Epidermal growth factor receptor (EGFR) is the prototypical member of the ErbB network comprised of four transmembrane receptor tyrosine kinases (EGFR/ErbB1, ErbB2, ErbB3, and ErbB4). These receptors bind EGF and heregulin family ligands in a dynamic, feedback-regulated fashion, activating multiple signal transduction pathways and ultimately affecting many cellular functions (Avraham and Yarden, 2011; Wilson et al., 2009). ErbB receptors and their ligands are overexpressed in a wide spectrum of hyperplastic epithelial disorders including psoriasis and cancers. Numerous monoclonal antibodies and receptor tyrosine kinase inhibitors have been designed to block their activation (Yarden and Pines, 2012), and are highly effective for treating colorectal and non-small cell lung cancers. However, skin toxicity is frequently observed in patients undergoing therapy with EGFR inhibitors (EGFRIs) (Lacouture, 2006), including papular and pustular eruptions of the face and trunk, nail changes, dry skin, itch, and hair loss (Lacouture and Lai, 2006). These side-effects are the major dose-limiting toxicity of EGFRI therapy (Boone et al., 2007) and emphasize the importance of understanding the mechanisms involved.
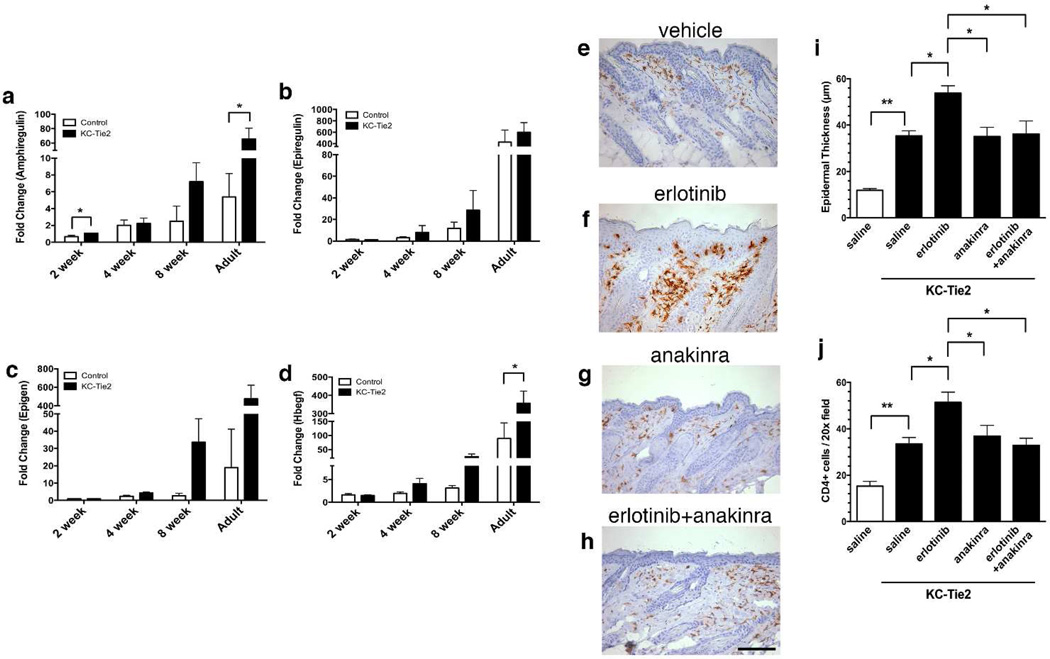
KC-Tie2 mice develop marked skin inflammation and epidermal hyperplasia which is suppressible by cyclosporin A (Wolfram et al., 2009), TNFα (Ward et al., 2011b) or vascular endothelial growth factor (VEGF) inhibition (Diaconu et al., 2013). In the course of experiments designed to identify the cause of this inflammatory hyperplasia, we observed a time-dependent increase in the expression of transcripts encoding the EGFR ligands amphiregulin, heparin-binding EGF-like growth factor (HB-EGF), epiregulin and epigen in full-thickness dorsal skin using qRT-PCR. These increases were much greater in KC-Tie2 double transgenic (DT) mice relative to wild-type (WT) or single transgenic (ST) littermate controls (Fig. 1a-d). To test the hypothesis that these EGFR ligands might be driving the observed inflammation, and that prevention of EGFR signaling would prevent phenotype development, we treated a group of KC-Tie2 DT mice and control littermates with intraperitoneal administration of erlotinib (10mg/kg erlotinib HCl, LC Laboratories) or vehicle for 28 days, beginning at postnatal day 10, emulsifying the erlotinib in Captisol (Cydex Pharmaceuticals) as described (Zerbe et al., 2008), and then analyzed the skin phenotype in a blinded manner as described previously (Wolfram et al., 2009). All mouse experiments were approved by the Institutional Animal Care and Use Committee of Case Western Reserve University School of Medicine. Despite the fact that we could demonstrate erlotinib inhibition of ultraviolet light-induced EGFR and ERK activation (data not shown), KC-Tie2 mice treated with erlotinib showed a marked increase in skin inflammation relative to saline-treated KC-Tie2 mice (Fig. 1), whereas erlotinib