Abstract
AIM: To investigate the effects of p57kip2, cyclinE protein and proliferating cell nuclear antigen (PCNA) on occurrence and progression of human pancreatic cancer.
METHODS: The expression of p57kip2, cyclinE protein and PCNA in tumor tissues and adjacent tissues from 32 patients with pancreatic cancer was detected by SP immunohistochemical technique.
RESULTS: The positive expression rate of p57kip2 protein in tumor tissues was 46.9%, which was lower than that in adjacent pancreatic tissues (χ2 = 5.317, P<0.05). p57kip2 protein positive expression remarkably correlated with tumor cell differentiation (P<0.05), but not with lymph node metastasis (P>0.05). The positive expression rate of cyclinE protein in tumor tissues was 68.8%, which was higher than that in adjacent pancreatic tissues (χ2 = 4.063, P<0.05). CyclinE protein positive expression significantly correlated with tumor cell differentiation and lymph node metastasis (P<0.05). The positive expression rate of PCNA in the tumor tissues was 71.9%, which was higher than that in adjacent pancreatic tissues (χ2 = 5.189, P<0.05). PCNA positive expression remarkably correlated with tumor cell differentiation and lymph node metastasis (P<0.05).
CONCLUSION: The decreased expression of p57kip2 and/or overexpression of cyclinE protein and PCNA may contribute to the occurrence and progression of pancreatic cancer. p57kip2, cyclinE protein, and PCNA play an important role in occurrence and progression of pancreatic cancer.
Keywords: p57kip2, CyclinE, PCNA, Human pancreatic cancer
INTRODUCTION
Abnormality in mammalian cell cycle regulation is an important cause of cell proliferation and oncogenesis[1]. Orderly progression of the cell cycle is controlled by a family of cyclins and cyclin-dependent kinases (CDKs), which are restrictively counterbalanced by CDK inhibitors (CDKIs)[2]. Two distinct families of CDKIs, INK4, and CIP/KIP families, which regulate the activity of cyclin-CDK complexes, have been described[3]. The CIP/KIP family, including p21, p27, and p57 proteins, harbors homologous CDK-binding domains or functions of cyclin-CDK complexes and causes the cell cycle to arrest in G1 phase. CyclinE protein is a positive regulator of cell cycle, which promotes the transfer from G1 to S phase. The expression of PCNA remarkably correlates with status of cell proliferation. There are few reports about the relationship between p57kip2 protein as negative factor of cell cycle regulation and pancreatic cancer. In this study, the expression of p57kip2, cyclinE protein, and PCNA in pancreatic cancer tissues and adjacent tissues was detected by immunohistochemical technique to investigate the effects of p57kip2, cyclinE protein, and PCNA on occurrence and progression of human pancreatic cancer.
MATERIALS AND METHODS
Patients and tumor samples
Thirty-two specimens of primary human pancreatic cancer were collected from pancreatic resection performed in the Department of General Surgery, General Hospital of Shenyang Military Command. There were 20 male and 12 female patients, with a mean age of 59.5 years (26-72 years). Nineteen patients had well-differentiated pancreatic cancer, 13 had moderately or poorly-differentiated pancreatic cancer, and 12 had lymph node metastasis. All patients were confirmed by clinicopathological diagnosis. These specimens were fixed in 10% buffered formalin and embedded in paraffin. All sections stained with hematoxylin and eosin were reviewed and kept for further studies.
