Abstract
Background.
Recent reports of Plasmodium vivax associated with severe syndromes and mortality from malaria endemic areas questions the “benign” course of non-falciparum malarias.
Methods.
We retrospectively analyzed data from patients reported to the US Centers for Disease Control and Prevention with a diagnosis of malaria parasite single-species infection between 1985 and 2011. Patients classified as having severe illness were further classified according to outcome (survival versus death) and clinical syndrome.
Results.
Among all cases, .9% of Plasmodium falciparum cases resulted in death and 9.3% were classified as severe, whereas .09% of P. vivax cases resulted in death and 1.3% were classified as severe. The odds ratios for severe illness among 15 272 diagnoses of P. falciparum relative to patients diagnosed with P. vivax (n = 12 152), Plasmodium malariae (n = 1254), or Plasmodium ovale (n = 903) was 7.5, 5.7, and 5.0, respectively (P < .0001 for all); in contrast, the corresponding odds ratios for death among those severely ill was 1.6, 1.1, and .8 (P > .1 for all), respectively. Compared with P. vivax (n = 163), the odds of P. falciparum cases classified as severely ill (n = 1416) were 1.9 (P = .0006), .5 (P = .001), and 1.3 times (P = .1) as likely to present as cerebral, acute respiratory distress, and renal syndromes, respectively.
Conclusions.
Although less common, patients presenting with non-falciparum even in the United States can develop severe illness, and severe illness in patients having malaria of any species threatens life.
Keywords: morbidity, mortality, Plasmodium, species, USA
The medical, scientific, and public health communities have long considered a diagnosis of malaria caused by Plasmodium vivax, Plasmodium malariae, or Plasmodium ovale rarely threatening to life. In contrast, Plasmodium falciparum is acknowledged as often life threatening. This difference is the basis of the widely applied clinical division of falciparum and non-falciparum malarias as representing malignant versus benign malarias. That dichotomy effectively defines not only how the global malaria problem is perceived and managed [1], but also influences strategies of prevention and treatment in individual patients. The perception of P. vivax as generally harmless with respect to mortality risk effectively fostered its neglect in research and public health for over six decades [2], despite heavy burdens of risk and disease [3].
Over the past decade, case reports and hospital- and village-based studies in endemic zones demonstrate that P. vivax often results in serious illness and fatal outcomes. Summarizing these studies, approximately 10%–20% of admissions with a primary diagnosis of P. falciparum or P. vivax were classified as severe, and approximately 5%–15% of those did not survive [1]. Both species appeared to be associated with significant risk of severe morbidity and mortality among hospitalized patients. Falciparum and vivax malarias were equally threatening to children in one village in Papua New Guinea [4] and to pregnant women and their fetuses in Indonesia and Thailand [5–7]. Historic experience with malaria therapy applying P. vivax in otherwise relatively healthy neurosyphilis patients resulted in a 5%–15% risk of mortality, and this result also refuted its benign identity [1].
In 2011, the largest number of cases of malaria since 1971 was reported to the US Centers for Disease Control and Prevention (CDC) from healthcare providers within the United States. Despite the apparent progress in reducing the number of malaria cases in regions that are endemic for malaria, international travel seems to be growing steadily, and use of appropriate prevention measures by travelers remains inadequate [8]. Plasmodium falciparum and P. vivax overwhelmingly dominate estimated global populations at risk and likely case burdens [3, 9–11]. The relative proportion among cases reported to the CDC reflect the global codominance of these two species over the other two, P. malariae and P. ovale [8].
Malaria incidence among travelers may be quite distinct compared with patients living in endemic zones in very important clinical, epidemiological, demographic, and socioeconomic respects [12, 13]. Nonetheless, malaria among travelers and endemic country residents share the same etiologic agents and their innate biological characteristics, including pathogenicity. Lacking the naturally acquired partial immunity to acute malaria common in endemic zones, nonimmune travelers who contract malaria for the first time are less likely to be protected from the innate pathogenic effects of the plasmodium species. On the other hand, travelers usually have greater access to higher levels of care compared with residents of endemic zones, but they may experience delays in malaria diagnosis and treatment due to the relative rarity of the infection.
After acknowledging these important confounding factors with regard to clinical consequence of malaria infection, we undertook an assessment of severe morbidity and mortality in travelers diagnosed with malaria during the 27 years from 1985 to 2011 in the United States. In particular, we aimed at evaluating the association between vivax malaria and severe illness and malaria mortality.
