Abstract
Mitochondria produce reactive oxygen species (mROS) as a natural by-product of electron transport chain activity. While initial studies focused on the damaging effects of reactive oxygen species, a recent paradigm shift has shown that mROS can act as signaling molecules to activate pro-growth responses. Cancer cells have long been observed to have increased production of ROS relative to normal cells, although the implications of this increase were not always clear. This is especially interesting considering cancer cells often also induce expression of antioxidant proteins. Here, we discuss how cancer-associated mutations and microenvironments can increase production of mROS, which can lead to activation of tumorigenic signaling and metabolic reprogramming. This tumorigenic signaling also increases expression of antioxidant proteins to balance the high production of ROS to maintain redox homeostasis. We also discuss how cancer-specific modifications to ROS and antioxidants may be targeted for therapy.
Keywords: Mitochondria reactive oxygen species, ROS, Cancer, Metabolism, Antioxidants, Oxidative stress
Review
Introduction
Mitochondrial-derived reactive oxygen species (mROS) have increasingly been appreciated to function as signaling molecules that modify cellular physiology. Increased production of ROS has long been observed to be a hallmark of many tumors and cancer cell lines [1]. Early investigations showed that ROS are capable of damaging proteins, lipids, and DNA, and thus it was believed that ROS can be tumorigenic by promoting genomic instability [2]. While high levels of ROS can promote DNA mutations and genetic instability, over the last 20 years a more nuanced view of the role of ROS in cancer has come to light. Specifically, cancer cells generate increased ROS; however, these ROS levels are still below that which cause overt damage. This range of ROS is capable of increasing tumorigenesis by activating signaling pathways that regulate cellular proliferation, metabolic alterations, and angiogenesis. Here, we will focus on the mechanisms of how mROS impact cellular physiology in cancer and the pathways by which cancer cells increase mROS.
Reactive oxygen species
The term reactive oxygen species covers several molecules derived from oxygen that have accepted extra electrons and can oxidize other molecules [3]. Most intracellular ROS are derived from the single electron reduction of oxygen (O2) to form the radical superoxide (O2·−). Two superoxide molecules can then be converted to one molecule of the non-radical ROS molecule hydrogen peroxide (H2O2) and one water molecule by superoxide dismutases. Hydrogen peroxide can also accept another electron from free Fe2+ by the Fenton reaction to become a hydroxyl radical (HO·). These three primary forms of ROS have different reactivities that can lead to differential effects on cellular physiology (Figure 1).
Figure 1.
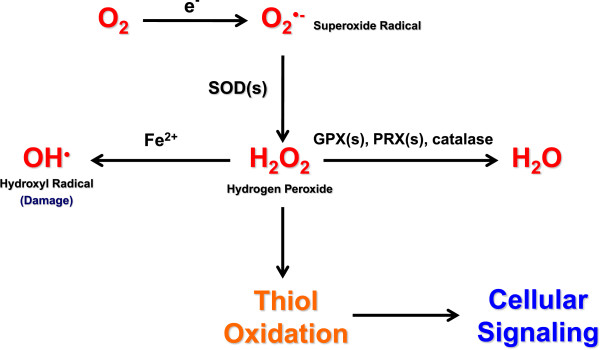
Production and interconversion of reactive oxygen species. O2 ·− is formed from molecular O2 by gaining a single electron from a NADPH oxidase (NOX) enzyme or from electron leak in the electron transport chain of the mitochondria. Superoxide dismutase (SOD) enzymes convert two superoxide molecules into a H2O2 and a water (H2O) molecule. Hydrogen peroxide can undergo Fenton chemistry with Fe2+ to form HO·, which is extremely reactive and can cause cellular damage. Hydrogen peroxide can also modify redox-sensitive cysteine residues to change cellular signaling. Alternatively, hydrogen peroxide can be reduced to water by glutathione peroxidases (GPXs), peroxiredoxins (PRXs), or catalase.
Seminal studies in the 1990s demonstrated that the primary signaling ROS molecule is hydrogen peroxide, which can act by inactivating phosphatases to allow for growth factor-dependent signaling [4, 5]. Hydrogen peroxide has the capacity to cross membranes and is significantly more stable than the radical ROS molecules. These attributes allow hydrogen peroxide to encounter susceptible residues on target molecules and display selectivity. One understood mechanism of hydrogen peroxide signaling is through the oxidation of cysteine residues on proteins. Cysteine residues exist in equilibrium between the protonated thiol (Cys-SH) and thiolate anion (Cys-S−) forms. Thiolate forms of cysteine are more susceptible to oxidation by hydrogen peroxide to form a sulfenic acid (Cys-SOH) residue [6]. In regulatory cysteine residues this can cause allosteric changes within the protein to modify activity or binding partners. Alternatively, oxidation of active site cysteines can inhibit activity and thus change signaling cascades. The likelihood of cysteine oxidation of a given protein is a combination of solvent accessibility, local hydrogen peroxide concentration, and cysteine pKa [7]. While hydrogen peroxide is the best described signaling ROS molecule, roles for superoxide as an independent signaling molecule have also been described [8]. In addition, other reactive oxidants such as peroxynitrite (ONOO−) can form from a reaction between superoxide and nitric oxide (·NO). These reactive nitrogen species likely have both overlapping and distinct mechanisms of mediating signaling changes with ROS since they are capable of both oxidizing and nitrating intracellular amino acids. Hydroxyl radicals likely do not play a signaling role since they are generally too reactive to display selectivity in reaction targets.
Sources of reactive oxygen species
One major source of intracellular ROS is the NADPH oxidases. NADPH oxidases catalyze the production of superoxide from O2 and NADPH. These enzymes were originally described in phagocytes, where they were shown to kill engulfed pathogens by creating locally high levels of oxidative stress [9]. Since this discovery, it has been observed that NADPH oxidase family members are present in many tissues in the body where they are important for non-immune functions as well [10, 11]. The presence of enzymes that specifically produce ROS validates the model that ROS serve a controlled function in the cell, rather than simply acting as toxic by-products. In addition, oncogenes can stimulate NADPH oxidase-dependent ROS production, which has been shown to be necessary for cell proliferation [12]. NADPH oxidases have been detected to be intracellularly localized to many organelles including the plasma membrane, nucleus, mitochondria, and endoplasmic reticulum. Interestingly, the endoplasmic reticulum has recently also been shown to also have NADPH oxidase-independent production of ROS as well [13]. While NADPH oxidases are well-described sources of intracellular ROS, when possible, this review will focus on the mechanisms and consequences of mitochondrial-derived ROS.
The largest contributor to cellular ROS is the mitochondria. It has been estimated that as much as 1% of the total mitochondrial O2 consumption is used to produce superoxide [14, 15]. The mitochondria have eight known sites that are capable of producing superoxide [16, 17]. The relative contribution of each of these sites to the total cellular ROS is unclear, however, ROS from complex I, II, and III have all been shown to have effects on cellular signaling [16]. Interestingly, while complexes I and II release ROS into the mitochondrial matrix, complex III has the ability to release ROS to both sides of the mitochondrial inner membrane [18]. Theoretically, releasing ROS to the inner membrane space would allow easier access to cytosolic targets. Consistent with this hypothesis, complex III-derived ROS have specifically been shown to be required for many biological processes including oxygen sensing, cell differentiation, and adaptive immunity [19]. Whether the other sources of mROS have individual or simply contributory roles to the total mROS signaling is unknown.
Antioxidant pathways balance ROS levels
Considering that mROS can modify proteins, regulation of the concentration of mROS is crucial for its ability to act as a signaling molecule. Levels of mROS are controlled both at the level of production (discussed below) and by degradation. The SOD proteins (SOD1-3) first convert two superoxide molecules into hydrogen peroxide and water, removing one reactive oxygen species per cycle. Hydrogen peroxide is then further reduced to water by a host of antioxidant enzymes including six PRXs, eight GPXs, and catalase in mammalian cells. PRXs are among the most abundant proteins in cells and have been calculated to degrade most of the intracellular hydrogen peroxide [20, 21]. GPXs also are highly active, although less abundant, and may be an important antioxidant mechanism at higher concentrations of hydrogen peroxide [22]. In the context of ROS signaling, there is accumulating evidence that antioxidant enzymes may be modified in complex ways to facilitate specific ROS signaling events. For example, in response to growth factor signaling membrane-bound PRX1 can be phosphorylated to inhibit degradation of hydrogen peroxide. This results in localized accumulation of hydrogen peroxide and increased growth factor signaling [23]. Similarly, GPX1 activity can be increased by phosphorylation by c-Abl and Arg to protect against high levels of oxidative stress [24]. These examples, as well as the high number of PRXs and GPXs, suggest that the regulation of ROS by antioxidant enzymes may be much more intricate than simply constitutive degradation activity.