Immunohistochemical study
Four-micrometer-thick sections from the tissues were cut for immunohistochemical study. The expression of p57kip2, cyclinE protein, and PCNA was assessed by SP immunohistochemical method using anti-human p57kip2 mAb (57P06), anti-human cyclinE protein mAb (13A3), anti-human PCNA mAb (PC10), and the UltraSensitiveTM SP kit (kit-9720). Immunohisto-chemical staining for these proteins was then performed according to the UltraSensitiveTM SP kit manual. All reagents were supplied by Maixin-Bio Co., Fuzhou, China. The cells with brown-yellow granules in the nuclei or cytoplasm were taken as positive. Five hundred cells on each slide were counted. The slides were classified as negative (-), positive (+), strong positive (++), and strongest positive (+++) according to the count of positive cells for p57kip2 and cyclinE proteins less than 10%, 10-25%, 25-50%, and more than 50%, respectively. The slides was distinguished as negative (-), and positive (+) when the count of positive cells were less than 50% and over 50% for PCNA resp-ectively.
Statistical analysis
The χ2 test and Fisher’s exact test were used in the analysis by SAS system statistical software (release 6.12). P<0.05 was considered statistically significant.
RESULTS
Expression of p57kip2 protein
The p57kip2 protein was located in the nuclei or cytoplasm of normal pancreatic cells and positive pancreatic cancer cells with brown-yellow granules (Figure 1A).The positive expression rate of p57kip2 protein in tumor tissues was 46.9%, which was lower than that in adjacent pancreatic tissues (χ2 = 5.317, P<0.05). The positive expression rate of p57kip2 protein in the moderately or poorly differentiated tumor tissues was 23.1%, which was lower than that in well-differentiated tumor tissues (χ2 = 4.979, P<0.05). The positive expression rate of p57kip2 protein in lymph node metastasis group was 25.0%, which was lower than that in non-lymph node metastasis group (P>0.05, Table 1).
Figure 1.
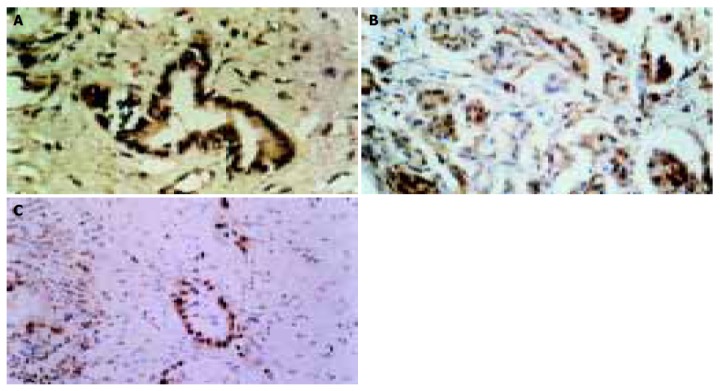
P57kip2 protein in well-differentiated pancreatic adenocarcinoma (A), cyclin E protein in poorly-differentiated pancreatic adenocarcinoma (B), and PCNA protein in moderately-differentiated pancreatic adenocarcinoma (C).
Table 1.
Expression of p57kip2 protein in pancreatic cancer tissues
|
p57kip2 protein expression |
|||||
| Characteristics | - | + | ++ | +++ | Rate (%) |
| Tumor tissue | 17 | 11 | 3 | 1 | 46.9a |
| Well-differentiated | 7 | 9 | 2 | 1 | 63.2c |
| Moderately or | |||||
| poorly-differentiated | 10 | 2 | 1 | 0 | 23.1c |
| Lymph node metastasis | 9 | 2 | 1 | 0 | 25.0e |
| Non-lymph node | |||||
| metastasis | 8 | 9 | 2 | 1 | 60.0e |
| Adjacent tissue | 8 | 13 | 6 | 5 | 75.0a |
P<0.05,
P<0.05,
P>0.05 vs others.
Expression of cyclinE protein
CyclinE protein was located in the nuclei or cytoplasm of normal pancreatic cells and positive pancreatic cancer cells with brown-yellow granules (Figure 1B). The positive expression rate of cyclinE protein in tumor tissues was 68.8%, which was higher than that in adjacent pancreatic tissues (χ2 = 4.063, P<0.05). The positive expression rate of cyclinE protein in moderately or poorly differentiated tumor tissues was 84.6%, which was higher than that in well-differentiated tumor tissues (χ2 = 5.128, P<0.05). The cyclinE protein positive expression rate in lymph node metastasis group was 91.7%, which was higher than that in non-lymph node metastasis group (χ2 = 4.693, P<0.05, Table 2).
