Abstract
Massive intraventricular hemorrhage (IVH) is nearly always associated with hydrocephalus and is often treated with prolonged external ventricular drainage (EVD); however this procedure can lead to bacterial ventriculitis and meningitis, which can worsen the clinical outcomes. Endoscopic burr hole surgery to remove the hematomas in lateral and third ventricles is an alternative treatment option. We describe the surgical techniques and benefits of endoscopic surgery for acute massive IVH in four patients and discuss the current published literature-related to this condition. Four patients were treated endoscopically for massive IVH. Three patients presented with secondary IVH due to vascular malformation, tumoral bleed and chronic hypertension, while one case presented as massive primary IVH. Endoscopic wash out and removal of hematomas was normally performed together with an endoscopic third ventriculostomy. Recombinant factor VIIa was only administered prior to surgery for IVH secondary to vascular malformation and for cases with postoperative rebleeding which required second endoscopic surgery. Weaning from ventilator and EVD commenced on day 4 postoperatively. All treated patients recovered and did not require further shunt surgery. Good outcomes obtained may be related to early removal of hematomas, creation of new cerebrospinal fluid diversion pathway after thorough wash-out, early weaning from ventilator and EVD. Endoscopic surgery is beneficial in treating poor grade IVH with Graeb score of more than 6.
Keywords: Endoscopy, factor VIIa, Graeb score, hydrocephalus, intraventricular haemorrhage
Background
Intraventricular hemorrhage (IVH) is a common neurosurgical emergency usually seen in cases of hypertensive intracerebral hemorrhage (ICH) with the extension into the ventricular system, termed as secondary IVH. The common locations for ICH are the putamen, lenticular nucleus, internal capsule and globus pallidus (50% of cases), followed by the thalamus (15% of cases). Less common locations in order of decreasing frequency are the pons, cerebellum, white matter of cerebrum and brainstem.[1,2] Because of the anatomical proximity of the basal ganglia structures and thalamus to the ventricular system, most hemorrhages rupture through the ventricular wall into the ventricular system, producing IVH. This occurs in almost 55% of cases.[2,3,4] Other possible causes of massive IVH are aneurysmal or arteriovenous malformation (AVM) bleed and tumoral bleed.
The prognosis in IVH is influenced by several factors. Several studies done previously have shown that ventricular dilatation, intraventricular hematoma volume and increased intracranial pressure (ICP) are indicators of poor outcome in patients with IVH.[2,5,6] Thus, the primary goal of IVH management in the acute phase should be early removal of intraventricular blood, rapid reversal of ventricular dilatation and normalization of ICP. These objectives seem attainable by treating massive or poor grade IVH (Graeb score > 6) cases with neuroendoscopy rather than by conventional treatment using external ventricular drainage (EVD) catheter. In this article, we describe four cases of massive IVH, surgical techniques employed and review of the literature on IVH and neuroendoscopy.
Case Reports
Case 1
A 41-year-old man, known case of the left atrial AVM presented in an unconscious state with Glasgow coma score (GCS) of 4 and Graeb score of 9 secondary to massive IVH [Figure 1a and b]. The motor response was decerebrate, and pupils were pinpoint bilaterally. No evidence of persistent hyperthermia or arrthymia were noted to suggest severe hypothalamic dysfunction. He was operated as an emergency case by using a 30° neuroendoscopy. One ampoule of recombinant factor VIIa was administered prior to surgery. He achieved a good recovery after 1 month of rehabilitation and subsequently referred for AVM radiosurgery.
Figure 1.