The predominant transcriptional response that increases the production of antioxidant proteins in cancer cells is through the activation of nuclear factor (erythroid-derived 2)-like 2 (NRF2) [25]. Stabilization of the labile transcription factor NRF2 by inhibition of its negative regulator Kelch-like ECH-associated protein 1 (KEAP1) allows it to increase expression of antioxidants including GPXs and glutathione synthesis and utilization genes [26, 27]. One mechanism of NRF2 stabilization is by ROS-mediated oxidation of sensitive cysteine residues on KEAP1 [28–30]. While increased ROS is a common feature in cancer cells, NRF2 has also been shown to be essential for tumorigenesis [31, 32]. It is thus likely that the requirement for NRF2 controls ROS levels in cancer cells to maintain homeostasis. Interestingly, while NRF2 loss inhibited tumor formation, mice deficient for the antioxidant PRX1 have increased ROS and display decreased life span due to hemolytic anemia and development of malignant cancers [33]. Thus, small molecule increases in ROS as a result of removing a single component of the antioxidant response may increase tumorigenesis while complete loss of the antioxidant response pathway, such as in NRF2 knockout mice, results in prohibitively high levels of ROS and decreases tumorigenesis. The distinction between small changes in ROS that promote tumorigenic signaling vs. large changes in ROS that cause oxidative stress to induce cell death is an important factor that will dictate the response to ROS stimuli (Figure 2).
Figure 2.
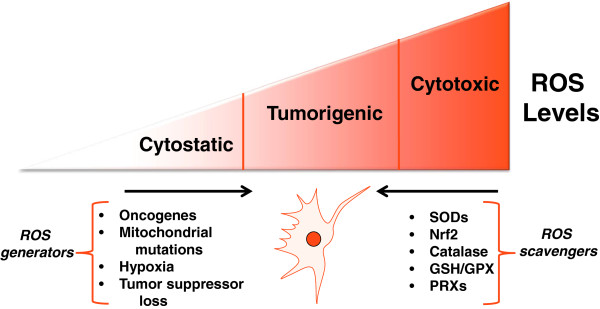
Balancing ROS generation and ROS scavenging allows cancer cells to remain in the tumorigenic range of ROS levels. Activation of mitochondrial ROS generation by oncogenes, mitochondrial mutations, hypoxia, or tumor suppressor loss increases ROS signaling to increase tumorigenicity. Tumor cells also express enhanced levels of antioxidant proteins that prevent increased ROS from reaching cytotoxic levels incompatible with growth.
Mitochondrial reactive oxygen species regulate signaling pathways
ROS enhance phosphoinositide 3-kinase signaling
The phosphoinositide 3-kinase (PI3K) pathway is a central growth factor response pathway that is hyper-activated in many cancers. Activation of this pathway has been shown to increase proliferation, promote survival, and increase cellular mobility [34]. Upon growth factor stimulation, growth factor receptors activate the catalytic subunit of PI3K, p110, through Ras activation or recruitment of the regulatory subunit, p85. Once activated, p110 phosphorylates phosphoinositides (PI) to generate PI (3, 4, 5) P3 (PIP3). PIP3 acts as a signaling lipid by binding to the pleckstrin homology (PH) domain of Akt, causing its localization to the plasma membrane. Akt is then activated by phosphorylation from another PH domain-containing kinase, phosphoinositide-dependent kinase-1 (PDK1). Activation of Akt is an important mediator of the PI3K pathway and leads to increased cell proliferation and suppression of apoptosis. The negative regulator of this pathway, phosphatase and tensin homolog deleted on chromosome ten (PTEN), has constitutive phosphatase activity on PIP3 to convert it to the inactive form, PIP2.
The intracellular level of ROS can affect the PI3K pathway. Treatment of cells with exogenous hydrogen peroxide is sufficient to activate Akt [35]. The primary known ROS target in the PI3K pathway is PTEN. ROS have been shown to oxidize the active site cysteine on PTEN (Cys124) resulting in a disulfide formation to another intraprotein cysteine (Cys71). This results in inactivation of PTEN and perpetual activation of the PI3K pathway [36, 37]. In addition to general ROS effects, mROS were specifically shown to inhibit PTEN and activate Akt [38, 39]. Aside from PTEN, ROS have been shown to inhibit other phosphatases, including protein phosphatase 2A (PP2A) and protein tyrosine phosphatase 1B (PTP1B) [40]. PP2A dephosphorylates Akt on threonine 308 and serine 493 resulting in Akt inactivation; however, PP2A dephosphorylation activity is inhibited by hydrogen peroxide [41]. PTP1B also suppresses Akt activity by dephosphorylation but, like PP2A, ROS inhibit PTP1B activity and increase Akt activity resulting in increased anchorage-independent growth [42, 43]. Thus, ROS inhibit phosphatases to dysregulate PI3K signaling resulting in increased Akt signaling and enhanced proliferation and survival (Figure 3).
Figure 3.
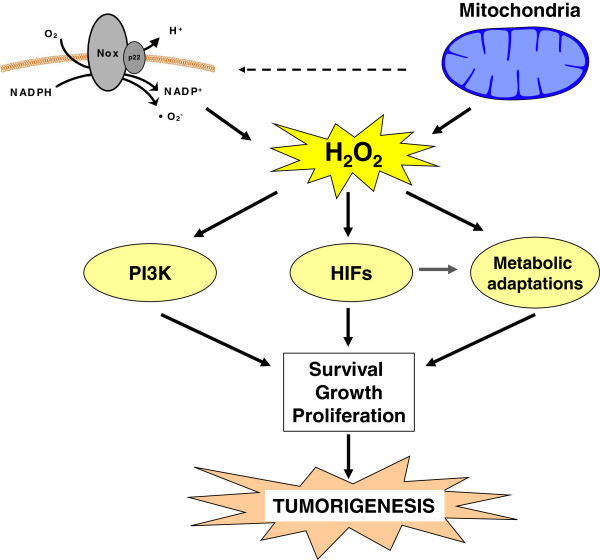
Reactive oxygen species modify cellular signaling. Hydrogen peroxide derived from either NOXs or the mitochondria can activate the PI3K pathway, the hypoxia-inducible factor (HIF) pathway, and metabolic adaptations. These modifications are essential to allowing the survival, growth, and proliferation fundamental to tumorigenesis.
Mitochondrial ROS activate hypoxia-inducible factors
One of the best characterized pathways shown to be responsive to mROS is the hypoxia-response pathway. Hypoxia is a prominent feature of tumor cells in vivo due to a mismatch between the high proliferative rate of tumor cells and the ability of the blood supply to provide nutrients including oxygen. Tumor cells activate hypoxia inducible factors (HIFs) to activate a transcriptional network to allow tumor cells to adapt to their diminished oxygen microenvironment. The pathway consists of three hypoxia-sensitive α subunits (HIF1α, HIF2α, and HIF3α) that, upon activation, heterodimerize with the constitutively expressed HIF1β and activate transcription from hypoxia-response elements (HREs) [44]. Under normoxic conditions (21% O2), HIFα subunits are rapidly hydroxylated on proline residues by prolyl hydroxylase domain-containing protein 2 (PHD2) which is recognized and targeted for degradation by the von Hippel-Lindau (VHL) E3 ubiquitin ligase pathway [45]. When cells are exposed to hypoxia, PHD2 hydroxylation of HIFα subunits is inhibited leading to HIFα accumulation, heterodimerization, and translocation to the nucleus. The HIF heterodimer interacts with the co-activators p300 and CBP to initiate transcription of hypoxia-response genes from HREs. Appropriately for cells under hypoxia, transcriptional targets of HIFs include genes that promote survival under hypoxia, shift metabolism to increased glycolysis, and activate angiogenesis [46].
Exposure to hypoxia increases mROS to stabilize HIFα subunits. Initial evidence for this mechanism stems from the observation that cells depleted of their mitochondrial DNA (ρ0 cells) are incapable of stabilizing HIFα subunits under hypoxia [47]. ρ0 cells do not exhibit mitochondrial oxygen consumption and do not produce mROS [48]. In addition, treatment of mitochondria-replete cells with the electron transport chain (ETC) inhibitors rotenone, myxothiazol, and stigmatellin can inhibit mROS production and inhibit stabilization of HIFα subunits under hypoxia [49]. In contrast, the ETC inhibitor antimycin A increases mROS and leads to increased HIFα stabilization. Further studies have identified that hypoxia increases the release of superoxide from complex III to the mitochondrial intermembrane space [50]. In complex III, electron transport is mediated by the Rieske-Fe-S protein (RISP), cytochrome b, and cytochrome c1. While the loss of RISP or cytochrome b eliminates mitochondrial oxygen consumption, the loss of RISP eliminates mROS production while the loss of cytochrome b retains mROS production. Importantly, cells depleted of RISP are incapable of stabilizing HIFα under hypoxia, whereas cells depleted of cytochrome b retain their ability to stabilize HIFα [51–54]. This indicates that in hypoxia the increased release of superoxide from complex III is responsible for the inhibition of PHD2 and stabilization of HIFα subunits. How these ROS inhibit PHD2 activity is not fully understood, however, one possibility is that ROS oxidize intracellular Fe2+, a cofactor required for PHD2 function [55]. Treatment of cells with mitochondrial-targeted antioxidants blocks the release of mitochondrial ROS and inhibits the stabilization of HIFα subunits under hypoxia [56]. Furthermore, a large chemical screen designed to uncover inhibitors of hypoxic activation of HIFs enriched for mitochondrial inhibitors of complex III [57]. Thus, mROS are both sufficient and required for hypoxic activation of HIFs (Figure 3). Interestingly, suppression of HIF1α by treatment with antioxidants has been shown to inhibit cancer cell proliferation in vitro and in vivo [58, 59].