treatment of WT and ST littermates displayed no changes compared to saline treatment (data not shown). The increase in epidermal thickness as well as CD4+ (Fig. 1) and F4/80+ immune cell infiltrates (Supp Fig. 1a) was accompanied by a 6.7-fold increase in Il1b mRNA in erlotinib-treated KC-Tie2 skin by qRT-PCR (p=0.035, Supp Fig. 2), with no effect in erlotinib-treated littermate controls (data not shown). These findings are consistent with prior reported adverse effects of EGFRI therapy (Lacouture, 2006) and a previous study of EGFRI-induced inflammation in mouse skin (Surguladze et al., 2009). Based upon these studies as well our observation of increased Il1b, we hypothesized that erlotinib-mediated inflammatory hyperplasia in KC-Tie2 mice is at least in part mediated by interleukin-1 (IL-1).
Figure 1.
EGFR ligand expression increases in KC-Tie2 mouse skin during development of the inflammatory phenotype and erlotinib treatment increases epidermal thickness and immunocyte infiltration into skin, which is abrogated following anakinra treatment.
EGFR ligand mRNA expression (amphiregulin (a), epiregulin (b), epigen (c) and HB-EGF (d)) increases during development of the in KC-Tie2 skin phenotype. mRNA expression values are normalized to 18S rRNA and are expressed relative to week 1. Erlotinib-treated KC-Tie2 mice have increases in epidermal thickness (f and i) and CD4+ T cell skin infiltration (f, j) compared to vehicle (e) and anakinra alone (g), and this increase is abrogated with concomitant anakinra treatment (h-j). Photomicrographs depict CD4+ T cell immunohistochemistry with diaminobenzidine (DAB) as the chromogen counterstained with hematoxylin; scale bars indicate 100 µm. Bars, mean + SEM (n=3–4 for a-d and n=7–22 for i and j). Statistical significance indicated by * p < 0.05, ** p < 0.005, using unpaired multiple t-tests with Holm-Sidak correction for multiple comparisons.
To test this hypothesis, mice were treated daily with vehicle, erlotinib (10mg/kg), the IL-1 receptor antagonist anakinra (100mg/kg) or erlotinib and anakinra together, using the same approach described above. These doses correspond to therapeutic human in vivo patient levels for both erlotinib and anakinra. Notably, the erlotinib-mediated increases in epidermal thickness and CD4+ T cells (Fig. 1) and F4/80+ macrophages (Supp Fig. 1) were abrogated by anakinra. While CD8+ T cells and CD11c+ dendritic cells were significantly increased in KC-Tie2 mouse skin compared to littermate controls, there was no significant further increase or decrease in response to erlotinib or anakinra, respectively (Supp Fig. 1). While anakinra was able to reverse the pro-inlfammatory effects of ertlotinib in KC-Tie2 mice, the lack of efficacy of erlotinib and anakinra as individual agents in resolving the psoriasiform skin inflammation that arises spontaneously in this model is consistent with the lack of reported efficacy of each of these inhibitors for treating psoriasis.