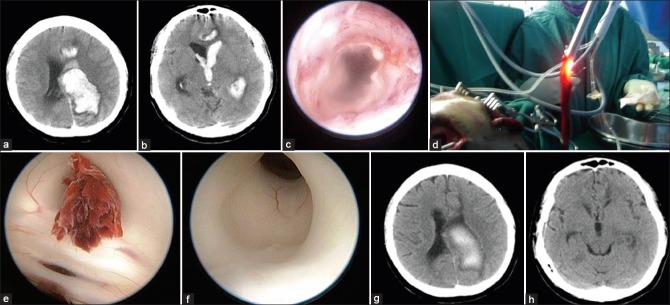
(a) Massive left lateral and paraventricular hemorrhage secondary to arteriovenous malformation. (b) The hemorrhage extends to the third ventricle. (c) The inner cortex is sometimes used to guide the surgeon. (d) Blood clot is removed. (e) Blood clot blocks the aqueduct. (f) Gentle suction successfully removed the clot. (g and h) Computed tomography brain after the surgery
Standard surgical technique
The patient was positioned supine with head fixed in Mayfield clamp, bringing the burr hole sites to the highest point. Bilateral burr holes were made at the frontal region away from the pathological site, and one burr hole was purposely made to get an easy access to the floor of the third ventricle. Cannulation of the lateral ventricles was initially done using an EVD. Both lateral ventricles were cannulated without difficulty due to an obvious ventriculomegaly. Neuronavigation was not used in this emergency surgery. By keeping at least one EVD in situ (change alternately with endoscope), the endoscope was introduced according to the trajectory and length estimated by the removed-EVD. An obvious marker was patched onto the endoscopic shaft commonly at 6 cm prior to its insertion. This marker reminds the neurosurgeon for not to pass beyond in order to avoid inadvertent injury to the deep structures. Endoscopic-inner-cortical view can also act as a navigation-guide to the neurosurgeon; whenever, the neurosurgeon felt uncertain about the exact location of the endoscopic tip, a gentle retraction to the inner cortex was done to orientate oneself and to ensure the actual operative depth [Figure 1c].
The hematomas were removed by using hand-controlled suction, aspirated via the center working channel. Three patterns of hemorrhage were commonly encountered; (a) liquefied clots which are easily aspirated and replaced with continuous infusion of warmed Hartmann's solution (b) semi-solid clots which require gentle shaking of the endoscope to aspirate the clots and (c) well-formed solid clots which can be removed by simultaneously aspirating the clots to the tip of the endoscope while withdrawing the endoscope [Figure 1d]. The other two endoscopic channels were used as an inlet and outlet for irrigating fluid. The outlet channel was also connected to the monitor for ICP monitoring. Further increase in ICP was prevented by maintaining the EVD at the opposite burr hole site.
Following clot removal, copious irrigation with warmed Hartman's solution is done for ventricular wash out, and this allows better visualization of ventricular anatomy. This subsequently facilitates clot removal in the third ventricle. After doing so, the floor of the third ventricle can now be clearly defined. In some cases, further inspection of the aqueduct inlet can be made by angulating the endoscope more posteriorly [Figure 1e and f]. This maneuver can be done without difficulty because of marked ventriculomegaly present in most cases with massive IVH. Frequently, the aqueduct is blocked and endoscopic inspection assists in decision making for endoscopic third ventriculostomy (ETV) at the same sitting to avoid prolonged-postoperative EVD. In this case, patency of aqueduct was observed after gentle suction of the blood clot at the surface of aqueduct entry as shown in Figure 1e and f, therefore, the ETV was not performed. The EVD was kept until day 3 postsurgery to facilitate drainage of residual liquefied hematomas and for ICP monitoring. Repeated postsurgery computed tomography (CT) brain is shown in Figure 1g and h. Serial CT brain of at least 2-3 times after the surgery was normally done to detect hydrocephalus. The patient recovered well and was discharged home without requiring further shunt surgery. He was referred to the neurocognitive outpatient rehabilitation unit as massive-IVH induced limbic system injury is common.