METHODS
Deidentified data from the US National Malaria Surveillance System (NMSS) from 1985 to 2011 were used in this analysis and have been described elsewhere [8]. In brief, malaria cases diagnosed by blood film, polymerase chain reaction, or rapid diagnostic tests are mandated to be reported to local and state health departments by healthcare providers or laboratory staff. Case investigations are conducted by local and state health departments, and reports are transmitted to CDC through the NMSS or through direct CDC consults. Malaria cases were categorized by infecting species: P. falciparum, P. vivax, P. malariae, and P. ovale. Cases infected with more than one species, typically referred to as mixed infections, were rare and not included in this analysis. In addition, cases where the species was not reported were not included in the main analyses. The zoonotic malaria from Southeast Asian macaques caused by Plasmodium knowlesi [15] has been reported only once in a returning traveler in the United States [16] and is not considered in the current analysis. Although species confirmatory services are available at the state and federal levels, most cases are not confirmed and species confirmation is usually only sought in nonplausible scenarios, eg, P. vivax acquired in Haiti.
Cases of severe malaria were defined as those with one or more of the following manifestations: death, neurologic symptoms, renal failure, severe anemia, acute respiratory distress syndrome (ARDS), jaundice, or ≥5% parasitemia. To attempt to include severe cases in which clinical criteria were not reported, persons who received a regimen recommended for the treatment of severe malaria (ie, parenteral artesunate or quinidine, and/or an exchange blood transfusion) despite having no specific severe manifestations reported also were counted as a severe case in this analysis. Neurologic symptoms or cerebral complications were defined as impaired consciousness, coma, or repeated generalized convulsions. For surveillance purposes, severe illness is categorized by CDC and not by the treating physician. In 2008, CDC revised the case report form to collect information on parasitemia density, treatment with artesunate, quinine or quinidine, and modified the anemia classification [17]. The anemia complication before 2008 was for those with hemoglobin (Hb) < 11 g/dL and hematocrit < 33%. Beginning in 2008, severe anemia was defined as Hb < 7 g/dL to be in line with the World Health Organization definition for severe cases. Beginning in 2010, CDC established a partnership with the Armed Forces Health Surveillance Center to identify additional cases occurring among military personnel that might not have been identified previously by local or state health departments or private healthcare providers [18]. Odds ratios (ORs) and 95% confidence intervals (CIs) were calculated using SAS version 9.3 (Cary, NC). P values < .05 were considered statistically significant.
With the varying number of cases with unknown species, a sensitivity analysis was performed to assess the impact of possible misclassification of these cases on the prevalence of severe malaria. The percentage of cases that were classified as being severe was recalculated for P. falciparum and for P. vivax under the assumption that all of the unknown cases were severe P. falciparum or P. vivax, respectively, to determine the range of possible ORs. In addition, to assess the differential impact of hyperparasitemia (>5%), which more commonly occurs with P. falciparum, the analyses were repeated excluding cases of severe malaria due to hyperparasitemia alone without another accompanying severe clinical syndrome.
RESULTS
All Cases: 1985–2011
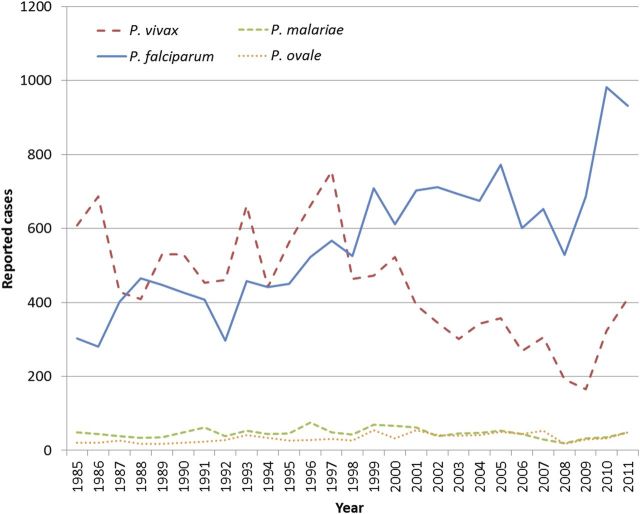
Figure 1 illustrates the total annual number of cases of malaria by species (excluding the 226 mixed infections and 5408 species not reported; averaging 8 and 200 cases per year, respectively) from 1985 through 2011. During 1985–1998, P. falciparum and P. vivax contributed roughly equal halves to total case numbers. However, since 1999, P. falciparum cases have increased substantially relative to P. vivax. Of note, the number of cases with no species data also steadily increased during this period (range, 50–575). During 2010 and 2011, reported cases of both species increased substantially due to a concerted effort to improve the completeness of reporting. Numbers of reported cases involving the two minority species, P. malariae and P. ovale, and mixed infections remained relatively constant over these years.