Mitochondrial ROS modify metabolism
The interplay between ROS levels and cellular metabolism is tightly regulated. Metabolic processes produce ROS, particularly in the mitochondria, thus metabolic fluxes need to be intimately controlled to maintain ROS homeostasis. One important mechanism of metabolic control is through HIF1α. Activation of HIF1α induces expression of glycolysis enzymes and transporters to increase glycolytic flux, as well as increases expression of PDK1 to divert glycolytic carbon away from the mitochondria [60]. In addition, HIF1α induction of NADH dehydrogenase (ubiquinone) 1 alpha subcomplex, 4-like 2 (NDUFA4L2) suppresses complex I activity and mROS [61]. HIF1α has also been shown to induce microRNA-210, which is sufficient to decrease expression of the iron-sulfur cluster assembly proteins ISCU1/2 and decrease mitochondrial oxygen consumption, increase lactate production, and increase ROS [62, 63]. Another method by which ROS can modify metabolism is through activating NRF2. Activation of NRF2 increases synthesis of anabolic enzymes and supports tumor growth by increasing production of NADPH increasing and purine biosynthesis [64]. ROS have also been shown to modify metabolism directly by oxidizing the glycolytic enzyme pyruvate kinase M2 (PKM2). In contrast to its constitutively active splice isoform PKM1, PKM2 is preferentially expressed in cancer cells and is unique due to its ability to be inhibited by a variety of stimuli [65, 66]. Interestingly, ROS have also been shown to inhibit PKM2 activity by directly oxidizing a cysteine residue on PKM2 [67]. Oxidation of this residue was shown to cause increased pentose phosphate pathway flux, increase glutathione levels, and increase proliferation under hypoxia. Importantly, inhibition of pyruvate kinase activity has been associated with increased tumorigenesis in vivo [68].
Cancer cells increase mitochondrial reactive oxygen species
Tumorigenic mutations increase mROS
Many cancer cells show increased levels of ROS, and the signaling events and mutations that increase ROS is an area of active research. Several oncogenes have been linked to increased ROS production (Figure 4). Exogenous expression of H-RasG12V has been shown to increase mitogenic activity of 3T3 fibroblasts, and this activity was dependent on increased ROS [12]. In murine embryonic fibroblasts (MEFs) immortalized by a dominant negative p53, expression of Myr-Akt, H-RasG12V, or K-RasG12D conferred increased mROS-dependent soft-agar colony formation [69]. In addition, deregulated expression of Myc has also been shown to modify ROS levels. Exogenous expression of Myc increased ROS production, leading to the transformation in some cells, but ROS induced apoptosis in others [70, 71]. This suggests that the ROS effects may be dependent on cell type, other mutations, and expression level of the oncogene. Interestingly, in mouse models of cancer, activation of physiological expression of K-RasG12D, B-RafV619E, or Myc suppressed steady state levels of ROS [31]. This suppression was shown to be mediated by induction of the NRF2 antioxidant program, and thus it is not clear if oncogenes in this context modify the ROS production or simply decrease steady state ROS by increased expression of antioxidant proteins. Another possibility is that NRF2 expression suppresses the total cell ROS levels, but localized increases in compartmentalized ROS (such as mROS) are maintained to promote tumorigenic signaling.
Figure 4.
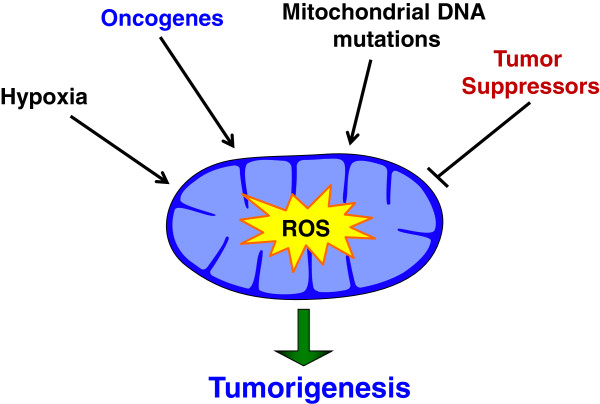
Pathways that modulate mitochondrial reactive oxygen species. Hypoxia, activation of oncogenes, mitochondrial DNA mutations, and loss of tumor suppressors have all been shown to lead to a mitochondrial ROS dependent increases in tumorigenesis.
Several tumor suppressors have been shown to have ROS inhibitory functions. The most common of them, the tumor suppressor p53, known as ‘the guardian of the genome’ is lost or mutated in approximately 50% of cancers [72]. Classically, it has been shown that in response to telomere erosion, oncogene activation, or genotoxic stress that activation of p53 suppresses cancer formation by inducing apoptosis and senescence [73]. However, recent evidence has shown that endogenous expression of p53 with mutations that prevent its ability to cause cell cycle arrest, apoptosis, or senescence still maintains its tumor suppressive function [74]. Interestingly, this mutated p53 retained its ability to control metabolic homeostasis and suppress ROS. In addition, treatment of xenografts with the antioxidant N-acetyl cysteine (NAC) suppressed tumor growth in p53 null cancer cells, but not p53 replete cells [75]. These data suggest that p53-mediated tumor suppression may be, in part, due to its ability to suppress ROS (Figure 4).
The sirtuins are a family of NAD+-dependent proteins that have been linked to control of metabolic state and cell signaling. Although disputable, several of the sirtuins, including SirT1, SirT2, SirT3, and SirT6, have been implicated to act as tumor suppressors [76]. SirT3, one of the three mitochondrial sirtuins, modulates mitochondrial function by deacetylation of proteins of the electron transport chain, the tricarboxylic acid (TCA) cycle, and antioxidant defense [77]. A survey of human tumors has shown that SirT3 protein expression is significantly decreased in tumors and deletion of at least one copy of SirT3 has been observed in 20%–30% of cancers [78]. Loss of SirT3 expression by genetic knockout or small hairpin RNA (shRNA) increased mROS, while overexpression of SirT3 suppressed mROS [78, 79]. These changes in mROS by SirT3 expression directly correlated with proliferation rate of cancer cells in vitro and in vivo and could also be modulated with antioxidants.
Mitochondrial mutations increase mROS
Mutations in mitochondrial DNA (mtDNA)-encoded ETC proteins have been reported in a wide variety of human tumors [80]. Considering cells contain thousands of copies of mtDNA per cell, these mutations typically occur in only a fraction of the total cellular mtDNA, a condition known as heteroplasmy. Heteroplasmic mutations have been observed to be enriched in tumors relative to normal tissue and have been implicated to confer a selective advantage in tumorigenesis [81]. Heteroplasmic mutations in complex I have been shown to increase mROS, increase colony formation in soft agar, and increase tumor formation in vivo [82]. Further, reconstitution of complex I activity using the yeast complex I analog NDI1 suppressed mROS, mROS-mediated activation of Akt and HIF1α, and colony formation in soft agar [83]. Perhaps the strongest evidence for the role of heteroplasmic mutations in tumorigenesis comes from a study in which the mtDNA from a poorly metastatic cell line was switched with that of a highly metastatic cell line. Upon acceptance of the new mtDNA, the recipient tumor cells acquired the metastatic characteristics of the opposite cell line [84]. Heteroplasmic mutations in the complex I subunit NADH dehydrogenase subunit 6 (ND6) were shown to increase metastatic potential through increased mROS production and activation of HIF1α. Furthermore, treatment of these cells with the antioxidant NAC inhibited this activity. While relatively low levels of heteroplasmic mutations (10%–60%) increase tumorigenesis, high level heteroplasmy or homoplasmic mutations in mtDNA will likely become detrimental to metabolism and thereby tumorigenicity upon sufficient loss. In support of this model, large levels of heteroplasmy sensitized cells to growth inhibition under low glucose [85]. Cancer cells with mitochondrial mutations resulting in homoplasmic loss of complex I function were unable to form xenografts [86]. In addition, loss of mitochondrial transcription factor A (TFAM), a transcription factor required for mtDNA replication, inhibited tumor formation in an in vivo mouse model of K-Ras-driven lung cancer [69]. However, heterozygosity for TFAM caused a mROS dependent increase in intestinal tumorigenesis in an APCmin/+ mouse model of cancer [87]. Thus, moderate amounts of heteroplasmy may be beneficial for tumorigenesis by increasing mROS while high heteroplasmic mutations or homoplasmic mutations may inhibit tumorigenesis by causing metabolic dysfunction (Figure 5).
Figure 5.
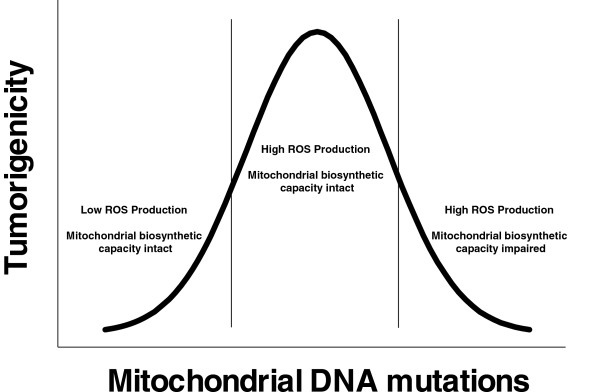
Heteroplasmic mutations in mitochondrial DNA increase tumorigenesis. Small amounts of heteroplasmic mutations increase tumorigenicity by increasing mROS levels while maintaining mitochondrial biosynthetic capacity. However, large amounts of mtDNA mutations eventually compromise mitochondrial biosynthetic capacity and will decrease tumorigenicity.
