed might activate AR was unexpected; however, in vitro studies previously have shown BPA agonism of AR at similar concentrations (1–10 nM) (23). In mammals, hypothalamic AR and AroB transcript expression is higher in prenatal males than in prenatal females, and thus our results linking BPA and AR-mediated AroB up-regulation with altered brain development might provide insight into why certain neurodevelopmental etiologies are male-specific (60, 61). Nevertheless, future studies are needed to determine the exact mechanism by which BPA modulates AR signaling and AroB up-regulation, leading to precocious neurogenesis.
In our model (Fig. S5), BPA activation of ARs results in the up-regulation of AroB transcription, presumably leading to increased estradiol production in the smooth endoplasmic reticulum and diffusion into the cytoplasm. The exact downstream targets of locally produced estradiol remain uncertain, because here we show that ERs, as likely targets of either BPA or locally produced estradiol, are not involved in BPA-mediated precocious neurogenesis. Alternatively, AroB also converts androstenedione to the estradiol intermediate estrone, which may play an unappreciated role in hypothalamic progenitors. Continued examination of the biological effects of local estrogen synthesis on hypothalamic neural progenitor cells may yield important insights into novel mechanisms of endocrine-disrupting chemicals.
Controversy regarding the true effects of low-dose BPA exposure on human health remains, probably reflecting the variability that accompanies different assays and model systems. Our study shows that low-dose BPA and BPS exposure has physiological effects. We purposefully chose the 0.0068-μM dose because this is the exact concentration of BPA measured in the Oldman River, a major Albertan waterway that serves as a life source to two major urban centers (34). For comparison, waterborne exposure of 1 μM (12 μg/kg) BPA is comparable to the BPA concentration normally found in human placental tissue (12.7 μg/kg) (32, 62); thus the 0.0068-μM doses used herein are magnitudes lower than levels found in human placenta and 100-fold lower than circulating levels measured in fetal serum (0.0101 μM) (62). When determining tolerable daily intake levels, government organizations such as USEPA, Health Canada, and the EFSA rely on linear dose–response relationships and so begin compound testing at high doses, then lowering the dose to the level at which no physiological effect is observed. However, many endocrine-disrupting compounds follow alternative U-shaped dose–response curves, whereby exposure to midrange concentrations activates physiological defense mechanisms against the compound, but at low-range concentrations, the compound mimics endogenous hormones (10). Thus, our finding that BPA at a very low dose (0.0068 μM) alters neurogenesis and that a moderate BPA (1 μM) dose did not affect neurogenesis significantly calls for change in government-sanctioned methods of assessing human tolerable daily intake levels.
Recently, manufacturers have turned to BPS with little proper toxicology testing to produce the “BPA-free” products (19) demanded by society. Indeed, there is a strong negative correlation between BPA and BPS levels in thermal receipts (20), showing that BPS is the primary replacement for BPA. Because there are many structurally similar bisphenol analogs [e.g., BPB, BPE, BPF, BPS, and 4-cumylphenol] (63) that are potential candidates for manufacturing, and given our data herein, a societal push to remove all bisphenols from our consumer goods is justified.
Materials and Methods
For further information on materials and methods, see SI Materials and Methods.
Zebrafish Husbandry and Contaminant Preparation.
All protocols and procedures were approved by the Health Science Animal Care Committee (protocol #M10079) at University of Calgary in compliance with the Guidelines of the Canadian Council of Animal Care. Wild-type zebrafish embryos were maintained at 28 °C in embryo medium (E3) as described by Westerfield, et al. (64). BPA (239658; Sigma), BPS (103039; Sigma), and 17β-estradiol (E8875; Sigma) contaminants as well as the pharmacological agents ICI 182,780 (I4409; Sigma), fadrozole (F3806; Sigma), flutamide (F9397; Sigma), GSK4716 (G6173; Sigma), and amiodarone (A8423; Sigma) were prepared in a 1:3 ratio of 0.002% (vol/vol) 1 M NaOH to 95% EtOH (vehicle) and were diluted to the final concentration in E3. DHT (D-073; Cerilliant) treatments and vehicle controls were prepared in 0.08% (vol/vol ) MeOH in E3. Zebrafish embryos were immersed in treatment within 3 hpf except where otherwise indicated. Chemical structures were designed using ChemBioDraw 13.0 (PerkinElmer) software.