Case 2
A 15-year-old boy with pineal region tumor presented acutely with GCS 6 secondary to massive IVH. Imaging findings revealed presence of a tumor in the pineal region with massive IVH [Figure 2a]. Emergency endoscopic surgery was performed for clot evacuation, biopsy of the lesion and ETV. The ETV was done as pineal region tumors are commonly associated with obstruction at aqueduct of Sylvius. The surgical technique employed is similar to the one described above with minor modification for one of the two burr hole sites. The right frontal burr hole was made at lower point than usual, 4 cm above the eyebrow and 3 cm away from the midline to allow for a direct trajectory to the pineal region for tumoral biopsy. The second burr hole on the left side was made at the standard landmark for the ETV, 2 cm away from the midline and 1-2 cm in front of the coronal suture. The surgery was uneventful; however, few hours after surgery, tumoral rebleed occurred, necessitating a second endoscopic surgery. Prior to the second surgery, one ampoule of recombinant factor VIIa was administered. The hemorrhage was evacuated, and the previous ETV site was inspected and its stoma was recreated [Figure 2b]. The patient recovered well and was independent at 1 month after the surgery. Figure 2c depicts his CT brain after the second surgery. The histological diagnosis was an intermediate grade of pineoblastoma.
Figure 2.
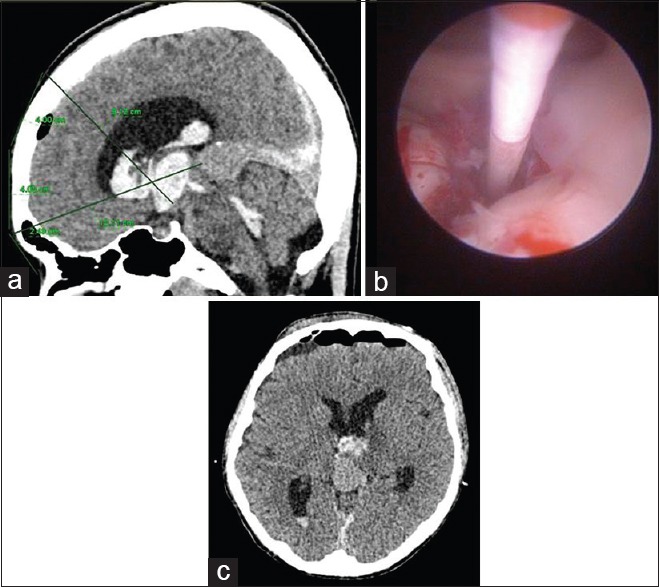
(a) Pineal region mass with massive intraventricular hemorrhage. (b) Endoscopic third ventriculostomy was reperformed after the second bleed. (c) Computed tomography brain after the second surgery
Case 3
A 58-year-old gentleman with a history of hypertension for 5 years, who defaulted treatment for 2 years, was found unconscious in the bathroom. His GCS on admission was 7 with normal flexion to pain for motor response. CT brain showed left basal ganglia ICB with IVH and hydrocephalus [Figure 3a]. He underwent standard endoscopic washout and ETV. In this and subsequent case, ETV was done as re-establishment of normal CSF pathway remained doubtful. In our opinion, once the floor of the third ventricle is well visualized, but the normal flow of CSF via the aqueduct cannot be ascertained, ETV should be performed using blunt technique. This procedure may reduce shunt dependency by creating a bypass for CSF flow and preventing obstructive hydrocephalus which may arise from a blockage along the aqueduct of Sylvius to the foramen of Magendie or Luschka. Postoperatively, he remained stable, and GCS improvement in motor response was noted. Repeat CT brain showed marked reduction in IVH [Figure 3b]. He was weaned off ventilation, and EVD was removed at postoperative day 4. He was discharged home well after 1 month of hospitalization.
Figure 3.