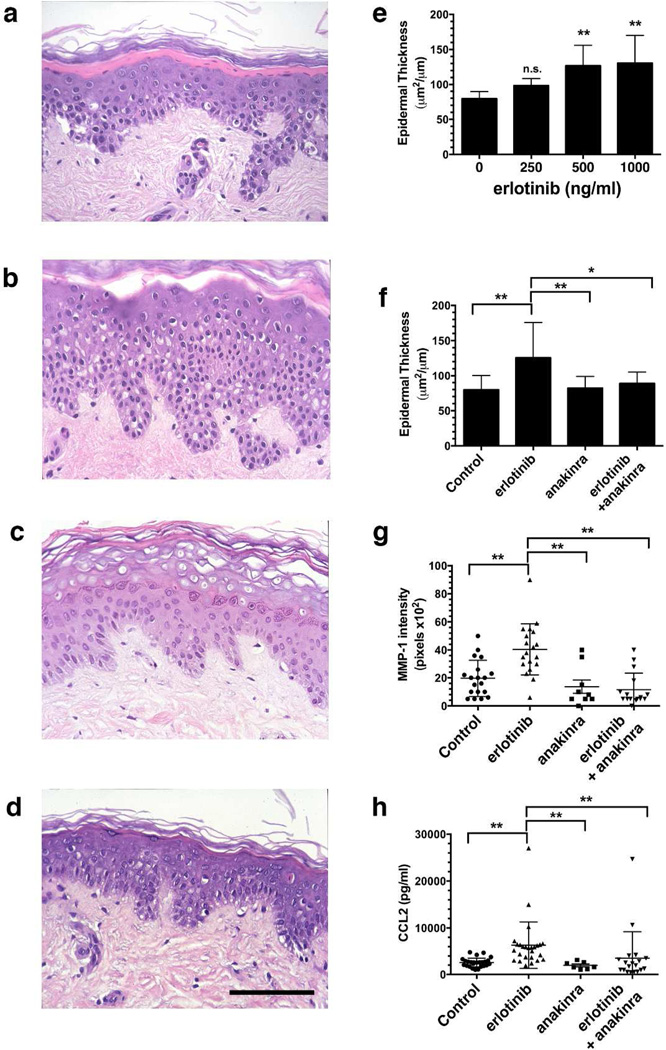
In parallel to the murine work, we conducted translational experiments using human skin organ cultures (Varani et al., 1993), examining the pro-inflammatory effects of erlotinib and the role of IL-1. The participation of human subjects in this study was approved by the University of Michigan Institutional Review Board and subjects provided written informed consent prior to inclusion in the study. Treatment of human skin organ cultures with erlotinib (2mm punch biopsies from hip skin of volunteers) led to significant increases in epidermal thickness (Fig. 2), which was dose-dependent and was prevented by 20µg/ml anakinra, and had no significant effect on its own (Fig. 2). EGFRIs potentiate the induction of chemokines such as CCL2, CCL5, and CXCL10 by TNF and IFN-γ in keratinocytes (Mascia et al., 2003) and CCL2 induction has been observed immunohistochemically in the skin of cancer patients treated with the EGFRI gefitinib (Yamaki et al., 2010). Accordingly, we found that erlotinib significantly increased elaboration of CCL2 and MMP-1 into the organ culture medium, and these increases were blocked by anakinra (Fig. 2g and 2h).
Figure 2.
Erlotinib-induced increases in epidermal thickness are blocked by anakinra in organ cultures of human skin.
Representative photomicrographs of H&E-stained sections for (a) control, (b) erlotinib (1000 ng/ml), (c) anakinra (20 µg/ml) and (d) erlotinib + anakinra treated cultures at day 7. Scale bars indicate 100 µm. Erlotinib induced a dose-dependent epidermal thickening in the human skin organ cultures (e). Treatment of human hip skin cultures with anakinra (20 µg/ml) lead to an attenuation of the erlotinib-induced epidermal thickening (f). Bars, mean + SEM, n=9–14 subjects for panel E and 8–27 subjects for panel f. Statistical significance denoted by * p < 0.05 and **p < 0.005 using two-tailed unpaired t-test with unequal variances and Welch’s correction (b). Anakinra also prevented production of MMP-1 (as determined by Western blotting; g) and CCL2 (measured by ELISA; h). Median ± 95% confidence interval shown. ** indicates p < 0.005, by Mann-Whitney U-test.