MO Analyses.
Mixtures of ATG and splice-blocking MOs (AroB-MOs) were engineered by Gene-Tools (Philomath) (5′-AGGCTTCCATCATCCCCAACTTCAT-3′), (5′-CGAGCCTGAGAGGACAACAAAGACA-3′) and were injected (2.6–4.6 nL) at the one-cell stage.
Locomotor Behavior Assays.
Larval zebrafish were maintained in 96-well plates immersed in respective treatments from 0–5 dpf, and the duration of hyperactivity bursts (33) was assayed by methods similar to those described by Saili, et al. (32).
Neurogenesis Assessments.
Contaminant-exposed embryos were pulsed-labeled with 0.01% (vol/vol) EdU (C10338; Molecular Probes) at 9, 12, 24, 36, and 48 hpf, corresponding to the window of neurogenesis. EdU was washed off after 30 min, and embryos were replaced in their respective treatments until they were killed at 5 dpf by overnight immersion in 4% paraformaldehyde (PFA) at 4 °C. Processed slides were imaged, and EdU+/α-HuC+ cells were counted by three independent persons to ensure accuracy. No differences in cell morphology or total number of cells were noted between treatments.
Sectioning and Immunohistochemistry.
After overnight fixation, larvae were cryoprotected, embedded in optimum cutting temperature (OCT) compound (Clear Frozen Section Compound; CA95057-838; VWR Scientific), snap frozen, and kept at −80 °C. Embedded larvae were cryosectioned coronally through the hypothalamus. After 20 min of antigen retrieval, slides were blocked in 5% normal goat serum (S1000; Vector Labs) for 1 h. Slides were incubated overnight with α-HuC (A21271; Molecular Probes) and then were incubated with Alexa Fluor 488 IgG (A11001; Molecular Probes) for 2 h. DAPI (D1308; Molecular Probes) was applied, and slides were treated with Click-iT EdU Kit (C10338; Molecular Probes).
Transcript Measurement Assays.
qRT-PCR and PCR reactions were performed on treated larvae at 48 hpf following the protocol in Kurrasch, et al. (65). Primer sequences are given in Table S1.
Statistical Analyses.
Assumptions of normality and equality of variance were met, and ANOVAs with Tukey’s honestly significant difference (HSD) and Student’s t test were performed where indicated using Prism 6 (GraphPad Software). qRT-PCR data were log transformed before analyses.
Supplementary Material
Acknowledgments
We thank Gaurav Kaushik and Natalia Klenin for technical assistance on this project. The research described in these studies was supported by National Sciences and Engineering Research Council of Canada (NSERC) Grant DG386445 (to D.M.K.), NSERC Discovery Grants Program–Individual Grant 156910 (to H.R.H.), and NSERC Postgraduate Scholarships–Doctoral (2-year) Program Grant 459881-2014 (to C.D.K.).
Footnotes
The authors declare no conflict of interest.
*This Direct Submission article had a prearranged editor.
This article contains supporting information online at www.pnas.org/lookup/suppl/doi:10.1073/pnas.1417731112/-/DCSupplemental.