Table 2.
Expression of cyclinE protein in pancreatic cancer tissues
| Characteristics |
CyclinE protein expression |
||||
| - | + | ++ | +++ | Rate (%) | |
| Tumor tissue | 10 | 7 | 9 | 6 | 68.8a |
| Well-differentiated | 9 | 5 | 4 | 1 | 52.6c |
| Moderately or | |||||
| poorly-differentiated | 1 | 1 | 6 | 5 | 84.6c |
| Lymph node metastasis | 1 | 1 | 5 | 5 | 91.7e |
| Non-lymph node | |||||
| metastasis | 9 | 6 | 4 | 1 | 55.0e |
| Adjacent tissue | 18 | 6 | 8 | 0 | 43.8a |
P<0.05,
P<0.05,
P<0.05 vs others.
Expression of PCNA
PCNA was located in the nuclei of normal pancreatic cells and positive pancreatic cancer cells with brown-yellow granules (Figure 1C). The positive expression rate of PCNA in tumor tissues was 71.9%, which was higher than that in adjacent pancreatic tissues (χ2 = 5.189, P<0.05). The positive expression rate of PCNA in moderately or poorly diffe-rentiated tumor tissues was 92.3%, which was higher than that in well-differentiated tumor tissues (χ2 = 4.522, P<0.05). The positive expression rate of PCNA in lymph node metastasis group was 100%, which was higher than that in non-lymph node metastasis group (χ2 = 7.513, P<0.05, Table 3).
Table 3.
Expression of PCNA in pancreatic cancer tissues
| Characteristics |
PCNA protein expression |
||
| - | + | Rate (%) | |
| Tumor tissue | 9 | 23 | 71.9a |
| Well-differentiated | 8 | 11 | 57.9c |
| Moderately or poorly-differentiated | 1 | 12 | 92.3c |
| Lymph node metastasis | 0 | 12 | 100e |
| Non-lymph node metastasis | 9 | 11 | 55.0e |
| Adjacent tissue | 18 | 14 | 43.8a |
P<0.05,
P<0.05,
P<0.05 vs others.
Relationship between expression of p57kip2 and cyclinE proteins
The cyclinE protein positive expression rate in tumor tissues of the p57kip2 protein positive expression group and the p57kip2 protein negative expression was 60.0% and 76.5% respectively. There was no significant correlation between the two groups (r = -0.11211, P>0.05, Table 4).
Table 4.
Relationship between expression of p57kip2 and cyclinE proteins
| p57kip2 |
CyclinE protein expression |
||||
| - | + | ++ | +++ | Rate (%) | |
| – | 4 | 4 | 6 | 3 | 76.5 |
| + | 4 | 3 | 2 | 2 | |
| ++ | 2 | 0 | 0 | 1 | 60.0 |
| +++ | 0 | 0 | 1 | 0 | |
DISCUSSION
Studies in recent years have shown that G1 phase regulation is a complex procedure[4-7]. p57kip2 gene is located in chromosome 11p15.5, and p57kip2 protein is a cell cycle inhibitor with molecular weight of 57 ku, which is included in the CIP/KIP family and similar to p21 and p27 proteins in functions[8,9]. Lee et al[10] suggested that the tumor suppressor mechanism of p57kip2 protein may be integrated with cyclin-CDK complexes and makes cell cycle to arrest in the G1 phase. Kondo et al[11] considered that paternal alleles of p57kip2 are imprinted, maternal alleles of p57kip2 are expressed in the normal status, loss of imprinting and imprinting mistakes of p57kip2 lead to a decrease at level of gene expression in tumors. Matsumoto et al[12] reported that p57kip2 protein positive expression rate is 43.3±3.2% in patients with esophageal squamous cell carcinoma. From then on, studies about p57kip2 