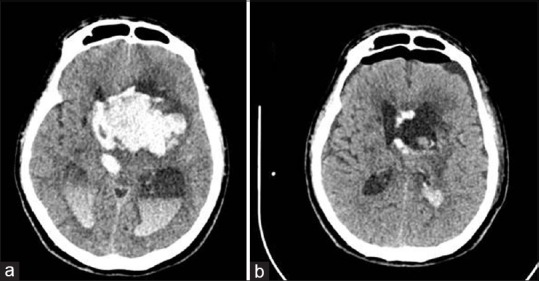
(a) Massive intraventriclar hemorrhage. (b) Computed tomography brain after endoscopic wash out and endoscopic third ventriculostomy
Case 4
A 49-year-old female with no previous comorbids, presented with intermittent headache for the past 2 months which was progressively worsening. She presented acutely with 5 episodes of vomiting and headache. On arrival, her initial GCS was 12, and subsequently dropped to 9. Her blood pressure was elevated at 198/100. The CT brain showed presence of IVH in bilateral lateral ventricles, third and fourth ventricles with hydrocephalus [Figure 4a and b]. She was subjected for emergency standard endoscopic washout, clot evacuation and ETV. On postoperative day 3, she was weaned off from the EVD catheter. Her GCS improved to obeying simple commands. Repeat CT brain showed minimal IVH without hydrocephalus [Figure 4c]. She was weaned off ventilation and management was directed toward the control of blood pressure and primary IVH.
Figure 4.
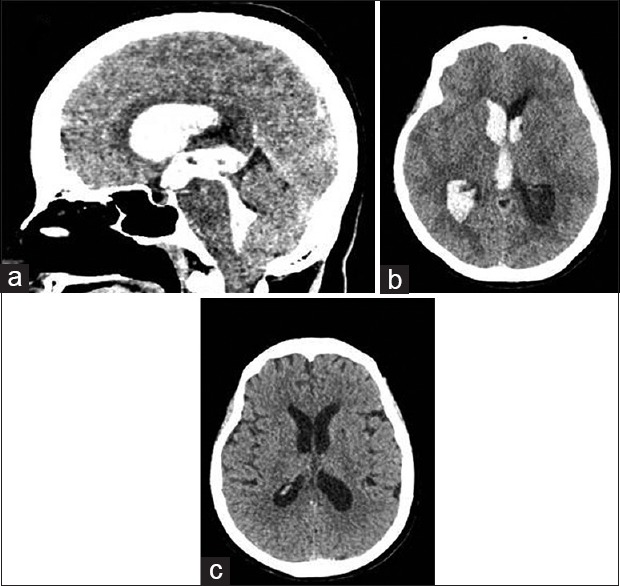
(a and b) Preoperative images of massive intraventricular hemorrhage. (c) Computed tomography brain after the surgery
Discussion
Intraventricular hemorrhage can be classified into primany or secondary. Primary IVH is confined to the ventricular system and immediate parenchymal lining of the ependymal cells. The bleed originates from an intraventricular source or from a lesion that is in close proximity with the ventricular system. Primary IVH consists of 30% of IVH, and some of the common causes are head injury, periventricular AVM and presence of coagulopathy. Secondary IVH, on the other hand, originates as an ‘extension’ of an intraparenchymal or subarachnoid hemorrhage into the ventricular system. These type of bleed consists of 70% of IVH.[2] Common causes of secondary IVH are hypertensive bleed (especially from the basal ganglia and thalamus), bleeding vascular malformations, hemorrhagic transformation of ischemic infarct and tumoral bleed.[2]
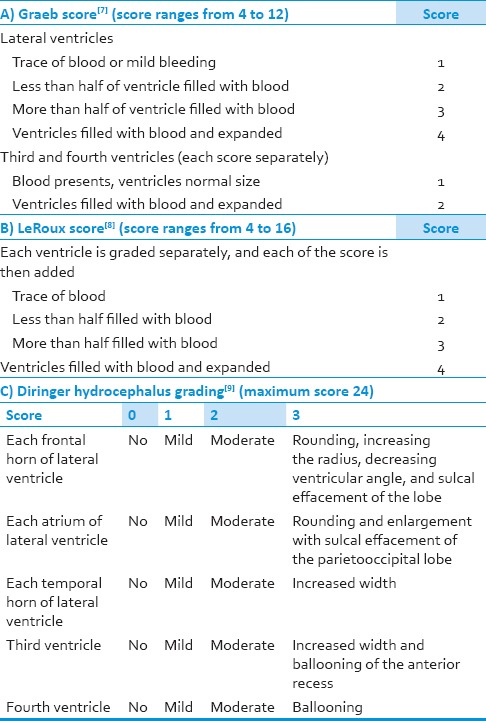
To grade the severity of IVH, there are two widely accepted classifications, Graeb grading and LeRoux grading. In Graeb grading of IVH, the score ranges from 4 to 12. The score is a product of the addition of the component scores that is given to the lateral ventricles, third and fourth ventricles [Table 1].[7] As for the LeRoux grading, all the ventricles are scored evenly, from 1 to 4 and then each of the component score is added to get the final score [Table 1].[8] To grade the severity of hydrocephalus, Diringer has formulated a scoring system which scores the lateral ventricle, third and fourth ventricles. As for the lateral ventricles, it is further scored in three different locations (frontal horn, atrium and temporal horn). Each of these components is given a score of 0-3, in which 0 represents normal shape of the ventricle, 1 indicates mild hydrocephalus, 2 for moderate and 3 when there is rounding of the ventricular wall or presence of sulcal effacement. The maximum score for the Diringer grading for hydrocephalus is 24 [Table 1].[9]
