Abstract
As the oceans absorb anthropogenic CO2 they become more acidic, a problem termed ocean acidification (OA). Since this increase in CO2 is occurring rapidly, OA may have profound implications for marine ecosystems. In the temperate northeast Pacific, fisheries play key economic and cultural roles and provide significant employment, especially in rural areas. In British Columbia (BC), sport (recreational) fishing generates more income than commercial fishing (including the expanding aquaculture industry). Salmon (fished recreationally and farmed) and Pacific Halibut are responsible for the majority of fishery-related income. This region naturally has relatively acidic (low pH) waters due to ocean circulation, and so may be particularly vulnerable to OA. We have analyzed available data to provide a current description of the marine ecosystem, focusing on vertical distributions of commercially harvested groups in BC in the context of local carbon and pH conditions. We then evaluated the potential impact of OA on this temperate marine system using currently available studies. Our results highlight significant knowledge gaps. Above trophic levels 2–3 (where most local fishery-income is generated), little is known about the direct impact of OA, and more importantly about the combined impact of multi-stressors, like temperature, that are also changing as our climate changes. There is evidence that OA may have indirect negative impacts on finfish through changes at lower trophic levels and in habitats. In particular, OA may lead to increased fish-killing algal blooms that can affect the lucrative salmon aquaculture industry. On the other hand, some species of locally farmed shellfish have been well-studied and exhibit significant negative direct impacts associated with OA, especially at the larval stage. We summarize the direct and indirect impacts of OA on all groups of marine organisms in this region and provide conclusions, ordered by immediacy and certainty.
Introduction
Fossil fuel burning and changes in land use by humankind have increased atmospheric carbon dioxide (CO2) at an unprecedented rate, causing our climate to change [1]. A significant portion of this anthropogenic CO2 (∼30%; [2]) has been absorbed by the ocean. When CO2 enters the ocean it combines with water (H2O), resulting in an increase in the concentration of hydrogen ions [H+] and an increase in acidity (decrease in pH [3, 4]. Therefore, as our climate changes, our oceans become more acidic due to anthropogenic contributions, a problem termed Ocean Acidification (OA) [5].
While anthropogenic atmospheric CO2 dominates contributions to OA on a global scale, other anthropogenic sources may be significant on a local scale [6]. For example, acid rain from vehicle emissions and industry cause an increase in ocean acidity, which is likely relevant, at least near (and downwind of) urbanized regions [7]. Any addition of organic carbon to the ocean, such as sewage, decomposes to dissolved inorganic carbon (DIC), and increases acidity. Agricultural run-off provides nutrients which then fuel (an anthropogenic) increase in production of organic carbon in the ocean [8], again increasing acidity.
Aquatic acidity is most commonly reported as pH. However, pH is difficult to determine accurately in saltwater because of the additional ions present in solution [9]. It is closely linked with carbonate chemistry in the ocean, which is complex. To quantify the carbon state (i.e. the concentration of each chemical form of DIC present) in seawater, two of four measured parameters—DIC, pH, total alkalinity (TA), and partial pressure of CO2 (P CO2)—must be known, in addition to temperature and salinity. To be more accurate, phosphate and silicic acid concentrations are also required [10]. In the past, pH has most often been determined from DIC and TA (e.g. [11]). (TA is the acid neutralizing capacity of the solution, which is not simply related to pH in seawater [10].) Thus, although one can generalize to say that high DIC is usually associated with low pH (or high P CO2), more information, e.g. TA, is required to be quantitative.
The carbon state is relevant to biology. Most of the DIC in the ocean occurs in the form of bicarbonate () and carbonate (), with less than 1% in the form of CO2. When pH decreases, the balance between and changes so that there is less . This shift has important implications for plants and animals that build calcium carbonate (CaCO3 structures (e.g. shellfish, corals) [12]. Two mineral forms of CaCO3 (aragonite and calcite) are common in biological structures. The aragonitic form is more soluble than calcite given the same environmental conditions [13]; therefore, creatures that use aragonite are more susceptible to OA than those that use calcite [12]. The ease with which these minerals are formed is quantified by the saturation state (Ω), such that as Ω decreases, dissolution increases [14]. The water is undersaturated with respect to CaCO3 when the chemical rate of dissolution exceeds the rate of formation [15]. For organisms that precipitate CaCO3, decreasing Ω means that more energy is required to build and maintain their carbonate structures [16, 17].
Marine organisms are also affected by carbon state (defined above) and OA in other ways. All marine animals need to rid themselves of metabolically produced CO2 through respiration. The effectiveness of this removal is dependent, in part, on the ambient P CO2 of the medium (e.g. [18]). Similarly, plants and animals rely on pH to regulate ion transport, and the energy they must expend to maintain intra- and extracellular pH depends on ambient pH (e.g. [19]). Thus, there is no one carbon parameter that best indicates OA impacts on all marine organisms, and so full knowledge of the complete carbon state is desirable (e.g. [20]).
A large and growing number of studies have been undertaken regarding OA (S1 Table). To understand and predict biological impacts, an increasing number of experiments have been completed that attempt to emulate future ocean conditions in the laboratory. Experimental conditions are usually defined by controlling either the P CO2 or the pH (e.g. S2 Table) and recently an internationally accepted guide has been published that describes the techniques used [21]. In most of these experiments, present-day conditions (the control) are set at either atmospheric P CO2 (∼400 μatm at the time of writing) or the estimated current global average pH of the surface ocean, which is 8.1 [5]. However, marine organisms in the natural environment may experience values that are significantly different depending on location and the depth that they occupy.
In the ocean, DIC (and P CO2) generally increase with depth while pH decreases. In other words, low pH conditions naturally occur at depth. This partitioning of inorganic carbon towards deeper parts of the ocean is due in large part to the ‘biological pump’ that allows the ocean to hold more carbon [22]. Photosynthesis in the surface draws down DIC (which increases pH) and produces organic forms of carbon. Some of this organic carbon falls to deeper levels, where it decays back to DIC (decreasing pH).
British Columbia—oceanography
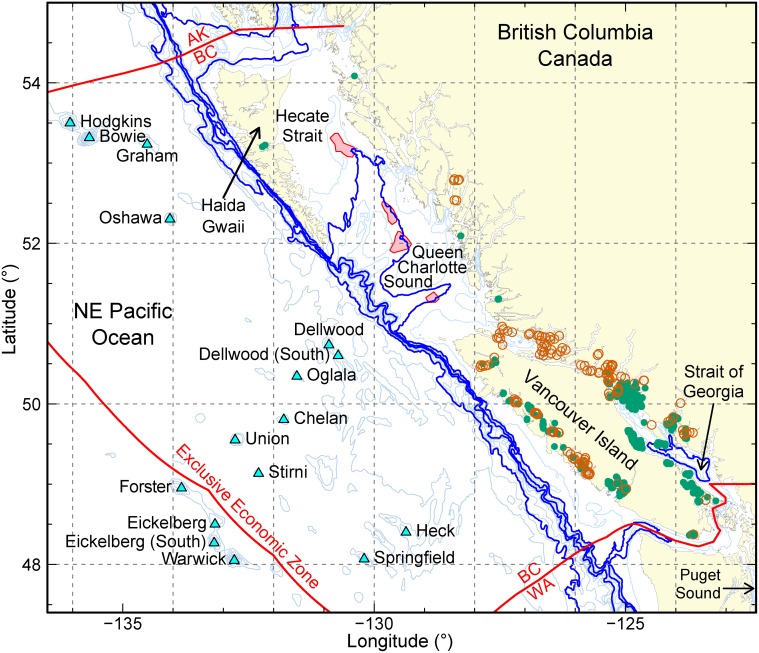
British Columbia (BC) makes up 27,000 km (17,000 mi) of the temperate northeast Pacific coastline. Circulation along this coast (Fig. 1) is dynamic so that large changes in carbon parameters occur both in space (e.g. [23]) and time (e.g. [24, 25]). Coastal upwelling along the west coast of Vancouver Island (WCVI) [26] brings subsurface water high in DIC into the surface mixed layer [27] so that low pH (e.g. 7.6) is found at relatively shallow depths, e.g. above 125 m (Fig. 2). Furthermore, these subsurface waters are enriched in DIC relative to waters at the same depth in other ocean basins, simply because north Pacific water is relatively ‘old’ and has had more time to receive organic matter [28, 29]. Upwelled waters are also rich in nutrients that are limiting to phytoplankton growth and so cause high primary production that increases pH at times. In fact, the WCVI enjoys the highest productivity of any zone on the northeast Pacific coast [30]. Consequently, present-day ranges in pH in the surface mixed layer along the outer BC coast span a remarkable range (7.8–8.4; Fig. 2). The low end of this range is significantly lower than the benchmark of present-day average global surface ocean pH (8.1).
Fig 1. British Columbia (BC) coastline and bathymetry (isobaths in metres: thin grey—100, 200, 300, …, 1000, 1250, 1500, 2000, 2500; thick blue—200, 500, 800, and 1600).
The continental slope along most of BC comprises steep slopes, especially along the west coasts of Haida Gwaii and northern Vancouver Island. Hecate Strait is largely dominated by shallow waters and a flat seafloor. Sponge reef core protected areas in Hecate Strait and Queen Charlotte Sound are shaded pink. The Strait of Georgia forms a large inland sea that is heavily influenced by river runoff and tidal currents. Saltwater finfish farm and hatchery sites are indicated by open red circles, commercial marine shellfish farms are indicated by solid green circles [345]. Select seamounts [346] are marked by blue triangles. Canada’s Exclusive Economic Zone (200-nautical miles offshore) is delimited in red. Map was prepared using PBSmapping in R [347]. The R code is provided as Supporting Information (S1 Code).
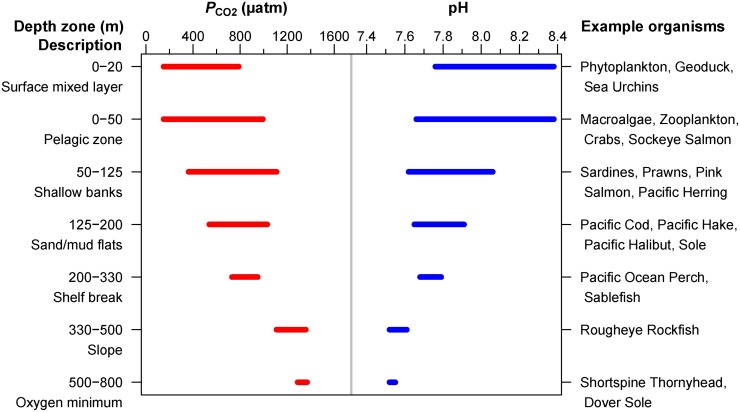
Fig 2. Estimated present-day ranges of P CO2 (red) and pH (blue) during spring [40] and summer [27] for various depth zones along the outer BC continental shelf, with typical species found in each zone (see Methods).
There are numerous data above 50m and few below 125 m. The number of values in each depth zone from top to bottom are: 70, 116, 33, 45, 5, 4 and 2, respectively. Above 50 m, the distributions of values are skewed, such that high P CO2 (low pH) extremes occur less often than the low P CO2 (high pH) extremes. Data and R code for this figure are provided as Supporting Information (S2 Code).
In protected waters (e.g. Strait of Georgia, Fig. 1) less data are available relative to the WCVI. These data show similar (or larger) ranges in surface pH and P CO2 (unpublished data, DI), which are also similar to values found just to the south in the protected waters of Puget Sound, Washington State (WA) [6, 31]. Again, a critical feature in these waterways is low surface pH (high P CO2) relative to global averages, especially during the winter season [32].
British Columbia—fishery
Fisheries and aquaculture play an important role in the BC economy, contributing over $650 million (we quote all dollar values in Canadian dollars) to the provincial gross domestic product (GDP) in 2011 [33]. Sport (or recreational) fishing, mainly for salmon and Pacific Halibut, is responsible for approximately 50% of this contribution, while the wild (or capture) fishery makes up ∼15% and aquaculture ∼10%. Marine ecosystems also play critical cultural roles in BC and their monetary value to tourism is only partially included in these totals (through sport fishing).
Over the past 20 years the wild fishery has declined in terms of both its contribution to the BC GDP and employment, although some individual components are increasing (e.g. prawns, Geoduck Clam, Pacific Halibut). Meanwhile aquaculture has nearly tripled its contribution to BC GDP in the same time frame [33]. As a result, published landed values associated with aquaculture are about the same as those from the wild fishery (see Results) and aquaculture now employs slightly more people than does the wild fishery [33].
The wild fishery is for the most part associated with the open coast (outer WCVI and Queen Charlotte Sound, Fig. 1) and is relatively diverse, with no one fishery dominating landed values (see Results). The most important contributors (Pacific Halibut, Geoduck Clams, prawns, crabs, tunas, Sablefish, rockfishes) currently each have landed values in the $20–50 million range [34]. Aquaculture occurs in protected waters: shellfish farming mainly in the northern Strait of Georgia and finfish farms and hatcheries mainly north of that on the northeastern side of Vancouver Island (Fig. 1). In BC, Atlantic Salmon aquaculture clearly dominates all other commercial fisheries (see Results).
Predicting biological impacts due to OA is a highly complex problem that has only become a concern relatively recently (primarily over the past decade). There have been excellent review papers outlining anticipated impacts on a general global scale (e.g. [3, 35]) as well as meta-analyses of existing work on the topic (e.g. [36]). Cooley and Doney [37] have provided the first estimate of the economic impact of OA, centred on the shellfish fishery, in the United States. However, few studies consider specific ecosystems, particularly in the context of local pH conditions and natural variability, and none focus on the temperate northeast Pacific.
Here, we examine the potential impact of OA on temperate coastal ecosystems in the northeast Pacific Ocean, with a focus on BC fisheries. To tackle this issue we:
describe the current marine ecosystem in BC (especially by depth, Fig. 3);
define the present-day carbon state with depth in local waters (Fig. 2);
assess the response by marine organisms in this region to OA by investigating existing biological OA impact studies (on local and non-local species) and comparing anticipated changes in acidity (P CO2) to those currently experienced along the BC coast.
We use the best information available at present to address this problem. The quantitative details, including treatments and measured carbon parameters, of all studies that we used are summarised in S2 Table. We provide specific conclusions ordered by immediacy and relevance to the BC fishery.
Fig 3. Depth-of-capture, expressed as quantile box plots of depth (m), from fisheries and survey data (where available) for species groups identified in Fig. 4.
For each quantile box, the upper whisker, box top, box delimiter (horizontal line), box bottom and lower whisker correspond to the 0.025, 0.25, 0.5, 0.75, and 0.975 quantiles, respectively. Depth quantiles that lie deeper than the figure limit are indicated along the bottom. Horizontal dashed lines correspond to depth zones in Fig. 2. See Methods for data sources. Data and R code for this figure are provided in Supporting Information (S1 Data and S3 Code, respectively).
Methods
Present state of the BC marine ecosystem
Marine organisms were assigned to taxonomic groups and sorted by trophic levels adapted from model-derived output for the BC shelf [38] (Fig. 4). We added several taxonomic groups that are commercially fished [34] (e.g. sardine, tuna) and unfished (e.g. seagrasses, glass sponges) to this list as necessary. To evaluate species abundance and distribution within these groups, we used published literature (both primary and secondary as cited) where available. When literature was not available we consulted Canadian Department of Fisheries and Oceans (DFO) databases and the expertise of individuals active in the field (see Results and Acknowledgements). Landed values of fished species were taken from [34] (or [39] for euphausiids).
Fig 4. Summary of ocean acidification effects on (A) wild, and (B) farmed species groups in BC waters, including landed value for those that are fished or farmed.
Species groups are arranged vertically by trophic level, adapted from output by Preikshot [38] (courtesy of D. Preikshot, Madrone Environmental Services, Duncan BC). Areas of circles are proportional to the landed values in 2011, based on data in [34] (and [39] for euphausiids). Squares represent species groups that are not commercially harvested. Solid colours represent the likely direct effects of ocean acidification (see Results for explanations). Stippling refers to possible effects. For species marked by an asterisk (*), colours represent indirect effects. Data and R code for this figure are provided as Supporting Information (S4 Code).
Species depth distributions (Fig. 3) were obtained from DFO databases (Pacific Biological Station, Nanaimo, Canada). Depths associated with commercially-caught groundfish (compiled by RH, May 1, 2014) and shellfish (compiled by Georg Jorgensen, May 6, 2014) are depths-at-capture, most often a mean of the minimum and maximum depths of fishing events (usually trawl or trap). For the commercial species groups (Fig. 4), depths were selected based on fishing methods specific to each group—Sea Urchins (dive), Euphausiids (nets), Sea Cucumbers (dive), Geoduck Clam (dive), Scallops (dive, trawl), Crabs (trap), Squid & Octopus (dive, trap), Prawns (trap), Shrimp (trawl), Pacific Cod (midwater & bottom trawl), Pollock (midwater & bottom trawl), Halibut (bottom trawl), Lingcod (bottom trawl), Sole (bottom trawl), Dogfish(bottom trawl), Hake (midwater trawl), Arrowtooth (bottom trawl), Rockfish (midwater & bottom trawl), Sablefish (bottom trawl). Depths associated with pelagic species (Herring, Sardines, and Salmon—Chinook, Chum, Coho, Sockeye, Pink) come from two sources: the WCVI Sardine Trawl Survey (spanning the WCVI, Fig. 1: −129.14°W to −124.56°W, 48.32°N to 51.14°N), which occurs mid-summer and is conducted during the night (data compiled by Linnea Flostrand, May 8, 2014), and the La Perouse Survey (spanning the BC coast, Fig. 1: −132.89°W to −123.07°W, 43.58°N to 54.64°N), which is a daytime acoustic trawl survey used to verify acoustic targets (data compiled by Jennifer Boldt, May 14, 2014). The two surveys did not capture any SARA-listed species. Mean depths-of-capture are summarised by quantile boxplots where the box represents 50% of the observations, and the region between the whiskers represents 95% (Fig. 3).
Commercial fishing in Canada is regulated by the Fishery Act. Specifically, Section 22 (http://laws-lois.justice.gc.ca/eng/regulations/SOR-93-53/page-6.html) identifies all license conditions that DFO uses to manage gear, monitoring, reporting, harvesting, allocation, and catch requirements. DFO’s Pacific Region Animal Care Committee requires animal-use protocols (Supplementary S1 text), but specifically exempts lethal sampling of fish and invertebrates for stock assessment and sampling from commercial operations where animals are dead or certain to die. Data used here were collected for stock assessments and are therefore exempt from protocols.
Local inorganic carbon distributions
Published inorganic carbon data (DIC, TA) from the outer BC coast in Queen Charlotte Sound (QCS) [40] and along the WCVI [27, 40] (Fig. 1) are used. These data (174 discrete samples) were collected over the continental shelf, slope and offshore, from the surface to 800 m with greater depth resolution in the top 50 m. The carbonate system was defined from TA and DIC (CO2SYS, [41]) and the constants of [42] with conductivity, temperature, depth and nutrient data that were collected concurrently. These data were sorted into depth intervals defined by local bathymetry relevant to local marine organisms (Fig. 2).
Responses of marine organisms to OA
We evaluated the potential impact (coded by colour in Fig. 4) of OA on each taxonomic group that occurs in BC, recognizing that uncertainty exists. We also identified the depth distributions that these groups of species occupy, along with associated OA conditions (Fig. 3). Similar to our description of the local marine system, we used published literature where available to assess direct and indirect effects of OA on taxonomic groups. When no publications were available in this rapidly emerging field, we consulted individuals who presented at recent conferences (in particular 2014 Ocean Sciences Meeting, Honolulu HI and 2014 Salish Sea Ecosystem Conference, Seattle WA), and we consulted many other experts in their respective fields (cited within Results and Acknowledgements).
Results
There are relatively few published carbon data in BC waters. We use these data [27, 40] to estimate present-day ranges in pH and P CO2 for depth intervals relevant to local marine organisms (Fig. 2). We then defined three relative P CO2 levels, which are based on the present-day ranges in Fig. 2, to group the experimental treatments presented in the literature relative to our local waters (Table 1). For example, Pink Salmon (Oncorhynchus gorbuscha) generally occupy depths in both the 0–50 m and the 50–125 m zone (Fig. 3) so for these fish present-day P CO2 in our region is ∼200–1000 μatm (pH ∼7.6–8.4) (Fig. 2) so that a P CO2 level of 5000 μatm would be the upper limit of an ‘elevated’ (Table 1) treatment.
Table 1. Terminology used in the text to quantify levels of P CO2 used in manipulation experiments.
S2 Table provides details for each treatment in each experiment cited.
| Terminology | PCO2 |
|---|---|
| present-day | depends on depth range (Fig. 2) |
| reduced | 0.5× present-day |
| elevated | 2–5× present-day |
| very elevated | 5–10× present-day |
Vertical distributions of marine organisms on the BC coast are presented with associated impacts of OA, ordered by trophic level (Fig. 4) in the following sections. Depending on trophic level and group, the amount of information available was variable. For many commercially harvested groups (represented by circles in Fig. 4) excellent data were available (e.g. finfish, Fig. 3). On the other hand, abundance and species composition of unfished groups are not well characterised, particularly at lower trophic levels (squares in Fig. 4, e.g. microzooplankton, corals). For many organisms important in the region, no published OA related studies exist (grey circles and squares in Fig. 4). Where necessary, we have adopted results from OA studies on species elsewhere that are similar to the ones found locally. These caveats are detailed in each section. Experimental details are summarised in S2 Table.
Phytoplankton
In the coastal northeast Pacific the predominant class of phytoplankton is diatoms, which are associated with high trophic transfer [43]. Many species (including the dominants: Skeletonema costatum, Thalassiosira spp., and Chaetoceros spp.) occur along the entire coast of BC [44–50]. Large blooms associated with coastal upwelling are often monospecific (e.g. [51]), but in our region they appear to be more diverse and occasionally include large numbers of photosynthetic dinoflagellates [46, 52]. Coccolithophorid blooms have been directly observed in more protected regions [50] and by satellite along the entire BC coast during summer [53]; however, coccolithophores (which calcify) are generally assumed to contribute minimally to overall productivity in the coastal zone (roughly landward of the 800 m isobath, Fig. 1) despite their importance further offshore [54]. Primary production by phytoplankton is exceptionally high in the region [27, 47, 55] and ultimately responsible for the high fish yields along our coast [30].
Phytoplankton species that are harmful to higher trophic levels are also common in the region. Large blooms of diatoms from the genus Pseudo-nitzschia occur on the outer coast (e.g. [56, 57]) while the dinoflagellate Alexandrium is more prolific in protected locations [58]. Both Pseudo-nitzschia and Alexandrium produce neurotoxins that bioaccumulate in higher trophic levels. These toxins can interfere with the reproductive success of fish, seabirds, and mammals and cause mass mortalities [59, 60]. They are also responsible for numerous seasonal shellfish closures in BC (http://www.pac.dfo-mpo.gc.ca/fm-gp/contamination/biotox/index-eng.html). Additionally, significant blooms of Heterosigma akashiwo occur in protected waterways [61, 62]. Heterosigma releases peroxide free radicals into the water [63], which damage fish gill tissue [64, 65] and cause significant mortality and monetary losses (millions of dollars per year) to salmon aquaculture in BC [66]. Thus, harmful algae already pose a threat to health and food safety along the BC coast [58].