Mutations in components of the nuclear-encoded mitochondrial metabolic enzyme succinate dehydrogenase (SDH) have been shown to lead to paraganglioma and pheochromocytoma [88]. The SDH complex is comprised of four subunits (SDHA, SDHB, SDHC, and SDHD) and is the only TCA cycle enzyme that is also a component of the ETC (complex II). Mutations in SDHB, SDHC, and SDHD are commonly associated with cancer formation, whereas mutations in SDHA are rarely associated. Interestingly, given the structure and mechanism of complex II, loss of SDHB, SDHC, and SDHD would allow for acceptance of an electron, but not progression along the ETC, and thus may increase ROS generation. In support of this model, loss of SDHB, but not SDHA increases mROS, HIF1α, and tumorigenicity [89]. In addition, mutations in SDHC are also been associated with increased mROS and tumorigenesis [90]. Thus, loss of components of the SDH complex may, in part, cause tumorigenesis by increasing mROS levels.
In hereditary leiomyomatosis and renal cell cancer (HLRCC), the loss of the TCA cycle enzyme fumarate hydratase (FH) leads to accumulation of the metabolite fumarate and renal cell cancer. FH-deficient cancer cells display pseudo-hypoxia with aberrant activation of HIF1α. Congruent with SDH mutations, this HIF1α activation was also shown to be ROS dependent [91]. However, the mechanism of ROS production is different than SDH mutations. Intracellular thiolate residues on cysteines can undergo a nucleophilic attack on the electrophilic alkene bond of fumarate to produce a ‘succination’ modification [92]. Accumulated fumarate in FH-deficient cells succinates the thiol residue on the intracellular antioxidant molecule glutathione to produce the metabolite succinated glutathione (GSF) [93]. The metabolism of GSF consumes NADPH, the primary reducing equivalent used in ROS detoxification reactions. Thus, GSF reduces overall NADPH antioxidant capacity resulting in increased mROS and HIF1α stabilization. Interestingly, FH-null cancer cells also display hyper-activation of the master antioxidant transcription factor NRF2. While ROS have been shown to stabilize NRF2, FH-deficient cancer cells primarily activate NRF2 by succination and inactivation of KEAP1 [93–95]. Depletion of NRF2 by shRNA in FH-null cells further increased ROS, increased HIF1α stabilization, and decreased proliferation, suggesting that NRF2 suppresses fumarate-mediated ROS to maintain a favorable homeostatic level compatible with proliferation [93].
Targeting ROS for therapy
Suppressing ROS to inhibit proliferation
ROS contribute to mitogenic signaling, and thus decreasing intracellular ROS levels is an attractive method for inhibiting cancer growth. With this in mind, several large-scale studies have investigated whether supplementation with antioxidant vitamins, including β-carotene and vitamin A or vitamin E can reduce cancer risk in humans. Contrary to the expected result, supplementation increased the risk of cancer in both cases [96, 97]. In agreement with these results, in genetic mouse models of K-Ras- or B-Raf-induced lung cancer, treatment with NAC or vitamin E markedly enhanced tumor growth and accelerated mortality [98]. These results show that the potential use of antioxidants for cancer therapy is complex and needs to be carefully validated before being applied. One possibility for the failure of these antioxidants as cancer treatments is their lack of specificity. Treatment of patients with general antioxidants may modulate many physiological processes that are relevant to cancer growth. For example, the immune system, an important modulator of cancer growth, has been shown to be sensitive to ROS levels [99]. Another possibility is that general antioxidants are differentially effective than targeted antioxidants. Mitochondrial-targeted versions of antioxidants have been shown to be potent inhibitors of cancer cell growth in vitro and in vivo [69, 100]. Thus, further investigation needs to be considered to determine if targeted antioxidants are a viable method to treat cancer.
Another approach for inhibiting ROS is to decrease production. Decreasing mROS production necessarily involves inhibition of the ETC and thus may not be a practical due to toxicity inherent in inhibiting mitochondrial respiration. However, patients taking the antidiabetic drug metformin have recently been shown to have a reduced risk of cancer incidence and mortality [101]. Metformin has been shown to act as an inhibitor of complex I of the ETC [102, 103]. We recently used a metformin insensitive complex I analog to confirm that the anticancer effect of metformin is primarily mediated by specific inhibition of complex I of cancer cells in vivo [104]. Interestingly, we also observed that treatment with metformin suppressed hypoxic activation of HIF1α, indicating that it may also decrease production of mROS under hypoxia. Whether this effect is important for the cancer suppressive effects of metformin requires further investigation. An alternative approach to decrease ROS production is by inhibiting NADPH oxidases. Indeed, loss of NADPH oxidase 4 has been shown to activate apoptosis in pancreatic cancer cells [105]. In addition, inhibitors of NADPH oxidase activity have been shown to have efficacy on mouse models of cancer in vivo [106, 107].
Increasing ROS to selectively kill cancer cells
Considering that cancer cells have increased ROS levels, they may be selectively sensitive to the damaging effects of further increasing ROS. Increasing ROS production specifically in cancer cells is likely difficult to accomplish, although it is one proposed mechanism for how many current chemotherapeutics function [108]. Alternatively, since cancer cells frequently have increased expression of antioxidants to maintain homeostasis, a promising therapeutic approach is to inhibit antioxidants to expose cancer cells to endogenously produced ROS [109]. In support of this model, several small molecule screens identifying compounds that specifically inhibit growth of transformed cells have converged upon glutathione utilization [110–112]. In all cases, treatment with the identified small molecules decreased glutathione levels, increased ROS, and could be rescued by treatment with NAC. In addition, inhibition of antioxidant pathways has also been shown to be effective for inhibiting cancer growth. Genetic knockout of NRF2 inhibited disease progression in mouse models of pancreatic and lung cancer [31, 32]. Inhibition of SOD1 by the small molecule ATN-224 was shown to cause ROS-dependent cancer cell death in vitro and decreased tumor burden in advanced K-Ras-driven lung cancers in vivo [113]. These recent examples provide further proof of principle that increasing ROS, whether by increasing production or inhibiting antioxidants, is a promising approach for targeting cancer cells (Figure 6). Further research is warranted to determine which components of the antioxidant pathway are selectively essential for tumor growth.
Figure 6.
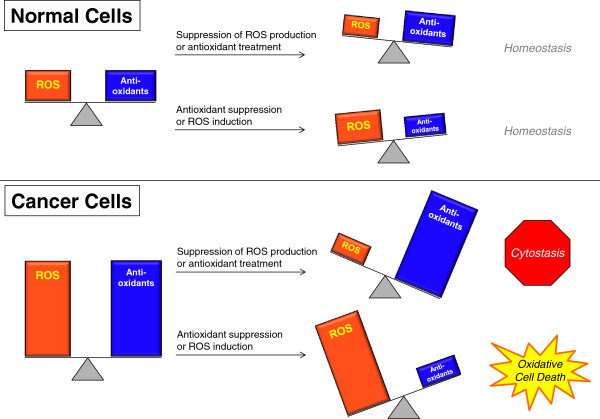
Targeting cancer cells by modifying ROS levels. Normal cells have decreased amounts of both ROS and antioxidants relative to cancer cells. Loss of either ROS or antioxidants therefore causes only small changes in ROS homeostasis, leaving cells viable and functional. However, since cancer cells have more ROS and antioxidants, they may be more susceptible to changes in ROS levels. Treatment with antioxidants or prevention of ROS generation will cause cells to lose sufficient ROS signaling to maintain growth. The result is cytostasis and possibly senescence. Alternatively, inhibition of antioxidants or increasing ROS generation will result in excess ROS in cancer cells and cause cancer-specific oxidative cell death.
Conclusions
It is becoming increasingly apparent that ROS play an important role in the biology of tumorigenesis. While several mechanisms have been presented here, the bulk of ROS-mediated signaling targets are largely unknown. However, the frequency of cancer-associated mutations that increase ROS levels suggests that increased production of ROS may be a common output of a large fraction of cancer-associated mutations in oncogenes and tumor suppressors. In addition, the apparent selection for mitochondrial mutations that increase ROS at the detriment of metabolic flexibility suggests that ROS are strongly selected for in these cancer cells. An emerging model is that cancer cells increase the production of ROS to activate localized pro-tumorigenic signaling but balance the increased ROS with elevated antioxidant activity to maintain redox balance. As with all studies in cancer, the final goal will be to design therapeutics that can take advantage of these discoveries. Both the suppression of ROS to prevent activation of pro-tumorigenic signaling pathways and the exacerbation of ROS by disabling antioxidants to induce cell death represent promising approaches in this regard. Future work is needed to better understand ROS-targeted pathways. In addition, future studies need to determine what sources of ROS and what specific antioxidants are required for homeostasis. With this knowledge, we can better understand cancer biology and design novel therapeutics to specifically treat cancer cells.
Acknowledgements
This work was supported by NIH grants RO1CA123067 (NSC).
Footnotes
Competing interests
The authors declare that they have no competing interests.
Authors' contributions
LS wrote the manuscript and prepared the figures. NC supervised the design of the review and wrote the manuscript. Both authors read and approved the final manuscript.
Contributor Information
Lucas B Sullivan, Email: lucasbs@mit.edu.
Navdeep S Chandel, Email: nav@northwestern.edu.