Table 1.
Intraventricular hemorrhage and its grading's
With regards to prognosis of massive IVH (Graeb > 6), the mortality rate is almost 50-75% with conservative management.[2,3,10] Many studies done previously showed that the volume of IVH, presence of hydrocephalus, patients age and admitting GCS score are among the predictors for mortality and functional outcome.[5,6,11,12] IVH contributes to morbidity in three main ways. First, hemorrhage in the ventricular system leads to blockage of ventricular conduits, producing acute hydrocephalus. If left untreated, acute hydrocephalus leads to elevation of ICP and progression to death. At present, the management of IVH with hydrocephalus involves placements of one or more EVD for drainage of blood and cerebrospinal fluid (CSF) from the ventricular system and normalization of ICP with or without intraventricular fibrinolysis.[13,14] However, this approach alone may not be sufficient in improving the outcomes of patients with IVH. One of the common problems encountered is maintaining the patency of the ventricular catheters. The catheters are easily blocked by blood clots defeating its purpose in draining the IVH and measuring the ICP. Even when drainage is successful, frequent administration of medication intraventricularly or persistent presence of the blood clot increases the duration for drainage, thus increasing the risk of ventriculitis; which is estimated to be about 50%, in the presence of IVH.[15,16]
Besides raised ICP, direct mass effect from the IVH may be another contributing pathophysiologic event that determines prognosis independent of ICP elevation. The prolonged presence of clots deep within the brain is related to ventriculomegaly, brain edema and inflammatory responses. EVD does not consistently improve either event, as drainage alone does not alter the size of ventricles, brain tissue edema, and the inflammation provoked by the presence of intraventricular blood, and neither does increase the rate of blood clot resolution.[11,17] Furthermore, placement of a drain is frequently complicated by infection such as meningitis and ventriculitis which also contributes to the formation of new hemorrhage. Communicating and obstructive hydrocephalus is another common complication of IVH.[18,19] Blood degradation products of IVH will flow via CSF pathways to the cisterns and arachnoid granulations. Prolonged contact of the blood breakdown products with the pathways, cisternal surfaces and arachnoid granulations leads to an inflammatory response that permanently scars the granulations and pathways and may alter the cisterns which leads to delayed development of communicating or obstructive hydrocephalus. When this condition occurs, treatment involves the placement of a shunt for CSF diversion.