Direct effects There have been numerous studies on phytoplankton related to OA (S1 Table) and a variety of responses have been observed depending on the species and the experimental treatment (e.g. [67–70]). Although natural conditions in most coastal environments, including the BC coast (Figs. 1 & 2), cover an exceptionally large range in carbon states and consequently pH (e.g. [6, 27, 71]), experiments in the field are challenging to complete. Thus, most studies have been conducted in the laboratory, often using a single strain of cultured phytoplankton. Also, because coccolithophores calcify (and at least some are easy to culture), they have been studied disproportionately. We sample a relatively small subset of this body of literature to summarise results of most relevance to the mixed, often diatom-dominated, community in the region and briefly describe the current understanding of the mechanisms involved.
Species specific responses by primary producers, including phytoplankton, to increases in ambient CO2 are highly dependent on their carbon-uptake mechanism. Carbon assimilation relies on the enzyme ribulose biphosphate carboxylase-oxygenase (RuBisCO) to fix CO2 [72], but this enzyme has a poor affinity for CO2 [72, 73]. Over geological times scales (i.e. the last 3.5 billions years), as newer phytoplankton species have evolved, their use of RuBisCO has become more effective [72]. Some have carbon-concentrating mechanisms (CCMs), e.g. diatoms [74], to help transport and accumulate CO2 to the active RuBisCO site [75]. The most important CCM for phytoplankton involves carbonic anhydrase to convert abundant to the limiting CO2 [76]. Despite CCMs, many photosynthetic phytoplankters, including some diatoms, appear to be carbon-limited under present-day conditions (e.g. [72]).
Because of these limitations in carbon uptake, it is anticipated that OA will increase overall production, which may provide more food to higher trophic levels. However, this increase does not appear to be large. Numerous mesocosm experiments, which use natural assemblages, suggest that regardless of species composition, there may be at most a 10–30% increase in primary production due to OA (e.g. [77–80]). In addition, a side-effect of elevated P CO2 (Table 1) is increased carbon to nitrogen (C:N) ratios in phytoplankton, effectively decreasing its nutritional quality [80].
While it is generally agreed that OA is likely to cause shifts in phytoplankton species composition, it remains unclear what these shifts will be [69]. It is reasonable to expect that species that do not have effective CCMs will do better than species that are already efficient with carbon uptake (diatoms in general). For example, the fish-killing raphidophyte Heterosigma akashiwo relies on passive diffusion to obtain CO2. As a result it responds strongly (increased rates of growth and primary productivity) to an increase in dissolved CO2 [81, 82] regardless of temperature [82]. In contrast, growth rates for some phytoplankton species reach a maximum value at the low end of present-day P CO2 in the upper mixed layer on the outer BC coast (Fig. 2) assuming a salinity of 31–32 [81]. For other species (including several diatoms) these rates remain invariant under elevated P CO2 [83].
Competition may be more subtle. For instance, some experiments have shown an increase in the proportion of diatoms relative to smaller phytoplankton with increased P CO2 (e.g. [84]) while others show the opposite effect (e.g. [85]). In addition, Tortell et al. [86] found that the prymnesiophyte Phaeocystis could outcompete diatoms at reduced P CO2 even though both groups have efficient CCMs. Finally, it has been suggested that at least one motile species (H. akashiwo) will swim faster under OA and deepen its vertical distribution [87], which may give it (and any species that can take advantage of its absence nearer to the surface) an additional competitive advantage.
Factors associated with climate change, including OA, are expected to increase the frequency and severity of harmful algal blooms [88]. In addition, the production of potent neurotoxins—domoic acid by common and sometimes prolific diatom species of Pseudo-nitzschia, and saxitoxin by dinoflagellate species of Alexandrium—has been shown to increase markedly under OA conditions [89–91]. In fact, domoic acid production in (at least some) Pseudo-nitzschia spp. increases dramatically (5–50× per cell) as P CO2 increases [92, 93].
Coccolithophores (prymnesiophytes) are the major calcifiers in the phytoplankton community [94, 95]. The most commonly studied species is Emiliania huxleyi, and although it appears to be less prevalent locally in the coastal zone (Fig. 1 in [94]), it plays an important role in the Alaskan Gyre [54]. Numerous experiments (most in vitro, some in situ) on Emiliania have been conducted to determine the effects of carbonate chemistry on calcification. Most (but not all, e.g. [96, 97]) suggest decreasing calcification at lower pH values (e.g. [70, 98]). Although much remains unknown (e.g. [20, 99]), the consensus is that OA will decrease calcification [69]. This observation is reinforced by mesocosm experiments that manipulate coccolithophore populations [67, 100] and by paleolithic records [101].
Phytoplankton synopsis We conclude that the overall impact on ecosystems and fisheries due to changes in the phytoplankton community in our region will be negative. While a modest increase in primary production is anticipated (so a direct positive benefit to phytoplankton, Fig. 4A), this increase is not likely to benefit higher trophic levels due to expected shifts in species composition (away from diatoms) and decreased nutritional value of the plankton. More importantly, the fish-killing alga Heterosigma akashiwo may gain a competitive advantage, which would seriously threaten salmon aquaculture. In addition, increasing P CO2 has been shown to alter the mix of neurotoxins produced by genera such as Pseudo-nitzschia and Alexandrium to favour the more potent forms, posing a significant threat to higher trophic levels and the shellfish industry as well as overall food safety.
Macroalgae
Three groups of macroalgae are delineated by their pigmentation: green, brown, and red algae, all of which are common in BC. In particular, brown algae constitute the majority of the biomass in intertidal and upper subtidal zones, and are dominated by kelps and rockweeds [102]. Brown algae have soft fleshy morphologies, and both green and red algal groups contain species with hard, calcified structures. Calcified red algae have two morphologies, crust-forming on substrate, and erect and branched. Both red and green algae are found in the intertidal and upper subtidal zones, but red algae extend down to the lower photic zone [103]. The large-blade (brown) macroalgae (e.g. Laminaria, Macrocystis) that form dense kelp forests along temperate coasts, common in BC, are the basis of some of the most productive ecosystems on Earth [103, 104]. These forests provide extensive shelter from predation, desiccation and wave action, as well as food, for hundreds of species with representatives from most taxonomic groups [105]. Calcified red algae provide similar protective structures, that are especially important for invertebrate species (e.g. urchins and anemones) [106].
Direct effects As with phytoplankton, many macroalgal species use carbon concentrating mechanisms (CCMs) to help transport and accumulate the CO2 required for carbon assimilation [107]. Those relatively rare species without CCMs (most of which are red algae) rely on passive diffusion of CO2 [108, 109] and so may experience enhanced photosynthesis and growth under OA, whereas those that have CCMs are likely to show no, or only small, positive effects due to reduced energy expenditure [107, 110]. Responses to elevated P CO2 (Table 1) may be more significant at depths where light levels are reduced because energy constrains photosynthesis and CCMs are energetically expensive, though these effects are likely to be species-specific [110]. In addition, UVB (Ultraviolet B, 280–315 nm) exposure near the water surface tends to be harmful to some macroalgae, reducing the positive response to elevated P CO2 [111]. The ultimate effects of OA on photosynthesis and growth of macroalgae will likely depend on interactions with light exposure, UV radiation, and other stressors. There has been less research concerning reproduction and life stages; however, it has been suggested that OA will result in reduced gametophyte growth of giant kelp [112].
For calcifying macroalgae, elevated P CO2 affects the ability to build and maintain the calcified component of their tissues [108]. For example, Hofmann et al. [113] observed reduced calcification and growth for a cosmopolitan species of red algae when exposed to elevated P CO2 over a 4-week period (S2 Table). Calcifying red algae are particularly sensitive to OA because unlike most calcifying green algae and invertebrates, red algae deposit a high-magnesium form of calcite into their cell walls, that is more soluble in acidified water than other forms of calcite [28]. However, Kroeker et al. [36] found no consistent change in calcification at elevated P CO2 levels for a suite of calcifying macroalgae, perhaps because many species are able to generate microenvironments suitable for calcification despite increases in ambient P CO2 [114–117]. Indeed, the observed reductions in growth with elevated P CO2 (e.g. [113]) may result from the increased dissolution of carbonate skeletons rather than reduced production [117]. These effects are likely to interact with other stressors, such as UV radiation and temperature [118]. For example, Gao and Zheng [119] suggest that the carbonate skeleton of the same red algal species serves as a protective layer against UV; thus, CO2 induced shell dissolution may increase vulnerability to detrimental effects of UV radiation [119].
Indirect effects Changes in macroalgal community composition are anticipated given the diversity of responses to OA among species. In general, non-calcifying macroalgae (especially those that rely on diffusion of CO2 instead of CCMs) are expected to experience increased competitive success compared with calcifying macroalgae [110], resulting in an overall shift of community composition toward non-calcifying species [36]. Furthermore, studies on CO2-enriched waters surrounding seafloor vents elsewhere support this hypothesis [120]. Most research has focused on losses of crust-forming calcified red algae in particular and replacement with non-calcifying turf-forming algal communities (i.e. species that reach heights of <15cm [121]) [36, 115, 122]. In BC, crust-forming red algae release chemical cues that play an important role in the settlement of some invertebrate larvae (e.g. abalone [123, 124]), and they bond substrata to provide stable habitats for other benthic species [106], but the resulting ecosystem effects under OA remain highly uncertain. Likewise, the ecological effects of possible declines in erect calcified red macroalgae and replacement by fleshy macroalgal species have received little attention (but see [113, 125]).
In addition to competition, herbivory is another key factor structuring macroalgal communities [126]. Rates of herbivory on macroalgae depend on palatability and the presence of hard carbonate structures for algal defence [127]. OA may reduce structural protection thereby increasing grazing on calcified species [115]. For non-calcified species, OA may increase C:N ratios possibly reducing palatability and hence grazing pressure [115]. However, OA will likely be detrimental to many herbivores, especially calcified species such as echinoderms and molluscs (see below), with resulting beneficial effects on some macroalgal species (e.g. [128], Mediterranean Sea, S3 Table).
Given these potential impacts, Harley et al. [115] suggest that in the California Current ecosystem, which includes the WCVI, OA may result in a shift from diverse nearshore communities consisting of kelp canopies, understory turf assemblages, crust-forming calcifying algae, and calcifying invertebrates (e.g. urchins), to communities dominated by kelp and macroalgal turfs. Where kelp canopies have been lost due to other natural or anthropogenic disturbances (e.g. indirect effects of commercial harvest of fish species as found for large regions of the northeast Pacific, [129]), OA may prevent kelp recovery by facilitating expansion of algal turfs which inhibit kelp recruitment [130], as found along the Australian coast [131]. Kelp is the dominant primary producer among macroalgal species in BC, providing food and habitat for commercially important fish species, such as Pacific salmon [132, 133]. However, because responses of benthic communities to OA are highly species-dependent, the results of these studies cannot be extrapolated to other regions with high confidence [115].
In addition to community-level effects from altered competition and herbivory, OA may slow decay rates of some kelp species including those commonly found in BC (e.g. bull kelp, Nereocystis leutkeana), which could indirectly affect detritivore consumption and nutrient cycling [111]. This delay may result in the accumulation of phytodetritus, possibly reducing food availability for consumers in nearshore waters.
Macroalgae synopsis The direct effect of OA is hypothesised to be positive on non-calcifying species due to enhanced availability of CO2 for carbon assimilation, but negative for calcifying species due to reduced growth and dissolution of protective shells (Fig. 4A). Community composition may shift from calcifying macroalgae species toward non-calcifying species, with an inhibition in the recovery of depleted kelp populations. However, community-level responses will depend on the extent of grazing on fleshy, non-calcifying species, possible changes in grazing due to OA-impacts on invertebrate herbivores, and the expansion of algal turfs. Responses of benthic communities to OA are highly species-dependent, limiting confidence in generalisations and extrapolations among regions and studies.
Seagrasses
Seagrasses belong to a small group of marine angiosperms comprising 60 species worldwide [134]. In BC, there are only two species of eelgrass—the native Zostera marina and the introduced species Z. japonica—and three species of surfgrass all belonging to the genus Phyllospadix [135]. Seagrass beds are well-known as nurseries for juvenile fish and invertebrates [136]. Another advantage conferred by seagrass beds is their ability to modify the seawater carbonate system, increasing aragonite saturation states within their confines [137], which might offer calcifying organisms refugia from the effects of OA.
In contrast to most macroalgae, seagrass cannot take advantage of the abundant [138] and so increase their photosynthetic rate when DIC becomes more abundant [139]. With more DIC, seagrass are better able to compensate for light attenuation [139]. As a result, increased P CO2 may foster the growth of seagrass beds, despite worldwide losses of seagrass ecosystems due to anthropogenic disturbances along coastal environments [134]. However, OA-related reductions in phenolic compounds [140], which protect seagrasses against herbivory, may result in increased grazing pressure under increased P CO2. The evidence for decreasing phenolics in seagrass under OA is limited and contrary to the trend of increasing phenolics in terrestrial angiosperms under increased atmospheric CO2 [140].
Seagrass synopsis Seagrasses will likely benefit from increased P CO2 because higher DIC helps them compensate for light limitation; however, a decrease in protective phenolic compounds may offset any benefit due to increased grazing. The net effect of increased OA will likely be neutral for seagrasses.
Microzooplankton
Microzooplankton (20–200 μm) include heterotrophic protists such as ciliates and non-photosynthetic dinoflagellates. Typical ciliate genera along the BC coast include Strombidium, Tintinnopsis and Strobilidium [141] while the heterotrophic dinoflagellate species belong chiefly to Protoperidinium, which feeds almost exclusively on diatoms [142], and Gyrodinium. In nearshore waters, microzooplankton can be very abundant, depending on the time of year and food source (e.g. [44]). More importantly, fluctuations in microzooplankton populations, tightly coupled to phytoplankton, can have a large effect on pelagic ecosystems [143] and can influence the success or failure of fish recruitment [144].
Direct effects There are no studies that test the direct effects of OA on individual microzooplankton species. That said, foraminifera are amoeboid protists that form CaCO3 shells and, like coccolithophores, are probably at risk from OA (e.g. [145]). There is also speculation that microzooplankton motility might be affected by OA [146], with the closest evidence coming from the study of the photosynthetic flagellate Heterosigma that demonstrated an increase in swimming speed and an increase in downward migration [87]. Large-scale mesocosm manipulations and on-board experiments that compare present-day and elevated P CO2 (Table 1) have found conflicting results—(i) no shifts in composition or abundance [147–149], (ii) almost identical succession patterns [150], and (iii) significant increases in heterotrophic dinoflagellate abundance [151, 152], although in the former (i.e. [151]) an increase in the prey species of diatoms was likely responsible.
Microzooplankton synopsis Based on the limited studies for microzooplankton, we expect that most species will be unaffected by OA, except through changes to their prey (phytoplankton). Direct OA effects will likely have a negative effect on foraminifera through reductions in CaCO3 shells.
Mesozooplankton
In our region, the zooplankton community is strongly dominated by calanoid copepods [153, 154]. Important species include Neocalanus plumchrus, Acartia longiremis and Pseudocalanus spp. [153, 154]. In protected regions like the Strait of Georgia (Fig. 1) Calanus pacificus is also important [154], while on the outer shelf Calanus marshalae is significant [153]. Some species spend part of their life cycles (that includes egg production) in relatively deep waters, >300–500 m (e.g. Neocalanus plumchrus and Calanus pacificus) while others, like Acartia longiremis, are always found above ∼50 m. Zooplankton productivity is variable and appears to be changing over time [153], with species composition dependent on temperature [154]. Mesozooplankton provide the main trophic link connecting phytoplankton and microzooplankton with larger oceanic predators [155]. They are critical for several commercially-valuable fish species that prey on them directly, such as Pacific Herring, Pacific Hake, Pacific Sardine, various salmon species, and Spiny Dogfish (Squalus acanthias) [155].
Direct effects Only Calanus pacificus has been studied locally so we include experiments on copepods found elsewhere from the common genera Acartia and Calanus. Although responses to acidic conditions can be species-specific, even within genera (e.g. [156]), our summary provides a general indication of possible effects on the mesozooplankton community in our region.
Most OA related mesozooplankton research involves eggs and/or survival rates within individual stages. Egg production rates of adult females appear unaffected by increased P CO2 (even under very elevated conditions, Table 1) [156–160], although P CO2-induced increases or decreases were observed depending on temperature [161]. On the other hand, egg hatching rates may decrease with OA [156–160], although increases have also been observed [161]. However, it is possible that hatching is simply delayed and so not observed in short-term experiments [160]. Effects of OA on overall egg hatching success are uncertain. In Puget Sound, WA (Fig. 1), egg hatching in Calanus pacificus is reduced under elevated P CO2 (Anna McLaskey, pers. comm., University of Alaska, Fairbanks AK), whereas egg hatching success in Calanus helgolandicus (found in the North Atlantic) appears unaffected [162]. For copepod embryos, survival rates appear unaffected by OA, while developmental rates may decline [163]. In adult copepods, survival rates are not significantly affected even under very elevated experimental conditions (except for one species) [156, 157, 159].
Although impacts on individual life stages may not be significantly different from a control scenario, the cumulative impacts may be significant. In addition, the studies thus far have been relatively short-term, and do not consider the possibility for copepods to respond to environmental changes through adaptive evolution [161]. The lack of detailed information on potential effects on zooplankton physiology “currently restricts our ability to reliably predict future impacts” [162].
Mesozooplankton synopsis For copepod species from the genera Acartia and Calanus, adult survival rates and egg productions rates appear unaffected by OA, even when P CO2 is ‘very elevated’ (Table 1), whereas egg hatching rates are negatively affected and egg hatching success remains uncertain. Cumulative impacts across life stages are unknown. Thus, the effects of OA on mesozooplankton will likely be neutral and possibly negative (Fig. 4A).
Pteropods
In BC waters only three species of pelagic snail, or pteropod, have been regularly observed [164]. Limacina helicina (shelled) is by far the most common of these three, occurring throughout most of the year, generally in the upper 100 m [164] and occasionally forming strong blooms (> 1000 m−3) which can dominate the plankton (M. Galbraith, pers. comm., Institute of Ocean Sciences, Sidney BC). Clione spp. (naked) is also often present, although at significantly lower numbers. These two species are common in the Strait of Georgia and less so in Hecate Strait (Fig. 1); they are also found on the outer BC shelf and in the Alaskan Gyre (M. Galbraith, pers. comm.). Clio pyramidata (shelled), a subtropical species, is present only episodically along the WCVI [165]. Pteropods are an important food source for fish (especially juvenile salmon [166]), birds and marine mammals [167, 168]. Most pteropods produce aragonitic shells [167] and those that don’t (naked pteropods) feed almost exclusively on the shelled species, making all pteropods susceptible to OA [164].
Direct effects It is difficult to keep pteropods in laboratory conditions [164] due to their delicate feeding structure [167]. Thus, few controlled experiments on live animals have been made until recently, and sample size remains limited. Most of these experiments have been conducted on (variants of) L. helicina harvested from Arctic and Antarctic waters (S2 Table).
Shells of dead pteropods dissolve in waters undersaturated with respect to aragonite, (e.g. [169, 170]) as expected. Live individuals, which may form protective biological coatings on the exterior of their shell [171] and/or actively counteract dissolution [170] also show evidence of dissolution when harvested from waters under, or near, saturation with respect to aragonite [172–174] (S3 Table). Similarly, live individuals incubated for short periods under the high end of present-day P CO2 (0–100 m, Fig. 2) and elevated P CO2 (Table 1) show reduced calcification (e.g. [170, 175]; S2 Table). In one experiment the larval state failed to calcify at all [176].
Despite the negative impacts on shell quality and maintenance, many (and in some cases all, e.g. [175]) animals studied survived their respective treatments (e.g. [170, 177]). However, the reduction of shell formation will impact the pteropods’ ability to control buoyancy and withstand predation [167]. In addition, as P CO2 rises, increased energetic costs associated with maintaining their shells are likely, particularly as temperature increases [170]. The ability to supply energy to perform these (and other) tasks may be suppressed, [178] although some pteropods are likely to be more resilient than others (e.g. [179], S2 Table).
Pteropod synopsis In summary, there is a clear cause for concern about the future of pteropods and the animals that depend on them. Although in the last several decades pteropods make up, on average, only about 5% of the average annual zooplankton biomass in BC waters (M. Galbraith, pers. comm.), they are an important food source for juvenile Pink Salmon [166] and are related to Pink Salmon survival [180] (see Fish—Indirect effects). Already in our region, where aragonite saturation horizons are frequently shallower than 100 m [11, 31, 32], numbers of the most common pteropod have declined significantly [164].
Molluscs
Molluscs comprise a diverse group of organisms that includes a variety of shellfish as well as predators such as squid and octopus (and pteropods, above). In the northeast Pacific, mussels dominate rocky intertidal zones (e.g. Mytilus californianus [181]) while oysters (mainly the Pacific Oyster, Crassostrea gigas), clams (family Veneridae) and cockles (family Cardiidae) are commonly found on beaches [182]. Geoduck Clams and scallops live significantly deeper (∼10–20 m and 15–45 m, respectively) as do squids and octopuses (∼15–140 m, Fig. 3). Shellfish consume plankton through filter-feeding and are able to significantly reduce plankton concentrations on a local scale (e.g. [183]), making them strong indicators of water quality [184, 185]. In turn, shellfish are preyed upon by many animals including sea otters, octopuses, seabirds and sea stars [186, 187].
The annual landed value of molluscs harvested from wild and farmed fisheries in BC is $63 million (Fig. 4), of which 66% is Geoduck Clam (Panopea abrupta). Other major harvested clams are Manila Clam (Venerupis philippinarum), Native Littleneck Clam (Leukoma staminea), Butter Clam (Saxidomus gigantea) and Varnish (Savoury) Clam (Nutallia obscurata) [188]. The Pacific Oyster was introduced into BC waters in the early 1900s and is used in aquaculture, while the native Olympia Oyster (Ostrea conchaphila) is no longer harvested [189, 190] and is listed as Special Concern under the Canadian Species at Risk Act (SARA). There are small fisheries for Pink Scallop (Chlamys rubida) and Spiny Scallop (Chlamys hastata) [191]; a commercially-developed hybrid called “Pacific Scallop” (Patinopecten caurinus x yessoensis) is used in aquaculture. There is a small but growing mussel industry, no harvest for Northern Abalone (Haliotis kamtschatkana) as it is listed by SARA as Endangered, and minor harvests for squid and octopus.
Direct effects Shelled molluscs calcify internally and actively increase pH at that site to do so, making them directly vulnerable to OA [16, 17]. Larval shells are particularly vulnerable since they are mostly composed of aragonite [192, 193] and for at least a few species the initial deposit is amorphous CaCO3 (the least stable form of CaCO3) [192]. By adulthood, shells are composed of aragonite and/or calcite, depending on the species [192, 193]; e.g., oyster shells are mainly calcite [194]. To deal with vulnerability at the larval stage (e.g. [195]), mollusc aquaculture in the northeast Pacific relies on hatcheries (often with controlled conditions) to rear larvae that are then distributed to growers.