References
- 1.Braun JM, et al. Prenatal bisphenol A exposure and early childhood behavior. Environ Health Perspect. 2009;117(12):1945–1952. doi: 10.1289/ehp.0900979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Braun JM, et al. Impact of early-life bisphenol A exposure on behavior and executive function in children. Pediatrics. 2011;128(5):873–882. doi: 10.1542/peds.2011-1335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Perera F, et al. Prenatal bisphenol a exposure and child behavior in an inner-city cohort. Environ Health Perspect. 2012;120(8):1190–1194. doi: 10.1289/ehp.1104492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Harley KG, et al. Prenatal and early childhood bisphenol A concentrations and behavior in school-aged children. Environ Res. 2013;126:43–50. doi: 10.1016/j.envres.2013.06.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Anderson OS, et al. Perinatal bisphenol A exposure promotes hyperactivity, lean body composition, and hormonal responses across the murine life course. FASEB J. 2013;27(4):1784–1792. doi: 10.1096/fj.12-223545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Tian YH, Baek JH, Lee SY, Jang CG. Prenatal and postnatal exposure to bisphenol a induces anxiolytic behaviors and cognitive deficits in mice. Synapse. 2010;64(6):432–439. doi: 10.1002/syn.20746. [DOI] [PubMed] [Google Scholar]
- 7.Evans SF, et al. Prenatal bisphenol A exposure and maternally reported behavior in boys and girls. Neurotoxicology. 2014;45C:91–99. doi: 10.1016/j.neuro.2014.10.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Krishnan AV, Stathis P, Permuth SF, Tokes L, Feldman D. Bisphenol-A: An estrogenic substance is released from polycarbonate flasks during autoclaving. Endocrinology. 1993;132(6):2279–2286. doi: 10.1210/endo.132.6.8504731. [DOI] [PubMed] [Google Scholar]
- 9.Liao C, Kannan K. Widespread occurrence of bisphenol A in paper and paper products: Implications for human exposure. Environ Sci Technol. 2011;45(21):9372–9379. doi: 10.1021/es202507f. [DOI] [PubMed] [Google Scholar]
- 10.Vandenberg LN, Maffini MV, Sonnenschein C, Rubin BS, Soto AM. Bisphenol-A and the great divide: A review of controversies in the field of endocrine disruption. Endocr Rev. 2009;30(1):75–95. doi: 10.1210/er.2008-0021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Nishikawa M, et al. Placental transfer of conjugated bisphenol A and subsequent reactivation in the rat fetus. Environ Health Perspect. 2010;118(9):1196–1203. doi: 10.1289/ehp.0901575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Adinolfi M. The development of the human blood-CSF-brain barrier. Dev Med Child Neurol. 1985;27(4):532–537. doi: 10.1111/j.1469-8749.1985.tb04581.x. [DOI] [PubMed] [Google Scholar]
- 13.Kim K, et al. Potencies of bisphenol A on the neuronal differentiation and hippocampal neurogenesis. J Toxicol Environ Health A. 2009;72(21-22):1343–1351. doi: 10.1080/15287390903212501. [DOI] [PubMed] [Google Scholar]
- 14.Komada M, et al. Maternal bisphenol A oral dosing relates to the acceleration of neurogenesis in the developing neocortex of mouse fetuses. Toxicology. 2012;295(1-3):31–38. doi: 10.1016/j.tox.2012.02.013. [DOI] [PubMed] [Google Scholar]
- 15.Itoh K, Yaoi T, Fushiki S. Bisphenol A, an endocrine-disrupting chemical, and brain development. Neuropathology. 2012;32(4):447–457. doi: 10.1111/j.1440-1789.2011.01287.x. [DOI] [PubMed] [Google Scholar]
- 16.US Environmental Protection Agency 2008 Child-specific exposure factors handbook, EPA/600/R-06/096F, September 2008 (National Center for Environmental Assessment, Office of Research and Development, Washington, DC). Available at www.epa.gov/ncea. Accessed Dec. 13, 2013. [PubMed]
- 17.Health Canada 2009 Survey of bisphenol A in canned drink products. Available at www.hc-sc.gc.ca/fn-an/securit/packagemball/bpa/bpa_survey-enquete-can-eng.php. Accessed December 13, 2013.
- 18.European Food Safety Authority 2007 EFSA re-evaluates safety of bisphenol A and sets tolerable daily intake. Available at www.efsa.europa.eu/en/press/news/afc070129.htm. Accessed December 13, 2013.