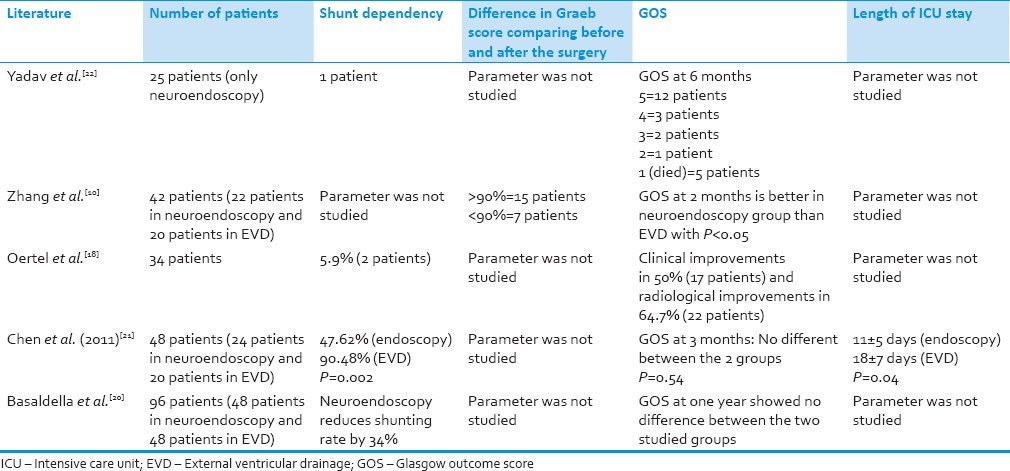
To address these issues, additional measures such as performing ETV and endoscopic washout for patients with IVH prior to the placement of EVD have been studied [Table 2]. Many previous studies have highlighted the benefits of endoscopic wash out for IVH.[10,18,20,21,22,23] In 2009, Oertel et al., did a study to define the role of endoscopy and ETV in IVH, and found that ETV represents a safe treatment option in IVH-related obstructive hydrocephalus yielding similar results to an EVD, but less risk of infection and very low subsequent shunt placement rate.[18] In a recent study conducted by Chen et al., comparing the use of endoscopic washout versus EVD for IVH, he concluded that there is a significant difference in the length of ICU stay in patients who underwent endoscopic washout and EVD (11 ± 5 days) when compared to those who only underwent EVD (18 ± 7 days), with P = 0.04. In this study, there was also a significant reduction in shunt dependency rate between the two groups, P = 0.002.[21] In another paper published by Zhang et al., they found that patients who underwent endoscopic washout for IVH had significantly good recovery at 2 months after hemorrhage compared to those patients who underwent EVD.[10] In 2007, Yadav and coworkers published their findings in which there was a resolution of hydrocephalus in 96% of patients who were treated via endoscopic washout.[23] Recently in 2012, Basaldella et al. studied 96 patients with IVH and found neuroendoscopy reduced the shunting rate by 34%.[20] These studies demonstrate the benefits of endoscopic washout in managing patients with IVH. Nonetheless, one must be mindful of the disadvantages associated with endoscopy and IVH. First, endoscopic management is normally incapable of treating the source of hemorrhage, requiring further investigation and management. Second, the endoscopic procedure itself is associated with a limited visual field and depth perception in massive IVH and difficulty in securing hemostasis. This was highlighted in case 2 where rebleed occurred after the first endoscopic surgery. Recombinant factor VIIa may be helpful in this scenario as well as in cases with known vascular malformation that cause massive IVH as reported in case 1. Finally, sudden increase in ICP during endoscopic washout is a threat to patients. Our common practice to overcome this problem are: (a) 2-burr holes technique with one EVD catheter left in situ inside the lateral ventricle to ensure optimal drainage of excess irrigation fluid and another burr hole for surgery (change alternately between EVD and endoscope); (b) monitoring the ICP continuously throughout the procedure via 2-ways outlet channel; cases with prolonged elevation in ICP and altered vital signs should have shorter intervention time; (c) since IVH is associated with neuro-inflammation which can lead to brain edema and raised ICP, administration of steroids for a short duration perioperatively is commonly practiced (note: The EVD is normally removed within 3-4 days after the surgery to avoid infectious complications). In order to perform endoscopic washout as a standard procedure for IVH, clinical trials are needed and more importantly, the neurosurgeons must be properly trained in neuroendoscopy to qualify him or herself managing massive IVH cases.