Experiments to quantify OA effects on shellfish have yielded a range of conclusions [36, 196]; however, with the advancement of the field, results are beginning to converge. Kroeker et al. [36] found that OA significantly reduced calcification (by 40%), growth (by 17%) and development (by 25%) in molluscs. Another recent review [197] found that 37 of 41 studies on calcification by molluscs reported significant negative effects following exposure to increased CO2 levels. Here we summarise experiments performed on species that are found in the northeast Pacific and elsewhere (e.g. scallops). There have been no studies on Geoduck Clams (despite their commercial importance), or on BC scallop species.
Experiments on fertilisation in Pacific Oyster have produced mixed results. Both sperm swimming speed and egg fertilisation success can be unaffected [198] or decline [199, 200] under elevated P CO2 (Table 1). Within two days of fertilisation, Pacific Oyster larvae precipitate >90% of their body weight as CaCO3, using limited energy reserves in eggs [17]. Early development (up to 8 h) remains unaffected at elevated P CO2 [201]; however, the number of embryos reaching the planktonic ‘D-veliger’ larval stage declines [199–201]. Elevated P CO2 increases the number of larvae with shells one day after fertilisation (due to an enhanced metabolic rate), yet decreases it three days after [202]. Larval survival of Pacific Oysters is unaffected by P CO2 after three and 16 days [202, 203]. Species that do exhibit a decline in larval survival are Northern Abalone [204] and Bay Scallop (Argopecten irradians) [205].
Metamorphosis from larvae to juveniles is affected differently for different species under elevated P CO2. For Olympia Oyster, the proportion of metamorphosing larvae declines [206, 207] and size at metamorphosis decreases [206]. Similar results, plus a delay in metamorphosis and reduction in survival, are usually seen for Bay Scallop [205, 208–210]. However, for Northern Abalone from the WCVI the proportion of metamorphosing larvae is unaffected [204]. Increased abnormalities in larvae have been observed under elevated P CO2 in Pacific Oyster [199–201] and Northern Abalone [204]. In the latter species, shell abnormalities increased substantially, occurring in 99% of larvae at P CO2 1800 μatm [204]. These abnormalities did not appear to affect survival rates in the laboratory, but in the field the abnormal larvae would be more susceptible to predation [204].
The size of D-veliger larvae of Pacific Oyster decreases [199–202, 211] and shell growth of later larval stages generally declines [199, 201] under elevated P CO2, though not always [199, 203]. Decreases in larvae shell growth also occur in Olympia Oyster [207, 212], Northern Abalone [204] and Bay Scallop [208–210]. Molecular analyses show that expression of proteins related to calcification and cytoskeleton production can be severely suppressed under high P CO2 [211]. For Northern Abalone larvae, settlement (attachment to the experimental container) is unaffected by P CO2 [204]. Additional effects on other larvae include decreased O2 consumption and feeding rates [203], and reduced lipid content [209, 210].
Shell growth and calcification of juvenile and adult molluscs under OA remains uncertain due to limited studies with contrasting results. Pacific Oyster juveniles exhibit increased expansion of shell area (but not thickness) under reduced pH, despite declines in O2 consumption and feeding rates of larvae [203]. In juvenile Bay Scallops, elevated P CO2 (Table 1) does not affect shell and tissue growth but does reduce survival [209]. Declines in calcification rates have been observed for Pacific Oyster juveniles and adults under elevated P CO2 [213] and for adult Zhikong Scallops (Chlamys farreri) under reduced pH [214].
The byssal threads that mussels use to attach themselves to rocks or vertical lines in aquaculture must be robust so that they do not drop off or get ripped off. The threads of the common mussel (Mytilus trossulus) have been shown to weaken under elevated P CO2 [215], although they may be more sensitive to temperature during during short-term fluctuations typical of local inlets (L. Newcomb, University of Washington, Seattle pers. comm.).
Metabolic rates of juveniles and adults appear to be generally unaffected by OA alone [216–218]. Also unaffected, at least in juvenile King Scallops, are clearance rates, growth rates, the ratio RNA:DNA (suggesting no effect on growth potential) [217], and various measures related to ‘clapping’ (rapid closing used for locomotion) by adults—frequency, recovery time between claps and clapping fatigue [218]. The latter study, however, did find a reduction in the force exerted by the clapping under elevated P CO2, which could reduce the scallops’ ability to escape predators.
As above, the larval stage is vulnerable to OA. South of BC, at a hatchery for Pacific Oyster in Oregon (USA), carbonate levels experience large fluctuations due to strong coastal upwelling [195]. Negative correlations were found between the aragonite saturation state (Ωarag of water in which larvae were spawned and reared, and the resulting larval production and mid-stage growth [195]. In the laboratory, the shell growth rate of juvenile Olympia Oysters depends on pH exposure at the larval stage but not at the juvenile stage [212]. To test such carry-over effects in a natural system, Olympia Oyster larvae were reared under different P CO2 levels, then transferred to field sites after metamorphosis [206]. Juvenile survival was not significantly different between the two larval treatments, but the elevated-P CO2 larvae yielded smaller juveniles, suggesting that they suffer irreversible damage (e.g. energy deficit, abnormality, inability for compensatory growth) [206].
Indirect effects Changes in species composition can be expected under OA. Few studies explore these changes for molluscs, however it has been shown that Eastern Oyster larvae (Crassostrea virginica) have higher survival rates than Bay Scallops under elevated P CO2, which is the opposite of the present-day P CO2 result (and in the absence of brown tides—in this study caused by a temperate phytoplankton species not found in the northeast Pacific) [210]. Thus, scallops may be affected by OA more than oysters. Scallops are also sensitive to other anthropogenic stressors, such as eutrophication [219], while the impact of these conditions on oysters and other shellfish was not investigated.
OA may increase the vulnerability of shelled molluscs to predation by thinning their protective shells and may also cause food web shifts. For example, Boring Sponges (Cliona celata) can bore twice the number of holes in Bay Scallop shells, and remove twice the weight of shell, at pH 7.8 compared to pH 8.1, despite taking longer to attach themselves to the shells [220]. Negative impacts on molluscs could also have large unintended consequences for other species [221]. Shell production and aggregation provide refuge for other organisms such as sponges and crabs, and introduce complexity and heterogeneity into benthic environments, with heterogeneity being important for maintaining species richness [221]. Thus, the direct effects of OA on molluscs may have detrimental effects at the ecosystem level.
Squid and octopus In BC, there are at least 30 species of squid and eight species of octopus [222], none of which have been studied for OA effects. Common species in BC waters are Opal Squid (Loligo opalescens) and Northern Giant Pacific Octopus (Enteroctopus dofleini). Similar to the otoliths of fish (see below), squids have internal calcified structures called statoliths used for sensing gravity and movement [223]. Under elevated P CO2 statoliths in embryos of the European Squid, Loligo vulgaris, are significantly larger than those formed under present-day P CO2 [224]. At higher P CO2 (still in the elevated range—Table 1), Kaplan et al. [225] observed reduced surface area, malformation, and abnormal crystalline structure in statoliths of Atlantic Longfin Squid, Doryteuthis pealeii. Aside from calcification, elevated P CO2 also leads to increased heavy metal retention in the protective eggshells and changes to the bioaccumulation of silver, mercury and cobalt in larval tissue [224]. Additionally, elevated P CO2 depresses metabolic rates in pelagic squids (e.g. [226]). The ultimate effect on fitness is not known.
Mollusc synopsis We conclude that the effects of OA on shelled molluscs will be negative based on available studies on oysters, scallops, abalone and mussels (Fig. 4). These negative effects occur at various life-history stages, and go beyond direct effects on calcification of larvae, e.g. reduced oxygen consumption and feeding rates of larvae and delayed behavioural responses of adults. It is generally anticipated that effects on larval survival rate and reproduction rate will directly influence population size, population distribution and community structure [227]. No experiments were found on local clam species (including geoducks) but given the results on other molluscs [36] we anticipate that they will also be negatively affected by OA, while effects on squid and octopus remain uncertain (Fig. 4).
Sponges and Coldwater Corals
Sponge reefs are globally unique to the northeast Pacific coast [228–230] and all four groups of cold-water corals: octocorals, stylasterids, stony and black corals, are present in the region. They occur where productivity and water flow are high (e.g. they are especially dense on seamounts and the heads of canyons, Fig. 1) and from the surface to depths >2000 m [231]. However, due in part to the depth range, very few benthic habitat mapping data exist along the BC coast (e.g. [232], Kim Conway, pers. comm., Pacific Geoscience Centre, Sidney, BC) and so we have used these data and the expertise of others to provide our own general description (below). Sponges and cold-water corals form important habitat for many marine organisms including species of fish that are commercially important (e.g. the rockfish Pacific Ocean Perch) in our region [233–236].
The coral and sponge contribution to the benthic fauna in BC appears to be patchy but diverse, based on: DFO trawl survey and observer records [237], comparison with neighbouring regions (e.g. [238, 239]), isolated studies (e.g. [229, 235]), anecdotal evidence (Lynne Yamanaka, pers. comm., Pacific Biological Station, Nanaimo, BC), and modelling work (e.g. [240]). This collection is likely dominated by siliceous sponges, and isolated stands of flexible corals with partly organic skeletons (octocorals), more specifically members of the diverse group Alcyonacea (e.g. large tree form coral) and pennatulaceans (sea pens and whips). Alcyonacea and solitary glass sponges occur on bedrock, mainly deeper than ∼200 m, while pennatulaceans and glass sponge reefs grow on flat sediment, generally shallower than ∼200 m [241].
Stylasterids (e.g. [242]) and stony corals (Scleractinia) also occur [237, 243], but primarily in small, solitary patches. The reef-forming scleractinian Lophelia pertusa has been found [244], but is rare, possibly influenced by the already low aragonite saturation states in this region [245]. Black corals, which do not calcify and are made of organic proteins, are also present below 500 m [237].
Direct effects OA studies have focused on stony corals, primarily Lophelia pertusa, which is entirely aragonitic. They show an increased energetic cost for calcification in L. pertusa with decreasing pH (and Ωarag [246, 247] (S2 Table); however, L. pertusa may adapt to moderate decreases in pH given sufficient time [248] (S2 Table). The holdfasts and some parts of the structure of many octocorals are also made of aragonite [249]. Similarly, some stylasterids precipitate aragonite as well as calcite [250]. However, neither octocorals nor stylasterids have been studied with respect to OA to date. Likewise, there are no OA studies specific to glass sponges.
Sponge and coral synopsis The OA response of the cold-water corals most common in our region (octocorals) has not yet been studied. While the skeletons of these corals are partly organic, they also calcify and so may be affected by OA at some level (Fig. 4A). There are no OA studies on glass sponges to date. Loss of coral and sponge habitat would have a negative impact on many fish species, particularly juvenile rockfish [233–235].
Echinoderms
Echinoderms form a marine set of invertebrate animals with ∼7000 known species worldwide [251] and 217 species recorded in BC [252], half of which occur exclusively at depths > 200 m [253]. The echinoderms comprise five classes: (i) echinoids (sea urchins and sand dollars), (ii) asteroids (sea stars), (iii) holothuroids (sea cucumbers), (iv) crinoids (sea lilies and feather stars), and (v) ophiuroids (brittle stars). A few are considered to be “keystone” species, such as the Purple Sea Star (Pisaster ochraceus) [254, 255], which is common along the BC coast. Echinoderms modify ecosystems (e.g. by mixing and transforming sediments, grazing kelp forests, preying on mussel beds) and provide food for carnivorous fish, shellfish, and marine mammals (e.g. sea otters prey heavily on sea urchins and sea cucumbers). In addition, sea stars and sea urchins act as important grazers in the sub-littoral zone [256].
Direct effects Green and Red Sea Urchins (Strongylocentrotus droebachiensis and S. franciscanus, respectively) harvested in BC generate significant income (Fig. 4A). Clark et al. [257] found that larval growth and skeletal calcification were reduced at lower pH levels for select species (see S2 Table) ranging from the tropics to the poles; no changes in skeletal morphology occurred. Studies on shell thickness are confounded by effects of diet and experiment length [125, 258], but urchins have higher growth rates when fed on calcifying algae and may derive some portion of essential elements (e.g. calcium, magnesium) from the algae [258]. Therefore, sea urchins may suffer as the proportion of calcifying macroalgae in their diet declines due to direct OA effects on these algae (see Macroalgae section above). In long-term studies, sea urchins have shown an ability to adapt to elevated P CO2 (Table 1); however, in the transition to new OA conditions, species may suffer from life-cycle carry-over effects. For instance, Dupont et al. [259] demonstrated that under elevated P CO2 females acclimated for four months experienced a 4.5 decrease in fecundity and produced offspring that suffered 95% juvenile mortality; however, these effects disappeared after acclimitisation for 16 months (S2 Table). OA may also influence reproduction in echinoderms. For example, as P CO2 increases under OA, higher sperm concentrations are necessary to achieve high fertilisation success in the sea urchin S. franciscanus, and the egg’s mechanism for blocking fertilisation by multiple sperm cells becomes slower [260].
A number of studies have used genetic markers to infer the possible physiological effects of OA in sea urchins (see S2 Table). O’Donnell et al. [261] measured the change in expression of a molecular helper-protein in S. franciscanus and suggested that the ability to handle temperature stress would be reduced under OA. Todgham and Hofmann [262] measured changes in ∼1000 genes of the sea star S. purpuratus and found reduced expression under elevated P CO2 in four categories—biomineralisation, cellular stress response, metabolism, and apoptosis (cell death). Also for this species, elevated P CO2 triggered changes in 40 functional classes of proteins, affecting biomineralisation, lipid metabolism, and ion homeostasis [263].
Giant Red Sea Cucumber (Parastichopus californicus) harvest also provides significant income in BC (sea cucumbers, Fig. 4A) but there are no studies on OA effects for this species. Elsewhere, a single study found that sperm motility of a reef-dwelling sea cucumber species (Holothuria sp.) was impaired at pH values <7.7 [264]. Elevated P CO2 and temperatures have been shown to have positive and additive effects on the relative growth of the keystone sea star Pisaster ochraceus [265]. Under increased P CO2, calcification is reduced [265]; however, growth rate remains unchanged as the endoskeleton is primarily composed of soft tissue with relatively small calcareous elements for rigidity and protection. Brittle stars (ophiuroids) are commonly found in the region, but the effects of OA have only been studied in species found elsewhere. In the eastern Atlantic Ocean, keystone brittle star Ophiothrix fragilis was found to be especially sensitive to small changes in pH [266], with 100% mortality of larvae at pH 7.9 vs. 30% mortality in the control (pH = 8.1). Finally, while Dupont et al. [251] found that echinoderms studied to date are relatively robust to OA effects, they conclude that the overall impact of OA on this group will be negative and suggest that associated ecosystem impacts may be more severe.
Indirect effects Declines in some echinoderms may affect the predators that depend on them, but ecosystem effects remain unknown. For example, on our coast, various nearshore rockfish and numerous flatfish prey on ophiuroids [267], although they only form an important component of the diet for China Rockfish (Sebastes nebulosus), Flathead Sole (Hippoglossoides elassodon), and Southern Rock Sole (Lepidopsetta bilineatus) [267]. Additionally, the deep-water rockfish Longspine Thornyhead (Sebastolobus altivelis) relies on brittle stars for a large proportion of its food [268]. In the eastern Atlantic, the inevitable decline in pH may lead to the disappearance of the keystone brittle star O. fragilis; the impact on the ecosystem is not really known [266].
Echinoderm synopsis Although many echinoderms have not been studied, the existing evidence indicates significant negative effects due to OA, especially at early life stages. Thus, we suggest that this group will be affected negatively (Fig. 4A). Of more concern are the anticipated negative impacts on ecosystems, e.g. declines in the population of a keystone species like the Purple Sea Star would have wide-ranging effects on the food web.
Crustaceans
Marine crustaceans are represented in BC by copepods [269], krill (euphausiids) [39], barnacles [270], shrimps, prawns and crabs [271]. Copepods (see Mesozooplankton) and krill form a substantial biomass in the oceans and provide an important source of food for upper trophic levels in temperate marine foodwebs and act as important grazers (e.g., [272]). Crabs are found in the upper 50 m, while adult prawns (Pandalus platyceros) and adult shrimp (mainly Smooth Pink—Pandalus jordani and Sidestripe—Pandalopsis dispar) are deeper (∼100 m and 120 m, respectively: Fig. 3). Krill, primarily Euphausia pacifica, perform strong diel vertical migration from the surface to depths exceeding 100 m. Krill is harvested on a limited basis in the Strait of Georgia and various inlets [39]. Prawns and shrimps, which are farmed extensively in other parts of the world, are only harvested from the wild in BC; the prawn fishery is substantial (∼$40 million, Fig. 4) [34]. The crab fishery in BC is also valuable (∼$33 million) [34, 273], with Dungeness Crab (Cancer magister) being the most important commercial species.
Direct effects Crustacean exoskeletons, composed of chitin and CaCO3 [274], are generally considered to be unaffected by OA. In fact, evidence suggests that this protective covering actually serves as a buffer to the corrosive nature of OA, and some crustaceans can use the increased DIC in seawater to fortify their shells through calcification [275]. This enhancement of the shell contrasts with shell dissolution in molluscs (see Molluscs Section), and is likely due to some crustaceans (crabs, lobsters) having an efficient proton-regulating mechanism [275]. Despite the advantage of localised pH-regulation, the calcification response appears to depend on a variety of additional factors: external organic coatings, skeletal mineralisation composition (e.g. magnesium content in calcite), and the degree to which amorphous CaCO3 (precursor to calcite/aragonite shells) is utilised [275–277].
Crustacean species’ ability to deal with increasing OA also depends on life-history strategies and habitat [278]. Active species or those in highly fluctuating environments (e.g. intertidal or estuarine) tend to utilise the oxygen-transporting protein haemocyanin, which also confers additional buffering capacity against high H+ concentrations. Sedentary species or those in stable environments (e.g. deep-sea or polar) tend to have less haemocyanin and consequently less buffering capacity. The latter group relies more on buffering and is probably more sensitive to OA.[278]
Recent studies on Alaskan King Crab (AKC, Paralithodes camtschaticus) and Tanner Crab (TC, Chionoecetes bairdi) in Alaskan waters highlight the vulnerability of the early life stages to OA [279, 280]. For AKC embryos and larvae, OA produces larger embryos (but not larger mass), smaller egg yolks, higher developmental rates, and higher calcium content [280]. In juveniles of both species, increased mortality occurs with elevated P CO2 (Table 1), with 100% mortality in their most extreme treatment (S2 Table) [279]. Differences between the two Alaskan crabs (decreased condition index in AKC but not TC and decreased calcium content in TC but not AKC) suggest that AKC puts more energy into osmoregulation and calcification than does TC [279]. Additionally, there is some preliminary evidence that adult AKC females fail to moult [280].
Initial studies are underway on the dominant local species of krill, Euphausia pacifica. A recent study in Puget Sound, WA (Fig. 1), found that elevated P CO2 slowed the development of hatched nauplii to the first feeding stage (Anna McLaskey, pers. comm., University of Alaska, Fairbanks AK). Also, under higher P CO2 the Antarctic krill species, Euphausia superba, experiences ingestion rates 3.5 times higher than those under present-day conditions, and consistently higher metabolic rates [281].
For the cold-water barnacle, Semibalanus balanoides (common in BC), experimental treatments at elevated CO2 (S2 Table) reduced adult survival and slowed embryonic development, which delayed the time of hatching by 19 days [282]. The cold-water shrimp, Pandalus borealis (common and commercially important in BC), also exhibited delayed juvenile development at reduced pH [283]. Other studies find no such delays [284–286], though significant effects have been observed when temperature and P CO2 interact [285]. The ability to tolerate OA also depends in part on prior exposure to habitats that experience highly fluctuating P CO2 [287].
Indirect effects Slow embryonic development [282] could potentially cause a timing mismatch between larval release and prey availability related to the spring phytoplankton bloom [288]. Potentially slower growth and lower fitness in juveniles and young adults may reduce egg production by females over their lifetime [279]. Despite the stability of adult exoskeletons, the post-moult calcification stage in crustaceans may be delayed significantly under elevated P CO2 [278], which may increase mortality due to predation on this defenseless life stage (e.g. [289]). Additionally, Kunkel et al. [290] hypothesise that OA may degrade the thin outer layer of calcite, which helps protect decapods from microbial attack. Finally, stock assessment models that incorporate reduced recruitment survival as a function of OA suggest that there can be a substantial socio-economic cost that is currently not recognised by decision makers [291].
Crustacean synopsis Generally, the crustaceans are expected to be sensitive to OA effects at early life cycle stages, while available studies suggest mixed results for adults. However, many local species, such as prawns, have not been studied (Fig. 4A). There is evidence that developmental anomalies in embryos and larvae occur at reduced pH, which may affect the fitness of juveniles and adults; however, the effects are species-specific and phenotypic adaptation is not known. Additionally, changes in growth rate and calcification may increase the susceptibility to predation, and delays in development may decouple life cycle timing between larval release and optimal foraging conditions.
Fish
In BC coastal waters, there are over 300 species of marine fish [292, 293]. The taxonomic groups represented in BC include jawless fish (e.g. hagfish (270–1010 m)), cartilaginous fish (e.g. ratfish (50–380 m), dogfish (50–430 m), sharks (90–1020 m), skates (50–860 m)), and bony fish. The latter group includes important contributors to BC fisheries—Pacific Herring (Clupea pallasi, 5–170 m), salmon (five species of Oncorhynchus, mostly in the surface 50 m but some species deeper than 100 m), Pacific Hake (Merluccius productus, 80–700 m), Pacific Cod (Gadus macrocephalus, 50–300 m), Walleye Pollock (Theragra chalcogramma, 50–300 m), rockfish (at least 36 species of Sebastes (70–470 m) and two species of Sebastolobus (160–1010 m)), Sablefish (Anoplopoma fimbria, 70–970 m), Lingcod (Ophiodon elongatus, 50–310 m), Arrowtooth Flounder (Atheresthes stomias, 60–600 m), soles and flounders (∼18 species, 50–860 m), and Pacific Halibut (Hippoglossus stenolepis, 50–490 m). Depth distributions for valuable BC fisheries (Fig. 4) appear in Fig. 3. Marine fish species are economically important (GDP of capture fisheries, aquaculture, and sport fishing in BC was over $340 million in 2011 [29]) and ecologically valuable because of their roles providing food sources to higher trophic levels (e.g. birds and mammals) and cycling nutrients to other ecosystems (e.g. salmon providing nutrients to coastal terrestrial ecosystems [294]).