Figure 1.
Annual number of cases of malaria reported in the United States from 1985 to 2011, by species.
Table 1 lists the numbers of cases classified as severe versus not for P. falciparum and P. vivax over the period 1985–2011. Of all cases, 122 of 13 971 (0.9%) of P. falciparum cases resulted in death compared with 10 of 11 456 (0.09%) of P. vivax cases (OR = 10.08; 95% CI = 5.29–19.22; P < .0001), and a diagnosis of P. falciparum was more likely to be classified as severe relative to that of P. vivax (OR = 7.5; 95% CI = 6.33–8.79; P < .0001). In all, 9.3% of P. falciparum patients versus 1.3% of P. vivax patients were classified as severely ill. Risk of death amongst the severely ill (8.6% vs 6.1%, respectively) was not significantly different (OR = 1.6; 95% CI = .82–3.12; P = .1649) between P. falciparum and P. vivax. Patients with a diagnosis of P. falciparum were slightly more likely to have cerebral complications compared with patients with a diagnosis of P. vivax (OR = 1.9; 95% CI = 1.32–2.84; P = .0006). In contrast, severely ill patients with a diagnosis of P. falciparum were less likely to experience ARDS (OR = .5; 95% CI = .35–.78; P = .0013) compared with patients with P. vivax. There was no statistical difference for renal complications (OR = 1.3; 95% CI = .93–1.88; P = .1235).
Table 1.
Severe Morbidity and Mortality With a Diagnosis of Plasmodium falciparum Relative to That of Plasmodium vivax, Plasmodium malariae, and Plasmodium ovale Among Travelers Presenting in the United States, 1985–2011
| Syndrome* |
P. falciparum |
P. vivax |
P. falciparum vs P. vivax |
||||||
|---|---|---|---|---|---|---|---|---|---|
| Syndrome (+) | Syndrome (−) | % | Syndrome (+) | Syndrome (−) | % | OR | 95% CI | P value | |
| Fatal among all cases† | 122 | 13 849 | 0.9% | 10 | 11 446 | 0.09% | 10.08 | 5.29, 19.22 | <.0001 |
| Severe | 1416 | 13 856 | 9.3% | 163 | 11 989 | 1.3% | 7.46 | 6.33, 8.79 | <.0001 |
| Fatal† | 122 | 1091 | 8.6% | 10 | 143 | 6.1% | 1.60 | .82, 3.12 | .1649 |
| Cerebral malaria | 514 | 902 | 36.3% | 37 | 126 | 22.7% | 1.94 | 1.32, 2.84 | .0006 |
| ARDS | 176 | 1240 | 12.4% | 35 | 128 | 21.5% | .52 | .35, .78 | .0013 |
| Renal | 503 | 913 | 35.5% | 48 | 115 | 29.4% | 1.32 | .93, 1.88 | .1235 |
| Severe anemia‡ | 140 | 434 | 24.4% | 16 | 45 | 26.2% | .91 | .50, 1.66 | .7512 |
| Syndrome* |
P. malariae |
P. falciparum vs P. malariae |
P. ovale |
P. falciparum vs P. ovale |
||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Syndrome (+) | Syndrome (−) | % | OR | 95% CI | P Value | Syndrome (+) | Syndrome (−) | % | OR | 95% CI | P Value | |
| Fatal among all cases† | 2 | 1195 | 0.2% | 5.26 | 1.30, 21.31 | .0092 | 2 | 807 | 0.3% | 3.55 | .88, 14.40 | .0577 |
| Severe | 22 | 1232 | 1.8% | 5.72 | 3.74, 8.75 | <.0001 | 18 | 885 | 2.0% | 5.02 | 3.14, 8.04 | <.0001 |
| Fatal† | 2 | 20 | 9.1% | 1.12 | .26, 4.84 | .8812 | 2 | 15 | 11.8% | .84 | .19, 3.71 | .8165 |
| Cerebral malaria | 4 | 18 | 18.2% | 2.56 | .86, 7.62 | .0791 | 2 | 16 | 11.1% | 4.56 | 1.04, 19.91 | .0270 |
| ARDS | 1 | 21 | 4.5% | 1.25 | .40, 22.30 | .2642 | 2 | 16 | 11.1% | 1.14 | .26, 4.98 | .8662 |
| Renal | 6 | 16 | 27.3% | 1.47 | .57, 3.78 | .4221 | 7 | 11 | 38.9% | .87 | .33, 2.25 | .7670 |
| Severe anemia‡ | 6 | 5 | 54.5% | .27 | .08, .89 | .0222 | 3 | 7 | 30.0% | .75 | .19, 2.95 | .6828 |
Abbreviations: ARDS, acute respiratory distress syndrome; CI, confidence interval; OR, odds ratio.