References
- 1.Szatrowski TP, Nathan CF. Production of large amounts of hydrogen peroxide by human tumor cells. Can Res. 1991;51(3):794–798. [PubMed] [Google Scholar]
- 2.Ames BN, Shigenaga MK, Hagen TM. Oxidants, antioxidants, and the degenerative diseases of aging. Proc Natl Acad Sci USA. 1993;90(17):7915–7922. doi: 10.1073/pnas.90.17.7915. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Cross CE, Halliwell B, Borish ET, Pryor WA, Ames BN, Saul RL, McCord JM, Harman D. Oxygen radicals and human disease. Ann Intern Med. 1987;107(4):526–545. doi: 10.7326/0003-4819-107-4-526. [DOI] [PubMed] [Google Scholar]
- 4.Sundaresan M, Yu ZX, Ferrans VJ, Irani K, Finkel T. Requirement for generation of H2O2 for platelet-derived growth factor signal transduction. Science. 1995;270(5234):296–299. doi: 10.1126/science.270.5234.296. [DOI] [PubMed] [Google Scholar]
- 5.Bae YS, Kang SW, Seo MS, Baines IC, Tekle E, Chock PB, Rhee SG. Epidermal growth factor (EGF)-induced generation of hydrogen peroxide. Role in EGF receptor-mediated tyrosine phosphorylation. J Biol Chem. 1997;272(1):217–221. doi: 10.1074/jbc.272.1.217. [DOI] [PubMed] [Google Scholar]
- 6.Finkel T. From sulfenylation to sulfhydration: what a thiolate needs to tolerate. Sci Signal. 2012;5(215):e10. doi: 10.1126/scisignal.2002943. [DOI] [PubMed] [Google Scholar]
- 7.Finkel T. Oxidant signals and oxidative stress. Curr Opin Cell Biol. 2003;15(2):247–254. doi: 10.1016/S0955-0674(03)00002-4. [DOI] [PubMed] [Google Scholar]
- 8.Buetler TM, Krauskopf A, Ruegg UT. Role of superoxide as a signaling molecule. News Physiol Sci. 2004;19:120–123. doi: 10.1152/nips.01514.2003. [DOI] [PubMed] [Google Scholar]
- 9.Babior BM. NADPH oxidase: an update. Blood. 1999;93(5):1464–1476. [PubMed] [Google Scholar]
- 10.Brown DI, Griendling KK. Nox proteins in signal transduction. Free Radic Biol Med. 2009;47(9):1239–1253. doi: 10.1016/j.freeradbiomed.2009.07.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Jiang F, Zhang Y, Dusting GJ. NADPH oxidase-mediated redox signaling: roles in cellular stress response, stress tolerance, and tissue repair. Pharmacol Rev. 2011;63(1):218–242. doi: 10.1124/pr.110.002980. [DOI] [PubMed] [Google Scholar]
- 12.Irani K, Xia Y, Zweier JL, Sollott SJ, Der CJ, Fearon ER, Sundaresan M, Finkel T, Goldschmidt-Clermont PJ. Mitogenic signaling mediated by oxidants in Ras-transformed fibroblasts. Science. 1997;275(5306):1649–1652. doi: 10.1126/science.275.5306.1649. [DOI] [PubMed] [Google Scholar]
- 13.Wang J, Pareja KA, Kaiser CA, Sevier CS. Redox signaling via the molecular chaperone BiP protects cells against endoplasmic reticulum-derived oxidative stress. Elife. 2014;3:e03496. doi: 10.7554/eLife.03496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Quinlan CL, Treberg JR, Perevoshchikova IV, Orr AL, Brand MD. Native rates of superoxide production from multiple sites in isolated mitochondria measured using endogenous reporters. Free Radic Biol Med. 2012;53(9):1807–1817. doi: 10.1016/j.freeradbiomed.2012.08.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Handy DE, Loscalzo J. Redox regulation of mitochondrial function. Antioxid Redox Signal. 2012;16(11):1323–1367. doi: 10.1089/ars.2011.4123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Murphy MP. How mitochondria produce reactive oxygen species. Biochem J. 2009;417(1):1–13. doi: 10.1042/BJ20081386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Brand MD. The sites and topology of mitochondrial superoxide production. Exp Gerontol. 2010;45(7–8):466–472. doi: 10.1016/j.exger.2010.01.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Muller FL, Liu Y, Van Remmen H. Complex III releases superoxide to both sides of the inner mitochondrial membrane. J Biol Chem. 2004;279(47):49064–49073. doi: 10.1074/jbc.M407715200. [DOI] [PubMed] [Google Scholar]
- 19.Sena LA, Chandel NS. Physiological roles of mitochondrial reactive oxygen species. Mol Cell. 2012;48(2):158–167. doi: 10.1016/j.molcel.2012.09.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Wood ZA, Schroder E, Robin Harris J, Poole LB. Structure, mechanism and regulation of peroxiredoxins. Trends Biochem Sci. 2003;28(1):32–40. doi: 10.1016/S0968-0004(02)00003-8. [DOI] [PubMed] [Google Scholar]
- 21.Cox AG, Winterbourn CC, Hampton MB. Mitochondrial peroxiredoxin involvement in antioxidant defence and redox signalling. Biochem J. 2010;425(2):313–325. doi: 10.1042/BJ20091541. [DOI] [PubMed] [Google Scholar]
- 22.Winterbourn CC, Hampton MB. Thiol chemistry and specificity in redox signaling. Free Radic Biol Med. 2008;45(5):549–561. doi: 10.1016/j.freeradbiomed.2008.05.004. [DOI] [PubMed] [Google Scholar]
- 23.Woo HA, Yim SH, Shin DH, Kang D, Yu DY, Rhee SG. Inactivation of peroxiredoxin I by phosphorylation allows localized H(2)O(2) accumulation for cell signaling. Cell. 2010;140(4):517–528. doi: 10.1016/j.cell.2010.01.009. [DOI] [PubMed] [Google Scholar]
- 24.Cao C, Leng Y, Huang W, Liu X, Kufe D. Glutathione peroxidase 1 is regulated by the c-Abl and Arg tyrosine kinases. J Biol Chem. 2003;278(41):39609–39614. doi: 10.1074/jbc.M305770200. [DOI] [PubMed] [Google Scholar]
- 25.Sporn MB, Liby KT. NRF2 and cancer: the good, the bad and the importance of context. Nature reviews Cancer. 2012;12(8):564–571. doi: 10.1038/nrc3278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Jaramillo MC, Zhang DD. The emerging role of the Nrf2-Keap1 signaling pathway in cancer. Genes Dev. 2013;27(20):2179–2191. doi: 10.1101/gad.225680.113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Malhotra D, Portales-Casamar E, Singh A, Srivastava S, Arenillas D, Happel C, Shyr C, Wakabayashi N, Kensler TW, Wasserman WW, Biswal S. Global mapping of binding sites for Nrf2 identifies novel targets in cell survival response through ChIP-Seq profiling and network analysis. Nucleic Acids Res. 2010;38(17):5718–5734. doi: 10.1093/nar/gkq212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Dinkova-Kostova AT, Holtzclaw WD, Cole RN, Itoh K, Wakabayashi N, Katoh Y, Yamamoto M, Talalay P. Direct evidence that sulfhydryl groups of Keap1 are the sensors regulating induction of phase 2 enzymes that protect against carcinogens and oxidants. Proc Natl Acad Sci USA. 2002;99(18):11908–11913. doi: 10.1073/pnas.172398899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Zhang DD, Hannink M. Distinct cysteine residues in Keap1 are required for Keap1-dependent ubiquitination of Nrf2 and for stabilization of Nrf2 by chemopreventive agents and oxidative stress. Mol Cell Biol. 2003;23(22):8137–8151. doi: 10.1128/MCB.23.22.8137-8151.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Fourquet S, Guerois R, Biard D, Toledano MB. Activation of NRF2 by nitrosative agents and H2O2 involves KEAP1 disulfide formation. J Biol Chem. 2010;285(11):8463–8471. doi: 10.1074/jbc.M109.051714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.DeNicola GM, Karreth FA, Humpton TJ, Gopinathan A, Wei C, Frese K, Mangal D, Yu KH, Yeo CJ, Calhoun ES, Scrimieri F, Winter JM, Hruban RH, Iacobuzio-Donahue C, Kern SE, Blair IA, Tuveson DA. Oncogene-induced Nrf2 transcription promotes ROS detoxification and tumorigenesis. Nature. 2011;475(7354):106–109. doi: 10.1038/nature10189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Satoh H, Moriguchi T, Takai J, Ebina M, Yamamoto M. Nrf2 prevents initiation but accelerates progression through the Kras signaling pathway during lung carcinogenesis. Cancer research. 2013;73(13):4158–4168. doi: 10.1158/0008-5472.CAN-12-4499. [DOI] [PubMed] [Google Scholar]
- 33.Neumann CA, Krause DS, Carman CV, Das S, Dubey DP, Abraham JL, Bronson RT, Fujiwara Y, Orkin SH, Van Etten RA. Essential role for the peroxiredoxin Prdx1 in erythrocyte antioxidant defence and tumour suppression. Nature. 2003;424(6948):561–565. doi: 10.1038/nature01819. [DOI] [PubMed] [Google Scholar]