Direct Effects In general, we expect that adult fish will be tolerant of OA because they can control ion concentrations through evolved regulatory mechanisms [295, 296]. In particular, active fish exhibit transient elevated metabolic rates and highly variable extracellular CO2 and proton concentrations. Acid-base imbalances are regulated by specialised gill epithelia, which compensate for pH disturbances caused by exposure to increased environmental P CO2 [296]. Although some studies suggest that aerobic performance of tropical fishes may decline under elevated P CO2 [297] (Table 1), detrimental effects were not found in a temperate species, Atlantic Cod, under elevated P CO2 (e.g. [295]).
The effects of lake acidification on diadromous fish (those migrating between marine and fresh water) are well known, but using these observations to suggest OA effects is potentially misleading due to (i) large physiochemical differences between fresh and acidified marine waters and (ii) high physiological variability between diadromous and marine species [298, 299]. Also, fluctuations in in [H+] seen in lake acidification are orders of magnitude greater than those in the ocean [298].
As with the invertebrates, OA effects in fish are expected to occur during the vulnerable developmental stage, and these effects appear to be species specific. The acid-base regulatory mechanisms of the larval stage remain rudimentary until gills have formed and respiration switches from cutaneous to branchial [300]. Developmental responses are thought to be more the result of CO2 toxicity rather than through pH acting alone [301, 302].
There are limited OA studies on fish species that occur in our region. Hurst et al. [303] showed that the effects of OA on the growth of Walleye Pollock larvae were minor and varied greatly within treatments (S2 Table, Fig. 4). Slightly higher growth rates in elevated P CO2 conditions (Table 1) proved non-significant. Other studies on Atlantic temperate fish species (cod and herring), closely related to those in BC waters, found no significant effects on sperm motility, embryogenesis, egg survival, or the development of skeletal, heart, and lung tissue [300, 304, 305]. Despite these benign effects, researchers have found some developmental anomalies. Franke and Clemmesen [305] showed an inverse relationship for Atlantic Herring between P CO2 and the ratio RNA/DNA at hatching, potentially reducing protein biosynthesis and growth. Frommel et al. [300] found significant tissue damage in liver, pancreas, kidney, eye, and gut of Atlantic Cod larvae under elevated P CO2 Baumann et al. [306] demonstrated that increasing P CO2 caused a 74% reduction in survival and an 18% reduction in length of embryos of a ubiquitous estuarine fish called Inland Silverside (Menidia beryllina). Any significant developmental effect could alter the abundance and diversity of marine fish populations.
Otoliths (ear bones) are aragonite-based structures that fish use to sense acceleration and orientation. In some species, otoliths grow larger when larval fish are exposed to elevated P CO2 (e.g. White Sea Bass, a species found in BC waters [307]; Atlantic Cod [308] and tropical clownfish [309]). Under elevated P CO2 pH is regulated in the endolymph sac surrounding the otolith resulting in increased CaCO3 precipitation and enhanced otolith growth for those species [309]. An increase in otolith size may enhance hearing range [310], which might help or harm fish depending on sensitivity to important auditory cues or disruptive background noise [310].
Behavioural responses have recently been documented at elevated P CO2 for larvae of tropical reef fish. In particular, behaviour to olfactory, auditory, and visual cues changes when larvae are selecting habitats and responding to predators [311–315]. Additionally, elevated P CO2 reduces learning abilities related to predator avoidance [316] and changes the propensity of larval reef fish to turn left or right (lateralisation) [317]. These behavioural changes can expose larval fish to increased mortality risk, which has important fitness consequences [313, 318]. Given possible behavioural effects on predators as well as prey under elevated P CO2 community-level responses are difficult to predict [318, 319].
Relatively few studies have investigated behavioural changes to OA in temperate species (three exceptions being [320–322]), and none have examined commercially important species in BC waters. The larvae of Threespine Stickleback (Gasterosteus aculeatus), a species found in marine and fresh water on the BC coast, exhibit behavioural disturbances (e.g. reduction in boldness and curiosity), compromised learning abilities, and declines in lateralisation when reared in elevated P CO2 [320]. These responses are surprising given the physiological plasticity of this species, which is expected to confer enhanced acclimatisation abilities to environmental challenges. These results suggest that sensitivity to OA is not limited to species occupying narrow ecological niches, such as tropical reef fish [320].
Elevated P CO2 can disrupt the functioning of GABAA (γ-Aminobutyric acid) receptors, the main inhibitory neurotransmitter receptors in the fish brain [323]. Normally, the opening of these receptors results in an inflow of Cl− and ions over the neuronal membrane, leading to inhibition of the neuron. When concentrations of intracellular Cl− and are altered (e.g, when fish with strong acid-base regulatory systems are exposed to higher environmental P CO2, the flow of ions can be reversed, resulting in neuronal excitation instead of inhibition. Such changes have been associated with dramatic shifts in behaviour and sensory preferences in larval tropical reef fish [323], but the effects on temperate species are unknown. Although these receptors are shared by many, if not most fish, the resulting behavioural responses will likely vary due to species-specific differences in acid-base regulatory systems [323].
Indirect effects Fish will likely be affected indirectly by OA through food-web interactions. Off the southern WCVI, the pelagic system is dominated by Pacific Hake, Pacific Herring, Spiny Dogfish, and Chinook Salmon (Oncorhynchus tshawytscha), all largely dependent on krill production in the region [324]. This area has also been described as a “toxic hot spot” due to consistently high levels of Pseudo-nitzschia species and the presence of domoic acid [325]. These neurotoxins are transferred to higher trophic levels [59], and as P CO2 increases under OA the toxicity of these blooms may also increase [93].
Many fish species of the north Pacific Ocean prey on shelled pteropods (e.g. cod, pollock, mackerel) and a decline in pteropod abundances may lead to a shift in diet toward greater predation on juvenile fish such as salmon [326]. Pteropods (see Pteropods—Indirect effects) are also an important food source for Pink Salmon in the first year of marine life [166]. Because pteropods often exhibit swarming behaviour, foraging costs are relatively low for Pink Salmon feeding on patches [166, 180], possibly enhancing growth in early marine life and increasing adult biomass [327]. Reductions in pteropod densities may therefore have significant impacts on Pink Salmon biomass (Fig. 4A).
Trophodynamic modelling can suggest possible impacts of OA on fish populations. One study [328] explored various scenarios under OA, one of which assumes a significant mortality on benthic shelled invertebrates (e.g. bivalves, corals, sea urchins, sea stars) that leads to a biomass reduction for fish that feed on these species. While both English Sole (Parophrys vetulus) and small demersal sharks (e.g. Spiny Dogfish) rely on these invertebrates for only 10% of their diet in the model, English Sole experiences a much bigger decline due to a lack of alternative prey items. Another OA scenario in [328] adds an additional mortality on large zooplankton and small phytoplankton, which leads to a large increase in microzooplankton, detritus, and bacteria. In this scenario, the model predicts various higher-order interactions: a reduction of Lingcod due to a decline in macrozooplanktonic prey; an increase in Canary Rockfish (Sebastes pinniger) due to an increase in sea urchins and shrimps; and the increase of nearshore rockfish due to a decline in one of its predators, Lingcod. While there are many possible outcomes using such modelling tools, they do highlight how effects from OA on any single biological component can affect the entire trophic web.
Fish synopsis In general, we expect that adult fish will be tolerant of OA because of their ability to control internal ion concentrations. However, OA may affect fish during vulnerable developmental stages, though evidence for these effects is weak for species in BC. Perhaps more importantly, behavioural responses to OA have been widely documented in tropical reef fish, resulting in reduced survival. Similar effects may occur in temperate species, though studies in this area are limited. OA-induced reductions in availability of some prey species may reduce fish growth and survival, though these effects may be tempered by prey-switching. Possible increases in HABs would have a negative impact on farmed fish and shellfish; wild fish might increasingly suffer the effects of biotoxin accumulation.
Marine mammals
British Columbia is host to a large and diverse group of marine mammals (∼30 species [329]), many of which have experienced dramatic population increases over the last century when hunting and culling practices were discontinued (e.g. on Grey Whales (Eschrichtius robustus) and Harbour Seals (Phoca vitulina), respectively) [330]. In addition to their role as top predator in the marine food web and their contribution to ecotourism, these mammals are iconic symbols of the region. Thus, they are valuable, but their value is difficult to assess (e.g. [331]).
In general, marine mammals cover an appreciable geographic range and many are able to dive to remarkable depths [332]. Their physiology is adapted to high pressures and they have an exceptional capacity for O2 [332]. Because they breathe at the surface, they are not susceptible to acidosis in the way that many other complex marine organisms will be as carbon levels increase (e.g. [302]). Therefore, direct impacts of OA on marine mammals are not expected, and have not been investigated (Fig. 4A). Indirect food web impacts are anticipated, e.g. for cetaceans that rely heavily on cephalopods or zooplankton such as pteropods [333]. In addition, underwater sound absorption at low frequencies (relevant for marine mammals) will decrease with OA [334]. However, this decrease is projected to be small (less than 0.2 dB) over the next few centuries and negligible in the context of the current noise associated with shipping [335].
Marine mammal synopsis Marine mammals will likely be affected by OA indirectly through food web changes, however direct impacts are not anticipated. While noise levels will increase with OA, this increase will not be large enough over the next few centuries to affect animals that rely on underwater sound.
Discussion
We have described the marine ecosystem in the temperate coastal northeast Pacific region at present, and then its response to OA. However, the available information is limited. For some organisms, no OA studies exist (e.g. Geoduck Clam, rockfish, S1 Table). In general there are more studies, with respect to distributions and OA impacts, on species that are easier to observe, are of commercial value (e.g. oysters, S1 Table) or that threaten human health (e.g. harmful algae, S1 Table). The results of studies like these are often adopted when similar research on native organisms is not available (as we have done), limiting the ability to predict responses with confidence. Furthermore, OA is only one aspect of climate change and predicting shifts in marine ecosystems, and the degree to which they are caused by natural or anthropogenic forcing, is a highly complex problem. In the following, we discuss these and other issues that influence our evaluation.
Caveats The number of studies related to OA is growing rapidly (e.g. S1 Table). While experiments in these studies are highly valuable, translating their results into changes in the real world is challenging. For example, wild populations of marine organisms will adapt (both physiologically in a single lifespan and genetically over multiple generations) to their changing environment, which is difficult or impossible to capture in vitro. However, using temperature-dependent adaptation as a guide, Kelly and Hofmann [336] caution that the ability to adapt to changing pH may be limited.
In addition, food-web interactions and responses to OA are extremely difficult to predict, but will influence marine populations and could tip the balance from an overall negative impact to a positive one for a given species if a key predator is removed. Ecosystem effects resulting from OA have previously been identified as a key knowledge gap [337]. Furthermore, different life stages, particularly the juvenile stage (e.g. echinoderms), often display increased susceptibility to OA, but the impact of exposure of one life stage to low pH conditions on the subsequent life stages has only rarely been studied (but see [206, 212]). Similarly, even in organisms that have been comparatively well studied, not all life stages have been considered and certainly not within the context of the variability in natural conditions (Fig. 2).
Manipulated experiments generally consider present-day atmospheric conditions (∼360–400 μatm) to be the control P CO2 level and all treatments above that to be ‘elevated’. Meanwhile P CO2 varies significantly with depth, and is naturally high in the north Pacific [28]. We quantify ‘elevated’ based on the local P CO2 levels at the depths of the organisms in question (Table 1). The combined effect of coastal upwelling, and local remineralisation of high production [27], results in exceptionally high (and variable) subsurface P CO2 on the outer BC shelf (Fig. 2). In local and connected inshore waters subsurface P CO2 is also high (unpublished data, DI; [6, 31]). Thus, many marine organisms in our region are currently experiencing conditions that are viewed as ‘elevated’ in the literature (Fig. 2; S2 Table). In addition, laboratory treatments often specify environmental conditions (e.g. temperature, P CO2) that do not occur in nature and are unlikely to occur, at least locally (e.g. [31]). Exposure time may also limit the interpretation of results, as there are distinct differences between treatments that are ‘shocked’ and those that are allowed to acclimate (e.g. [87, 248]).
Finally, defining the carbon state in seawater is not trivial [10] and requires that at least two of the four carbon parameters (DIC, TA, P CO2, pH) be measured. The quality of the measurements and manipulation in the laboratory work cited here is variable. While the high degree of accuracy and precision required by chemical oceanographers [10] is in general not necessary to obtain insight from biological manipulation experiments, the equations that define the carbon system lead to compounding errors when calculating one of the unknowns. Thus, a moderate uncertainty in P CO2 may translate to an estimated pH that has little, or no meaning. We urge the reader to consult S2 Table where all available detail for each experiment cited has been summarised.
Climate change—the whole picture The ocean has absorbed a significant portion of the anthropogenically produced carbon [2] and that has caused on average a 30% change in surface ocean acidity [5]. However the annual variability in surface P CO2 and pH in dynamic regions like the BC [24] and WA [31] coasts is generally more than two orders of magnitude greater than the annual atmospheric increase in CO2. In other words, we expect the OA trend to be present, but overlaid is a signal with large amplitude.
Climate change may alter this dynamic natural cycle so that negative impacts associated with high acidity are experienced earlier in the coastal northeast Pacific than elsewhere, regardless of OA. There are critical times during the year when carbon conditions (particularly in the upper mixed layer; 20–30 m on the outer coast; ∼10 m or less in protected waterways) change dramatically. For example, the spring bloom in the Strait of Georgia causes a large and rapid increase in surface pH (Ben Moore Maley pers. comm., University of British Columbia, Vancouver BC) and the timing of this event varies significantly from year to year [338]. On the outer shelf, the onset of summer upwelling brings lower pH water over the continental shelf and decreases pH (on average) throughout the entire water column. Climate change may alter the strength, timing [339–341], or even the variability in the timing, of such events. Thus, the influence of climate change on weather may play a critical role, that will only be exacerbated as OA progresses.
In addition to changing weather, sea surface temperatures are expected to increase and subsurface O2 is expected to decrease (leading to increased occurrence of hypoxia) with climate change, concurrent with OA. Temperature has a large effect on marine organisms because metabolism increases as the ocean warms, consequently increasing energetic costs. As a result, changes in present-day distributions of marine organisms have already been linked to changes in temperature [342]. Thus, a ‘multi-stressor’ approach is required to understand the net effect of climate change on marine organisms. The net effect of all three stressors (warming, hypoxia and OA) may be synergistic and has been generally described as a narrowing of the thermal ranges in which organisms can perform well, and a decrease in maximal performance [343]. Lastly, changes in human behaviour (e.g. fishing) as climate change and OA progress may also play an important, and possibly additive, role in shaping future marine ecosystems (e.g. [344]).
Conclusions
There remain significant knowledge gaps with respect to the biological impacts of OA on marine ecosystems globally, and locally. The most critical impacts will likely be indirect as a result of food web changes, and so are highly complex and difficult to predict even with extensive study. Furthermore, OA related changes will occur in concert with other climate change impacts that may be even more severe (see above). In particular, increasing temperature and decreasing dissolved oxygen are likely to produce synergistic effects.
The northeast Pacific region naturally has waters low in pH (undersaturated with respect to aragonite) near the surface. Thus, it is potentially more vulnerable to OA than other regions. We summarise the most relevant risks and identify key knowledge gaps, given present-day knowledge, to Pacific Canadian fisheries and marine ecosystems in the order of immediacy and certainty.
Shellfish aquaculture is highly susceptible to OA due to the direct impact of OA on shell formation and the dependence of the industry on hatchery production. These impacts are already experienced in BC (and WA). Wild shellfish experience similar difficulties but have the opportunity to adapt (e.g. [197]) and so will likely not be affected as rapidly and severely.
There are no studies on Geoduck Clams, which are responsible for a lucrative wild fishery and a growing aquaculture industry in BC (although the latter is still in its infancy).
The commercial BC fishery is dominated monetarily by salmon aquaculture. While uncertainty remains low, it is anticipated that the fish-killing alga Heterosigma akashiwo will gain a competitive advantage under OA, making blooms more frequent. Such blooms are already a significant issue for this industry in BC.
Neurotoxins produced by other harmful algae are expected to become more potent under OA. Such blooms already cause shellfish closures in BC. If this increase in toxicity occurs, the shellfish industry will be affected. In addition, these toxins may cause decreased reproductive success, and even mass mortality, at higher trophic levels including fish, seabirds and marine mammals.
Food web changes due to OA (e.g. in BC changes in the species composition of phytoplankton and decline of pteropods) are anticipated but remain unknown, as are the impacts of these lower level changes on higher trophic levels.
Finfish are likely to experience OA impacts through foodweb changes. In BC examples include: the decline of pteropods, that are directly preyed upon by some fish (particularly Pink Salmon), and the anticipated decline of some echinoderms, that are eaten by various species of rockfish and flatfish.
Habitat changes may also have a critical negative impact, in particular for juvenile fish. While these impacts remain highly uncertain, there may be a shift from upright macroalgae to algal turf. Also, local coral species (in BC primarily octocorals) that provide vertical structure may decline. Direct impacts of OA on finfish may also occur, but only at relatively high levels of CO2.
There are few direct OA studies on local finfish species and none on Pacific Halibut and salmon, which drive the sport fishing industry. Similarly there are no studies on the adaptation of these local species to OA and multiple stressors, like temperature and O2, that will be changing at the same time. Because sport fishing dominates fishery related income in BC, this knowledge gap is significant.
Behavioural changes at various trophic levels have been observed (e.g. increased downward swimming in phytoflagellates, decreased detection and avoidance of predators in larval fish) and postulated (e.g. increased movement to OA refugia such as eelgrass meadows). Such behavioural changes might alter the structure of marine communities in BC, and present another knowledge gap.
Crabs may experience negative impacts under OA while other crustaceans significant to the harvest fishery in BC, like prawns, have not been well studied but appear to be more strongly sensitive to temperature than OA. In general, the juvenile stages of crustaceans are most vulnerable to OA, growing more slowly because they need to expend more energy under OA.
Supporting Information
Number of hits by Web of Science for OA-related studies on different groups of animals in March 2013 and March 2014.
(PDF)
The experimental details for all manipulation experiments referred to in this document.
(PDF)
Details (e.g. species, location, and carbon state) for OA-related in situ studies of marine organisms.
(PDF)
Marine animal use protocols in British Columbia, Canada.
(PDF)
Acknowledgments
We thank the following people for their helpful conversations and correspondence: Richard Beamish, Jim Boutillier, Stephen Cairns, Kim Conway, Carol Cooper, Paul Covert, Lyanne Curtis, Nancy Davis, Jason Dunham, Jessica Finney, John Ford, Ian Forster, Moira Galbraith, Nicky Haigh, Christopher Harley, John Holmes, Catriona Hurd, Hyewon Kim, Joanne Lessard, Sally Leys, Dave Mackas, Shayne MacLellan, Patrick Mahoux, Erin McClelland, Kristi Miller, James Murray, Linda Nichol, Miriam O, Chris Pearce, Angelica Peña, Ian Perry, Dave Preikshot, Peter Ross, George Somero, Bob Stone, Karyn Suchy, Terri Sutherland, Curtis Suttle, Ron Tanasichuk, Phil Tortell, Verena Tunnicliffe, and Lynne Yamanaka. Additionally, we thank the following people for their help with data: Leslie Barton, Jennifer Boldt, Linnea Flostrand, Moira Galbraith, Georg Jorgensen, Lisa Lacko, Norm Olsen, and Dave Preikshot. The manuscript benefited significantly from the helpful comments of two anonymous reveiwers. This work results from the project ‘Ocean Acidification Impacts on Marine Ecosystems’, funded by Fisheries and Oceans Canada’s International Governance Strategy program.
Data Availability
Fig. 1: Saltwater finfish and hatchery sites and commercial marine shellfish farms (referenced in Figure caption) are available at: https://apps.gov.bc.ca/pub/geometadata/. R code used to make the figure is available as a Supporting Information file titled S1 Code. Fig 2: Values of PCO2 and pH are calculated from published data (refs 27 and 40). We include these data in the R code used to make the figure in S2 Code. Fig. 3: We include the data used to make this figure—S1 Data—and the R code used to make this figure—S3 Code—as supporting information. Fig. 4: Landed values are available publicly from http://www.env.gov.bc.ca/omfd/reports/Seafood-YIR-2011.pdf (ref 34) and (for euphausiids) from http://www.pac.dfo-mpo.gc.ca/fm-gp/mplans/2013/krill-sm-2013-17-eng.pdf (ref 39). We include the data in the R code used to make this figure in S4 Code.