*The total number of cases do not sum to the total number of severe cases due to missing outcome data.
†Cases could have more than one syndrome or clinical complication in the course of their illness, thus the number of cases that are syndrome (+) do not sum to the total classified as severe. Analyses of individual syndromes are among those who are classified as having severe disease.
‡2008–2011 reporting only.
Excluding cases classified as severe based on hyperparasitemia alone from the analysis did not significantly change the findings that P. falciparum was more likely to cause severe disease compared to P. vivax (OR = 1.9; 95% CI = 1.32–2.84; P = .0006). Likewise, the risk of death (OR = 1.6; 95% CI = .82–3.13; P = .1619), cerebral malaria (OR = 1.97; 95% CI = 1.34–2.88; P = .0004), ARDS (OR = .52; 95% CI = .35–.78; P = .0015), and renal failure (OR = 1.34; 95% CI = .94–1.90; P = .1098) in those with severe illness remained essentially unchanged.
Table 1 also lists the same statistical summaries for diagnoses of P. malariae and P. ovale compared with P. falciparum. Similarly, P. malariae and P. ovale resulted in fewer deaths overall than P. falciparum. Apart from risk of a classification of severe disease for falciparum malaria relative to these malarias (OR = 5.7; 95% CI = 3.74–8.75; P < .0001 for P. falciparum vs P. malariae and OR = 5.0; 95% CI = 3.14–8.04; P < .0001 for P. falciparum vs P. ovale), no significant differences appeared for risk of death, cerebral malaria, ARDS, or renal failure for either species diagnosis. The relatively few cases of these species (n = 1254 for P. malariae and n = 903 for P. ovale), along with relatively low frequencies of severe illness (1.8% and 2.0%), resulted in poor statistical power to analyze relative risk of specific severe disease syndromes. Nonetheless, the OR for death among cases with severe illness for P. falciparum relative to that of P. malariae or P. ovale were 1.1 (95% CI = .26–4.84; P = .8812) and .8 (95% CI = .19–3.71, P = .8165), respectively.
Cases 2008–2011
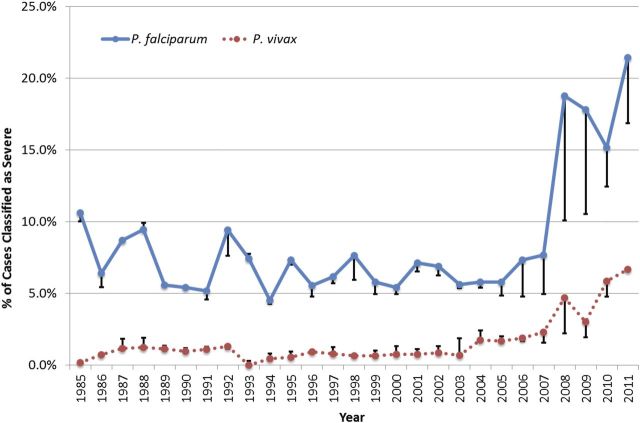
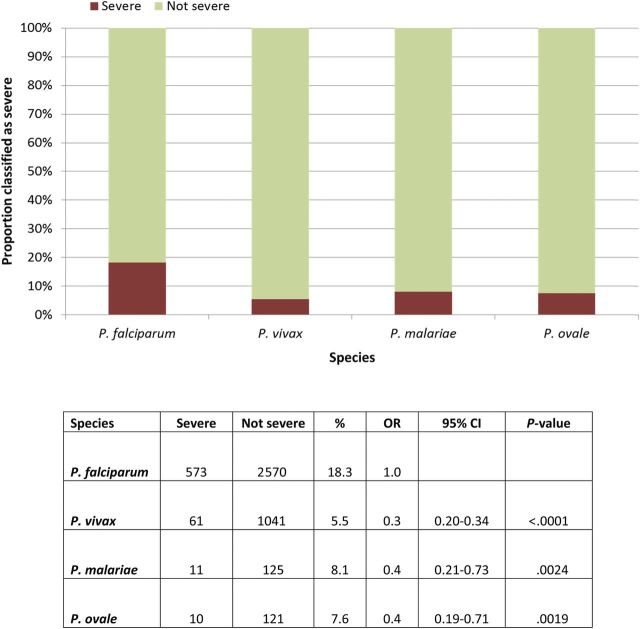
Limiting the analysis to the time period using current surveillance forms and improved reporting completeness, the years 2008–2011 were analyzed separately. Figure 2 illustrates the annual proportion of patients classified as having severe illness with a diagnosis of P. falciparum or P. vivax. Before 2008, fewer than 10% and 5% of cases having these respective diagnoses were classified as severely ill. Since that year, these rates rose sharply to 21% and 7%, respectively, by 2011. This sharp rise, although less notable, nonetheless persists if P. vivax or P. falciparum were imputed with equal probability in all cases without a species diagnosis. Figure 3 illustrates the relative proportions of severe illness among cases of falciparum and the other species [P. vivax (OR = .3; 95% CI = .20–.34; P < .0001); P. malariae (OR = .4; 95% CI = .21–.73; P = .0024); P. ovale (OR = .4; 95% CI = .19–.71; P = .0019)] during this period. Data for severe anemia were only available from 2008 to 2011. There was no statistical difference for severe anemia among patients classified as severely ill with a diagnosis of falciparum versus vivax (OR = .9; 95% CI = .50–1.66; P = .7512) or ovale malaria (OR = .75; 95% CI = .19–2.95; P = .6828) (Table 1). Severe anemia amongst those severely ill was less common with P. falciparum than P. malariae (OR = .3; 95% CI = .08–.89; P = .0222).