Table 2.
Literature review on endoscopic management of patients with intraventricular hemorrhage and its outcomes
Conclusions
The management of patients with massive IVH via endoscopic washout has various proven benefits such as reduced shunt dependency, lesser morbidity and mortality as compared to standard EVD. However, disadvantages such as limited visual field, difficulty in securing hemostasis and persistent increase in ICP may still be present. Therefore, the use of neuroendoscopy has to be coupled with skillful surgical techniques and thorough knowledge of this procedure.
Footnotes
Source of Support: Nil
Conflict of Interest: None declared.
References
- 1.Sahni R, Weinberger J. Management of intracerebral hemorrhage. Vasc Health Risk Manag. 2007;3:701–9. [PMC free article] [PubMed] [Google Scholar]
- 2.Hanley DF, Naff NJ, Harris DM. Intraventricular hemorrhage: Presentation and management options. Semin Cerebrovasc Dis Stroke. 2005;5:209–16. [Google Scholar]
- 3.Chen CC, Lin HL, Cho DY. Endoscopic surgery for thalamic hemorrhage: A technical note. Surg Neurol. 2007;68:438–42. doi: 10.1016/j.surneu.2006.11.054. [DOI] [PubMed] [Google Scholar]
- 4.Hsieh PC. Endoscopic removal of thalamic hematoma: A technical note. Minim Invasive Neurosurg. 2003;46:369–71. doi: 10.1055/s-2003-812505. [DOI] [PubMed] [Google Scholar]
- 5.Karnik R, Valentin A, Ammerer HP, Hochfelner A, Donath P, Slany J. Outcome in patients with intracerebral hemorrhage: Predictors of survival. Wien Klin Wochenschr. 2000;112:169–73. [PubMed] [Google Scholar]
- 6.Tuhrim S, Horowitz DR, Sacher M, Godbold JH. Volume of ventricular blood is an important determinant of outcome in supratentorial intracerebral hemorrhage. Crit Care Med. 1999;27:617–21. doi: 10.1097/00003246-199903000-00045. [DOI] [PubMed] [Google Scholar]
- 7.Graeb DA, Robertson WD, Lapointe JS, Nugent RA, Harrison PB. Computed tomographic diagnosis of intraventricular hemorrhage. Etiology and prognosis. Radiology. 1982;143:91–6. doi: 10.1148/radiology.143.1.6977795. [DOI] [PubMed] [Google Scholar]
- 8.LeRoux PD, Haglund MM, Newell DW, Grady MS, Winn HR. Intraventricular hemorrhage in blunt head trauma: An analysis of 43 cases. Neurosurgery. 1992;31:678–84. doi: 10.1227/00006123-199210000-00010. [DOI] [PubMed] [Google Scholar]
- 9.Diringer MN, Edwards DF, Zazulia AR. Hydrocephalus: A previously unrecognized predictor of poor outcome from supratentorial intracerebral hemorrhage. Stroke. 1998;29:1352–7. doi: 10.1161/01.str.29.7.1352. [DOI] [PubMed] [Google Scholar]
- 10.Zhang Z, Li X, Liu Y, Shao Y, Xu S, Yang Y. Application of neuroendoscopy in the treatment of intraventricular hemorrhage. Cerebrovasc Dis. 2007;24:91–6. doi: 10.1159/000103122. [DOI] [PubMed] [Google Scholar]
- 11.Young WB, Lee KP, Pessin MS, Kwan ES, Rand WM, Caplan LR. Prognostic significance of ventricular blood in supratentorial hemorrhage: A volumetric study. Neurology. 1990;40:616–9. doi: 10.1212/wnl.40.4.616. [DOI] [PubMed] [Google Scholar]
- 12.Martí-Fàbregas J, Piles S, Guardia E, Martí-Vilalta JL. Spontaneous primary intraventricular hemorrhage: Clinical data, etiology and outcome. J Neurol. 1999;246:287–91. doi: 10.1007/s004150050348. [DOI] [PubMed] [Google Scholar]