- 34.Cantley LC. The phosphoinositide 3-kinase pathway. Science. 2002;296(5573):1655–1657. doi: 10.1126/science.296.5573.1655. [DOI] [PubMed] [Google Scholar]
- 35.Nemoto S, Finkel T. Redox regulation of forkhead proteins through a p66shc-dependent signaling pathway. Science. 2002;295(5564):2450–2452. doi: 10.1126/science.1069004. [DOI] [PubMed] [Google Scholar]
- 36.Lee SR, Yang KS, Kwon J, Lee C, Jeong W, Rhee SG. Reversible inactivation of the tumor suppressor PTEN by H2O2. J Biol Chem. 2002;277(23):20336–20342. doi: 10.1074/jbc.M111899200. [DOI] [PubMed] [Google Scholar]
- 37.Leslie NR, Bennett D, Lindsay YE, Stewart H, Gray A, Downes CP. Redox regulation of PI 3-kinase signalling via inactivation of PTEN. EMBO J. 2003;22(20):5501–5510. doi: 10.1093/emboj/cdg513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Connor KM, Subbaram S, Regan KJ, Nelson KK, Mazurkiewicz JE, Bartholomew PJ, Aplin AE, Tai YT, Aguirre-Ghiso J, Flores SC, Melendez JA. Mitochondrial H2O2 regulates the angiogenic phenotype via PTEN oxidation. J Biol Chem. 2005;280(17):16916–16924. doi: 10.1074/jbc.M410690200. [DOI] [PubMed] [Google Scholar]
- 39.Pelicano H, Xu RH, Du M, Feng L, Sasaki R, Carew JS, Hu Y, Ramdas L, Hu L, Keating MJ, Zhang W, Plunkett W, Huang P. Mitochondrial respiration defects in cancer cells cause activation of Akt survival pathway through a redox-mediated mechanism. J Cell Biol. 2006;175(6):913–923. doi: 10.1083/jcb.200512100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Ostman A, Frijhoff J, Sandin A, Bohmer FD. Regulation of protein tyrosine phosphatases by reversible oxidation. J Biochem. 2011;150(4):345–356. doi: 10.1093/jb/mvr104. [DOI] [PubMed] [Google Scholar]
- 41.Rao RK, Clayton LW. Regulation of protein phosphatase 2A by hydrogen peroxide and glutathionylation. Biochem Biophys Res Commun. 2002;293(1):610–616. doi: 10.1016/S0006-291X(02)00268-1. [DOI] [PubMed] [Google Scholar]
- 42.Salmeen A, Andersen JN, Myers MP, Meng TC, Hinks JA, Tonks NK, Barford D. Redox regulation of protein tyrosine phosphatase 1B involves a sulphenyl-amide intermediate. Nature. 2003;423(6941):769–773. doi: 10.1038/nature01680. [DOI] [PubMed] [Google Scholar]
- 43.Lou YW, Chen YY, Hsu SF, Chen RK, Lee CL, Khoo KH, Tonks NK, Meng TC. Redox regulation of the protein tyrosine phosphatase PTP1B in cancer cells. FEBS J. 2008;275(1):69–88. doi: 10.1111/j.1742-4658.2007.06173.x. [DOI] [PubMed] [Google Scholar]
- 44.Semenza GL. Hypoxia-inducible factors in physiology and medicine. Cell. 2012;148(3):399–408. doi: 10.1016/j.cell.2012.01.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Kaelin WG, Jr, Ratcliffe PJ. Oxygen sensing by metazoans: the central role of the HIF hydroxylase pathway. Mol Cell. 2008;30(4):393–402. doi: 10.1016/j.molcel.2008.04.009. [DOI] [PubMed] [Google Scholar]
- 46.Semenza GL. Targeting HIF-1 for cancer therapy. Nature reviews Cancer. 2003;3(10):721–732. doi: 10.1038/nrc1187. [DOI] [PubMed] [Google Scholar]
- 47.Chandel NS, Maltepe E, Goldwasser E, Mathieu CE, Simon MC, Schumacker PT. Mitochondrial reactive oxygen species trigger hypoxia-induced transcription. Proc Natl Acad Sci USA. 1998;95(20):11715–11720. doi: 10.1073/pnas.95.20.11715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Chandel NS, Schumacker PT. Cells depleted of mitochondrial DNA (rho0) yield insight into physiological mechanisms. FEBS Lett. 1999;454(3):173–176. doi: 10.1016/S0014-5793(99)00783-8. [DOI] [PubMed] [Google Scholar]
- 49.Chandel NS, McClintock DS, Feliciano CE, Wood TM, Melendez JA, Rodriguez AM, Schumacker PT. Reactive oxygen species generated at mitochondrial complex III stabilize hypoxia-inducible factor-1alpha during hypoxia: a mechanism of O2 sensing. J Biol Chem. 2000;275(33):25130–25138. doi: 10.1074/jbc.M001914200. [DOI] [PubMed] [Google Scholar]
- 50.Waypa GB, Marks JD, Guzy R, Mungai PT, Schriewer J, Dokic D, Schumacker PT. Hypoxia triggers subcellular compartmental redox signaling in vascular smooth muscle cells. Circ Res. 2010;106(3):526–535. doi: 10.1161/CIRCRESAHA.109.206334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Guzy RD, Hoyos B, Robin E, Chen H, Liu L, Mansfield KD, Simon MC, Hammerling U, Schumacker PT. Mitochondrial complex III is required for hypoxia-induced ROS production and cellular oxygen sensing. Cell Metab. 2005;1(6):401–408. doi: 10.1016/j.cmet.2005.05.001. [DOI] [PubMed] [Google Scholar]
- 52.Brunelle JK, Bell EL, Quesada NM, Vercauteren K, Tiranti V, Zeviani M, Scarpulla RC, Chandel NS. Oxygen sensing requires mitochondrial ROS but not oxidative phosphorylation. Cell Metab. 2005;1(6):409–414. doi: 10.1016/j.cmet.2005.05.002. [DOI] [PubMed] [Google Scholar]
- 53.Mansfield KD, Guzy RD, Pan Y, Young RM, Cash TP, Schumacker PT, Simon MC. Mitochondrial dysfunction resulting from loss of cytochrome c impairs cellular oxygen sensing and hypoxic HIF-alpha activation. Cell Metab. 2005;1(6):393–399. doi: 10.1016/j.cmet.2005.05.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Bell EL, Klimova TA, Eisenbart J, Moraes CT, Murphy MP, Budinger GR, Chandel NS. The Qo site of the mitochondrial complex III is required for the transduction of hypoxic signaling via reactive oxygen species production. J Cell Biol. 2007;177(6):1029–1036. doi: 10.1083/jcb.200609074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Gerald D, Berra E, Frapart YM, Chan DA, Giaccia AJ, Mansuy D, Pouyssegur J, Yaniv M, Mechta-Grigoriou F. JunD reduces tumor angiogenesis by protecting cells from oxidative stress. Cell. 2004;118(6):781–794. doi: 10.1016/j.cell.2004.08.025. [DOI] [PubMed] [Google Scholar]
- 56.Sanjuan-Pla A, Cervera AM, Apostolova N, Garcia-Bou R, Victor VM, Murphy MP, McCreath KJ. A targeted antioxidant reveals the importance of mitochondrial reactive oxygen species in the hypoxic signaling of HIF-1alpha. FEBS Lett. 2005;579(12):2669–2674. doi: 10.1016/j.febslet.2005.03.088. [DOI] [PubMed] [Google Scholar]
- 57.Lin X, David CA, Donnelly JB, Michaelides M, Chandel NS, Huang X, Warrior U, Weinberg F, Tormos KV, Fesik SW, Shen Y. A chemical genomics screen highlights the essential role of mitochondria in HIF-1 regulation. Proc Natl Acad Sci USA. 2008;105(1):174–179. doi: 10.1073/pnas.0706585104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Ma Q, Cavallin LE, Yan B, Zhu S, Duran EM, Wang H, Hale LP, Dong C, Cesarman E, Mesri EA, Goldschmidt-Clermont PJ. Antitumorigenesis of antioxidants in a transgenic Rac1 model of Kaposi's sarcoma. Proc Natl Acad Sci U S A. 2009;106(21):8683–8688. doi: 10.1073/pnas.0812688106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Gao P, Zhang H, Dinavahi R, Li F, Xiang Y, Raman V, Bhujwalla ZM, Felsher DW, Cheng L, Pevsner J, Lee LA, Semenza GL, Dang CV. HIF-dependent antitumorigenic effect of antioxidants in vivo. Can Cell. 2007;12(3):230–238. doi: 10.1016/j.ccr.2007.08.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Kim JW, Tchernyshyov I, Semenza GL, Dang CV. HIF-1-mediated expression of pyruvate dehydrogenase kinase: a metabolic switch required for cellular adaptation to hypoxia. Cell Metab. 2006;3(3):177–185. doi: 10.1016/j.cmet.2006.02.002. [DOI] [PubMed] [Google Scholar]
- 61.Tello D, Balsa E, Acosta-Iborra B, Fuertes-Yebra E, Elorza A, Ordóñez Á, Corral-Escariz M, Soro I, López-Bernardo E, Perales-Clemente E, Martínez-Ruiz A, Enríquez JA, Aragonés J, Cadenas S, Landázuri MO. Induction of the mitochondrial NDUFA4L2 protein by HIF-1α decreases oxygen consumption by inhibiting complex I activity. Cell Metab. 2011;14(6):768–779. doi: 10.1016/j.cmet.2011.10.008. [DOI] [PubMed] [Google Scholar]
- 62.Chen Z, Li Y, Zhang H, Huang P, Luthra R. Hypoxia-regulated microRNA-210 modulates mitochondrial function and decreases ISCU and COX10 expression. Oncogene. 2010;29(30):4362–4368. doi: 10.1038/onc.2010.193. [DOI] [PubMed] [Google Scholar]
