Abstract
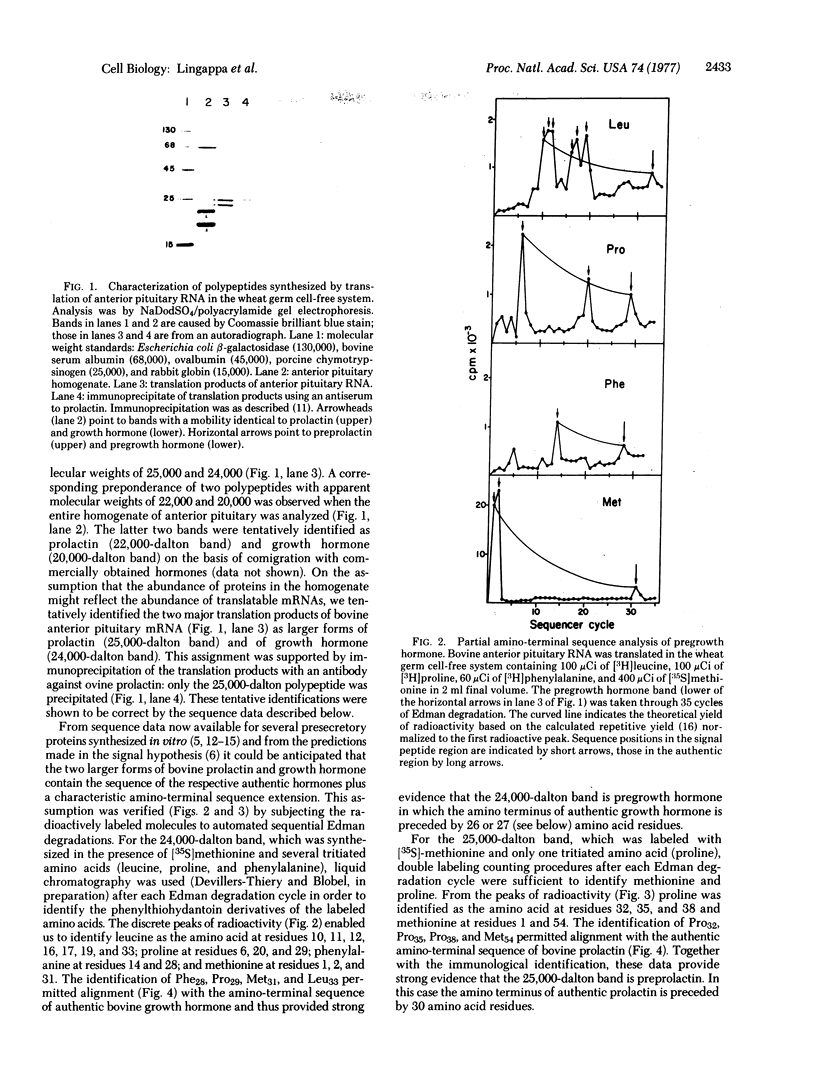
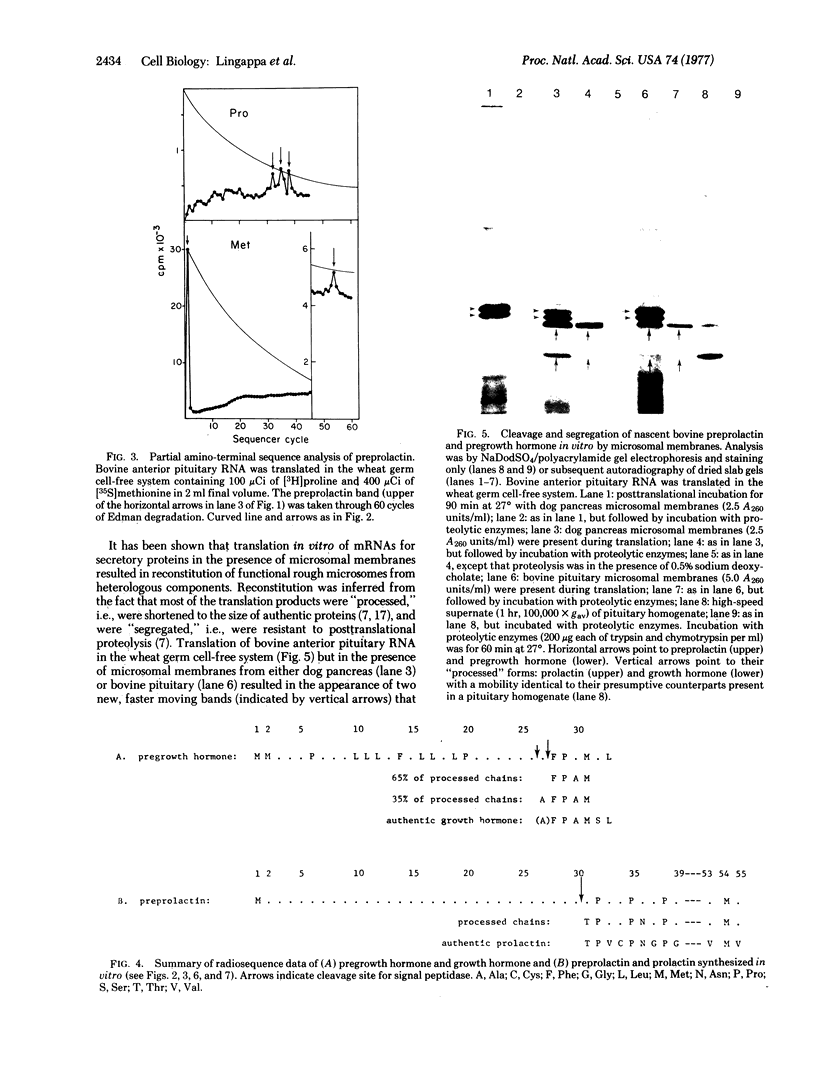
Major translation products of bovine pituitary RNA in a wheat germ cell-free system were identified as larger forms (prehormones) of growth hormone and prolactin containing amino-terminal extensions of 26 or 27 and 30 amino acid residues, respectively. However, translation of bovine pituitary RNA in the wheat germ cell-free system in the presence of microsomal membranes prepared from canine pancreas or bovine pituitary yielded products that were of the same size as authentic growth hormone and prolactin; by partial amino-terminal sequence analysis they were shown to contain the correct unique amino-terminal sequence of prolactin and the two correct amino termini of authentic growth hormone; moreover, they were found to be segregated within the microsomal vesicles in that they were largely inaccessible to degradation by proteolytic enzymes. When microsomal membranes were present after rather than during translation, prehormones were neither cleaved nor segregated. These results strongly suggest that the synthesis and segregation of the authentic hormone observed in the presence of membranes proceeds via a nascent prehormone rather than a completed prehormone.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Birken S., Smith D. L., Canfield R. E., Boime I. Partial amino acid sequence of human placental lactogen precursor and its mature hormone form produced by membrane-associated enzyme activity. Biochem Biophys Res Commun. 1977 Jan 10;74(1):106–112. doi: 10.1016/0006-291x(77)91381-x. [DOI] [PubMed] [Google Scholar]
- Blobel G., Dobberstein B. Transfer of proteins across membranes. I. Presence of proteolytically processed and unprocessed nascent immunoglobulin light chains on membrane-bound ribosomes of murine myeloma. J Cell Biol. 1975 Dec;67(3):835–851. doi: 10.1083/jcb.67.3.835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blobel G., Dobberstein B. Transfer of proteins across membranes. II. Reconstitution of functional rough microsomes from heterologous components. J Cell Biol. 1975 Dec;67(3):852–862. doi: 10.1083/jcb.67.3.852. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burstein Y., Schechter I. Amino acid-sequence variability at the N-terminal extra piece of mouse immunoglobulin light-chain precursors of the same and different subgroups. Biochem J. 1976 Jul 1;157(1):145–151. doi: 10.1042/bj1570145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chan S. J., Keim P., Steiner D. F. Cell-free synthesis of rat preproinsulins: characterization and partial amino acid sequence determination. Proc Natl Acad Sci U S A. 1976 Jun;73(6):1964–1968. doi: 10.1073/pnas.73.6.1964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dannies P. S., Tashjian A. H., Jr Thyrotropin-releasing hormone increases prolactin mRNA activity in the cytoplasm of GH-cells as measured by translation in a wheat germ cell-free system. Biochem Biophys Res Commun. 1976 Jun 21;70(4):1180–1189. doi: 10.1016/0006-291x(76)91027-5. [DOI] [PubMed] [Google Scholar]
- Devillers-Thiery A., Kindt T., Scheele G., Blobel G. Homology in amino-terminal sequence of precursors to pancreatic secretory proteins. Proc Natl Acad Sci U S A. 1975 Dec;72(12):5016–5020. doi: 10.1073/pnas.72.12.5016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dobberstein B., Blobel G., Chua N. H. In vitro synthesis and processing of a putative precursor for the small subunit of ribulose-1,5-bisphosphate carboxylase of Chlamydomonas reinhardtii. Proc Natl Acad Sci U S A. 1977 Mar;74(3):1082–1085. doi: 10.1073/pnas.74.3.1082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dobberstein B., Blobel G. Functional interaction of plant ribosomes with animal microsomal membranes. Biochem Biophys Res Commun. 1977 Feb 21;74(4):1675–1682. doi: 10.1016/0006-291x(77)90637-4. [DOI] [PubMed] [Google Scholar]