Funding Statement
The authors were funded by Fisheries and Oceans Canada through the Inter-Governance Strategy (IGS) funding. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1. Intergovernmental Panel on Climate Change (2013) Climate Change 2013: The Physical Science Basis. Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change. Cambridge, UK and New York, USA: Cambridge University Press, 1535 pp. [Google Scholar]
- 2. Canadell JG, Le Quere C, Raupach MR, Field CB, Buitenhuis ET, et al. (2007) Contributions to accelerating atmospheric CO2 growth from economic activity, carbon intensity, and efficiency of natural sinks. Proc Natl Acad Sci USA 104: 18866–18870. 10.1073/pnas.0702737104 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Doney SC, Feely VJFRA, Kleypas JA (2009) Ocean acidification: the other CO2 problem. Ann Rev Mar Sci 1: 169–192. 10.1146/annurev.marine.010908.163834 [DOI] [PubMed] [Google Scholar]
- 4. Intergovernmental Panel on Climate Change (2011) Workshop Report of the Intergovernmental Panel on Climate Change Workshop on Impacts of Ocean Acidification on Marine Biology and Ecosystems. Stanford, USA: Working Group II Technical Support Unit, Carnegie Institution, 164 pp. [Google Scholar]
- 5. Raven J, Caldeira K, Elderfield H, Hoegh-Guldberg O, Liss P, et al. (2005) Ocean acidification due to increasing carbon dioxide. The Royal Society 12/05: viii + 60p. [Google Scholar]
- 6. Feely RA, Alin SR, Sabine CL, Warner M, Devol A, et al. (2010) The combined effects of ocean acidification, mixing and respiration on pH and carbonate saturation in an urbanized estuary. Estuar Coast Shelf Sci 88: 442–449. 10.1016/j.ecss.2010.05.004 [DOI] [Google Scholar]
- 7. Doney SC, Mahowald N, Lima I, Feely RA, Mackenzie FT, et al. (2007) Impact of anthropogenic atmospheric nitrogen and sulfur deposition on ocean acidification and the inorganic carbon system. Proc Natl Acad Sci USA 104: 14580–14585. 10.1073/pnas.0702218104 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Howarth RW, Sharpley A, Walker D (2002) Sources of nutrient pollution to the coastal waters in the United States: Implications for achieving coastal water quality goals. Estuaries 25: 656–676. 10.1007/BF02804898 [DOI] [Google Scholar]
- 9. Brewer PG (2013) A short history of ocean acidification science in the 20th century: a chemist’s view. Biogeosciences 10: 7411–7422. 10.5194/bg-10-7411-2013 [DOI] [Google Scholar]
- 10. Dickson AG, Sabine CL, Christian JR (2007) Determination of dissolved organic carbon and total dissolved nitrogen in sea water. In: Guide to best practices of ocean CO2 measurements, PICES Special Publication 3. [Google Scholar]
- 11. Feely RA, Sabine CL, Hernandez-Ayon JM, Ianson D, Hales B (2008) Evidence for upwelling of corrosive ‘acidified’ water onto the continental shelf. Science 320: 1490–1492. 10.1126/science.1155676 [DOI] [PubMed] [Google Scholar]
- 12. Kleypas J, Buddemeier RW, Archer D, Gattuso JP, Langdon C, et al. (1999) Geochemical consequences of increased atmospheric carbon dioxide on coral reefs. Science 284: 118–120. 10.1126/science.284.5411.118 [DOI] [PubMed] [Google Scholar]
- 13. Mucci A (1983) The solubility of calcite and aragonite in seawater at various salinities, temperatures and 1 atmosphere total pressure. Am J Sci 238: 780–799. 10.2475/ajs.283.7.780 [DOI] [Google Scholar]
- 14. Sarmiento JL, Gruber N (2006) Ocean Biogeochemical Dynamics. Princeton University Press, 503 pp. [Google Scholar]
- 15. Feely RA, Byrne RH, Acker JG, Beltzer PR, Chen CTA, et al. (1998) Winter-summer variations of calcite and aragonite saturation in the northeast Pacific. Mar Chem 25: 227–241. 10.1016/0304-4203(88)90052-7 [DOI] [Google Scholar]
- 16. Weiner S, Dove PM (2003) An overview of biomineralization processes and the problem of the vital effect, Mineralog. Soc. Am. Geochem. Soc., volume 54 pp. 1–29. [Google Scholar]
- 17. Waldbusser GG, Brunner EL, Haley BA, Hales B, Langdon CJ, et al. (2013) A developmental and energetic basis linking larval oyster shell formation to acidification sensitivity. Geophys Res Lett 40: 2171–2176. 10.1002/grl.50449 [DOI] [Google Scholar]
- 18. Hofmann AF, Peltzer ET, Brewer PG (2013) Kinetic bottlenecks to chemical exchange rates for deep-sea animals—Part 2: Carbon dioxide. Biogeosciences 10: 2409–2425. 10.5194/bg-10-5049-2013 [DOI] [Google Scholar]
- 19. Claiborne JB, Edwards SL, Morrison-Shetlar AI (2002) Acid-base regulation in fishes: cellular and molecular mechanisms. J Exp Zool 293: 302–319. 10.1002/jez.10125 [DOI] [PubMed] [Google Scholar]
- 20. Bach LT, Riebesell U, Shulz KG (2011) Distinguishing between the effects of ocean acidification and ocean carbonation in the coccolithophore Emiliania huxleyi . Limnol Oceanogr 56: 2040–2050. 10.4319/lo.2011.56.6.2040 [DOI] [Google Scholar]
- 21. Riebesell U, Fabry VJ, L H, Gattuso JP (2011) Guide to best practices for ocean acidification research and data reporting. Publications Office of the European Union, Luxembourg: EPOCA—European Project on OCean Acidification, 260 pp. [Google Scholar]
- 22. Volk T, Hoffert MI (1985) Ocean carbon pumps: analysis of relative strengths and efficiencies in ocean-driven atmospheric pCO2 changes. In: Sundquist ET, Broecker WS, editors, The carbon cycle and atmospheric CO2, natural variations archean to present. Washington, DC, volume AGU Monograph 32, pp. 99–110. [Google Scholar]
- 23. Nemcek N, Ianson D, Tortell PD (2008) A high-resolution survey of DMS, CO2, and O2/Ar distributions in productive coastal waters. Global Biogeochem Cycles 22: GB2009 10.1029/2006GB002879 [DOI] [Google Scholar]
- 24. Ianson D, Allen SE (2002) A two-dimensional nitrogen and carbon flux model in a coastal upwelling region. Glob Biogeochem Cycles 16: 10.1029/2001GB001451 10.1029/2001GB001451 [DOI] [Google Scholar]
- 25. Tortell PD, Merzouk A, Ianson D, Pawlowicz R, Yelland DR (2012) Influence of regional climate forcing on surface water pCO2, δO2/Ar and dimethylsulfide (DMS) along the southern British Columbia coast. Cont Shelf Res 47: 119–132. 10.1016/j.csr.2012.07.007 [DOI] [Google Scholar]
- 26. Thomson RE (1981) Oceanography of the British Columbia Coast. 56 Ottawa: Canadian Special Publication of Fisheries and Aquatic Sciences, 291 pp. [Google Scholar]
- 27. Ianson D, Harris S, Allen SE, Orians K, Varela D, et al. (2003) The inorganic carbon system in the coastal upwelling region west of Vancouver Island, Canada. Deep Sea Res I 50: 1023–1042. 10.1016/S0967-0637(03)00114-6 [DOI] [Google Scholar]
- 28. Feely RA, Sabine CL, Lee K, Berelson W, Kleypas J, et al. (2004) Impact of anthropogenic CO2 on the CaCO3 system in the oceans. Science 305: 362 10.1126/science.1097329 [DOI] [PubMed] [Google Scholar]
- 29. Feely RA, Sabine CL, Byrne RH, Millero FJ, Dickson AG, et al. (2012) Decadal changes in the aragonite and calcite saturation state of the Pacific ocean. Global Biogeochem Cycles 26: GB3001 10.1029/2011GB004157 [DOI] [Google Scholar]
- 30. Ware DM, Thomson RE (2005) Bottom-up ecosystem trophic dynamics determine fish production in the Northeast Pacific. Science 308: 1280–1284. 10.1126/science.1109049 [DOI] [PubMed] [Google Scholar]
- 31. Reum JCP, Alin SR, Feely RA, Newton J, Warner M, et al. (2014) Seasonal carbonate chemistry covariation with temperature, oxygen, and salinity in a fjord estuary: Implications for the design of ocean acidification experiments. PLoS ONE 9: e89619 10.1371/journal.pone.0089619 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Ianson D (2013) The increase in carbon along the Canadian Pacific coast. In: Christian JR, Foreman MGG, editors, Climate Trends and Projections for the Pacific Large Area Basin. volume 3032 of Can. Tech. Rep. Fish. Aquat. Sci., pp. xi + 113p. [Google Scholar]
- 33. Stroomer C, Wilson M (2013) British Columbia’s fisheries and aquaculture sector, 2012 Edition. British Columbia Statistics, Canada, 104 pp. [Google Scholar]
- 34.BC Ministry of Agriculture (2012) British Columbia Seafood Industry 2011 Year in Review. British Columbia Ministry of Agriculture, ii + 14 pp. URL http://www.env.gov.bc.ca/omfd/reports/Seafood-YIR-2011.pdf.
- 35. Fabry VJ, Seibel BA, Feely RA, Orr JC (2008) Impacts of ocean acidification on marine fauna and ecosystem processes. ICES J Mar Sci 65: 414–432. 10.1093/icesjms/fsn048 [DOI] [Google Scholar]
- 36. Kroeker KJ, Kordas RL, Crim R, Hendriks IE, Ramajo L, et al. (2013) Impacts of ocean acidification on marine organisms: quantifying sensitivities and interaction with warming. Glob Change Biol 19: 1884–1896. 10.1111/gcb.12179 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Cooley SR, Doney SC (2009) Anticipating ocean acidification’s economic consequences for commercial fisheries. Environ Res Lett 4: 024007 10.1088/1748-9326/4/2/024007 [DOI] [Google Scholar]
- 38.Preikshot DB (2007) The Influence of Geographic Scale, Climate and Trophic Dynamics upon North Pacific Oceanic Ecosystem Models. Ph.D. thesis, University of British Columbia, Canada.
- 39.Fisheries and Oceans Canada (2013) Pacific Region Integrated Fisheries Management Plan—Euphausiids—January 1, 2013 to December 31, 2017. http://www.pac.dfo-mpo.gc.ca/fm-gp/mplans/2013/krill-sm-2013-17-eng.pdf, 52 pp.
- 40. Ianson D, Feely RA, Sabine CL, Juranek L (2009) Features of coastal upwelling regions that determine net air-sea CO2 flux. J Oceanogr 65: 677–687. 10.1007/s10872-009-0059-z [DOI] [Google Scholar]
- 41. Lewis ER, Wallace DWR (1998) Program Developed for CO2 System Calculations. DOE (U.S. Department of Energy) ORNL/CDIAC-105. [Google Scholar]
- 42. Lueker TJ, Dickson AG, Keeling CD (2000) Ocean pCO2 calculated from dissolved inorganic carbon, alkalinity, and equations for K1 and K2: validation based on laboratory measurements of CO2 in gas and seawater at equilibrium. Marine Chemistry 70: 105–119. 10.1016/S0304-4203(00)00022-0 [DOI] [Google Scholar]
- 43. Ryther JH (1969) Photosynthesis and fish production in the sea. Science 166: 72–76. 10.1126/science.166.3901.72 [DOI] [PubMed] [Google Scholar]
- 44. Haigh R, Taylor FJR (1991) Mosaicism of microplankton communities in the northern Strait of Georgia, British Columbia. Mar Biol 110: 301–314. 10.1007/BF01313717 [DOI] [Google Scholar]
- 45. Haigh R, Taylor FJR, Sutherland TF (1992) Phytoplankton ecology of Sechelt Inlet, a fjord system on the British Columbia coast. I. General features of the nano- and microplankton. Mar Ecol Prog Ser 89: 117–134. 10.3354/meps089117 [DOI] [Google Scholar]
- 46. Harris SJ, Varela DE, Whitney FW, Harrison PJ (2009) Nutrient and phytoplankton dynamics off the west coast of Vancouver Island during the 1997/98 ENSO event. Deep Sea Res II 56: 2487–2502. 10.1016/j.dsr2.2009.02.009 [DOI] [Google Scholar]
- 47. Harrison PJ, Fulton JD, Taylor FJR, Parsons TR (1983) Review of the biological oceanography of the Strait of Georgia: pelagic environment. Can J Fish Aquat Sci 40: 1064–1094. 10.1139/f83-129 [DOI] [Google Scholar]
- 48. Ianson D, Pond S, Parsons TR (2001) The spring phytoplankton bloom in the coastal temperate ocean: growth criteria and seeding from shallow embayments. J Oceanogr 57: 723–734. 10.1023/A:1021288510407 [DOI] [Google Scholar]
- 49. Perry RI, Dilke BR, Parsons TR (1983) Tidal mixing and summer plankton distributions in Hecate Strait, British Columbia. Can J Fish Aquat Sci 40: 871–887. 10.1139/f83-114 [DOI] [Google Scholar]
- 50. Peterson TD, Toews HNJ, Robinson CLK, Harrison PJ (2007) Nutrient and phytoplankton dynamics in the Queen Charlotte Islands (Canada) during the summer upwelling seasons of 2001–2002. J Plankton Res 29: 219–239. 10.1093/plankt/fbm010 [DOI] [Google Scholar]
- 51. Lassiter AM, Wilkerson FP, Dugdale RC, Hogue VE (2006) Phytoplankton assemblages in the CoOP-WEST coastal upwelling area. Deep Sea Res II 53: 3063–3077. 10.1016/j.dsr2.2006.07.013 [DOI] [Google Scholar]
- 52. Olson MB, Lessard EJ, Wong CHJ, Bernhardt MJ (2006) Copepod feeding selectivity on microzooplankton, including the toxigenic diatoms Pseudo-nitzschia spp., in the coastal Pacific Northwest. Mar Ecol Prog Ser 326: 207–220. 10.3354/meps326207 [DOI] [Google Scholar]
- 53. Gower J (2013) Phytoplankton blooms on the BC coast. In: Irvine JR, Crawford WR, editors, State of physical, biological, and selected fishery resources of Pacific Canadian marine ecosystems in 2012, Canadian Science Advisory Secretariat, Research Document 2013/032. pp. 60–61. [Google Scholar]
- 54. Lipsen MS, Crawford DW, Gower J, Harrison PJ (2007) Spatial and temporal variability in coccolithophore abundance and production of PIC and POC in the NE subarctic Pacific during El Niño (1998), La Niña (1999) and 2000 during wind-driven coastal upwelling. Prog Oceanogr 75: 304–325. 10.1016/j.pocean.2007.08.004 [DOI] [Google Scholar]
- 55. Hickey BM, Banas NS (2008) Why is the northern end of the California current system so productive? Oceanography 21: 90–107. 10.5670/oceanog.2008.07 [DOI] [Google Scholar]
- 56. Hickey BM, Trainer VL, Kosro PM, Adams NG, Connolly TP, et al. (2013) A springtime source of toxic Pseudo-nitzschia cells on razor clam beaches in the Pacific Northwest. Harmful Algae 25: 1–14. 10.1016/j.hal.2013.01.006 [DOI] [Google Scholar]
- 57. Trainer VL, Wells ML, Cochlan WP, Trick CG, Bill BD, et al. (2009) An ecological study of a massive bloom of toxigenic Pseudo-nitzschia cuspidata off the Washington State coast. Limnol Oceanogr 54: 1461–1474. 10.4319/lo.2009.54.5.1461 [DOI] [Google Scholar]
- 58. Taylor FJR, Harrison PJ (2002) Harmful algal blooms in western Canadian coastal waters. In: Taylor FJR, Trainer VL, editors, Harmful Algal Blooms in the PICES Region of the North Pacific, North Pacific Marine Science Organization, PICES Scientific Report 23 pp. 77–88. [Google Scholar]
- 59. Bejarano AC, VanDola FM, Gulland FM, Rowles TK, Schwacke LH (2008) Production and toxicity of the marine biotoxin domoic acid and its effects on wildlife: a review. Hum Ecol Risk Assess 14: 544–567. 10.1080/10807030802074220 [DOI] [Google Scholar]
- 60. Deeds JR, Landsberg JH, Etheridge SM, Pitcher GC, Longan SW (2008) Non-traditional vectors for paralytic shellfish poisoning. Mar Drugs 6: 308–348. 10.3390/md20080015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61. Taylor FJR, Haigh R (1993) The ecology of fish-killing blooms of the chloromonad flagellate Heterosigma in the Strait of Georgia and adjacent waters. In: Smayda TJ, Shimuzu Y, editors, Toxic Phytoplankton Blooms in the Sea, Elsevier Science. pp. 705–710. [Google Scholar]
- 62. Rensel JEJ (2007) Fish kills from the harmful alga Heterosigma akashiwo in Puget Sound: recent blooms and review. Technical report, National Oceanic and Atmospheric Administration (NOAA), Center for Sponsored Coastal Ocean Research (CSCOR), v+58 p. [Google Scholar]
- 63. Twiner MJ, Trick CG (2000) Possible physiological mechanisms for production of hydrogen peroxide by the ichthyotoxic flagellate Heterosigma akashiwo . J Plankton Res 22: 1961–1975. 10.1093/plankt/22.10.1961 [DOI] [Google Scholar]
- 64. Yang CZ, Albright LJ, Yousif AN (1995) Oxygen-radical-mediated effects of the toxic phytoplankter Heterosigma carterae on juvenile rainbow trout Oncorhynchus mykiss . Dis Aquat Org 23: 101–108. 10.3354/dao023101 [DOI] [Google Scholar]
- 65. Kim D, Nakamura A, Okamoto T, Komatsu N, Oda T, et al. (1999) Toxic potential of the raphidophyte Olisthodiscus luteus: mediation by reactive oxygen species. J Plankton Res 21: 1017–1027. 10.1093/plankt/21.6.1017 [DOI] [Google Scholar]
- 66. Haigh N, Esenkulova S (2013) Economic losses to the British Columbia salmon aquaculture industry due to harmful algal blooms 2009–2012. In: Workshop 6. Economic impacts of harmful algal blooms on fisheries and aquaculture. North Pacific Marine Science Organization (PICES), Nanaimo, BC, October 11–20, 2013. [Google Scholar]
- 67. Engel A, Zondervan I, Aerts K, Beaufort L, Benthien A, et al. (2005) Testing the direct effect of CO2 concentration on a bloom of the coccolithophorid Emiliania huxleyi in mesocosm experiments. Limnol Oceanogr 50: 493–507. 10.4319/lo.2005.50.2.0493 [DOI] [Google Scholar]
- 68. Nielsen LT, Jakobsen HH, Hansen PJ (2010) High resilience of two coastal plankton communities to twenty-first century seawater acidification: evidence from microcosm studies. Mar Biol Res 6: 542–555. 10.1080/17451000903476941 [DOI] [Google Scholar]
- 69. Riebesell UM, Tortell PD (2011) Effects of ocean acidification on pelagic organisms and ecosystems. In: Gattuso JP, Hansson L, editors, Ocean Acidification, Oxford University Press; pp. 99–121. [Google Scholar]
- 70. Riebesell U, Zondervan I, Rost B, Tortell PD, Zeebe RE, et al. (2000) Reduced calcification of marine plankton in response to increased atmospheric CO2 . Nature 407: 364–367. 10.1038/35030078 [DOI] [PubMed] [Google Scholar]
- 71. Hinga KR (2002) Effects of pH on coastal marine phytoplankton. Mar Ecol Prog Ser 238: 281–300. 10.3354/meps238281 [DOI] [Google Scholar]
- 72. Beardall J, Raven JA (2004) The potential effects of global climate change on microalgal photosynthesis, growth and ecology. Phycologia 43: 26–40. 10.2216/i0031-8884-43-1-26.1 [DOI] [Google Scholar]
- 73. Moroney JV, Somanchi A (1999) How do algae concentrate CO2 to increase the efficiency of photosynthetic carbon fixation. Plant Phys 119: 9–16. 10.1104/pp.119.1.9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74. Tortell PD, Rau GH, Morel FMM (2000) Inorganic carbon acquisition in coastal Pacific phytoplankton communities. Limnol Oceanogr 45: 1485–1500. 10.4319/lo.2000.45.7.1485 [DOI] [Google Scholar]
- 75. Raven JA, Cockell CS, de la Rocha CL (2008) The evolution of inorganic carbon concentrating mechanisms in photosynthesis. Phil Trans R Soc Lond B, Biol Sci 363: 2641–2650. 10.1098/rstb.2008.0020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76. Nimer NA, Iglesias-Rodriguez MD, Merrett MJ (1997) Bicarbonate utilization by marine phytoplankton species. J Phycol 33: 625–631. 10.1111/j.0022-3646.1997.00625.x [DOI] [Google Scholar]
- 77. Bellerby RGJ, Schulz KG, Riebesell U, Neill C, Nondal G, et al. (2008) Marine ecosystem community carbon and nutrient uptake stoichiometry under varying ocean acidification during the PeECE III experiment. Biogeosciences 5: 1517–1527. 10.5194/bg-5-1517-2008 [DOI] [Google Scholar]
- 78. Egge JK, Thingstad TF, Larsen A, Engel A, Wohlers J, et al. (2009) Primary production during nutrient-induced blooms at elevated CO2 concentrations. Biogeosciences 6: 877–885. 10.5194/bg-6-877-2009 [DOI] [Google Scholar]
- 79. Hein M, Sand-Jensen K (1997) CO2 increases oceanic primary production. Nature 388: 526–527. 10.1038/41457 [DOI] [Google Scholar]
- 80. Riebesell U, Schulz KG, Bellerby RGJ, Botros M, Fritsche P, et al. (2007) Enhanced biological carbon consumption in a high CO2 ocean. Nature 450: 545–548. 10.1038/nature06267 [DOI] [PubMed] [Google Scholar]
- 81. Clark DR, Flynn KJ (2000) The relationship between the dissolved inorganic carbon concentration and growth rate in marine phytoplankton. Proc R Soc Lond, B, Biol Sci 267: 953–959. 10.1098/rspb.2000.1096 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82. Fu FX, Zhang Y, Warner ME, Feng Y, Sun J, et al. (2008) A comparison of future increased CO2 and temperature effects on sympatric Heterosigma akashiwo and Prorocentrum minimum . Harmful Algae 7: 76–90. 10.1016/j.hal.2007.05.006 [DOI] [Google Scholar]
- 83. Burkhardt S, Riebesell U, Zondervan I (1999) Effects of growth rate, CO2 concentration, and cell size on the stable carbon isotope fractionation in marine phytoplankton. Geochim Cosmochim Acta 63: 3729–3741. 10.1016/S0016-7037(99)00217-3 [DOI] [Google Scholar]
- 84. Tortell PD, Payne CD, Li Y, Trimborn S, Rost B, et al. (2008) CO2 sensitivity of Southern Ocean phytoplankton. Geophys Res Lett 35: L04605 10.1029/2007GL032583 [DOI] [Google Scholar]
- 85. Hare CE, Leblanc K, DiTullio GR, Kudela RM, Zhang Y, et al. (2007) Consequences of increased temperature and CO2 for phytoplankton community structure in the Bering Sea. Mar Ecol Prog Ser 352: 9–16. 10.3354/meps07182 [DOI] [Google Scholar]
- 86. Tortell PD, DiTullio GR, Sigman DM, Morel FMM (2002) CO2 effects on taxonomic composition and nutrient utilization in an equatorial Pacific phytoplankton assemblage. Mar Ecol Prog Ser 236: 37–43. 10.3354/meps236037 [DOI] [Google Scholar]
- 87. Kim H, Spivack AJ, Menden-Deuer S (2013) pH alters the swimming behaviors of the raphidophyte Heterosigma akashiwo: implications for bloom formation in an acidified ocean. Harmful Algae 26: 1–11. 10.1016/j.hal.2013.03.004 [DOI] [Google Scholar]
- 88. Hallegraeff GM (2010) Ocean climate change, phytoplankton community responses, and harmful algal blooms: a formidable predictive challenge. J Phycol 46: 220–235. 10.1111/j.1529-8817.2010.00815.x [DOI] [Google Scholar]
- 89. Hwang DF, Lu YH (2000) Influence of environmental and nutritional factors on growth, toxicity, and toxin profile of dinoflagellate Alexandrium minutum . Toxicon 38: 1491–1503. 10.1016/S0041-0101(00)00080-5 [DOI] [PubMed] [Google Scholar]
- 90. Fu FX, Place AR, Garcia NS, Hutchins DA (2010) CO2 and phosphate availability control the toxicity of the harmful bloom dinoflagellate Karlodinium veneficum . Aquat Microb Ecol 59: 55–65. 10.3354/ame01396 [DOI] [Google Scholar]
- 91. Tatters AO, Flewelling LJ, Fu F, Granholm AA, Hutchins DA (2013) High CO2 promotes the production of paralytic shellfish poisoning toxins by Alexandrium catenella from Southern California waters. Harmful Algae 30: 37–43. 10.1016/j.hal.2013.08.007 [DOI] [Google Scholar]
- 92. Sun J, Hutchins DA, Feng Y, Seubert EL, Caron DA, et al. (2011) Effects of changing pCO2 and phosphate availability on domoic acid production and physiology of the marine harmful bloom diatom Pseudo-nitzschia multiseries. Limnol Oceanogr 56: 829–840. 10.4319/lo.2011.56.3.0829 [DOI] [Google Scholar]
- 93. Tatters AO, Fu FX, Hutchins DA (2012) High CO2 and silicate limitation synergistically increase the toxicity of Pseudo-nitzschia fraudulenta . PLoS ONE 7: e32116 10.1371/journal.pone.0032116 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94. Brown CW, Yoder JA (1994) Coccolithophorid blooms in the global ocean. J Geophys Res 99: 7467–7482. 10.1029/93JC02156 [DOI] [Google Scholar]