One final question concerns why erlotinib-mediated inflammatory events occur in organ cultures of normal human skin, whereas they do not occur in phenotypically-normal WT or ST mouse skin lacking Tie2 overexpression. While KC constitutively express angiopoietin (Larcher et al., 2003; Ward et al., 2011a), they express little to no endogenous Tie2 (Voskas et al., 2005; Wolfram et al., 2009). The expression of both genes in the epidermal compartment has been shown to activate the cutaneous neurovascular unit (CNU) (Ward et al., 2011a), at least in part by activating an autocrine signal transduction cascade in KCs that is not normally present (Wolfram et al., 2009). Many nerves and vessels are present in human skin, richly investing the hair follicles and eccrine glands. We would speculate that the CNU becomes activated in human skin organ cultures due to biopsy-induced trauma to the CNU, whereas it becomes activated in response to Ang1-Tie2 signaling in KC-Tie2 mice (Ward et al., 2011a). It is possible that an altered skin homeostasis involving the CNU may also be present in the subset of erlotinib-treated patients in whom skin rash develops.
Our results suggest that the cutaneous pro-inflammatory effects of erlotinib are IL-1-mediated in human and mouse skin. These findings support the notion that IL-1 inhibition may serve as a useful tool for either preventing or attenuating the dose-limiting side effects, specifically the skin toxicity, observed in patients undergoing therapy with EGFRIs (Lacouture, 2006). Our results also suggest that a simple autocrine EGFR-ligand-driven loop is unlikely to drive keratinocyte proliferation in either mouse or human skin, as envisioned in some earlier models of epidermal hyperplasia (Elder et al., 1989). Rather, EGFR signaling appears to be anti-inflammatory, with the increases in EGFR ligand expression that are observed in psoriasis (Johnston et al., 2011) and in KC-Tie2 mice (Wolfram et al., 2009) serving as a negative feedback mechanism. If so, this may explain why no successful controlled trials of EGFRIs as a therapeutic modality in psoriasis have appeared, despite their availability. Further exploration of other models in which skin homeostasis is altered is warranted to test this hypothesis in other contexts.
Supplementary Material
Acknowledgements
This work was supported in part by the National Institute for Arthritis, Musculoskeletal and Skin Disease (NIAMS), National Institutes of Health: R01 AR052889 (JTE); P30 AR39750, P50 AR05508, R01 AR063437; R01 AR062546; R21 AR063852 (NLW); K01 AR064765 (AJ); K08 AR060802 (JEG); and by awards to NLW, AJ, SL and JEG from the National Psoriasis Foundation, Dermatology Foundation, and the American Skin Association. JTE is supported by the Ann Arbor Veterans Affairs Hospital. JEG is an A. Alfred Taubman Medical Research Institute Kenneth and Frances Eisenberg Emerging Scholar Awardee. JTE, AJ, SL and JEG acknowledge the support of the Babcock Memorial Trust. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Abbreviations
- CNU
cutaneous neurovascular unit
- DT
double transgenic
- EGFR
epidermal growth factor receptor
- EGFRI
epidermal growth factor receptor inhibitor
- ErbB
avian erythroblastosis oncogene B
- HB-EGF
heparin binding EGF-like growth factor
- IL-1
interleukin-1
- qRT-PCR
quantitative reverse transcription-polymerase chain reaction
- ST
single transgenic
Footnotes
Conflict of Interest
The authors state no conflict of interest
References
- Avraham R, Yarden Y. Feedback regulation of EGFR signalling: decision making by early and delayed loops. Nat Rev Mol Cell Biol. 2011;12:104–117. doi: 10.1038/nrm3048. [DOI] [PubMed] [Google Scholar]
- Boone SL, Rademaker A, Liu D, et al. Impact and management of skin toxicity associated with anti-epidermal growth factor receptor therapy: survey results. Oncology. 2007;72:152–159. doi: 10.1159/000112795. [DOI] [PubMed] [Google Scholar]