- Evans G. A., Rosenfeld M. G. Cell-free synthesis of a prolactin precursor directed by mRNA from cultured rat pituitary cells. J Biol Chem. 1976 May 10;251(9):2842–2847. [PubMed] [Google Scholar]
- Kemper B., Habener J. F., Ernst M. D., Potts J. T., Jr, Rich A. Pre-proparathyroid hormone: analysis of radioactive tryptic peptides and amino acid sequence. Biochemistry. 1976 Jan 13;15(1):15–19. doi: 10.1021/bi00646a003. [DOI] [PubMed] [Google Scholar]
- Maurer R. A., Gorski J., McKean D. J. Partial amino acid sequence of rat pre-prolactin. Biochem J. 1977 Jan 1;161(1):189–192. doi: 10.1042/bj1610189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maurer R. A., Stone R., Gorski J. Cell-free synthesis of a large translation product of prolactin messenger RNA. J Biol Chem. 1976 May 10;251(9):2801–2807. [PubMed] [Google Scholar]
- Palade G. Intracellular aspects of the process of protein synthesis. Science. 1975 Aug 1;189(4200):347–358. doi: 10.1126/science.1096303. [DOI] [PubMed] [Google Scholar]
- Roman R., Brooker J. D., Seal S. N., Marcus A. Inhibition of the transition of a 40 S ribosome-Met-tRNA-i-Met complex to an 80 S ribosome-Met-tRNA-i-Met- complex by 7-Methylguanosine-5'-phosphate. Nature. 1976 Mar 25;260(5549):359–360. doi: 10.1038/260359a0. [DOI] [PubMed] [Google Scholar]
- Sussman P. M., Tushinski R. J., Bancroft F. C. Pregrowth hormone: product of the translation in vitro of messenger RNA coding for growth hormone. Proc Natl Acad Sci U S A. 1976 Jan;73(1):29–33. doi: 10.1073/pnas.73.1.29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Szczesna E., Boime I. mRNA-dependent synthesis of authentic precursor to human placental lactogen: conversion to its mature hormone form in ascites cell-free extracts. Proc Natl Acad Sci U S A. 1976 Apr;73(4):1179–1183. doi: 10.1073/pnas.73.4.1179. [DOI] [PMC free article] [PubMed] [Google Scholar]