- 19.Grignard E, Lapenna S, Bremer S. Weak estrogenic transcriptional activities of Bisphenol A and Bisphenol S. Toxicol In Vitro. 2012;26(5):727–731. doi: 10.1016/j.tiv.2012.03.013. [DOI] [PubMed] [Google Scholar]
- 20.Liao C, Liu F, Kannan K. Bisphenol s, a new bisphenol analogue, in paper products and currency bills and its association with bisphenol a residues. Environ Sci Technol. 2012;46(12):6515–6522. doi: 10.1021/es300876n. [DOI] [PubMed] [Google Scholar]
- 21.Liao C, et al. Bisphenol S in urine from the United States and seven Asian countries: Occurrence and human exposures. Environ Sci Technol. 2012;46(12):6860–6866. doi: 10.1021/es301334j. [DOI] [PubMed] [Google Scholar]
- 22.Bourguignon JP, et al. Neuroendocrine disruption of pubertal timing and interactions between homeostasis of reproduction and energy balance. Mol Cell Endocrinol. 2010;324(1-2):110–120. doi: 10.1016/j.mce.2010.02.033. [DOI] [PubMed] [Google Scholar]
- 23.Wetherill YB, et al. In vitro molecular mechanisms of bisphenol A action. Reprod Toxicol. 2007;24(2):178–198. doi: 10.1016/j.reprotox.2007.05.010. [DOI] [PubMed] [Google Scholar]
- 24.MacLusky NJ, Lieberburg I, McEwen BS. The development of estrogen receptor systems in the rat brain: Perinatal development. Brain Res. 1979;178(1):129–142. doi: 10.1016/0006-8993(79)90093-3. [DOI] [PubMed] [Google Scholar]
- 25.Gorelick DA, Watson W, Halpern ME. Androgen receptor gene expression in the developing and adult zebrafish brain. Dev Dyn. 2008;237(10):2987–2995. doi: 10.1002/dvdy.21700. [DOI] [PubMed] [Google Scholar]
- 26.Martínez-Cerdeño V, Noctor SC, Kriegstein AR. Estradiol stimulates progenitor cell division in the ventricular and subventricular zones of the embryonic neocortex. Eur J Neurosci. 2006;24(12):3475–3488. doi: 10.1111/j.1460-9568.2006.05239.x. [DOI] [PubMed] [Google Scholar]
- 27.Fowler CD, Liu Y, Wang Z. Estrogen and adult neurogenesis in the amygdala and hypothalamus. Brain Res Brain Res Rev. 2008;57(2):342–351. doi: 10.1016/j.brainresrev.2007.06.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Vosges M, et al. 17α-Ethinylestradiol and nonylphenol affect the development of forebrain GnRH neurons through an estrogen receptors-dependent pathway. Reprod Toxicol. 2012;33(2):198–204. doi: 10.1016/j.reprotox.2011.04.005. [DOI] [PubMed] [Google Scholar]
- 29.Diotel N, et al. Effects of estradiol in adult neurogenesis and brain repair in zebrafish. Horm Behav. 2013;63(2):193–207. doi: 10.1016/j.yhbeh.2012.04.003. [DOI] [PubMed] [Google Scholar]
- 30.Yolton K, et al. Prenatal exposure to bisphenol A and phthalates and infant neurobehavior. Neurotoxicol Teratol. 2011;33(5):558–566. doi: 10.1016/j.ntt.2011.08.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Miodovnik A, et al. Endocrine disruptors and childhood social impairment. Neurotoxicology. 2011;32(2):261–267. doi: 10.1016/j.neuro.2010.12.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Saili KS, et al. Neurodevelopmental low-dose bisphenol A exposure leads to early life-stage hyperactivity and learning deficits in adult zebrafish. Toxicology. 2012;291(1-3):83–92. doi: 10.1016/j.tox.2011.11.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Kalueff AV, et al. Zebrafish Neuroscience Research Consortium Towards a comprehensive catalog of zebrafish behavior 1.0 and beyond. Zebrafish. 2013;10(1):70–86. doi: 10.1089/zeb.2012.0861. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Sosiak A, Hebben T. A Preliminary Survey of Pharmaceuticals and Endocrine Disrupting Compounds in Treated Municipal Wastewaters and Receiving Rivers of Alberta. Technical Report AE T/773. Alberta Environment; Edmonton, AB, Canada: 2005. [Google Scholar]