- 13.Gaberel T, Magheru C, Parienti JJ, Huttner HB, Vivien D, Emery E. Intraventricular fibrinolysis versus external ventricular drainage alone in intraventricular hemorrhage: A meta-analysis. Stroke. 2011;42:2776–81. doi: 10.1161/STROKEAHA.111.615724. [DOI] [PubMed] [Google Scholar]
- 14.Naff NJ, Carhuapoma JR, Williams MA, Bhardwaj A, Ulatowski JA, Bederson J, et al. Treatment of intraventricular hemorrhage with urokinase: Effects on 30-Day survival. Stroke. 2000;31:841–7. doi: 10.1161/01.str.31.4.841. [DOI] [PubMed] [Google Scholar]
- 15.Hasan D, Vermeulen M, Wijdicks EF, Hijdra A, van Gijn J. Management problems in acute hydrocephalus after subarachnoid hemorrhage. Stroke. 1989;20:747–53. doi: 10.1161/01.str.20.6.747. [DOI] [PubMed] [Google Scholar]
- 16.Pfausler B, Beer R, Engelhardt K, Kemmler G, Mohsenipour I, Schmutzhard E. Cell index – a new parameter for the early diagnosis of ventriculostomy (external ventricular drainage)-related ventriculitis in patients with intraventricular hemorrhage? Acta Neurochir (Wien) 2004;146:477–81. doi: 10.1007/s00701-004-0258-8. [DOI] [PubMed] [Google Scholar]
- 17.Adams RE, Diringer MN. Response to external ventricular drainage in spontaneous intracerebral hemorrhage with hydrocephalus. Neurology. 1998;50:519–23. doi: 10.1212/wnl.50.2.519. [DOI] [PubMed] [Google Scholar]
- 18.Oertel JM, Mondorf Y, Baldauf J, Schroeder HW, Gaab MR. Endoscopic third ventriculostomy for obstructive hydrocephalus due to intracranial hemorrhage with intraventricular extension. J Neurosurg. 2009;111:1119–26. doi: 10.3171/2009.4.JNS081149. [DOI] [PubMed] [Google Scholar]
- 19.Burstein J, Papile LA, Burstein R. Intraventricular hemorrhage and hydrocephalus in premature newborns: A prospective study with CT. AJR Am J Roentgenol. 1979;132:631–5. doi: 10.2214/ajr.132.4.631. [DOI] [PubMed] [Google Scholar]
- 20.Basaldella L, Marton E, Fiorindi A, Scarpa B, Badreddine H, Longatti P. External ventricular drainage alone versus endoscopic surgery for severe intraventricular hemorrhage: A comparative retrospective analysis on outcome and shunt dependency. Neurosurg Focus. 2012;32:E4. doi: 10.3171/2012.1.FOCUS11349. [DOI] [PubMed] [Google Scholar]
- 21.Chen CC, Liu CL, Tung YN, Lee HC, Chuang HC, Lin SZ, et al. Endoscopic surgery for intraventricular hemorrhage (IVH) caused by thalamic hemorrhage: Comparisons of endoscopic surgery and external ventricular drainage (EVD) surgery. World Neurosurg. 2011;75:264–8. doi: 10.1016/j.wneu.2010.07.041. [DOI] [PubMed] [Google Scholar]
- 22.Longatti P, Fiorindi A, Martinuzzi A. Neuroendoscopic aspiration of hematocephalus totalis: Technical note. Neurosurgery. 2005;57:E409. doi: 10.1227/01.neu.0000176702.26810.b7. [DOI] [PubMed] [Google Scholar]
- 23.Yadav YR, Mukerji G, Shenoy R, Basoor A, Jain G, Nelson A. Endoscopic management of hypertensive intraventricular haemorrhage with obstructive hydrocephalus. BMC Neurol. 2007;7:9. doi: 10.1186/1471-2377-7-1. [DOI] [PMC free article] [PubMed] [Google Scholar]