Figure 2.
Percentage of Plasmodium falciparum and P. vivax cases reported in the United States classified as severe, 1985–2011. Bars above and below points represent the error bars of a sensitivity analysis where cases of unknown species daignosis were counted as either all P. falciparum or all P. vivax.
Figure 3.
Proportion of all cases between 2008 and 2011 classified as severe, and the odds ratio for that classification in Plasmodium falciparum compared with P. vivax, P. malariae, or P. ovale.
Severe Illness OR Trend, 1985–2011
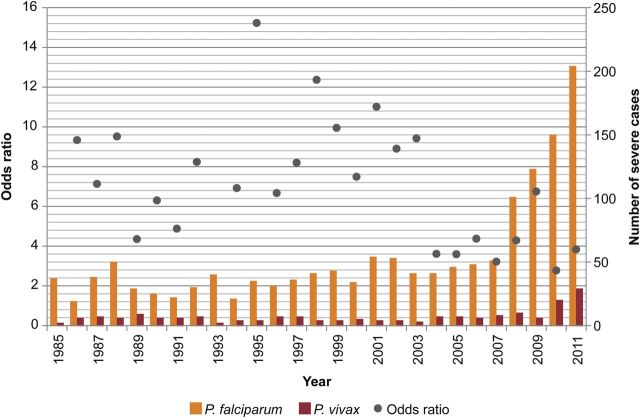
Figure 4 illustrates the OR for severe illness with a diagnosis of P. falciparum relative to P. vivax for each year from 1985 to 2011 as well as the number of severe cases. Excluding rare outliers, during 1985 to 2003, the annual ORs ranged from approximately four to 15, but during 2004 to 2011 this number typically ranged between three and four. Although the OR has trended downwards, these trends should be interpreted with caution because reporting format and quality varied during this time period.
Figure 4.
Annual number of severe Plasmodium falciparum and Plasmodium vivax cases, and odds ratio for a classification of severe illness with diagnosis of P. falciparum compared with P. vivax among cases in the United States between 1985 and 2011. The odds ratio for 1985 and 1993 were 82.2 and 61.3, respectively, and are not plotted.
DISCUSSION
P. vivax has been increasingly reported as an important cause of severe disease syndromes. When we analyzed all 29 489 single-species cases reported to the US CDC from 1985 to 2011, P. falciparum was ten times as likely to cause death and seven times as likely to be classified as severe compared with P. vivax. However, among the severely ill, the odds of death were not significantly different (8.6% and 6.1%; P = .1649) between the two species. Moreover, the odds of being classified as severely ill with a diagnosis of P. falciparum relative to P. vivax during 2008–2011, a period of improved data quality and reporting coverage, fell sharply to an OR of 3.8 (P < .0001). In 2008, the US CDC began applying substantially modified case detection, reporting, and illness classification algorithms (see METHODS). We consider those changes very probably responsible for the sharp increases in the proportion of reported cases classified as severe (see Figure 2). The greater sensitivity of the now current methods for detection and classification of severe illness, however, applied equally to all species of diagnosis. Therefore, these changes to surveillance methods were unlikely to have confounded the measured risks of severe illness with a diagnosis of one species relative to another in any given year between 2008 and 2011. Comparisons of trends across years, especially before and after 2008, should be interpreted with caution. We do not conclude, for example, the rising odds of severe illness in P. vivax relative to P. falciparum as evidence of increasingly threatening parasite behavior.