- Diaconu D, Fritz Y, Dawes SM, et al. VEGF-Trap decreases CD4(+) T cells, Th17 cytokines improving psoriasis-like skin inflammation in KC-Tie2 mice. Journal of dermatological science. 2013;72:322–325. doi: 10.1016/j.jdermsci.2013.08.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elder JT, Fisher GJ, Lindquist PB, et al. Overexpression of transforming growth factor alpha in psoriatic epidermis. Science. 1989;243:811–814. doi: 10.1126/science.2916128. [DOI] [PubMed] [Google Scholar]
- Johnston A, Gudjonsson JE, Aphale A, et al. EGFR and IL-1 signaling synergistically promote keratinocyte antimicrobial defenses in a differentiationdependent manner. J Invest Dermatol. 2011;131:329–337. doi: 10.1038/jid.2010.313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lacouture ME. Mechanisms of cutaneous toxicities to EGFR inhibitors. Nat Rev Cancer. 2006;6:803–812. doi: 10.1038/nrc1970. [DOI] [PubMed] [Google Scholar]
- Lacouture ME, Lai SE. The PRIDE (Papulopustules and/or paronychia, Regulatory abnormalities of hair growth, Itching, and Dryness due to Epidermal growth factor receptor inhibitors) syndrome. The British journal of dermatology. 2006;155:852–854. doi: 10.1111/j.1365-2133.2006.07452.x. [DOI] [PubMed] [Google Scholar]
- Larcher F, Franco M, Bolontrade M, et al. Modulation of the angiogenesis response through Ha-ras control, placenta growth factor, and angiopoietin expression in mouse skin carcinogenesis. Molecular carcinogenesis. 2003;37:83–90. doi: 10.1002/mc.10126. [DOI] [PubMed] [Google Scholar]
- Mascia F, Mariani V, Girolomoni G, et al. Blockade of the EGF receptor induces a deranged chemokine expression in keratinocytes leading to enhanced skin inflammation. Am J Pathol. 2003;163:303–312. doi: 10.1016/S0002-9440(10)63654-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Surguladze D, Deevi D, Claros N, et al. Tumor necrosis factor-alpha and interleukin-1 antagonists alleviate inflammatory skin changes associated with epidermal growth factor receptor antibody therapy in mice. Cancer Res. 2009;69:5643–5647. doi: 10.1158/0008-5472.CAN-09-0487. [DOI] [PubMed] [Google Scholar]
- Varani J, Fligiel SE, Schuger L, et al. Effects of all-trans retinoic acid and Ca++ on human skin in organ culture. Am J Pathol. 1993;142:189–198. [PMC free article] [PubMed] [Google Scholar]
- Voskas D, Jones N, Van Slyke P, et al. A cyclosporine-sensitive psoriasis-like disease produced in Tie2 transgenic mice. Am J Pathol. 2005;166:843–855. doi: 10.1016/S0002-9440(10)62305-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ward NL, Hatala DA, Wolfram JA, et al. Cutaneous manipulation of vascular growth factors leads to alterations in immunocytes, blood vessels and nerves: Evidence for a cutaneous neurovascular unit. Journal of dermatological science. 2011a;61:14–22. doi: 10.1016/j.jdermsci.2010.11.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ward NL, Loyd CM, Wolfram JA, et al. Depletion of antigen-presenting cells by clodronate liposomes reverses the psoriatic skin phenotype in KC-Tie2 mice. The British journal of dermatology. 2011b;164:750–758. [Google Scholar]
- Wilson KJ, Gilmore JL, Foley J, et al. Functional selectivity of EGF family peptide growth factors: implications for cancer. Pharmacol Ther. 2009;122:1–8. doi: 10.1016/j.pharmthera.2008.11.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wolfram JA, Diaconu D, Hatala DA, et al. Keratinocyte but not endothelial cellspecific overexpression of Tie2 leads to the development of psoriasis. Am J Pathol. 2009;174:1443–1458. doi: 10.2353/ajpath.2009.080858. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamaki M, Sugiura K, Muro Y, et al. Epidermal growth factor receptor tyrosine kinase inhibitors induce CCL2 and CCL5 via reduction in IL-1R2 in keratinocytes. Experimental dermatology. 2010;19:730–735. doi: 10.1111/j.1600-0625.2010.01108.x. [DOI] [PubMed] [Google Scholar]
- Yarden Y, Pines G. The ERBB network: at last, cancer therapy meets systems biology. Nat Rev Cancer. 2012;12:553–563. doi: 10.1038/nrc3309. [DOI] [PubMed] [Google Scholar]
- Zerbe LK, Dwyer-Nield LD, Fritz JM, et al. Inhibition by erlotinib of primary lung adenocarcinoma at an early stage in male mice. Cancer chemotherapy and pharmacology. 2008;62:605–620. doi: 10.1007/s00280-007-0644-z. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.