- 95. Westbroek P, Young JR, Linschooten K (1989) Coccolith production (biomineralization) in the marine alga Emiliania huxleyi . J Protozool 36: 368–373. 10.1111/j.1550-7408.1989.tb05528.x [DOI] [Google Scholar]
- 96. Iglesias-Rodriguez MD, Halloran PR, Rickaby REM, Hall IR, Colmenero-Hidalgo E, et al. (2008) Phytoplankton calcification in a high-CO2 world. Science 320: 336–340. 10.1126/science.1154122 [DOI] [PubMed] [Google Scholar]
- 97. Shi D, Xu Y, Morel FMM (2009) Effects of the pH/pCO2 control method on medium chemistry and phytoplankton growth. Biogeosciences 6: 1199–1207. 10.5194/bg-6-1199-2009 [DOI] [Google Scholar]
- 98. Zondervan I, Zeebe RE, Rost B, Riebesell U (2001) Decreasing marine biogenic calcification: a negative feedback on rising atmospheric pCO2 . Glob Biogeochem Cycles 5: 507–516. 10.1029/2000GB001321 [DOI] [Google Scholar]
- 99. Ridgwell A, Schmidt DN, Turley C, Brownlee C, Maldonado MT, et al. (2009) From laboratory manipulations to Earth system models: scaling calcification impacts of ocean acidification. Biogeosciences 6: 2611–2623. 10.5194/bg-6-2611-2009 [DOI] [Google Scholar]
- 100. Delille B, Harlay J, Zondervan I, Jacquet S, Chou L, et al. (2005) Response of primary production and calcification to changes of pCO2 during experimental blooms of the coccolithophorid Emiliania huxleyi . Glob Biogeochem Cycles 19: GB2023, 1–14. 10.1029/2004GB002318 [DOI] [Google Scholar]
- 101. Beaufort L, Probert I, de Garidel-Thoron T, Bendif EM, Ruiz-Pino D, et al. (2011) Sensitivity of coccolithophores to carbonate chemistry and ocean acidification. Nature 476: 80–83. 10.1038/nature10295 [DOI] [PubMed] [Google Scholar]
- 102. Bates C (2004) E-flora atlas of flora of British Columbia. An introduction to the (macro) algae of British Columbia. In: Klinkenberg B, editor, E-Flora BC: Atlas of the Plants of British Columbia [www.eflora.bc.ca]. Lab for Advanced Spatial Analysis, Department of Geography, University of British Columbia, Vancouver: [Date Accessed: 2013-06-06], Royal British Columbia Museum. URL http://www.geog.ubc.ca/biodiversity/eflora/algae.html. [Google Scholar]
- 103. Druehl LD (2000) Pacific Seaweeds: A Guide to Common Seaweeds of the West Coast. Harbour Publishing, Madeira Park BC. [Google Scholar]
- 104. Mann KH (1973) Seaweeds: their productivity and strategy for growth. Science 182: 975–981. 10.1126/science.182.4116.975 [DOI] [PubMed] [Google Scholar]
- 105. Foster MS, Schiel DR (1985) The ecology of giant kelp forests in California: a community profile. US Fish Wildl Serv Biol Rep 85 (7.2): xv+152 p. [Google Scholar]
- 106. Nelson WA (2009) Calcified macroalgae—critical to coastal ecosystems and vulnerable to change: a review. Mar Freshw Res 60: 787–801. 10.1071/MF08335 [DOI] [Google Scholar]
- 107. Cornwall CE, Hepburn CD, Pritchard D, Currie KI, McGraw CM, et al. (2012) Carbon-use strategies in macroalgae: differential responses to lowered pH and implications for ocean acidification. J Phycol 48: 137–144. 10.1111/j.1529-8817.2011.01085.x [DOI] [PubMed] [Google Scholar]
- 108. Hurd CL, Hepburn CD, Currie KI, Raven JA, Hunter KA (2009) Testing the effects of ocean acidification on algal metabolism: considerations for experimental designs. J Phycol 45: 1236–1251. 10.1111/j.1529-8817.2009.00768.x [DOI] [PubMed] [Google Scholar]
- 109. Kubler JE, Johnston AM, Raven JA (1999) The effects of reduced and elevated CO2 and O2 on the seaweed Lomentaria articulata . Plant Cell Environ 22: 1303–1310. 10.1046/j.1365-3040.1999.00492.x [DOI] [Google Scholar]
- 110. Hepburn CD, Pritchard DW, Cornwall CE, McLeod RJ, Beardall J, et al. (2011) Diversity of carbon use strategies in a kelp forest community: implications for a high CO2 ocean. Glob Change Biol 17: 2488–2497. 10.1111/j.1365-2486.2011.02411.x [DOI] [Google Scholar]
- 111. Swanson AK, Fox CH (2007) Altered kelp (Laminariales) phlorotannins and growth under elevated carbon dioxide and ultraviolet-B treatments can influence associated intertidal food webs. Glob Change Biol 13: 1696–1709. 10.1111/j.1365-2486.2007.01384.x [DOI] [Google Scholar]
- 112. Roleda MY, Morris JN, McGraw CM, Hurd CL (2012) Ocean acidification and seaweed reproduction: increased CO2 ameliorates the negative effect of lowered pH on meiospore germination in the giant kelp Macrocystis pyrifera (Laminariales, Phaeophyceae). Glob Change Biol 18: 854–864. 10.1111/j.1365-2486.2011.02594.x [DOI] [Google Scholar]
- 113. Hofmann LC, Straub S, Bischof K (2012) Competition between calcifying and noncalcifying temperate marine macroalgae under elevated CO2 levels. Mar Ecol Prog Ser 464: 89–105. 10.3354/meps09892 [DOI] [Google Scholar]
- 114. Cornwall CE, Hepburn CD, Pilditch CA, Hurd CL (2013) Concentration boundary layers around complex assemblages of macroalgae: Implications for the effects of ocean acidification on understory coralline algae. Limnol Oceanogr 58: 121–130. 10.4319/lo.2013.58.1.0121 [DOI] [Google Scholar]
- 115. Harley CDG, Anderson KM, Demes KW, Jorve JP, Kordas RL, et al. (2012) Effects of climate change on global seaweed communities. J Phycol 48: 1064–1078. 10.1111/j.1529-8817.2012.01224.x [DOI] [PubMed] [Google Scholar]
- 116. Hurd CL, Cornwall CE, Currie K, Hepburn CD, McGraw CM, et al. (2011) Metabolically induced pH fluctuations by some coastal calcifiers exceed projected 22nd century ocean acidification: a mechanism for differential susceptibility? Glob Change Biol 17: 3254–3262. 10.1111/j.1365-2486.2011.02473.x [DOI] [Google Scholar]
- 117. Roleda MY, Boyd PW, Hurd CL (2012) Before ocean acidification: calcifier chemistry lessons. J Phycol 48: 840–843. 10.1111/j.1529-8817.2012.01195.x [DOI] [PubMed] [Google Scholar]
- 118. Gao KS, Helbling EW, Hader DP, Hutchins DA (2012) Responses of marine primary producers to interactions between ocean acidification, solar radiation, and warming. Mar Ecol Prog Ser 470: 167–189. 10.3354/meps10043 [DOI] [Google Scholar]
- 119. Gao KS, Zheng YQ (2010) Combined effects of ocean acidification and solar UV radiation on photosynthesis, growth, pigmentation and calcification of the coralline alga Corallina sessilis (Rhodophyta). Glob Change Biol 16: 2388–2398. 10.1111/j.1365-2486.2009.02113.x [DOI] [Google Scholar]
- 120. Koch M, Bowes G, Ross C, Zhang XH (2013) Climate change and ocean acidification effects on seagrasses and marine macroalgae. Glob Change Biol 19: 103–132. 10.1111/j.1365-2486.2012.02791.x [DOI] [PubMed] [Google Scholar]
- 121. Lessard J, Campbell A (2007) Describing northern abalone, Haliotis kamtschatkana, habitat: focusing rebuilding efforts in British Columbia, Canada. J Shell Res 26: 677–686. 10.2983/0730-8000(2007)26[677:DNAHKH]2.0.CO;2 [DOI] [Google Scholar]
- 122. Kuffner IB, Andersson AJ, Jokiel PL, Rodgers KS, Mackenzie FT (2008) Decreased abundance of crustose coralline algae due to ocean acidification. Nat Geosci 1: 114–117. 10.1038/ngeo100 [DOI] [Google Scholar]
- 123. Roberts R (2001) A review of settlement cues for larval abalone (Haliotis spp.). J Shellfish Res 20: 571–586. [Google Scholar]
- 124. Morse ANC (1991) How do planktonic larvae know where to settle? Am Sci 79: 154–167. [Google Scholar]
- 125. Asnaghi V, Chiantore M, Mangialajo L, Gazeau F, Francour P, et al. (2013) Cascading effects of ocean acidification in a rocky subtidal community. PLoS ONE 8: e61978 10.1371/journal.pone.0061978 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126. Lubchenco J, Gaines SD (1981) A unified approach to marine plant-herbivore interactions. I. Populations and communities. Annu Rev Ecol Syst 12: 405–437. 10.1146/annurev.es.12.110181.002201 [DOI] [Google Scholar]
- 127. Duffy JE, Hay ME (1990) Seaweed adaptations to herbivory—chemical, structural, and morphological defenses are often adjusted to spatial or temporal patterns of attack. Bioscience 40: 368–375. 10.2307/1311214 [DOI] [Google Scholar]
- 128. Johnson VR, Russell BD, Fabricius KE, Brownlee C, Hall-Spencer JM (2012) Temperate and tropical brown macroalgae thrive, despite decalcification, along natural CO2 gradients. Glob Change Biol 18: 2792–2803. 10.1111/j.1365-2486.2012.02716.x [DOI] [PubMed] [Google Scholar]
- 129. Jackson JBC, Kirby MX, Berger WH, Bjorndal KA, Botsford LW, et al. (2001) Historical overfishing and the recent collapse of coastal ecosystems. Science 293: 629–637. 10.1126/science.1059199 [DOI] [PubMed] [Google Scholar]
- 130. Connell S, Kroeker KJ, Fabricius KE, Kline DI, Russell BD (2013) The other ocean acidification problem: CO2 as a resource among competitors for ecosystem dominance. Phil Trans R Soc Lond B, Biol Sci 368: 20120442 10.1098/rstb.2012.0442 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131. Connell SD, Russell BD (2010) The direct effects of increasing CO2 and temperature on non-calcifying organisms: increasing the potential for phase shifts in kelp forests. Proc R Soc Lond, B, Biol Sci 277: 1409–1415. 10.1098/rspb.2009.2069 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Shaffer J (2004) Preferential use of nearshore kelp habitats by juvenile salmon and forage fish. In: Toni D, Fraser DA, editors, Proceedings of the 2003 Georgia Basin / Puget Sound Research Conference, 31 March–3 April, Vancouver, British Columbia. pp. 1–11. URL http://www.caseinlet.org/uploads/SalmonKelp_Shaffer_1_.pdf.
- 133. Duggins DO, Simenstad CA, Estes JA (1989) Magnification of secondary production by kelp detritus in coastal marine ecosystems. Science 245: 170–173. 10.1126/science.245.4914.170 [DOI] [PubMed] [Google Scholar]
- 134. Orth RJ, Carruthers TJB, Dennison WC, Duarte CM, Fourqurean JW, et al. (2006) A global crisis for seagrass ecosystems. Bioscience 56: 987–996. 10.1641/0006-3568(2006)56[987:AGCFSE]2.0.CO;2 [DOI] [Google Scholar]
- 135. Lucas BG, Johannessen D, Lindstrom S (2007) Appendix E: Marine Plants. In: Lucas BG, Verrin S, Brown R, editors, Ecosystem Overview: Pacific North Coast Integrated Management Area (PNCIMA), Canadian Technical Report of Fisheries and Aquatic Sciences 2667, iv + 23 p. [Google Scholar]
- 136. Beck MW, KLH Jr, Able KW, Childers DL, Eggleston DB, et al. (2001) The identification, conservation, and management of estuarine and marine nurseries for fish and invertebrates. BioSci 51: 633–641. 10.1641/0006-3568(2001)051[0633:TICAMO]2.0.CO;2 [DOI] [Google Scholar]
- 137. Hendriks IE, Olsen YS, Ramajo L, Basso L, Steckbauer A, et al. (2014) Photosynthetic activity buffers ocean acidification in seagrass meadows. Biogeosci 11: 333–346. 10.5194/bg-11-333-2014 [DOI] [Google Scholar]
- 138. Beer S (1989) Photosynthesis and photorespiration of marine angiosperms. Aquat Bot 34: 153–166. 10.1016/0304-3770(89)90054-5 [DOI] [Google Scholar]
- 139. Zimmerman RC, Kohrs DG, Steller DL, Alberte RS (1997) Impacts of CO2 enrichment on productivity and light requirements of eelgrass. Plant Physiol 115: 599–607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140. Arnold T, Mealey C, Leahey H, Miller AW, Hall-Spencer JM, et al. (2012) Ocean acidification and the loss of phenolic substances in marine plants. PLoS ONE 7: e35107 10.1371/journal.pone.0035107 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141. Martin AJ, Montagnes DJS (1993) Winter ciliates in a British Columbian fjord: six new species and an analysis of ciliate putative prey. J Eukaryot Microbiol 40: 535–549. 10.1111/j.1550-7408.1993.tb06105.x [DOI] [Google Scholar]
- 142. Jacobson DM, Anderson DM (1986) Thecate heterotrophic dinoflagellates: feeding behavior and mechanisms. J Phycol 22: 249–258. 10.1111/j.1529-8817.1986.tb00021.x [DOI] [Google Scholar]
- 143. Irigoien X, Flynn KJ, Harris RP (2005) Phytoplankton blooms: a ‘loophole’ in microzooplankton grazing impact? J Plankton Res 27: 313–321. 10.1093/plankt/fbi011 [DOI] [Google Scholar]
- 144. Bakun A, Broad K (2003) Environmental ‘loopholes’ and fish population dynamics: comparative pattern recognition with focus on El Niño effects in the Pacific. Fish Oceanogr 12: 458–473. 10.1046/j.1365-2419.2003.00258.x [DOI] [Google Scholar]
- 145. Moy AD, Howard WR, Bray SG, Trull TW (2009) Reduced calcification in modern Southern Ocean planktonic foraminifera. Nat Geosci 2: 276–280. 10.1038/ngeo460 [DOI] [Google Scholar]
- 146. Caron DA, Hutchins DA (2013) The effects of changing climate on microzooplankton grazing and community structure: drivers, predictions and knowledge gaps. J Plankton Res 35: 235–252. 10.1093/plankt/fbs091 [DOI] [Google Scholar]
- 147. Suffrian K, Simonelli P, Nejstgaard JC, Putzeys S, Carotenuto Y, et al. (2008) Microzooplankton grazing and phytoplankton growth in marine mesocosms with increased CO2 levels. Biogeosciences 5: 1145–1156. 10.5194/bg-5-1145-2008 [DOI] [Google Scholar]
- 148. Aberle N, Schulz KG, Stuhr A, Malzahn AM, Ludwig A, et al. (2013) High tolerance of microzooplankton to ocean acidification in an Arctic coastal plankton community. Biogeosciences 10: 1471–1481. 10.5194/bg-10-1471-2013 [DOI] [Google Scholar]
- 149. Rossoll D, Sommer U, Winder M (2013) Community interactions dampen acidification effects in a coastal plankton system. Mar Ecol Prog Ser 486: 37–46. 10.3354/meps10352 [DOI] [Google Scholar]
- 150. Rose JM, Feng Y, Gobler CJ, Gutierrez R, Hare CE, et al. (2009) Effects of increased pCO2 and temperature on the North Atlantic spring bloom. II. Microzooplankton abundance and grazing. Mar Ecol Prog Ser 388: 27–40. 10.3354/meps08134 [DOI] [Google Scholar]
- 151. Kim JM, Lee K, Yang EJ, Shin K, Noh JH, et al. (2010) Enhanced production of oceanic dimethylsulfide resulting from CO2-induced grazing activity in a high CO2 world. Environ Sci Technol 44: 8140–8143. 10.1021/es102028k [DOI] [PubMed] [Google Scholar]
- 152.Gravinese PM, Foy M, Lessard E, Murray JW (2014) The effects of elevated pCO2 on microzooplankton biomass, abundance, and community structure—a mesocosm study in the Salish Sea. In: Ocean Sciences Meeting, Session 033, presented Feb. 25, 2014, Honolulu, HI.
- 153. Mackas DL, Tsuda A (1999) Mesozooplankton in the eastern and western subarctic Pacific: community structure, seasonal life histories, and interannual variability. Prog Oceanogr 43: 335–363. 10.1016/S0079-6611(99)00012-9 [DOI] [Google Scholar]
- 154. Mackas D, Galbraith M, Faust D, Masson D, Young K, et al. (2013) Zooplankton time series from the Strait of Georgia: results from year-round sampling at deep water locations, 1990–2010. Prog Oceanogr 115: 129–159. 10.1016/j.pocean.2013.05.019 [DOI] [Google Scholar]
- 155. Mackas DL, Thomson RE, Galbraith M (2001) Changes in the zooplankton community of the British Columbia continental margin, 1985–1999, and their covariation with oceanographic conditions. Can J Fish Aquat Sci 58: 685–702. 10.1139/f01-009 [DOI] [Google Scholar]
- 156. Zhang D, Li S, Wang G, Guo D (2011) Impacts of CO2-driven seawater acidification on survival, egg production rate and hatching success of four marine copepods. Acta Oceanol Sin 30: 86–94. 10.1007/s13131-011-0165-9 [DOI] [Google Scholar]
- 157. Kurihara H, Shimode S, Shirayama Y (2004) Effects of raised CO2 concentration on the egg production rate and early development of two marine copepods (Acartia steueri and Acartia erythraea). Mar Pollut Bull 49: 721–727. 10.1016/j.marpolbul.2004.05.005 [DOI] [PubMed] [Google Scholar]
- 158. Mayor DJ, Matthews C, Cook K, Zuur AF, Hay S (2007) CO2-induced acidification affects hatching success in Calanus finmarchicus . Mar Ecol Prog Ser 350: 91–97. 10.3354/meps07142 [DOI] [Google Scholar]
- 159. Kurihara H, Ishimatsu A (2008) Effects of high CO2 seawater on the copepod (Acartia tsuensis) through all life stages and subsequent generations. Mar Pollut Bull 56: 1086–1090. 10.1016/j.marpolbul.2008.03.023 [DOI] [PubMed] [Google Scholar]
- 160. Weydmann A, Søreide JE, Kwasniewski S, Widdicombe S (2012) Influence of CO2-induced acidification on the reproduction of a key Arctic copepod Calanus glacialis . J Exp Mar Biol Ecol 428: 39–42. 10.1016/j.jembe.2012.06.002 [DOI] [Google Scholar]
- 161. Vehmaa A, Brutemark A, Engström-Öst J (2012) Maternal effects may act as an adaptation mechanism for copepods facing pH and temperature changes. PLoS ONE 7: e48538 10.1371/journal.pone.0048538 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162. Mayor DJ, Everett NR, Cook KB (2012) End of century ocean warming and acidification effects on reproductive success in a temperate marine copepod. J Plankton Res 34: 258–262. 10.1093/plankt/fbr107 [DOI] [Google Scholar]
- 163. Pedersen SA, Hansen BH, Altin D, Olsen AJ (2013) Medium-term exposure of the North Atlantic copepod Calanus finmarchicus (Gunnerus, 1770) to CO2-acidified seawater: effects on survival and development. Biogeosciences 10: 7481–7491. 10.5194/bg-10-7481-2013 [DOI] [Google Scholar]
- 164. Mackas DL, Galbraith MD (2012) Pteropod time-series from the NE Pacific. ICES J Mar Sci 69: 448–459. 10.1093/icesjms/fsr163 [DOI] [Google Scholar]
- 165. Mackas DL, Tsurumi M, Galbraith MD, Yelland D (2005) Zooplankton distribution and dynamics in a North Pacific eddy of coastal origin. 2. Mechanisms of eddy colonization by and retention of offshore species. Deep Sea Res II 52: 1011–1035. 10.1016/j.dsr2.2005.02.008 [DOI] [Google Scholar]
- 166. Armstrong JL, Boldt JL, Cross AD, Moss JH, David ND, et al. (2005) Distribution, size and interannual, seasonal and diel food habits of northern Gulf of Alaska juvenile pink salmon Oncorhynchus gorbuscha . Deep Sea Res II 52: 247–265. 10.1016/j.dsr2.2004.09.019 [DOI] [Google Scholar]
- 167. Lalli CM, Gilmer RW (1989) Pelagic Snails: The Biology of Holoplanktonic and Gastropod Mollusks. Stanford, CA: Stanford University Press, 259 pp. [Google Scholar]
- 168. Hunt BP, Pakhomov EA, Hosie GW, Seigel W, Ward P, et al. (2008) Pteropods in Southern Ocean ecosystems. Prog Oceanogr 78: 193–221. 10.1016/j.pocean.2008.06.001 [DOI] [Google Scholar]
- 169. Byrne RH, Acker JG, Betzer PR, Feely RA, Cates MH (1984) Water column dissolution of aragonite in the Pacific Ocean. Nature 312: 321–326. 10.1038/312321a0 [DOI] [Google Scholar]
- 170. Lischka S, Riebesell U (2012) Synergistic effects of ocean acidification and warming on overwintering pteropods in the Arctic. Glob Change Biol 18: 3517–3528. 10.1111/gcb.12020 [DOI] [Google Scholar]
- 171. Sato-Okoshi W, Okoshi K, H S, Akiha F (2010) Shell structure characteristics of pelagic and benthic molluscs from Antarctic waters. Polar Sci 4: 257–261. 10.1016/j.polar.2010.05.006 [DOI] [Google Scholar]
- 172. Bednarsek N, Tarling GA, Bakker DCE, Fielding S, Jones EM, et al. (2012) Extensive dissolution of live pteropods in the Southern Ocean. Nat Geosci 5: 881–885. 10.1038/ngeo1635 [DOI] [Google Scholar]
- 173. Roger LM, Richardson AJ, McKinnon AD, Knott B, Matear R, et al. (2012) Comparison of the shell structure of two tropical Thecosomata (Creseis acicula and Diacavolinia longirostris) from 1963 to 2009: potential implications of declining aragonite saturation. ICES J Mar Sci 69: 465–474. 10.1093/icesjms/fsr171 [DOI] [Google Scholar]
- 174. Bednarsek N, Feely RA, Reum JCP, Peterson B, Menkel J, et al. (2014) Limacina helicina shell dissolution as an indicator of declining habitat suitability owing to ocean acidification in the California Current Ecosystem. Proc R Soc Lond, B, Biol Sci 281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175. Comeau S, Gorsky G, Jeffree R, Teyssié JL, Gattuso JP (2009) Impact of ocean acidification on a key arctic pelagic mollusc (Limacina helicina). Biogeosciences 6: 1877–1882. 10.5194/bg-6-1877-2009 [DOI] [Google Scholar]
- 176. Comeau S, Gorsky G, Alliouane S, Gattuso JP (2010) Larvae of the pteropod Cavolinia inflexa exposed to aragonite undersaturation are viable but shell-less. Mar Biol 78: 2341–2345. 10.1007/s00227-010-1493-6 [DOI] [Google Scholar]
- 177. Comeau S, Gattuso JP, Nisumaa AM, Orr J (2012) Impact of aragonite saturation state changes on migratory pteropods. Proc R Soc Lond, B, Biol Sci 279: 732–738. 10.1098/rspb.2011.0910 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178. Seibel BA, Mass AE, Dierssen HM (2012) Energetic plasticity underlies a variable response to ocean acidification in the pteropod Limacina helicina antarctica . PLoS ONE 7: e30464 10.1371/journal.pone.0030464 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179. Maas AE, Wishner KF, Seibel BA (2012) The metabolic response of pteropods to acidification reflects natural CO2-exposure in oxygen minimum zones. Biogeosciences 9: 747–757. 10.5194/bg-9-747-2012 [DOI] [Google Scholar]
- 180. Armstrong JL, Myers KW, Beauchamp DA, Davis ND, Walker RV, et al. (2008) Interannual and spatial feeding patterns of hatchery and wild juvenile pink salmon in the Gulf of Alaska in years of high and low survival. Trans Am Fish Soc 137: 1299–1316. 10.1577/T07-196.1 [DOI] [Google Scholar]
- 181. Helmuth B, Harley C, Halpin PM, O’Donnell M, Hoffmann G (2002) Climate change and latitudinal patterns of intertidal thermal stress. Science 298: 1015–1017. 10.1126/science.1076814 [DOI] [PubMed] [Google Scholar]
- 182. Harbo R (1996) Shells and Shellfish of the Pacific northwest: a field guide. Madiera Park, BC: Harbour Publishing. [Google Scholar]
- 183. Bacher C, Grant J, Hawkins A, Fang J, Zhu M, et al. (2003) Modelling the effect of food depletion on scallop growth in Sungo Bay China. Auat Living Resour 16: 10–24. 10.1016/S0990-7440(03)00003-2 [DOI] [Google Scholar]
- 184. Wilson JG (1994) The role of bioindicators in estuarine management. Estuaries 17: 94–101. 10.2307/1352337 [DOI] [Google Scholar]
- 185. Lindahl O, Hart R, Hernroth B, Kollberg S, Loo LO, et al. (2005) Improving marine water quality by mussel farming: a profitable solution for Swedish society. Ambio 34: 131–138. 10.1579/0044-7447-34.2.131 [DOI] [PubMed] [Google Scholar]
- 186. Johnson AM (1982) Status of Alaska sea otter populations and developing conflicts with fisheries. In: Sabol K, editor, Transactions of the 47th North American Wildlife and natural resources conference. Washington DC, USA, volume 42, pp. 293–299. [Google Scholar]
- 187. Baker P (1995) Review of ecology and fishery of the Olympia oyster Ostrea lurida with annotated bibliography. J Shellfish Res 14: 501–518. [Google Scholar]
- 188.Fisheries and Oceans Canada (2013) Pacific Region Integrated Fisheries Management Plan—Intertidal Clams—January 1, 2013 to December 31, 2015. http://www.pac.dfo-mpo.gc.ca/fm-gp/mplans/2013/intertidal_clam-palourde_intercotidale-2013-15-eng.pdf, 100 pp.