The downward trend in OR for risk of severe illness with P. falciparum versus P. vivax (see Figure 4) seems to be driven by other factors. These factors could include changes to the reporting form as well as increased provider awareness of the severe complications possible with P. vivax illness. Although rising resistance to chloroquine by P. vivax may also be a factor [19], this phenomenon has not been seen thus far as a common problem in the management of post-travel vivax malaria in the United States. Furthermore, pernicious virulence in P. vivax had been evident in neurosyphilis patients treated with repeated severe paroxysms of vivax malaria during the 1920s and 1930s [1]. The character and frequency of severe morbidity and mortality in those patients was essentially similar to that in hospitalized patients in endemic zones today—typically manifest as severe anemia, pulmonary distress, altered consciousness, renal and hepatic dysfunction, and circulatory collapse. Similar clinical syndromes were observed in patients diagnosed with P. vivax and treated in US hospitals during 1985–2011.
Although the reported rates of deaths and severe illness with a diagnosis of P. vivax or P. falciparum were substantially lower in travelers in the United States than those typically reported from hospitals in malaria endemic areas [1], case fatality rates amongst those with severe illness were comparable. Those rates for P. falciparum and P. vivax were 9.3% and 1.3% for severe illness, and among those with severe illness, 8.6% and 6.1% case fatality, respectively. Although from a wide range of settings with differing data, quality rates of severe illness with these two diagnoses from hospitals in malaria endemic areas widely ranged between approximately 10% and 30% of admissions, and case fatality was typically between 5% and 15% [1]. The higher rates of severe illness observed in malaria endemic areas can likely be attributed to higher probabilities of comorbidities and the multitude of other factors associated with the poverty typical of most heavily malarious zones: endemic infections, geographic isolation and limited access to care, reluctance to seek expensive care, poor nutrition, etc. Although comorbidities can be present among international travelers, they typically represent a relatively healthy and wealthy subpopulation. These distinctions did not appear to impact the risk of death after the onset of severe complications.
Vivax malaria has long been thought to be a relatively benign condition, resulting in the occasional infarct or rupture of the spleen. Like many reports from endemic areas over the past decade [1, 2, 20–27], the findings in this analysis of malaria cases diagnosed and treated in the United States, especially in the period 2008–2011, challenge the dogma of vivax malaria being relatively harmless and not causing serious complications such as severe anemia, respiratory distress, shock, altered consciousness, and other syndromes typically linked to falciparum malaria. Although a diagnosis of P. falciparum carried a higher risk of severe illness relative to that of P. vivax (18.3% vs 5.5%; P < .0001) during 2008–2011, the risk of severe illness with vivax malaria should not be considered rare or limited to injury to the spleen. Although less likely to run a malignant course than P. falciparum, P. vivax also seems to be an inherently pernicious species capable of a fatal course in travelers and residents of endemic areas alike.
As with all routine surveillance systems, limitations around underreporting, data accuracy, and completeness exist in the NMSS [28]. However, these limitations are not species specific. It is possible that frontline clinicians define severity differently or could have a differential perception of severity by species, but severe illness and specific syndrome classification is completed by the CDC based on the provided information. Although most syndromes are not species specific, hyperparasitemia, defined as >5%, is more relevant and common for P. falciparum. However, the sensitivity analysis excluding hyperparasitemia alone as qualifying as severe illness showed that the initial results were robust. Although the inclusion of the use of parenteral artesunate or quinidine alone without other manifestations in the classification of severe illness could have overestimated the overall number of severe illness, this bias is not species specific and is not likely to impact the comparative results.
Authoritative guidances from the US CDC and others have recommended prophylactic antimalarials for prevention of malaria in travelers [29–31]. This strategy mostly aims at killing blood stages of the plasmodia as they emerge into the bloodstream, and these drugs (doxycycline, chloroquine, and mefloquine) have no effect on the latent liver stages of P. vivax responsible for repeated clinical attacks in the many months (up to about two years) following primary infection. Suppressive prophylaxis coupled with immediate post-travel presumptive anti-relapse therapy with primaquine is recommended for persons departing from vivax and ovale endemic areas after a period of extended exposure. In addition, the US CDC now recommends the use of primaquine for causal prophylaxis (a daily dose of 30 mg (approximately 0.5 mg/kg for the average adult) commencing with travel and ceasing seven days following travel to vivax-endemic areas) [29], which may currently be the only reliably effective chemoprophylaxis option for preventing both primary and secondary attacks by P. vivax [32–34].