- 35.Nesan D, Vijayan MM. Role of glucocorticoid in developmental programming: Evidence from zebrafish. Gen Comp Endocrinol. 2013;181:35–44. doi: 10.1016/j.ygcen.2012.10.006. [DOI] [PubMed] [Google Scholar]
- 36.Matthews JB, Twomey K, Zacharewski TR. In vitro and in vivo interactions of bisphenol A and its metabolite, bisphenol A glucuronide, with estrogen receptors alpha and beta. Chem Res Toxicol. 2001;14(2):149–157. doi: 10.1021/tx0001833. [DOI] [PubMed] [Google Scholar]
- 37.Teng C, et al. Bisphenol A affects androgen receptor function via multiple mechanisms. Chem Biol Interact. 2013;203(3):556–564. doi: 10.1016/j.cbi.2013.03.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Schiller V, et al. Transcriptome alterations in zebrafish embryos after exposure to environmental estrogens and anti-androgens can reveal endocrine disruption. Reprod Toxicol. 2013;42:210–223. doi: 10.1016/j.reprotox.2013.09.003. [DOI] [PubMed] [Google Scholar]
- 39.Liu YW, Chan WK. Thyroid hormones are important for embryonic to larval transitory phase in zebrafish. Differentiation. 2002;70(1):36–45. doi: 10.1046/j.1432-0436.2002.700104.x. [DOI] [PubMed] [Google Scholar]
- 40.Grino PB, Griffin JE, Wilson JD. Testosterone at high concentrations interacts with the human androgen receptor similarly to dihydrotestosterone. Endocrinology. 1990;126(2):1165–1172. doi: 10.1210/endo-126-2-1165. [DOI] [PubMed] [Google Scholar]
- 41.Mouriec K, et al. Androgens upregulate cyp19a1b (aromatase B) gene expression in the brain of zebrafish (Danio rerio) through estrogen receptors. Biol Reprod. 2009;80(5):889–896. doi: 10.1095/biolreprod.108.073643. [DOI] [PubMed] [Google Scholar]
- 42.Roselli CE, Salisbury RL, Resko JA. Genetic evidence for androgen-dependent and independent control of aromatase activity in the rat brain. Endocrinology. 1987;121(6):2205–2210. doi: 10.1210/endo-121-6-2205. [DOI] [PubMed] [Google Scholar]
- 43.Roselli CE, Liu M, Hurn PD. Brain aromatization: Classic roles and new perspectives. Semin Reprod Med. 2009;27(3):207–217. doi: 10.1055/s-0029-1216274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Tong SK, Chung BC. Analysis of zebrafish cyp19 promoters. J Steroid Biochem Mol Biol. 2003;86(3-5):381–386. doi: 10.1016/s0960-0760(03)00347-9. [DOI] [PubMed] [Google Scholar]
- 45.Chung E, Genco MC, Megrelis L, Ruderman JV. Effects of bisphenol A and triclocarban on brain-specific expression of aromatase in early zebrafish embryos. Proc Natl Acad Sci USA. 2011;108(43):17732–17737. doi: 10.1073/pnas.1115187108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Ji K, Hong S, Kho Y, Choi K. Effects of bisphenol s exposure on endocrine functions and reproduction of zebrafish. Environ Sci Technol. 2013;47(15):8793–8800. doi: 10.1021/es400329t. [DOI] [PubMed] [Google Scholar]
- 47.Itoh K, Yaoi T, Fushiki S. Bisphenol A, an endocrine-disrupting chemical, and brain development. Neuropathology. 2012;32(4):447–457. doi: 10.1111/j.1440-1789.2011.01287.x. [DOI] [PubMed] [Google Scholar]
- 48.MacLusky NJ, Chaptal C, McEwen BS. The development of estrogen receptor systems in the rat brain and pituitary: Postnatal development. Brain Res. 1979;178(1):143–160. doi: 10.1016/0006-8993(79)90094-5. [DOI] [PubMed] [Google Scholar]
- 49.Young WJ, Chang C. Ontogeny and autoregulation of androgen receptor mRNA expression in the nervous system. Endocrine. 1998;9(1):79–88. doi: 10.1385/ENDO:9:1:79. [DOI] [PubMed] [Google Scholar]
- 50.Toran-Allerand CD. Sex steroids and the development of the newborn mouse hypothalamus and preoptic area in vitro: Implications for sexual differentiation. Brain Res. 1976;106(2):407–412. doi: 10.1016/0006-8993(76)91038-6. [DOI] [PubMed] [Google Scholar]