- 189. Gillespie GE (1999) Status of the Olympia Oyster, Ostrea conchaphila, in Canada. Can Stock Assess Sec Res Doc 99/150: 36 p. [Google Scholar]
- 190. Gillespie GE, Bower SM, Marcus KL, Kieser D (2012) Biological synopsises for three exotic molluscs, Manila Clam (Venerupis philippinarum), Pacific Oyster (Crassostrea gigas) and Japanese Scallop (Mizuhopecten yessoensis) licensed for Aquaculture in British Columbia. Can Sci Advis Sec Res Doc 2012/013: v + 97 p. [Google Scholar]
- 191.Fisheries and Oceans Canada (2013) Pacific Region Exploratory Fishery Guidelines—Pink and Spiny Scallop by Trawl—August 1, 2013 to July 31, 2014. http://www.pac.dfo-mpo.gc.ca/fm-gp/mplans/2013/scallop-petoncle-2013-eng.pdf, 30 pp.
- 192. Weiss IM, Tuross N, Addadi L, Weiner S (2002) Mollusc larval shell formation: amorphous calcium carbonate is a precursor phase for aragonite. J Exp Zool 293: 478–491. 10.1002/jez.90004 [DOI] [PubMed] [Google Scholar]
- 193. Wilt FH (2005) Developmental biology meets materials science: morphogenesis of biomineralized structures. Dev Biol 280: 15–25. 10.1016/j.ydbio.2005.01.019 [DOI] [PubMed] [Google Scholar]
- 194. Stenzel HB (1963) Aragonite and calcite as constituents of adult oyster shells. Science 142: 232–233. 10.1126/science.142.3589.232 [DOI] [PubMed] [Google Scholar]
- 195. Barton A, Hales B, Waldbusser GG, Langdon C, Feely RA (2012) The Pacific oyster, Crassostrea gigas, shows negative correlation to naturally elevated carbon dioxide levels: Implications for near-term ocean acidification effects. Limnol Oceanogr 57: 698–710. 10.4319/lo.2012.57.3.0698 [DOI] [Google Scholar]
- 196. Kroeker KJ, Kordas RL, Crim RN, Singh GG (2010) Meta-analysis reveals negative yet variable effects of ocean acidification on marine organisms. Ecol Lett 13: 1419–1434. 10.1111/j.1461-0248.2010.01518.x [DOI] [PubMed] [Google Scholar]
- 197. Parker LM, Ross PM, O’Connor WA, Pörtner HO, Scanes E, et al. (2013) Predicting the response of molluscs to the impact of ocean acidification. Biology 2: 651–692. 10.3390/biology2020651 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198. Havenhand JN, Schlegel P (2009) Near-future levels of ocean acidification do not affect sperm motility and fertilization kinetics in the oyster Crassostrea gigas . Biogeosciences 6: 3009–3015. 10.5194/bg-6-3009-2009 [DOI] [Google Scholar]
- 199. Parker LM, Ross PM, O’Connor WA (2010) Comparing the effect of elevated pCO2 and temperature on the fertilization and early development of two species of oysters. Mar Biol 157: 2435–2452. 10.1007/s00227-010-1508-3 [DOI] [Google Scholar]
- 200. Barros P, Sobral P, Range P, Chícharo L, Matias D (2013) Effects of sea-water acidification on fertilization and larval development of the oyster Crassostrea gigas . J Exp Mar Biol Ecol 440: 200–206. 10.1016/j.jembe.2012.12.014 [DOI] [Google Scholar]
- 201. Kurihara H, Kato S, Ishimatsu A (2007) Effects of increased seawater pCO2 on early development of the oyster Crassostrea gigas . Aquat Biol 1: 91–98. 10.3354/ab00009 [DOI] [Google Scholar]
- 202. Timmins-Schiffman E, O’Donnell MJ, Friedman CS, Roberts SB (2013) Elevated pCO2 causes developmental delay in early larval Pacific oysters, Crassostrea gigas . Mar Biol 160: 1973–1982. 10.1007/s00227-012-2055-x [DOI] [Google Scholar]
- 203. Ginger KWK, Vera CBS, R D, Dennis CKS, Adela LJ, et al. (2013) Larval and post-larval stages of Pacific Oyster (Crassostrea gigas) are resistant to elevated CO2 . PLoS ONE 8: e64147 10.1371/journal.pone.0064147 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204. Crim RN, Sunday JM, Harley CDG (2011) Elevated seawater CO2 concentrations impair larval development and reduce larval survival in endangered northern abalone (Haliotis kamtschatkana). J Exp Mar Biol Ecol 400: 272–277. 10.1016/j.jembe.2011.02.002 [DOI] [Google Scholar]
- 205. Gobler CJ, DePasquale EL, Griffith AW, Baumann H (2014) Hypoxia and acidification have additive and synergistic negative effects on the growth, survival, and metamorphosis of early life stage bivalves. PLoS ONE 9: e83648 10.1371/journal.pone.0083648 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206. Hettinger A, Sanford E, Hill TM, Lenz EA, Russell AD, et al. (2013) Larval carry-over effects from ocean acidification persist in the natural environment. Glob Change Biol 19: 3317–3326. [DOI] [PubMed] [Google Scholar]
- 207. Hettinger A, Sanford E, Hill TM, Hosfelt JD, Russell AD, et al. (2013) The influence of food supply on the response of Olympia oyster larvae to ocean acidification. Biogeosciences 10: 6629–6638. 10.5194/bg-10-6629-2013 [DOI] [Google Scholar]
- 208. Talmage SC, Gobler CJ (2009) The effects of elevated carbon dioxide concentrations on the metamorphosis, size, and survival of larval hard clams (Mercenaria mercenaria), bay scallops (Argopecten irradians), and Eastern oysters (Crassostrea virginica). Limnol Oceanogr 54: 2072–2080. 10.4319/lo.2009.54.6.2072 [DOI] [Google Scholar]
- 209. Talmage SC, Gobler CJ (2011) Effects of elevated temperature and carbon dioxide on the growth and survival of larvae and juveniles of three species of Northwest Atlantic bivalves. PLoS ONE 6: e26941 10.1371/journal.pone.0026941 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210. Talmage SC, Gobler CJ (2012) Effects of CO2 and the harmful alga Aureococcus anophagefferens on growth and survival of oyster and scallop larvae. Mar Ecol Prog Ser 464: 121–134. 10.3354/meps09867 [DOI] [Google Scholar]
- 211. Dineshram R, Wong KKW, Xiao S, Yu Z, Qian PY, et al. (2012) Analysis of Pacific oyster larval proteome and its response to high-CO2 . Mar Pollut Bull 64: 2160–2167. 10.1016/j.marpolbul.2012.07.043 [DOI] [PubMed] [Google Scholar]
- 212. Hettinger A, Sanford E, Hill TM, Russell AD, Sato KNS, et al. (2012) Persistent carry-over effects of planktonic exposure to ocean acidification in the Olympia oyster. Ecology 93: 2758–2768. 10.1890/12-0567.1 [DOI] [PubMed] [Google Scholar]
- 213. Gazeau F, Quiblier C, Jansen JM, Gattuso JP, Middelburg JJ, et al. (2007) Impact of elevated CO2 on shellfish calcification. Geophys Res Lett 34: L07603 10.1029/2006GL028554 [DOI] [Google Scholar]
- 214. Mingliang Z, Jianguang F, Jihong Z, Bin L, Shengmin R, et al. (2011) Effect of marine acidification on calcification and respiration of Chlamys farreri . J Shellfish Res 30: 267–271. 10.2983/035.030.0211 [DOI] [Google Scholar]
- 215. O’Donnell MJ, George MN, Carrington E (2013) Mussel byssus attachment weakened by ocean acidification. Nature Clim Change 3: 1471–1481. [Google Scholar]
- 216. Lannig G, Eilers S, Pörtner HO, Sokolova IM, Bock C (2010) Impact of ocean acidification on energy metabolism of oyster, Crassostrea gigas—changes in metabolic pathways and thermal response. Mar Drugs 8: 2318–2339. 10.3390/md8082318 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 217. Sanders MB, Bean TP, Hutchinson TH, Le Quesne WJF (2013) Juvenile king scallop, Pecten maximus, is potentially tolerant to low levels of ocean acidification when food is unrestricted. PLoS ONE 8: e74118 10.1371/journal.pone.0074118 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 218. Schalkhausser B, Bock C, Stemmer K, Brey T, Pörtner HO, et al. (2013) Impact of ocean acidification on escape performance of the king scallop, Pecten maximus, from Norway. Mar Biol 160: 1995–2006. 10.1007/s00227-012-2057-8 [DOI] [Google Scholar]
- 219. Zhai WD, Zhao HD, Zheng N, Xu Y (2012) Coastal acidification in summer bottom oxygen-depleted waters in northwestern-northern Bohai Sea from June to August in 2011. Chin Sci Bull 57: 1062–1068. 10.1007/s11434-011-4949-2 [DOI] [Google Scholar]
- 220. Duckworth AR, Peterson BJ (2013) Effects of seawater temperature and pH on the boring rates of the sponge Cliona celata in scallop shells. Mar Biol 160: 27–35. 10.1007/s00227-012-2053-z [DOI] [Google Scholar]
- 221. Gutiérrez JL, Jones CG, Strayer DL, Iribarne OO (2003) Mollusks as ecosystem engineers: the role of shell production in aquatic habitats. Oikos 101: 79–90. 10.1034/j.1600-0706.2003.12322.x [DOI] [Google Scholar]
- 222. Cosgrove J (2009) Checklist of cephalopods (octopuses and squids) of British Columbia (November, 2009). In: Klinkenberg B, editor, E-Fauna BC: Electronic Atlas of the Fauna of British Columbia [www.efauna.bc.ca]. Lab for Advanced Spatial Analysis, Department of Geography, University of British Columbia, Vancouver: [Date Accessed: 2014-03-06], jacosgrove@telus.net: Royal British Columbia Museum. URL http://www.royalbcmusem.bc.ca/. [Google Scholar]
- 223. Arkhipkin AI, Bizikov VA (2000) Role of the statolith in functioning of the acceleration receptor system in squids and sepioids. Journal of Zoology 250: 31–55. 10.1111/j.1469-7998.2000.tb00575.x [DOI] [Google Scholar]
- 224. Lacoue-Labarthe T, Réveillac E, Oberhänsli F, Teyssié JL, Jeffree R, et al. (2011) Effects of ocean acidification on trace element accumulation in the early-life stages of squid Loligo vulgaris . Aquat Toxicol 105: 166–176. 10.1016/j.aquatox.2011.05.021 [DOI] [PubMed] [Google Scholar]
- 225. Kaplan MB, Mooney TA, McCorkle DC, Cohen AL (2013) Adverse effects of ocean acidification on early development of squid (Doryteuthis pealeii). PLoS ONE 8: e63714 10.1371/journal.pone.0063714 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 226. Rosa R, Seibel BA (2008) Synergistic effects of climate-related variables suggest future physiological impairment in a top oceanic predator. Proc Natl Acad Sci USA 105: 20776–20780. 10.1073/pnas.0806886105 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 227. Kurihara H (2008) Effects of CO2-driven ocean acidification on the early developmental stages of invertebrates. Mar Ecol Prog Ser 373: 275–284. 10.3354/meps07802 [DOI] [Google Scholar]
- 228. Conway KW, Barrie JV, Austin WC, Luternauer JL (1991) Holocene sponge bioherms on the western Canadian continental shelf. Cont Shelf Res 11: 771–790. 10.1016/0278-4343(91)90079-L [DOI] [Google Scholar]
- 229. Leys SP, Wilson K, Holeton C, Reiswig HM, Austin WC, et al. (2004) Patterns of glass sponge (Porifera, Hexactinellida) distribution in coastal waters of British Columbia, Canada. Mar Ecol Prog Ser 283: 133–149. 10.3354/meps283133 [DOI] [Google Scholar]
- 230. Stone RP, Conway DJ, Barrie JV (2013) The boundary reefs: glass sponge reefs on the international border between Canada and the United States. NOAA Tech Mem NMFS-AFSC-264: 31. [Google Scholar]
- 231. Roberts JM, Wheeler AJ, Freiwald A (2006) Reefs of the deep: the biology and geology of cold-water coral ecosystems. Science 312: 543–547. 10.1126/science.1119861 [DOI] [PubMed] [Google Scholar]
- 232. Jamieson GS, Pellegrin N, Jessen S (2006) Taxonomy and zoogeography of cold water corals in explored areas of coastal British Columbia. Can Sci Advis Sec Res Doc 2006/062: ii + 45 p. [Google Scholar]
- 233. Stone RP (2006) Coral habitat in the Aleutian Islands of Alaska: depth distribution, fine-scale species associations. Coral Reefs 25: 229–238. 10.1007/s00338-006-0091-z [DOI] [Google Scholar]
- 234. Rooper CN, Boldt JL, Zimmermann M (2007) An assessment of juvenile Pacific Ocean perch (Sebastes alutus) habitat use in a deepwater nursery. Estuar Coast Shelf Sci 75: 371–380. 10.1016/j.ecss.2007.05.006 [DOI] [Google Scholar]
- 235. Du Preez C, Tunnicliffe V (2011) Shortspine thornyhead and rockfish (Scorpaenidae) distribution in response to substratum, biogenic structures and trawling. Mar Ecol Prog Ser 425: 217–231. 10.3354/meps09005 [DOI] [Google Scholar]
- 236. Miller RJ, Hocevar J, Sone RP, Fedorov DV (2012) Structure-forming corals and sponges and their use as fish habitat in Bering Sea submarine canyons. PLoS ONE 7: e33885 10.1371/journal.pone.0033885 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 237. Finney JL, Boutillier P (2010) Distribution of cold-water coral, sponges and sponge reefs in British Columbia with options for identifying significant encounters. Can Sci Advis Sec Res Doc 2010/090: vi + 9 p. [Google Scholar]
- 238. Stone RP, Shotwell SK (2007) State of deep coral ecosystems in Alaska region: Gulf of Alaska, Bering Sea and Aleutian Islands. In: Lumsden SE, Hourigan TF, Bruckner AW, Dorr G, editors, The State of Deep Coral Ecosystems of the United States, NOAA Technical Memorandum CRCP-3; Silver Spring MD: pp. 65–108. [Google Scholar]
- 239. Cairns SD (2011) A revision of the Primoidae (Octocorallia: Alcyoncea) from the Aleutian Islands and Bering Sea. Smithson Contrib Zool 634: 55. [Google Scholar]
- 240. Finney JL (2010) Overlap of predicted cold-water coral habitat and bottom-contact fisheries in British Columbia. Can Sci Advis Sec Res Doc 2010/067: vi + 26 p. [Google Scholar]
- 241. Conway KW, Krautter M, Barrie JV, Whitney F, Thomson RE, et al. (2005) Sponge reefs in the Queen Charlotte Basin, Canada: controls on distribution, growth and development. In: Freiwald A, Roberts JM, editors, Coldwater Corals and Ecosystems, Springer Berlin Heidelberg; pp. 605–621. [Google Scholar]
- 242. Cairns SD, Lindner A (2011) A revision of the Stylasteridae (Cnidarian, Hydrozoa, Filifera) from Alaska and adjacent waters. ZooKeys 158: 1–88. 10.3897/zookeys.158.1910 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 243. Cairns SD (1994) Scleractinia of the temperate North Pacific. Smithson Contrib Zool 557: 150. [Google Scholar]
- 244. Conway KW, Barrie JV, Hill PR, Austin WC, Pickard K (2007) Mapping sensitive benthic habitats in the Strait of Georgia, coastal British Columbia: deep-water sponge and coral reefs. Geol Surv Can, Curr Res A2: 6p. [Google Scholar]
- 245. Guinotte JM, Orr J, Cairns SD, Freiwald A, Morgan L, et al. (2006) Will human-induced changes in seawater chemistry alter the distribution of deep-sea scleractinian corals? Front Ecol Environ 4: 141–146. 10.1890/1540-9295(2006)004[0141:WHCISC]2.0.CO;2 [DOI] [Google Scholar]
- 246. McCulloch M, Trotter J, Montagna P, Falter J, Dunbar R, et al. (2012) Resilience of cold-water scleractinian corals to ocean acidification: Boron isotopic systematics of pH and saturation state up-regulation. Geochim Cosmochim Acta 87: 21–34. 10.1016/j.gca.2012.03.027 [DOI] [Google Scholar]
- 247. Maier C, Hegeman J, Weinbauer MG, Gattuso JP (2009) Calcification of the cold-water Lophelia pertusa under ambient and reduced pH. Biogeosciences 6: 1671–1680. 10.5194/bg-6-1671-2009 [DOI] [Google Scholar]
- 248. Form AU, Riebesell U (2012) Acclimation to ocean acidification during long-term CO2 exposure in the cold-water coral Lophelia pertusa . Glob Change Biol 18: 843–853. 10.1111/j.1365-2486.2011.02583.x [DOI] [Google Scholar]
- 249. Bayer FM, Macintyre IG (2001) The mineral component of the axis and holdfast of some gorgonacean octocorals (Coelenterata: Anthozoa), with special reference to the family Gorgoniidae. Proc Biol Soc Wash 114: 309–345. [Google Scholar]
- 250. Cairns SD, Macintyre IG (1992) Phylogenetic implications of calcium carbonate mineralogy in the Stylasteridae (Cnidaria: Hydrozoa). Palaios 7: 96–107. 10.2307/3514799 [DOI] [Google Scholar]
- 251. Dupont S, Ortega-Martínez O, Thorndyke M (2010) Impact of near-future ocean acidification on echinoderms. Ecotoxicology 19: 449–462. 10.1007/s10646-010-0463-6 [DOI] [PubMed] [Google Scholar]
- 252.Lambert P (2007) Checklist of the echinoderms of British Columbia (April 2007). In: Klinkenberg, B, editor, E-Fauna BC: Electronic Atlas of the Fauna of British Columbia [www.efauna.bc.ca]. Lab for Advanced Spatial Analysis, Department of Geography, University of British Columbia, Vancouver. [Date Accessed: 2013-12-02], plambert@pacificcoast.net: Royal British Columbia Museum. URL http://www.royalbcmusem.bc.ca/.