- 63.Chan SY, Zhang YY, Hemann C, Mahoney CE, Zweier JL, Loscalzo J. MicroRNA-210 controls mitochondrial metabolism during hypoxia by repressing the iron-sulfur cluster assembly proteins ISCU1/2. Cell Metab. 2009;10(4):273–284. doi: 10.1016/j.cmet.2009.08.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Mitsuishi Y, Taguchi K, Kawatani Y, Shibata T, Nukiwa T, Aburatani H, Yamamoto M, Motohashi H. Nrf2 redirects glucose and glutamine into anabolic pathways in metabolic reprogramming. Can Cell. 2012;22(1):66–79. doi: 10.1016/j.ccr.2012.05.016. [DOI] [PubMed] [Google Scholar]
- 65.Hitosugi T, Kang S, Vander Heiden MG, Chung TW, Elf S, Lythgoe K, Dong S, Lonial S, Wang X, Chen GZ, Xie J, Gu TL, Polakiewicz RD, Roesel JL, Boggon TJ, Khuri FR, Gilliland DG, Cantley LC, Kaufman J, Chen J. Tyrosine phosphorylation inhibits PKM2 to promote the Warburg effect and tumor growth. Sci Signal. 2009;2(97):ra73. doi: 10.1126/scisignal.2000431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Christofk HR, Vander Heiden MG, Wu N, Asara JM, Cantley LC. Pyruvate kinase M2 is a phosphotyrosine-binding protein. Nature. 2008;452(7184):181–186. doi: 10.1038/nature06667. [DOI] [PubMed] [Google Scholar]
- 67.Anastasiou D, Poulogiannis G, Asara JM, Boxer MB, Jiang JK, Shen M, Bellinger G, Sasaki AT, Locasale JW, Auld DS, Thomas CJ, Vander Heiden MG, Cantley LC. Inhibition of pyruvate kinase M2 by reactive oxygen species contributes to cellular antioxidant responses. Science. 2011;334(6060):1278–1283. doi: 10.1126/science.1211485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Israelsen WJ, Dayton TL, Davidson SM, Fiske BP, Hosios AM, Bellinger G, Li J, Yu Y, Sasaki M, Horner JW, Burga LN, Xie J, Jurczak MJ, DePinho RA, Clish CB, Jacks T, Kibbey RG, Wulf GM, Di Vizio D, Mills GB, Cantley LC, Vander Heiden MG. PKM2 isoform-specific deletion reveals a differential requirement for pyruvate kinase in tumor cells. Cell. 2013;155(2):397–409. doi: 10.1016/j.cell.2013.09.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Weinberg F, Hamanaka R, Wheaton WW, Weinberg S, Joseph J, Lopez M, Kalyanaraman B, Mutlu GM, Budinger GR, Chandel NS. Mitochondrial metabolism and ROS generation are essential for Kras-mediated tumorigenicity. Proc Natl Acad Sci USA. 2010;107(19):8788–8793. doi: 10.1073/pnas.1003428107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Vafa O, Wade M, Kern S, Beeche M, Pandita TK, Hampton GM, Wahl GM. c-Myc can induce DNA damage, increase reactive oxygen species, and mitigate p53 function: a mechanism for oncogene-induced genetic instability. Mol Cell. 2002;9(5):1031–1044. doi: 10.1016/S1097-2765(02)00520-8. [DOI] [PubMed] [Google Scholar]
- 71.Tanaka H, Matsumura I, Ezoe S, Satoh Y, Sakamaki T, Albanese C, Machii T, Pestell RG, Kanakura Y. E2F1 and c-Myc potentiate apoptosis through inhibition of NF-kappaB activity that facilitates MnSOD-mediated ROS elimination. Mol Cell. 2002;9(5):1017–1029. doi: 10.1016/S1097-2765(02)00522-1. [DOI] [PubMed] [Google Scholar]
- 72.Levine AJ, Oren M. The first 30 years of p53: growing ever more complex. Nature reviews Cancer. 2009;9(10):749–758. doi: 10.1038/nrc2723. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Vousden KH, Prives C. Blinded by the light: the growing complexity of p53. Cell. 2009;137(3):413–431. doi: 10.1016/j.cell.2009.04.037. [DOI] [PubMed] [Google Scholar]
- 74.Li T, Kon N, Jiang L, Tan M, Ludwig T, Zhao Y, Baer R, Gu W. Tumor suppression in the absence of p53-mediated cell-cycle arrest, apoptosis, and senescence. Cell. 2012;149(6):1269–1283. doi: 10.1016/j.cell.2012.04.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Sablina AA, Budanov AV, Ilyinskaya GV, Agapova LS, Kravchenko JE, Chumakov PM. The antioxidant function of the p53 tumor suppressor. Nat Med. 2005;11(12):1306–1313. doi: 10.1038/nm1320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Roth M, Chen WY. Sorting out functions of sirtuins in cancer. Oncogene. 2014;33(13):1609–1620. doi: 10.1038/onc.2013.120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Bell EL, Guarente L. The SirT3 divining rod points to oxidative stress. Mol Cell. 2011;42(5):561–568. doi: 10.1016/j.molcel.2011.05.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Finley LW, Carracedo A, Lee J, Souza A, Egia A, Zhang J, Teruya-Feldstein J, Moreira PI, Cardoso SM, Clish CB, Pandolfi PP, Haigis MC. SIRT3 opposes reprogramming of cancer cell metabolism through HIF1α destabilization. Can Cell. 2011;19(3):416–428. doi: 10.1016/j.ccr.2011.02.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Bell EL, Emerling BM, Ricoult SJ, Guarente L. SirT3 suppresses hypoxia inducible factor 1alpha and tumor growth by inhibiting mitochondrial ROS production. Oncogene. 2011;30(26):2986–2996. doi: 10.1038/onc.2011.37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Chatterjee A, Mambo E, Sidransky D. Mitochondrial DNA mutations in human cancer. Oncogene. 2006;25(34):4663–4674. doi: 10.1038/sj.onc.1209604. [DOI] [PubMed] [Google Scholar]
- 81.Larman TC, DePalma SR, Hadjipanayis AG, Protopopov A, Zhang J, Gabriel SB, Chin L, Seidman CE, Kucherlapati R, Seidman JG, Cancer Genome Atlas Research Network Spectrum of somatic mitochondrial mutations in five cancers. Proc Natl Acad Sci U S A. 2012;109(35):14087–14091. doi: 10.1073/pnas.1211502109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Park JS, Sharma LK, Li H, Xiang R, Holstein D, Wu J, Lechleiter J, Naylor SL, Deng JJ, Lu J, Bai Y. A heteroplasmic, not homoplasmic, mitochondrial DNA mutation promotes tumorigenesis via alteration in reactive oxygen species generation and apoptosis. Hum Mol Genet. 2009;18(9):1578–1589. doi: 10.1093/hmg/ddp069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Sharma LK, Fang H, Liu J, Vartak R, Deng J, Bai Y. Mitochondrial respiratory complex I dysfunction promotes tumorigenesis through ROS alteration and AKT activation. Hum Mol Genet. 2011;20(23):4605–4616. doi: 10.1093/hmg/ddr395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Ishikawa K, Takenaga K, Akimoto M, Koshikawa N, Yamaguchi A, Imanishi H, Nakada K, Honma Y, Hayashi J. ROS-generating mitochondrial DNA mutations can regulate tumor cell metastasis. Science. 2008;320(5876):661–664. doi: 10.1126/science.1156906. [DOI] [PubMed] [Google Scholar]
- 85.Birsoy K, Possemato R, Lorbeer FK, Bayraktar EC, Thiru P, Yucel B, Wang T, Chen WW, Clish CB, Sabatini DM. Metabolic determinants of cancer cell sensitivity to glucose limitation and biguanides. Nature. 2014;508(7494):108–112. doi: 10.1038/nature13110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Iommarini L, Kurelac I, Capristo M, Calvaruso MA, Giorgio V, Bergamini C, Ghelli A, Nanni P, De Giovanni C, Carelli V, Fato R, Lollini PL, Rugolo M, Gasparre G, Porcelli AM. Different mtDNA mutations modify tumor progression in dependence of the degree of respiratory complex I impairment. Hum Mol Genet. 2014;23(6):1453–1466. doi: 10.1093/hmg/ddt533. [DOI] [PubMed] [Google Scholar]
- 87.Woo DK, Green PD, Santos JH, D'Souza AD, Walther Z, Martin WD, Christian BE, Chandel NS, Shadel GS. Mitochondrial genome instability and ROS enhance intestinal tumorigenesis in APC(Min/+) mice. Am J Pathol. 2012;180(1):24–31. doi: 10.1016/j.ajpath.2011.10.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Dahia PL. Pheochromocytoma and paraganglioma pathogenesis: learning from genetic heterogeneity. Nature reviews Cancer. 2014;14(2):108–119. doi: 10.1038/nrc3648. [DOI] [PubMed] [Google Scholar]