- 51.Murashov AK, Pak ES, Hendricks WA, Tatko LM. 17beta-Estradiol enhances neuronal differentiation of mouse embryonic stem cells. FEBS Lett. 2004;569(1-3):165–168. doi: 10.1016/j.febslet.2004.05.042. [DOI] [PubMed] [Google Scholar]
- 52.Diotel N, et al. Aromatase in the brain of teleost fish: Expression, regulation and putative functions. Front Neuroendocrinol. 2010;31(2):172–192. doi: 10.1016/j.yfrne.2010.01.003. [DOI] [PubMed] [Google Scholar]
- 53.Mouriec K, et al. Early regulation of brain aromatase (cyp19a1b) by estrogen receptors during zebrafish development. Dev Dyn. 2009;238(10):2641–2651. doi: 10.1002/dvdy.22069. [DOI] [PubMed] [Google Scholar]
- 54.Zhang Y, et al. Androgen rather than estrogen up-regulates brain-type cytochrome P450 aromatase (cyp19a1b) gene via tissue-specific promoters in the hermaphrodite teleost ricefield eel Monopterus albus. Mol Cell Endocrinol. 2012;350(1):125–135. doi: 10.1016/j.mce.2011.12.001. [DOI] [PubMed] [Google Scholar]
- 55.Lassiter CS, Linney E. Embryonic expression and steroid regulation of brain aromatase cyp19a1b in zebrafish (Danio rerio) Zebrafish. 2007;4(1):49–57. doi: 10.1089/zeb.2006.9995. [DOI] [PubMed] [Google Scholar]
- 56.Kazeto Y, Trant JM. Molecular biology of channel catfish brain cytochrome P450 aromatase (CYP19A2): Cloning, preovulatory induction of gene expression, hormonal gene regulation and analysis of promoter region. J Mol Endocrinol. 2005;35(3):571–583. doi: 10.1677/jme.1.01805. [DOI] [PubMed] [Google Scholar]
- 57.Karolczak M, Küppers E, Beyer C. Developmental expression and regulation of aromatase- and 5alpha-reductase type I mRNA in the male and female mouse hypothalamus. J Neuroendocrinol. 1998;10(4):267–274. doi: 10.1046/j.1365-2826.1998.00200.x. [DOI] [PubMed] [Google Scholar]
- 58.Abe-Dohmae S, Tanaka R, Harada N. Cell type- and region-specific expression of aromatase mRNA in cultured brain cells. Brain Res Mol Brain Res. 1994;24(1-4):153–158. doi: 10.1016/0169-328x(94)90127-9. [DOI] [PubMed] [Google Scholar]
- 59.Lephart ED, Simpson ER, Ojeda SR. Effects of Cyclic AMP and Andre-gens on in vitro Brain Aromatase Enzyme Activity During Prenatal Development in the Rat. J Neuroendocrinol. 1992;4(1):29–36. doi: 10.1111/j.1365-2826.1992.tb00341.x. [DOI] [PubMed] [Google Scholar]
- 60.Negri-Cesi P, Colciago A, Motta M, Martini L, Celotti F. Aromatase expression and activity in male and female cultured rat hypothalamic neurons: Effect of androgens. Mol Cell Endocrinol. 2001;178(1-2):1–10. doi: 10.1016/s0303-7207(01)00442-7. [DOI] [PubMed] [Google Scholar]
- 61.Beyer C, Hutchison JB. Androgens stimulate the morphological maturation of embryonic hypothalamic aromatase-immunoreactive neurons in the mouse. Brain Res Dev Brain Res. 1997;98(1):74–81. doi: 10.1016/s0165-3806(96)00170-8. [DOI] [PubMed] [Google Scholar]
- 62.Schönfelder G, et al. Parent bisphenol A accumulation in the human maternal-fetal-placental unit. Environ Health Perspect. 2002;110(11):A703–A707. doi: 10.1289/ehp.110-1241091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Rosenmai AK, et al. Are structural analogues to bisphenol a safe alternatives? Toxicol Sci. 2014;139(1):35–47. doi: 10.1093/toxsci/kfu030. [DOI] [PubMed] [Google Scholar]
- 64.Westerfield M. The Zebrafish Book. A Guide for the Laboratory Use of Zebrafish (Danio rerio) 4th Ed Univ of Oregon Press; Eugene, OR: 2000. [Google Scholar]
- 65.Kurrasch DM, Nevin LM, Wong JS, Baier H, Ingraham HA. Neuroendocrine transcriptional programs adapt dynamically to the supply and demand for neuropeptides as revealed in NSF mutant zebrafish. Neural Dev. 2009;4(22):1–16. doi: 10.1186/1749-8104-4-22. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.