protein expression in human colorectal carcinoma[13], hepatocellular carcinoma[14,15], prostate tumor[16], neoplastic thyroid tissues[17], epithelial ovarian tumor[18], extrahepatic bile duct carcinoma and intrahepatic cholangiocellular carcinoma have been reported[19,20], but few reports on the relationship between p57kip2 protein expression and pancreatic cancer are available[21]. In this study, we found that the positive expression rate of p57kip2 protein in pancreatic cancer tissues was significantly lower than that in adjacent pancreatic tissues. The worse the cancer cell differentiation, the lower was the p57kip2 protein expression, and there was no correlation between the reduced expression of p57kip2 protein and lymph node metastasis. The results suggest that reduced expression of p57kip2 protein correlates with the occurrence and malignant degree of pancreatic cancer. CyclinE protein is a positive regulating factor in the cell cycle and promotes the genesis and progression of tumors[22-24]. Our results suggest that overexpression of cyclinE is associated with the genesis and malignant degree, as well as lymph node metastasis of pancreatic cancer. PCNA is a δ-assistant factor of DNA synthetase, takes part in DNA biological synthesis and regulates cell cycle and cell proliferation by tetramer with cyclin, CDK and p21. Overexpression of PCNA is associated with a variety of tumors of digestive system including human colorectal cancer[25], gastric cancer[26], hepatocellular carcinoma[27], pancreas tumor[28,29]. The results in the present study suggest that overexpression of PCNA is associated with the occurrence and progression of pancreatic cancer, and malignant proliferation status of pancreatic cancer determined by expression of PCNA is of practical value. Our results suggest that cell proliferative activity is high for the negative or reduced expression of p57kip2 protein. Furthermore, p57kip2 protein plays a role in suppressing cell proliferation. Our findings are in accordance with the results of previous studies[5].
In summary, decreased expression of p57kip2 and/or overexpression of cyclinE protein and PCNA might contribute to the occurrence and progression of pancreatic cancer. The p57kip2, cyclinE protein, and PCNA might play an important role in occurrence and progression of pancreatic cancer.
Footnotes
Science Editor Wang XL and Guo SY Language Editor Elsevier HK
References
- 1.Clurman BE, Roberts JM. Cell cycle and cancer. J Natl Cancer Inst. 1995;87:1499–1501. doi: 10.1093/jnci/87.20.1499. [DOI] [PubMed] [Google Scholar]
- 2.Graña X, Reddy EP. Cell cycle control in mammalian cells: role of cyclins, cyclin dependent kinases (CDKs), growth suppressor genes and cyclin-dependent kinase inhibitors (CKIs) Oncogene. 1995;11:211–219. [PubMed] [Google Scholar]
- 3.Sherr CJ. G1 phase progression: cycling on cue. Cell. 1994;79:551–555. doi: 10.1016/0092-8674(94)90540-1. [DOI] [PubMed] [Google Scholar]
- 4.Sherr CJ. Cancer cell cycles. Science. 1996;274:1672–1677. doi: 10.1126/science.274.5293.1672. [DOI] [PubMed] [Google Scholar]