In summary, although 1985–2011 malaria surveillance data affirms P. falciparum as the primary threat to travelers presenting in the United States both in terms of numbers of cases and relative risk of severe illness and fatal outcomes, P. vivax also constituted a significant clinical threat in all of these regards. Thus, appropriate caution is warranted in dealing with the prevention and treatment of vivax malaria. Although relatively very few cases of infection by P. malariae and P. ovale have been reported in the United States, the available data suggest these species could pose risk of severe and fatal illnesses. Providers recommending chemoprophylaxis or managing malaria in travelers should consider no species of Plasmodium inherently benign or clinically inconsequential. The clinical dichotomy of falciparum versus non-falciparum malaria, as a prognostic classification that practically separates threatening from nonthreatening malarias, ought to be abandoned as misleading and potentially dangerous to the patient.
Notes
Disclaimer. The findings and conclusions in this report are those of the authors and do not necessarily represent the official position of the Centers for Disease Control and Prevention.
Financial support. J. K. B. is supported by the Wellcome Trust, Grant Number B9RJIXO.
Potential conflicts of interest. All authors have submitted the ICMJE Form for Disclosure of Potential Conflicts of Interest. Conflicts that the editors consider relevant to the content of the manuscript have been disclosed.
References
- 1.Baird JK. Evidence and implications of mortality associated with acute Plasmodium vivax malaria. Clin Microbiol Rev. 2013;26:36–57. doi: 10.1128/CMR.00074-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Price RN, Tjitra E, Guerra CA, et al. Vivax malaria: neglected and not benign. Am J Trop Med Hyg. 2007;77(6 Suppl):79–87. [PMC free article] [PubMed] [Google Scholar]
- 3.Gething PW, Elyazar IR, Moyes CL, et al. A long neglected world malaria map: Plasmodium vivax endemicity in 2010. PLoS Negl Trop Dis. 2012;6:e1814. doi: 10.1371/journal.pntd.0001814. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Genton B, D'Acremont V, Rare L, et al. Plasmodium vivax and mixed infections are associated with severe malaria in children: a prospective cohort study from Papua New Guinea. PLoS Med. 2008;5:e127. doi: 10.1371/journal.pmed.0050127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.McGready R, Lee SJ, Wiladphaingern J, et al. Adverse effects of falciparum and vivax malaria and the safety of antimalarial treatment in early pregnancy: a population-based study. Lancet Infect Dis. 2012;12:388–96. doi: 10.1016/S1473-3099(11)70339-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Poespoprodjo JR, Fobia W, Kenangalem E, et al. Vivax malaria: a major cause of morbidity in early infancy. Clin Infect Dis. 2009;48:1704–12. doi: 10.1086/599041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Poespoprodjo JR, Fobia W, Kenangalem E, et al. Adverse pregnancy outcomes in an area where multidrug-resistant plasmodium vivax and Plasmodium falciparum infections are endemic. Clin Infect Dis. 2008;46:1374–81. doi: 10.1086/586743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Cullen KA, Arguin PM Division of Parasitic Diseases and Malaria, Center for Global Health, Centers for Disease Control and Prevention (CDC) Malaria surveillance--United States, 2011. MMWR Surveill Summ. 2013;62:1–17. [PubMed] [Google Scholar]
- 9.Guerra CA, Howes RE, Patil AP, et al. The international limits and population at risk of Plasmodium vivax transmission in 2009. PLoS Negl Trop Dis. 2010;4:e774. doi: 10.1371/journal.pntd.0000774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Hay SI, Okiro EA, Gething PW, et al. Estimating the global clinical burden of Plasmodium falciparum malaria in 2007. PLoS Med. 2010;7:e1000290. doi: 10.1371/journal.pmed.1000290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.World Health Organization. Geneva: World Health Organization; 2013. World Malaria Report 2013. [Google Scholar]
- 12.Baird JK. Elimination therapy for the endemic malarias. Curr Infect Dis Rep. 2012;14:227–37. doi: 10.1007/s11908-012-0250-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Wellems TE, Miller LH. Two worlds of malaria. N Engl J Med. 2003;349:1496–98. doi: 10.1056/NEJMp038127. [DOI] [PubMed] [Google Scholar]
