Abstract
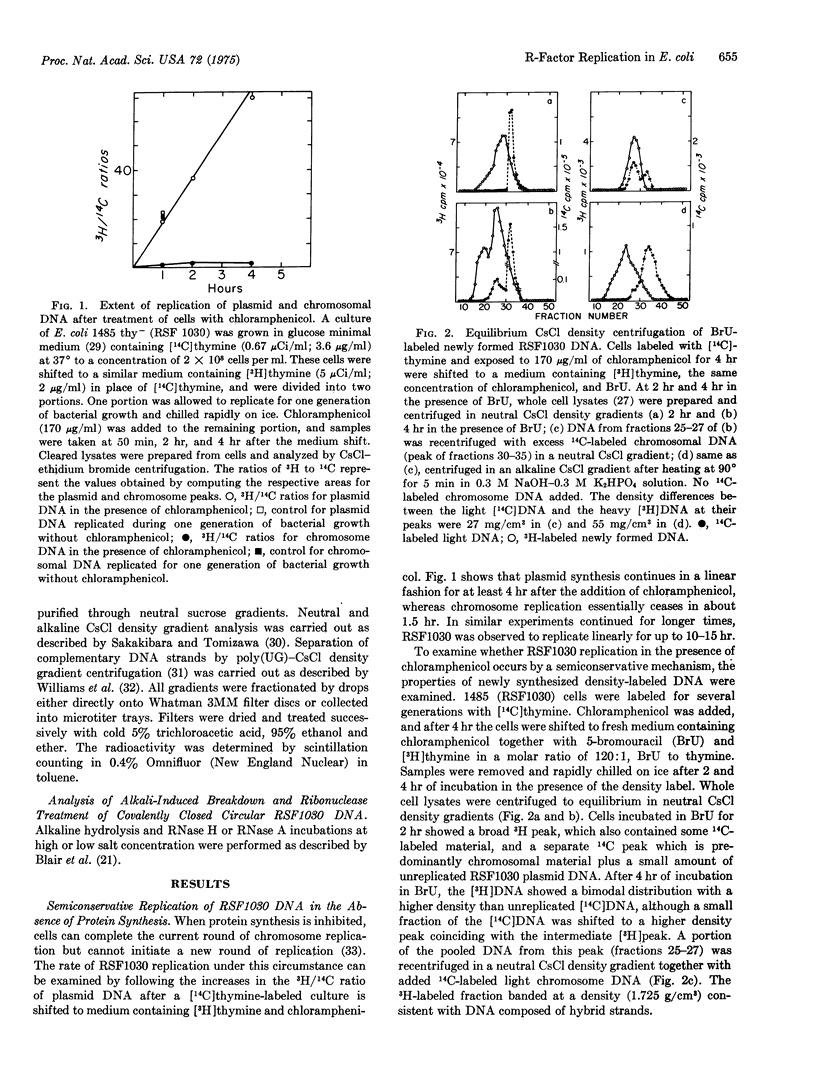
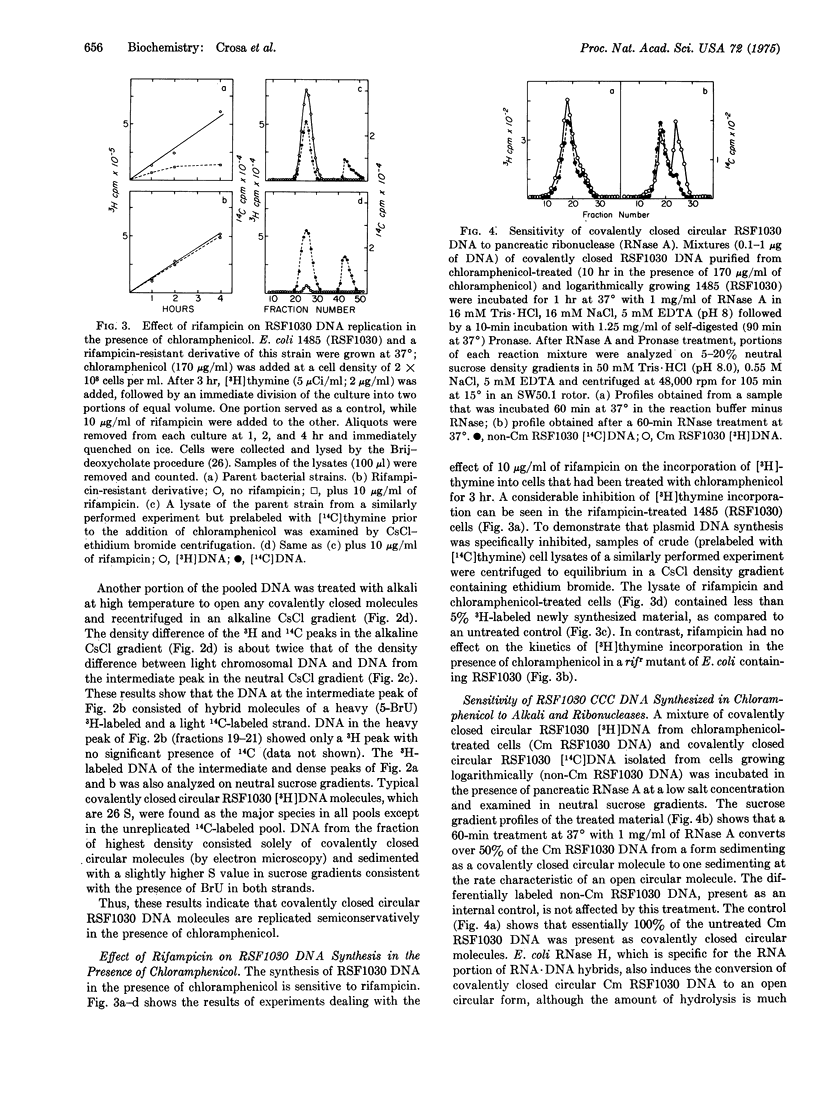
Covalently closed circular deoxyribonucleic acid molecules of RSF1030, a nonconjugative R-factor, initiate and complete rounds of semiconservative replication in the absence of protein synthesis long after the bacterial chromosome has ceased its replication. RSF1030 replication under these conditions is sensitive to the inhibitor of ribonucleic acid synthesis, rifampicin. The product of this replication, a covalently closed DNA molecule, shows, in contrast to those molecules produced during the replication in a logarithmically growing culture of Escherichia coli, a transition to the open circular form upon treatment with ribonucleases of alkali. Analysis of the product resulting from alkali treatment indicates that there is a single break in one strand of the original circular duplex. This alkali-sensitive site occurs with equal probability in either of the complementary strands. These results are interpreted as a requirement for an RNA primer for the initiation of RSF1030 DNA synthesis and as showing that its removal from the covalently closed molecule is inhibited in the absence of protein synthesis.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Anderson E. S., Natkin E. Transduction of resistance determinants and R factors of the transfer systems by phage Plkc. Mol Gen Genet. 1972;114(3):261–265. doi: 10.1007/BF01788895. [DOI] [PubMed] [Google Scholar]
- Bazaral M., Helinski D. R. Circular DNA forms of colicinogenic factors E1, E2 and E3 from Escherichia coli. J Mol Biol. 1968 Sep 14;36(2):185–194. doi: 10.1016/0022-2836(68)90374-4. [DOI] [PubMed] [Google Scholar]
- Blair D. G., Sherratt D. J., Clewell D. B., Helinski D. R. Isolation of supercoiled colicinogenic factor E 1 DNA sensitive to ribonuclease and alkali. Proc Natl Acad Sci U S A. 1972 Sep;69(9):2518–2522. doi: 10.1073/pnas.69.9.2518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brutlag D., Schekman R., Kornberg A. A possible role for RNA polymerase in the initiation of M13 DNA synthesis. Proc Natl Acad Sci U S A. 1971 Nov;68(11):2826–2829. doi: 10.1073/pnas.68.11.2826. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chabbert Y. A., Scavizzi M. R., Witchitz J. L., Gerbaud G. R., Bouanchaud D. H. Incompatibility groups and the classification of fi - resistance factors. J Bacteriol. 1972 Nov;112(2):666–675. doi: 10.1128/jb.112.2.666-675.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clewell D. B., Evenchik B., Cranston J. W. Direct inhibition of Col E 1 plasmid DNA replication in Escherichia coli by rifampicin. Nat New Biol. 1972 May 3;237(70):29–31. doi: 10.1038/newbio237029a0. [DOI] [PubMed] [Google Scholar]
- Clewell D. B., Helinski D. R. Supercoiled circular DNA-protein complex in Escherichia coli: purification and induced conversion to an opern circular DNA form. Proc Natl Acad Sci U S A. 1969 Apr;62(4):1159–1166. doi: 10.1073/pnas.62.4.1159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clewell D. B. Nature of Col E 1 plasmid replication in Escherichia coli in the presence of the chloramphenicol. J Bacteriol. 1972 May;110(2):667–676. doi: 10.1128/jb.110.2.667-676.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cohen S. N., Chang A. C., Hsu L. Nonchromosomal antibiotic resistance in bacteria: genetic transformation of Escherichia coli by R-factor DNA. Proc Natl Acad Sci U S A. 1972 Aug;69(8):2110–2114. doi: 10.1073/pnas.69.8.2110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Flügel R. M., Wells R. D. Nucleotides at the RNA-DNA covalent bonds formed in the endogenous reaction by the avian myeloblastosis virus DNA polymerase. Virology. 1972 May;48(2):394–401. doi: 10.1016/0042-6822(72)90050-5. [DOI] [PubMed] [Google Scholar]
- Grossman L. I., Watson R., Vinograd J. The presence of ribonucleotides in mature closed-circular mitochondrial DNA. Proc Natl Acad Sci U S A. 1973 Dec;70(12):3339–3343. doi: 10.1073/pnas.70.12.3339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kline B. C. Inhibition of plasmid DNA replication by rifampin in Salmonella pullorum. Biochem Biophys Res Commun. 1972 Mar 24;46(6):2019–2025. doi: 10.1016/0006-291x(72)90753-x. [DOI] [PubMed] [Google Scholar]
- Kline B. C. Role of DNA transcription in the initiation of Escherichia coli sex factor (F) DNA replication. Biochem Biophys Res Commun. 1973 Jan 23;50(2):280–288. doi: 10.1016/0006-291x(73)90837-1. [DOI] [PubMed] [Google Scholar]