- 5.Kamb A. Cell-cycle regulators and cancer. Trends Genet. 1995;11:136–140. doi: 10.1016/s0168-9525(00)89027-7. [DOI] [PubMed] [Google Scholar]
- 6.Hunter T, Pines J. Cyclins and cancer. II: Cyclin D and CDK inhibitors come of age. Cell. 1994;79:573–582. doi: 10.1016/0092-8674(94)90543-6. [DOI] [PubMed] [Google Scholar]
- 7.Kamb A, Gruis NA, Weaver-Feldhaus J, Liu Q, Harshman K, Tavtigian SV, Stockert E, Day RS, Johnson BE, Skolnick MH. A cell cycle regulator potentially involved in genesis of many tumor types. Science. 1994;264:436–440. doi: 10.1126/science.8153634. [DOI] [PubMed] [Google Scholar]
- 8.Matsuoka S, Edwards MC, Bai C, Parker S, Zhang P, Baldini A, Harper JW, Elledge SJ. p57KIP2, a structurally distinct member of the p21CIP1 Cdk inhibitor family, is a candidate tumor suppressor gene. Genes Dev. 1995;9:650–662. doi: 10.1101/gad.9.6.650. [DOI] [PubMed] [Google Scholar]
- 9.Orlow I, Iavarone A, Crider-Miller SJ, Bonilla F, Latres E, Lee MH, Gerald WL, Massagué J, Weissman BE, Cordón-Cardó C. Cyclin-dependent kinase inhibitor p57KIP2 in soft tissue sarcomas and Wilms'tumors. Cancer Res. 1996;56:1219–1221. [PubMed] [Google Scholar]
- 10.Lee MH, Reynisdottir I, Massague J. Cloning of p57KIP2, a cyclin dependent kinase inhibitor with unique domain structure and tissue distribution. Genes Dev. 1995;9:639–649. doi: 10.1101/gad.9.6.639. [DOI] [PubMed] [Google Scholar]
- 11.Kondo M, Matsuoka S, Uchida K, Osada H, Nagatake M, Takagi K, Harper JW, Takahashi T, Elledge SJ, Takahashi T. Selective maternal-allele loss in human lung cancers of the maternally expressed p57KIP2 gene at 11p15.5. Oncogene. 1996;12:1365–1368. [PubMed] [Google Scholar]
- 12.Matsumoto M, Furihata M, Ohtsuki Y, Sasaguri S, Ogoshi S. Immunohistochemical characterization of p57KIP2 expression in human esophageal squamous cell carcinoma. Anticancer Res. 2000;20:1947–1952. [PubMed] [Google Scholar]
- 13.Noura S, Yamamoto H, Sekimoto M, Takemasa I, Miyake Y, Ikenaga M, Matsuura N, Monden M. Expression of second class of KIP protein p57KIP2 in human colorectal carcinoma. Int J Oncol. 2001;19:39–47. doi: 10.3892/ijo.19.1.39. [DOI] [PubMed] [Google Scholar]
- 14.Ito Y, Takeda T, Sakon M, Tsujimoto M, Monden M, Matsuura N. Expression of p57/Kip2 protein in hepatocellular carcinoma. Oncology. 2001;61:221–225. doi: 10.1159/000055378. [DOI] [PubMed] [Google Scholar]
- 15.Nakai S, Masaki T, Shiratori Y, Ohgi T, Morishita A, Kurokohchi K, Watanabe S, Kuriyama S. Expression of p57(KIP2) in hepatocellular carcinoma: relationship between tumor differentiation and patient survival. Int J Oncol. 2002;20:769–775. [PubMed] [Google Scholar]
- 16.Schwarze SR, Shi Y, Fu VX, Watson PA, Jarrard DF. Role of cyclin-dependent kinase inhibitors in the growth arrest at senescence in human prostate epithelial and uroepithelial cells. Oncogene. 2001;20:8184–8192. doi: 10.1038/sj.onc.1205049. [DOI] [PubMed] [Google Scholar]
- 17.Ito Y, Yoshida H, Nakano K, Kobayashi K, Yokozawa T, Hirai K, Matsuzuka F, Matsuura N, Kuma K, Miyauchi A. Expression of p57/Kip2 protein in normal and neoplastic thyroid tissues. Int J Mol Med. 2002;9:373–376. [PubMed] [Google Scholar]