- 14.Doolan DL, Dobano C, Baird JK. Acquired immunity to malaria. Clin Microbiol Rev. 2009;22:13–36. doi: 10.1128/CMR.00025-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Singh B, Daneshvar C. Human infections and detection of Plasmodium knowlesi. Clin Microbiol Rev. 2013;26:165–84. doi: 10.1128/CMR.00079-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ennis J, Teal AE, Habura A, et al. Simian Malaria in a U.S. Traveler---New York, 2008. Morb Mortal Wkly Rep. 2009;58:229–32. [PubMed] [Google Scholar]
- 17.Mali S, Steele S, Slutsker L, et al. Malaria surveillance - United States, 2008. MMWR Surveill Summ. 2010;59:1–15. [PubMed] [Google Scholar]
- 18.Mali S, Kachur SP, Arguin PM Division of Parasitic Diseases and Malaria, Center for Global Health; Centers for Disease Control and Prevention (CDC) Malaria surveillance--United States, 2010. MMWR Surveill Summ. 2012;61:1–17. [PubMed] [Google Scholar]
- 19.Price RN, Douglas NM, Anstey NM. New developments in Plasmodium vivax malaria: severe disease and the rise of chloroquine resistance. Curr Opin Infect Dis. 2009;22:430–5. doi: 10.1097/QCO.0b013e32832f14c1. [DOI] [PubMed] [Google Scholar]
- 20.Andrade BB, Reis-Filho A, Souza-Neto SM, et al. Severe Plasmodium vivax malaria exhibits marked inflammatory imbalance. Malar J. 2010;9:13. doi: 10.1186/1475-2875-9-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Barcus MJ, Basri H, Picarima H, et al. Demographic risk factors for severe and fatal vivax and falciparum malaria among hospital admissions in northeastern Indonesian Papua. Am J Trop Med Hyg. 2007;77:984–91. [PubMed] [Google Scholar]
- 22.Kochar DK, Saxena V, Singh N, et al. Plasmodium vivax malaria. Emerg Infect Dis. 2005;11:132–4. doi: 10.3201/eid1101.040519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Kochar DK, Tanwar GS, Khatri PC, et al. Clinical features of children hospitalized with malaria--a study from Bikaner, northwest India. Am J Trop Med Hyg. 2010;83:981–9. doi: 10.4269/ajtmh.2010.09-0633. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Manning L, Laman M, Law I, et al. Features and prognosis of severe malaria caused by Plasmodium falciparum, Plasmodium vivax and mixed Plasmodium species in Papua New Guinean children. PLoS One. 2011;6:e29203. doi: 10.1371/journal.pone.0029203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Nurleila S, Syafruddin D, Elyazar IR, Baird JK. Serious and fatal illness associated with falciparum and vivax malaria among patients admitted to hospital at West Sumba in eastern Indonesia. Am J Trop Med Hyg. 2012;87:41–9. doi: 10.4269/ajtmh.2012.11-0577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Shaikh S, Memon H, Iohano B, et al. Severe disease in children hospitalized with a diagnosis of Plasmodium vivax in south-eastern Pakistan. Malar J. 2012;11:144. doi: 10.1186/1475-2875-11-144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Tjitra E, Anstey NM, Sugiarto P, et al. Multidrug-resistant Plasmodium vivax associated with severe and fatal malaria: a prospective study in Papua, Indonesia. PLoS Med. 2008;5:e128. doi: 10.1371/journal.pmed.0050128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Hwang J, McClintock S, Kachur SP, et al. Comparison of national malaria surveillance system with the national notifiable diseases surveillance system in the United States. J Public Health Manag Pract. 2009;15:345–51. doi: 10.1097/PHH.0b013e31819d816a. [DOI] [PubMed] [Google Scholar]
- 29.Brunette G, editor. Centers for Disease Control and Prevention. CDC Health Information for International Travel 2014 (The Yellow Book) 2014. [Google Scholar]
- 30.Steinhardt LC, Magill AJ, Arguin PM. Review: Malaria chemoprophylaxis for travelers to Latin America. Am J Trop Med Hyg. 2011;85:1015–24. doi: 10.4269/ajtmh.2011.11-0464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.World Health Organization. Guidelines for the treatment of malaria. 2nd ed. Geneva: World Health Organization; 2010. [PubMed] [Google Scholar]
- 32.Hill DR, Baird JK, Parise ME, et al. Primaquine: report from CDC expert meeting on malaria chemoprophylaxis I. Am J Trop Med Hyg. 2006;75:402–15. [PubMed] [Google Scholar]
- 33.Schwartz E, Parise M, Kozarsky P, et al. Delayed onset of malaria--implications for chemoprophylaxis in travelers. N Engl J Med. 2003;349:1510–6. doi: 10.1056/NEJMoa021592. [DOI] [PubMed] [Google Scholar]
- 34.Schwartz E, Regev-Yochay G. Primaquine as prophylaxis for malaria for nonimmune travelers: A comparison with mefloquine and doxycycline. Clin Infect Dis. 1999;29:1502–6. doi: 10.1086/313527. [DOI] [PubMed] [Google Scholar]