- Lark K. G. Evidence for the direct involvement of RNA in the initiation of DNA replication in Escherichia coli 15T. J Mol Biol. 1972 Feb 28;64(1):47–60. doi: 10.1016/0022-2836(72)90320-8. [DOI] [PubMed] [Google Scholar]
- MAALOE O., HANAWALT P. C. Thymine deficiency and the normal DNA replication cycle. I. J Mol Biol. 1961 Apr;3:144–155. doi: 10.1016/s0022-2836(61)80041-7. [DOI] [PubMed] [Google Scholar]
- Magnusson G., Pigiet V., Winnacker E. L., Abrams R., Reichard P. RNA-linked short DNA fragments during polyoma replication. Proc Natl Acad Sci U S A. 1973 Feb;70(2):412–415. doi: 10.1073/pnas.70.2.412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Messing J., Staudenbauer W. L., Hofschneider P. H. Inhibition of minicircular DNA replication in Escherichia coli 15 by rifampicin. Nat New Biol. 1972 Aug 16;238(85):202–203. doi: 10.1038/newbio238202a0. [DOI] [PubMed] [Google Scholar]
- Miyaki M., Koide K., Ono T. RNase and alkali sensitivity of closed circular mitochondrial DNA of rat ascites hepatoma cells. Biochem Biophys Res Commun. 1973 Jan 23;50(2):252–258. doi: 10.1016/0006-291x(73)90833-4. [DOI] [PubMed] [Google Scholar]
- Radloff R., Bauer W., Vinograd J. A dye-buoyant-density method for the detection and isolation of closed circular duplex DNA: the closed circular DNA in HeLa cells. Proc Natl Acad Sci U S A. 1967 May;57(5):1514–1521. doi: 10.1073/pnas.57.5.1514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rosenkranz H. S. RNA in coliphage T5. Nature. 1973 Mar 30;242(5396):327–329. doi: 10.1038/242327a0. [DOI] [PubMed] [Google Scholar]
- Sakakibara Y., Tomizawa J. I. Replication of colicin E1 plasmid DNA in cell extracts. Proc Natl Acad Sci U S A. 1974 Mar;71(3):802–806. doi: 10.1073/pnas.71.3.802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sato S., Ariake S., Saito M., Sugimura T. RNA bound to nascent DNA in Ehrlich ascites tumor cells. Biochem Biophys Res Commun. 1972 Nov 1;49(3):827–834. doi: 10.1016/0006-291x(72)90485-8. [DOI] [PubMed] [Google Scholar]