- 18.Rosenberg E, Demopoulos RI, Zeleniuch-Jacquotte A, Yee H, Sorich J, Speyer JL, Newcomb EW. Expression of cell cycle regulators p57(KIP2), cyclin D1, and cyclin E in epithelial ovarian tumors and survival. Hum Pathol. 2001;32:808–813. doi: 10.1053/hupa.2001.26462. [DOI] [PubMed] [Google Scholar]
- 19.Ito Y, Takeda T, Sasaki Y, Sakon M, Yamada T, Ishiguro S, Imaoka S, Tsujimoto M, Monden M, Matsuura N. Expression of p57/Kip2 protein in extrahepatic bile duct carcinoma and intrahepatic cholangiocellular carcinoma. Liver. 2002;22:145–149. doi: 10.1034/j.1600-0676.2002.01532.x. [DOI] [PubMed] [Google Scholar]
- 20.Lee MH, Yang HY. Negative regulators of cyclin-dependent kinases and their roles in cancers. Cell Mol Life Sci. 2001;58:1907–1922. doi: 10.1007/PL00000826. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ito Y, Takeda T, Wakasa K, Tsujimoto M, Matsuura N. Expression of p57/Kip2 protein in pancreatic adenocarcinoma. Pancreas. 2001;23:246–250. doi: 10.1097/00006676-200110000-00004. [DOI] [PubMed] [Google Scholar]
- 22.Keyomarsi K, Tucker SL, Buchholz TA, Callister M, Ding Y, Hortobagyi GN, Bedrosian I, Knickerbocker C, Toyofuku W, Lowe M, et al. Cyclin E and survival in patients with breast cancer. N Engl J Med. 2002;347:1566–1575. doi: 10.1056/NEJMoa021153. [DOI] [PubMed] [Google Scholar]
- 23.Georgieva J, Sinha P, Schadendorf D. Expression of cyclins and cyclin dependent kinases in human benign and malignant melanocytic lesions. J Clin Pathol. 2001;54:229–235. doi: 10.1136/jcp.54.3.229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Oshita T, Shigemasa K, Nagai N, Ohama K. p27, cyclin E, and CDK2 expression in normal and cancerous endometrium. Int J Oncol. 2002;21:737–743. doi: 10.3892/ijo.21.4.737. [DOI] [PubMed] [Google Scholar]
- 25.Kunimoto Y, Nakamura T, Ohno M, Kuroda Y. Relationship between immunohistochemical evaluation of thymidylate synthase and proliferating cell nuclear antigen labeling index in gastrointestinal carcinoma. Oncol Rep. 2004;12:1163–1167. [PubMed] [Google Scholar]
- 26.Czyzewska J, Guzińska-Ustymowicz K, Lebelt A, Zalewski B, Kemona A. Evaluation of proliferating markers Ki-67, PCNA in gastric cancers. Rocz Akad Med Bialymst. 2004;49 Suppl 1:64–66. [PubMed] [Google Scholar]
- 27.Tsuboi Y, Ichida T, Sugitani S, Genda T, Inayoshi J, Takamura M, Matsuda Y, Nomoto M, Aoyagi Y. Overexpression of extracellular signal-regulated protein kinase and its correlation with proliferation in human hepatocellular carcinoma. Liver Int. 2004;24:432–436. doi: 10.1111/j.1478-3231.2004.0940.x. [DOI] [PubMed] [Google Scholar]
- 28.Sato T, Konishi K, Kimura H, Maeda K, Yabushita K, Tsuji M, Miwa A. Evaluation of PCNA, p53, K-ras and LOH in endocrine pancreas tumors. Hepatogastroenterology. 2000;47:875–879. [PubMed] [Google Scholar]
- 29.Niijima M, Yamaguchi T, Ishihara T, Hara T, Kato K, Kondo F, Saisho H. Immunohistochemical analysis and in situ hybridization of cyclooxygenase-2 expression in intraductal papillary-mucinous tumors of the pancreas. Cancer. 2002;94:1565–1573. doi: 10.1002/cncr.10358. [DOI] [PubMed] [Google Scholar]