- 89.Guzy RD, Sharma B, Bell E, Chandel NS, Schumacker PT. Loss of the SdhB, but not the SdhA, subunit of complex II triggers reactive oxygen species-dependent hypoxia-inducible factor activation and tumorigenesis. Mol Cell Biol. 2008;28(2):718–731. doi: 10.1128/MCB.01338-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Ishii T, Yasuda K, Akatsuka A, Hino O, Hartman PS, Ishii N. A mutation in the SDHC gene of complex II increases oxidative stress, resulting in apoptosis and tumorigenesis. Cancer Res. 2005;65(1):203–209. [PubMed] [Google Scholar]
- 91.Sudarshan S, Sourbier C, Kong HS, Block K, Valera Romero VA, Yang Y, Galindo C, Mollapour M, Scroggins B, Goode N, Lee MJ, Gourlay CW, Trepel J, Linehan WM, Neckers L. Fumarate hydratase deficiency in renal cancer induces glycolytic addiction and hypoxia-inducible transcription factor 1alpha stabilization by glucose-dependent generation of reactive oxygen species. Mol Cell Biol. 2009;29(15):4080–4090. doi: 10.1128/MCB.00483-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Nagai R, Brock JW, Blatnik M, Baatz JE, Bethard J, Walla MD, Thorpe SR, Baynes JW, Frizzell N. Succination of protein thiols during adipocyte maturation: a biomarker of mitochondrial stress. J Biol Chem. 2007;282(47):34219–34228. doi: 10.1074/jbc.M703551200. [DOI] [PubMed] [Google Scholar]
- 93.Sullivan LB, Martinez-Garcia E, Nguyen H, Mullen AR, Dufour E, Sudarshan S, Licht JD, Deberardinis RJ, Chandel NS. The proto-oncometabolite fumarate binds glutathione to amplify ROS-dependent signaling. Mol cell. 2013;51(2):236–248. doi: 10.1016/j.molcel.2013.05.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Adam J, Hatipoglu E, O'Flaherty L, Ternette N, Sahgal N, Lockstone H, Baban D, Nye E, Stamp GW, Wolhuter K, Stevens M, Fischer R, Carmeliet P, Maxwell PH, Pugh CW, Frizzell N, Soga T, Kessler BM, El-Bahrawy M, Ratcliffe PJ, Pollard PJ. Renal cyst formation in Fh1-deficient mice is independent of the Hif/Phd pathway: roles for fumarate in KEAP1 succination and Nrf2 signaling. Cancer Cell. 2011;20(4):524–537. doi: 10.1016/j.ccr.2011.09.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Ooi A, Wong JC, Petillo D, Roossien D, Perrier-Trudova V, Whitten D, Min BW, Tan MH, Zhang Z, Yang XJ, Zhou M, Gardie B, Molinié V, Richard S, Tan PH, Teh BT, Furge KA. An antioxidant response phenotype shared between hereditary and sporadic type 2 papillary renal cell carcinoma. Cancer Cell. 2011;20(4):511–523. doi: 10.1016/j.ccr.2011.08.024. [DOI] [PubMed] [Google Scholar]
- 96.Omenn GS, Goodman GE, Thornquist MD, Balmes J, Cullen MR, Glass A, Keogh JP, Meyskens FL, Valanis B, Williams JH, Barnhart S, Hammar S. Effects of a combination of beta carotene and vitamin A on lung cancer and cardiovascular disease. N Engl J Med. 1996;334(18):1150–1155. doi: 10.1056/NEJM199605023341802. [DOI] [PubMed] [Google Scholar]
- 97.Klein EA, Thompson IM, Jr, Tangen CM, Crowley JJ, Lucia MS, Goodman PJ, Minasian LM, Ford LG, Parnes HL, Gaziano JM, Karp DD, Lieber MM, Walther PJ, Klotz L, Parsons JK, Chin JL, Darke AK, Lippman SM, Goodman GE, Meyskens FL, Jr, Baker LH. Vitamin E and the risk of prostate cancer: the Selenium and Vitamin E Cancer Prevention Trial (SELECT) JAMA. 2011;306(14):1549–1556. doi: 10.1001/jama.2011.1437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Sayin VI, Ibrahim MX, Larsson E, Nilsson JA, Lindahl P, Bergo MO. Antioxidants accelerate lung cancer progression in mice. Sci Transl Med. 2014;6(221):221–ra215. doi: 10.1126/scitranslmed.3007653. [DOI] [PubMed] [Google Scholar]
- 99.Sena LA, Li S, Jairaman A, Prakriya M, Ezponda T, Hildeman DA, Wang CR, Schumacker PT, Licht JD, Perlman H, Bryce PJ, Chandel NS. Mitochondria are required for antigen-specific T cell activation through reactive oxygen species signaling. Immunity. 2013;38(2):225–236. doi: 10.1016/j.immuni.2012.10.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Cheng G, Zielonka J, McAllister DM, Mackinnon AC, Jr, Joseph J, Dwinell MB, Kalyanaraman B. Mitochondria-targeted vitamin E analogs inhibit breast cancer cell energy metabolism and promote cell death. BMC cancer. 2013;13:285. doi: 10.1186/1471-2407-13-285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Noto H, Goto A, Tsujimoto T, Noda M. Cancer risk in diabetic patients treated with metformin: a systematic review and meta-analysis. PloS one. 2012;7(3):e33411. doi: 10.1371/journal.pone.0033411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.El-Mir MY, Nogueira V, Fontaine E, Averet N, Rigoulet M, Leverve X. Dimethylbiguanide inhibits cell respiration via an indirect effect targeted on the respiratory chain complex I. J Biol Chem. 2000;275(1):223–228. doi: 10.1074/jbc.275.1.223. [DOI] [PubMed] [Google Scholar]
- 103.Owen MR, Doran E, Halestrap AP. Evidence that metformin exerts its anti-diabetic effects through inhibition of complex 1 of the mitochondrial respiratory chain. Biochem J. 2000;348(Pt 3):607–614. doi: 10.1042/bj3480607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Wheaton WW, Weinberg SE, Hamanaka RB, Soberanes S, Sullivan LB, Anso E, Glasauer A, Dufour E, Mutlu GM, Budinger GR, Chandel NS: Metformin inhibits mitochondrial complex I of cancer cells to reduce tumorigenesis.eLife 2014.,3(e02242): [DOI] [PMC free article] [PubMed]
- 105.Mochizuki T, Furuta S, Mitsushita J, Shang WH, Ito M, Yokoo Y, Yamaura M, Ishizone S, Nakayama J, Konagai A, Hirose K, Kiyosawa K, Kamata T. Inhibition of NADPH oxidase 4 activates apoptosis via the AKT/apoptosis signal-regulating kinase 1 pathway in pancreatic cancer PANC-1 cells. Oncogene. 2006;25(26):3699–3707. doi: 10.1038/sj.onc.1209406. [DOI] [PubMed] [Google Scholar]
- 106.Bhandarkar SS, Jaconi M, Fried LE, Bonner MY, Lefkove B, Govindarajan B, Perry BN, Parhar R, Mackelfresh J, Sohn A, Stouffs M, Knaus U, Yancopoulos G, Reiss Y, Benest AV, Augustin HG, Arbiser JL. Fulvene-5 potently inhibits NADPH oxidase 4 and blocks the growth of endothelial tumors in mice. J Clin Invest. 2009;119(8):2359–2365. doi: 10.1172/JCI33877. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Munson JM, Fried L, Rowson SA, Bonner MY, Karumbaiah L, Diaz B, Courtneidge SA, Knaus UG, Brat DJ, Arbiser JL, Bellamkonda RV. Anti-invasive adjuvant therapy with imipramine blue enhances chemotherapeutic efficacy against glioma. Sci Transl Med. 2012;4(127):127ra36. doi: 10.1126/scitranslmed.3003016. [DOI] [PubMed] [Google Scholar]
- 108.Conklin KA. Chemotherapy-associated oxidative stress: impact on chemotherapeutic effectiveness. Integr Cancer Ther. 2004;3(4):294–300. doi: 10.1177/1534735404270335. [DOI] [PubMed] [Google Scholar]
- 109.Gorrini C, Harris IS, Mak TW. Modulation of oxidative stress as an anticancer strategy. Nature reviews Drug discovery. 2013;12(12):931–947. doi: 10.1038/nrd4002. [DOI] [PubMed] [Google Scholar]
- 110.Raj L, Ide T, Gurkar AU, Foley M, Schenone M, Li X, Tolliday NJ, Golub TR, Carr SA, Shamji AF, Stern AM, Mandinova A, Schreiber SL, Lee SW. Selective killing of cancer cells by a small molecule targeting the stress response to ROS. Nature. 2011;475(7355):231–234. doi: 10.1038/nature10167. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 111.Yang WS, Sriramaratnam R, Welsch ME, Shimada K, Skouta R, Viswanathan VS, Cheah JH, Clemons PA, Shamji AF, Clish CB, Brown LM, Girotti AW, Cornish VW, Schreiber SL, Stockwell BR. Regulation of ferroptotic cancer cell death by GPX4. Cell. 2014;156(1–2):317–331. doi: 10.1016/j.cell.2013.12.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Trachootham D, Zhou Y, Zhang H, Demizu Y, Chen Z, Pelicano H, Chiao PJ, Achanta G, Arlinghaus RB, Liu J, Huang P. Selective killing of oncogenically transformed cells through a ROS-mediated mechanism by beta-phenylethyl isothiocyanate. Cancer cell. 2006;10(3):241–252. doi: 10.1016/j.ccr.2006.08.009. [DOI] [PubMed] [Google Scholar]
- 113.Glasauer A, Sena LA, Diebold LP, Mazar AP, Chandel NS. Targeting SOD1 reduces experimental non-small-cell lung cancer. J Clin Invest. 2014;124(1):117–128. doi: 10.1172/JCI71714. [DOI] [PMC free article] [PubMed] [Google Scholar]