- Schekman R., Wickner W., Westergaard O., Brutlag D., Geider K., Bertsch L. L., Kornberg A. Initiation of DNA synthesis: synthesis of phiX174 replicative form requires RNA synthesis resistant to rifampicin. Proc Natl Acad Sci U S A. 1972 Sep;69(9):2691–2695. doi: 10.1073/pnas.69.9.2691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Silver R. P., Falkow S. Specific labeling and physical characterization of R-factor deoxyribonucleic acid in Escherichia coli. J Bacteriol. 1970 Oct;104(1):331–339. doi: 10.1128/jb.104.1.331-339.1970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Speyer J. F., Chao J., Chao L. Ribonucleotides covalently linked to deoxyribonucleic acid in T4 bacteriophage. J Virol. 1972 Nov;10(5):902–908. doi: 10.1128/jvi.10.5.902-908.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sugino A., Hirose S., Okazaki R. RNA-linked nascent DNA fragments in Escherichia coli. Proc Natl Acad Sci U S A. 1972 Jul;69(7):1863–1867. doi: 10.1073/pnas.69.7.1863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sugino A., Okazaki R. RNA-linked DNA fragments in vitro. Proc Natl Acad Sci U S A. 1973 Jan;70(1):88–92. doi: 10.1073/pnas.70.1.88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Veltkamp E., Barendsen W., Nijkamp H. J. Influence of protein and ribonucleic acid synthesis on the replication of the bacteriocinogenic factor Clo DF13 in Escherichia coli cells and minicells. J Bacteriol. 1974 Apr;118(1):165–174. doi: 10.1128/jb.118.1.165-174.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wickner W., Brutlag D., Schekman R., Kornberg A. RNA synthesis initiates in vitro conversion of M13 DNA to its replicative form. Proc Natl Acad Sci U S A. 1972 Apr;69(4):965–969. doi: 10.1073/pnas.69.4.965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williams H., Boyer H. W., Helsinki D. R. Size and base composition of RNA in supercoiled plasmid DNA. Proc Natl Acad Sci U S A. 1973 Dec;70(12):3744–3748. doi: 10.1073/pnas.70.12.3744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Young E. T., 2nd, Sinsheimer R. L. Vegetative bacteriophage lambda-DNA. II. Physical characterization and replication. J Mol Biol. 1967 Nov 28;30(1):165–200. doi: 10.1016/0022-2836(67)90251-3. [DOI] [PubMed] [Google Scholar]