- 253. Lambert P, Boutillier J (2011) Deep-sea echinodermata of British Columbia, Canada. Can Tech Rep Fish Aquat Sci 2929: viii + 143 p. [Google Scholar]
- 254. Paine RT (1966) Food web complexity and species diversity. Am Nat 100: 65–75. 10.1086/282400 [DOI] [Google Scholar]
- 255. Paine RT (1969) A note on trophic complexity and community stability. Am Nat 103: 91–93. 10.1086/282586 [DOI] [Google Scholar]
- 256. Lawrence JM (1975) On the relationship between marine plants and sea urchins. Oceanogr Mar Biol Ann Rev 13: 213–286. [Google Scholar]
- 257. Clark D, Lamare M, Barker M (2009) Response of sea urchin pluteus larvae (Echinodermata: Echinoidea) to reduced seawater pH: a comparison among a tropical, temperate, and a polar species. Mar Biol 156: 1125–1137. 10.1007/s00227-009-1155-8 [DOI] [Google Scholar]
- 258. Asnaghi V, Mangialajo L, Gattuso JP, Francour P, Privitera D, et al. (2014) Effects of ocean acidification and diet on thickness and carbonate elemental composition of the test of juvenile sea urchins. Mar Environ Res 93: 78–84. 10.1016/j.marenvres.2013.08.005 [DOI] [PubMed] [Google Scholar]
- 259. Dupont S, Dorey N, Stumpp M, Melzner F, Thorndyke M (2013) Long-term and trans-life-cycle effects of exposure to ocean acidification in the green sea urchin Strongylocentrotus droebachiensis . Mar Biol 160: 1835–1843. 10.1007/s00227-012-1921-x [DOI] [Google Scholar]
- 260. Reuter KE, Lotterhos KE, Crim RN, Thompson CA, Harley CDG (2011) Elevated pCO2 increases sperm limitation and risk of polyspermy in the red sea urchin Strongylocentrotus franciscanus . Glob Change Biol 17: 163–171. 10.1111/j.1365-2486.2010.02216.x [DOI] [Google Scholar]
- 261. O’Donnell MJ, Hammond LM, Hofmann GE (2009) Predicted impact of ocean acidification on a marine invertebrate: elevated CO2 alters response to thermal stress in sea urchin larvae. Mar Biol 156: 439–446. 10.1007/s00227-008-1097-6 [DOI] [Google Scholar]
- 262. Todgham AE, Hofmann GE (2009) Transcriptomic response of sea urchin larvae Strongylocentrotus purpuratus to CO2-driven seawater acidification. J Exp Biol 212: 2579–2594. 10.1242/jeb.032540 [DOI] [PubMed] [Google Scholar]
- 263. Pespeni MH, Sanford E, Gaylord B, Hill TM, Hosfelt JD, et al. (2013) Evolutionary change during experimental ocean acidification. Proc Natl Acad Sci USA 110: 6937–6942. 10.1073/pnas.1220673110 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 264. Morita M, Suwa R, Iguchi A, Nakamura M, Shimada K, et al. (2010) Ocean acidification reduces sperm flagellar motility in broadcast spawning reef invertebrates. Zygote 18: 103–107. 10.1017/S0967199409990177 [DOI] [PubMed] [Google Scholar]
- 265. Gooding RA, Harley CDG, Tang E (2009) Elevated water temperature and carbon dioxide concentration increase the growth of a keystone echinoderm. Proc Natl Acad Sci USA 106: 9316–9321. 10.1073/pnas.0811143106 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 266. Dupont S, Havenhand J, Thorndyke W, Peck L, Thorndyke M (2008) Near-future level of CO2-driven ocean acidification radically affects larval survival and development in the brittlestar Ophiothrix fragilis . Mar Ecol Prog Ser 373: 285–294. 10.3354/meps07800 [DOI] [Google Scholar]
- 267. Love MS (2011) Certainly More Than You Want to Know About The Fishes of The Pacific Coast: A Postmodern Experience. Really Big Press, Santa Barbara, California. [Google Scholar]
- 268. Smith KL Jr, Brown NO (1983) Oxygen consumption of pelagic juveniles and demersal adults of the deep-sea fish Sebastolobus altivelis, measured at depth. Mar Biol 76: 325–332. 10.1007/BF00393036 [DOI] [Google Scholar]
- 269. Gardner GA, Szabo I (1982) British Columbia pelagic marine copepoda: an identification manual and annotated bibliography. Can Spec Publ Fish Aquat Sci 62: 536 p. [Google Scholar]
- 270.Baldwin A (2009) Checklist of the barnacles of British Columbia (updated October 2009). In: Klinkenberg, B, editor, E-Fauna BC: Electronic Atlas of the Fauna of British Columbia [www.efauna.bc.ca]. Lab for Advanced Spatial Analysis, Department of Geography, University of British Columbia, Vancouver. [Date Accessed: 2014-02-05], plambert@pacificcoast.net: Royal British Columbia Museum. URL http://www.royalbcmusem.bc.ca/.
- 271.Baldwin A (2011) Checklist of the shrimps, crabs, lobsters and crayfish of British Columbia 2011 (order Decapoda). In: Klinkenberg B, editor, E-Fauna BC: Electronic Atlas of the Fauna of British Columbia [www.efauna.bc.ca]. Lab for Advanced Spatial Analysis, Department of Geography, University of British Columbia, Vancouver. [Date Accessed: 2014-02-03], plambert@pacificcoast.net: Royal British Columbia Museum. URL http://www.royalbcmusem.bc.ca/.
- 272. Nakagawa Y, Endo Y, Taki K (2001) Diet of Euphausia pacifica hansen in Sanriku waters off northeastern Japan. Plankton Biol Ecol 48: 68–77. [Google Scholar]
- 273.Fisheries and Oceans Canada (2014) Pacific Region Integrated Fisheries Management Plan—Crab by Trap—January 1, 2014 to December 31, 2014. http://www.pac.dfo-mpo.gc.ca/fm-gp/mplans/2014/crab-crabe-2014-eng.pdf, 227 pp.
- 274. Muzzarelli RAA (1977) Chitin. Pergamon Press, Oxford. [Google Scholar]
- 275. Ries JB, Cohen AL, McCorkle DC (2009) Marine calcifiers exhibit mixed responses to CO2-induced ocean acidification. Geology 37: 1131–1134. 10.1130/G30210A.1 [DOI] [Google Scholar]
- 276. Ries JB (2011) Skeletal mineralogy in a high-CO2 world. J Exp Mar Biol Ecol 403: 54–64. 10.1016/j.jembe.2011.04.006 [DOI] [Google Scholar]
- 277. Findlay HS, Wood HL, Kendall MA, Spicer JI, Twitchett RJ, et al. (2011) Comparing the impact of high co2 on calcium carbonate structures in different marine organisms. Mar Biol Res 7: 565–575. 10.1080/17451000.2010.547200 [DOI] [Google Scholar]
- 278. Whiteley NM (2011) Physiological and ecological responses of crustaceans to ocean acidification. Mar Ecol Prog Ser 430: 257–271. 10.3354/meps09185 [DOI] [Google Scholar]
- 279. Long WC, Swiney KM, Harris C, Page HN, Foy RJ (2013) Effects of ocean acidification on juvenile Red King Crab (Paralithodes camtschaticus) and Tanner crab (Chionoecetes bairdi) growth, condition calcification and survival. PLOS ONE 8: e60959 10.1371/journal.pone.0060959 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 280. Long WC, Swiney KM, Foy RJ (2013) Effects of ocean acidification on the embryos and larvae of Red King Crab (Paralithodes camtschaticus). Mar Poll Bull 69: 38–47. 10.1016/j.marpolbul.2013.01.011 [DOI] [PubMed] [Google Scholar]
- 281. Saba GK, Schofield O, Torres JJ, Ombres EH, Steinberg DK (2012) Increased feeding and nutrient excretion of adult Antarctic krill, Euphausia superba, exposed to enhanced carbon dioxide (CO2). PLoS ONE 7: e52224 10.1371/journal.pone.0052224 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 282. Findlay HS, Kendall MA, Spicer JI, Widdicombe S (2009) Future high CO2 in the intertidal may compromise adult barnacle Semibalanus balanoides survival and embryonic development rate. Mar Ecol Prog Ser 389: 193–202. 10.3354/meps08141 [DOI] [Google Scholar]
- 283. Bechmann RK, Taban IC, Westerlund S, Godal BF, Arnberg M, et al. (2011) Effects of ocean acidification on early life stages of shrimp (Pandalus borealis) and mussel (Mytilus edulis). J Toxicol Environ Health A 74: 424–438. 10.1080/15287394.2011.550460 [DOI] [PubMed] [Google Scholar]
- 284. McDonald MR, McClintock JB, Amsler CD, Rittschof D, Angus RA, et al. (2009) Effects of ocean acidification over the life history of the barnacle Amphibalanus amphitrite . Mar Ecol Prog Ser 385: 179–187. 10.3354/meps08099 [DOI] [Google Scholar]
- 285. Pansch C, Nasrolahi A, Appelhans YS, Wahl M (2012) Impacts of ocean warming and acidification on the larval development of the barnacle Amphibalanus improvisus . J Exp Mar Biol Ecol 420–421: 48–55. 10.1016/j.jembe.2012.03.023 [DOI] [Google Scholar]
- 286. Pansch C, Schlegel P, Havenhand J (2013) Larval development of the barnacle Amphibalanus improvisus responds variably but robustly to near-future ocean acidification. ICES J Mar Sci 70: 805–811. 10.1093/icesjms/fst092 [DOI] [Google Scholar]
- 287. Pansch C, Schaub I, Havenhand J, Wahl M (2014) Habitat traits and food availability determine the response of marine invertebrates to ocean acidification. Glob Change Biol 20: 765–777. 10.1111/gcb.12478 [DOI] [PubMed] [Google Scholar]
- 288. Cushing DH (1969) The regularity of the spawning season of some fishes. ICES J Mar Sci 33: 81–92. 10.1093/icesjms/33.1.81 [DOI] [Google Scholar]
- 289. Ryer CH, van Montfrans J, Moody KE (1997) Cannibalism, refugia and the molting blue crab. Mar Ecol Prog Ser 147: 77–85. 10.3354/meps147077 [DOI] [Google Scholar]
- 290. Kunkel JG, Nagel W, Jercinovic MJ (2012) Mineral fine structure of the American lobster cuticle. J Shellfish Res 31: 515–526. 10.2983/035.031.0211 [DOI] [Google Scholar]
- 291. Punt AE, Poljak D, Dalton MG, Foy RJ (2014) Evaluating the impact of ocean acidification on fishery yields and profits: The example of the red king crab in Bristol Bay. Ecol Mod 285: 39–53. 10.1016/j.ecolmodel.2014.04.017 [DOI] [Google Scholar]
- 292. Hart JL (1973) Pacific fishes of Canada. Bull Fish Res Bd Can 180: ix + 740 p. [Google Scholar]
- 293.Peden A (2002) An introduction to the marine fish of British Columbia. In: Klinkenberg B, editor, E-Fauna BC: Electronic Atlas of the Fauna of British Columbia [www.efauna.bc.ca], Lab for Advanced Spatial Analysis, Department of Geography, University of British Columbia, Vancouver. [Date Accessed: 2013-11-27].
- 294. Naiman RJ, Bilby RE, Schindler DE, Helfield JM (2002) Pacific salmon, nutrients, and the dynamics of freshwater and riparian ecosystems. Ecosystems 5: 399–417. 10.1007/s10021-001-0083-3 [DOI] [Google Scholar]
- 295. Melzner F, Göbel S, Langenbuch M, Gutowska MA, Pörtner HO, et al. (2009) Swimming performance in Atlantic Cod (Gadus morhua) following long-term (4–12 months) acclimation to elevated seawater P CO2 . Aquat Toxicol 92: 30–37. 10.1016/j.aquatox.2008.12.011 [DOI] [PubMed] [Google Scholar]
- 296. Melzner F, Gutowska MA, Langenbuch M, Dupont S, Lucassen M, et al. (2009) Physiological basis for high CO2 tolerance in marine ectothermic animals: pre-adaptation through lifestyle and ontogeny? Biogeosciences 6: 2313–2331. 10.5194/bg-6-2313-2009 [DOI] [Google Scholar]
- 297. Munday PL, Crawley NE, Nilsson GE (2009) Interacting effects of elevated temperature and ocean acidification on the aerobic performance of coral reef fishes. Mar Ecol Prog Ser 388: 235–242. 10.3354/meps08137 [DOI] [Google Scholar]
- 298. Ishimatsu A, Hayashi M, Kikkawa T (2008) Fishes in high-CO2 and acidified oceans. Mar Ecol Prog Ser 373: 295–302. 10.3354/meps07823 [DOI] [Google Scholar]
- 299. Leduc AOHC, Munday PL, Brown GE, Ferrari MCO (2013) Effects of acidification on olfactory-mediated behaviour in freshwater and marine ecosystems: a synthesis. Phil Trans R Soc Lond B, Biol Sci 368: 20120447 10.1098/rstb.2012.0447 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 300. Frommel AY, Maneja R, Lowe D, Malzahn AM, Geffen AJ, et al. (2012) Severe tissue damage in Atlantic cod larvae under increasing ocean acidification. Nature Clim Change 2: 42–46. 10.1038/nclimate1324 [DOI] [Google Scholar]
- 301. Kikkawa T, Kita J, Ishimatsu A (2004) Comparison of the lethal effect of CO2 and acidification on red sea bream (Pagrus major) during the early developmental stages. Mar Pollut Bull 48: 108–110. 10.1016/S0025-326X(03)00367-9 [DOI] [PubMed] [Google Scholar]
- 302. Ishimatsu A, Kikkawa T, Hayashi M, Lee KS, Kita J (2004) Effects of CO2 on marine fish: larvae and adults. J Oceanogr 60: 731–741. 10.1007/s10872-004-5765-y [DOI] [Google Scholar]
- 303. Hurst TP, Fernandez ER, Mathis JT (2013) Effects of ocean acidification on hatch size and larval growth of walleye pollock (Theragra chalcogramma). ICES J Mar Sci 70: 812–822. 10.1093/icesjms/fst053 [DOI] [Google Scholar]
- 304. Frommel AY, Stiebens V, Clemmesen C, Havenhand J (2010) Effect of ocean acidification on marine fish sperm (Baltic cod: Gadus morhua). Biogeosciences 7: 3915–3919. 10.5194/bg-7-3915-2010 [DOI] [Google Scholar]
- 305. Franke A, Clemmesen C (2011) Effect of ocean acidification on early life stages of Atlantic herring (Clupea harengus L.). Biogeosciences 8: 3697–3707. 10.5194/bg-8-3697-2011 [DOI] [Google Scholar]
- 306. Baumann H, Talmage SC, Gobler CJ (2012) Reduced early life growth and survival in a fish in direct response to increased carbon dioxide. Nature Clim Change 2: 38–41. 10.1038/nclimate1291 [DOI] [Google Scholar]
- 307. Checkley DMJ, Dickson AG, Takahashi M, Radich JA, Eisenkolb N, et al. (2009) Elevated CO2 enhances otolith growth in young fish. Science 324: 1683–1683. 10.1126/science.1169806 [DOI] [PubMed] [Google Scholar]
- 308. Maneja RH, Frommel AY, Geffen AJ, Folkvord A, Piatkowski U, et al. (2013) Effects of ocean acidification on the calcification of otoliths of larval Atlantic cod Gadus morhua . Mar Ecol Prog Ser 477: 251–258. 10.3354/meps10146 [DOI] [Google Scholar]
- 309. Munday PL, Hernaman V, Dixson DL, Thorrold SR (2011) Effect of ocean acidification on otolith development in larvae of a tropical marine fish. Biogeosciences 8: 1631–1641. 10.5194/bg-8-1631-2011 [DOI] [Google Scholar]
- 310. Bignami S, Enochs IC, Manzello DP, Sponaugle S, Cowen RK (2013) Ocean acidification alters the otoliths of a pantropical fish species with implications for sensory function. Proc Natl Acad Sci USA 110: 7366–7370. 10.1073/pnas.1301365110 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 311. Dixson DL, Munday PL, Jones GP (2010) Ocean acidification disrupts the innate ability of fish to detect predator olfactory cues. Ecol Lett 13: 68–75. 10.1111/j.1461-0248.2009.01400.x [DOI] [PubMed] [Google Scholar]
- 312. Ferrari MCO, McCormick MI, Munday PL, Meekan MG, Dixson DL, et al. (2012) Effects of ocean acidification on visual risk assessment in coral reef fishes. Funct Ecol 26: 553–558. 10.1111/j.1365-2435.2011.01951.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 313. Munday PL, Dixson DL, McCormick MI, Meekan M, Ferrari MCO, et al. (2010) Replenishment of fish populations is threatened by ocean acidification. Proc Natl Acad Sci USA 107: 12930–12934. 10.1073/pnas.1004519107 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 314. Munday PL, Dixson DL, Donelson JM, Jones GP, Pratchett MS, et al. (2009) Ocean acidification impairs olfactory discrimination and homing ability of a marine fish. Proc Natl Acad Sci USA 106: 1848–1852. 10.1073/pnas.0809996106 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 315. Simpson SD, Munday PL, Wittenrich ML, Manassa R, Dixson DL, et al. (2011) Ocean acidification erodes crucial auditory behaviour in a marine fish. Biol Lett 7: 917–920. 10.1098/rsbl.2011.0293 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 316. Ferrari MCO, Manassa RP, Dixson DL, Munday PL, McCormick MI, et al. (2012) Effects of ocean acidification on learning in coral reef fishes. PLoS ONE 7: e31478 10.1371/journal.pone.0031478 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 317. Domenici P, Allan B, McCormick MI, Munday PL (2012) Elevated carbon dioxide affects behavioural lateralization in a coral reef fish. Biol Lett 8: 78–81. 10.1098/rsbl.2011.0591 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 318. Ferrari MCO, McCormick MI, Munday PL, Meekan MG, Dixson DL, et al. (2011) Putting prey and predator into the CO2 equation—qualitative and quantitative effects of ocean acidification on predator-prey interactions. Ecol Lett 14: 1143–1148. 10.1111/j.1461-0248.2011.01683.x [DOI] [PubMed] [Google Scholar]
- 319. Cripps IL, Munday PL, McCormick MI (2011) Ocean acidification affects prey detection by a predatory reef fish. PLoS ONE 6: e22736 10.1371/journal.pone.0022736 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 320. Jutfelt F, de Souza KB, Vuylsteke A, Sturve J (2013) Behavioural disturbances in a temperate fish exposed to sustained high-CO2 levels. PLoS ONE 8: e65825 10.1371/journal.pone.0065825 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 321. Maneja RH, Frommel AY, Browman HI, Clemmesen C, Geffen AJ, et al. (2013) The swimming kinematics of larval Atlantic cod, Gadus morhua L., are resilient to elevated seawater pCO2 . Mar Biol 160: 1963–1972. 10.1007/s00227-012-2054-y [DOI] [Google Scholar]
- 322. Dixson DL, Jennings AR, Munday PL (2014) Odor tracking in sharks is reduced under future ocean acidification conditions. Glob Change Biol. [DOI] [PubMed] [Google Scholar]
- 323. Nilsson GE, Dixson DL, Domenici P, McCormick MI, Sorensen C, et al. (2012) Near-future carbon dioxide levels alter fish behaviour by interfering with neurotransmitter function. Nature Clim Change 2: 201–204. 10.1038/nclimate1352 [DOI] [Google Scholar]
- 324. McFarlane GA, Ware DM, Thomson RE, Mackas DL, Robinson CLK (1997) Physical, biological and fisheries oceanography of a large ecosystem (west coast of Vancouver Island) and implications for management. Oceanologica Acta 20: 191–200. [Google Scholar]
- 325. Trainer VL, Hickey BM, Lessard EJ, Cochlan WP, Trick CG, et al. (2009) Variability of Pseudo-nitzschia and domoic acid in the Juan de Fuca eddy region and its adjacent shelves. Limnol Oceanogr 54: 289–308. 10.4319/lo.2009.54.1.0289 [DOI] [Google Scholar]
- 326. Willette TM, Cooney RT, Patrick V, Mason DM, Thomas GL, et al. (2001) Ecological processes influencing mortality of juvenile pink salmon (Oncorhynchus gorbuscha) in Prince William Sound, Alaska. Fish Oceanogr 10: 14–41. 10.1046/j.1054-6006.2001.00043.x [DOI] [Google Scholar]
- 327. Aydin KY, McFarlane GA, King JR, Megrey BA, Myers KW (2005) Linking oceanic food webs to coastal production and growth rates of Pacific salmon (Oncorhynchus spp.), using models on three scales. Deep Sea Research Part II: Topical Studies in Oceanography 52: 757–780. 10.1016/j.dsr2.2004.12.017 [DOI] [Google Scholar]
- 328. Kaplan IC, Levin PS, Burden M, Fulton EA (2010) Fishing catch shares in the face of global change: a framework for integrating cumulative impacts and single species management. Can J Fish Aquat Sci 67: 1968–1982. 10.1139/F10-118 [DOI] [Google Scholar]
- 329.Nagorsen D (2009) Mammals of British Columbia (2009). In: Klinkenberg B, editor, E-Fauna BC: Electronic Atlas of the Fauna of British Columbia [www.efauna.bc.ca]. Lab for Advanced Spatial Analysis, Department of Geography, University of British Columbia, Vancouver. [Date Accessed: 2014-04-14], plambert@pacificcoast.net: Royal British Columbia Museum. URL http://www.royalbcmusem.bc.ca/.
- 330. Ianson D, Flostrand L (2010) Coastal waters off the west coast of Vancouver Island, British Columbia. In: Ecosystem and Status Trends Report. Can Sci Advis Sec Res Doc, p. 56. [Google Scholar]
- 331. Loomis J, Larson D (1994) Total economic values of increasing gray whale populations: results from a contingent valuation survey of visitors and households. Marine Resource Economics 9: 275–286. [Google Scholar]
- 332. Ford JK (2014) Marine Mammals of British Columbia. Victoria, Canada: Royal BC Museum, Victoria Canada, 460 pp. [Google Scholar]
- 333.Bass CL, Simmonds MP, Isaac SJ (2006) An overview of the potential consequences for cetaceans of oceanic acidification. IWC, Scientific Committee SC/58/E10: 6 p.
- 334. Hester KC, Peltzer ET, Kirkwood WJ, Brewer PG (2008) Unanticipated consequences of ocean acidification: a noisier ocean at lower pH. Geophys Res Lett 35: L19601 10.1029/2008GL034913 [DOI] [Google Scholar]
- 335. Joseph JE, Chiu C (2010) A computational assessment of the sensitivity of ambient noise level to ocean acidification. J Acoust Soc Am 128: 3. [DOI] [PubMed] [Google Scholar]
- 336. Kelly MW, Hofmann GE (2012) Adaptation and the physiology of ocean acidification. Funct Ecol 27: 980–990. 10.1111/j.1365-2435.2012.02061.x [DOI] [Google Scholar]
- 337. Godbold JA, Calosi P (2013) Ocean acidification and climate change: advances in ecology and evolution. Phil Trans R Soc Lond B, Biol Sci 368: 20120448 10.1098/rstb.2012.0448 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 338. Allen SE, Wolfe MA (2013) Hindcast of the timing of the spring phytoplankton bloom in the Strait of Georgia, 1968–2010. Prog Oceanogr 26: 81–87. [Google Scholar]
- 339. Merryfield WJ, Pal B, Foreman MGG (2009) Projected future changes in surface marine winds off the west coast of Canada. J Geophys Res 114: C06008. [Google Scholar]
- 340. Foreman MGG, Pal B, Merryfield WJ (2011) Trends in upwelling and downwelling winds along the British Columbia shelf. J Geophys Res 116: C10023 10.1029/2011JC006995 [DOI] [Google Scholar]
- 341. Bylhouwer B, Ianson D, Kohfeld K (2013) Changes in the onset and intensity of wind-driven coastal upwelling and downwelling along the North American Pacific coast. J Geophys Res Oceans 118: 1–16. [Google Scholar]
- 342. Beaugrand G (2009) Decadal changes in climate and ecosystems in the North Atlantic Ocean and adjacent seas. Deep Sea Research II 56: 656–673. 10.1016/j.dsr2.2008.12.022 [DOI] [Google Scholar]
- 343. Pörtner HO (2009) Ecosystem effects of ocean acidification in times of ocean warming: a physiologist’s view. Mar Ecol Prog Ser 373: 203–217. [Google Scholar]
- 344. Griffith GP, Fulton EA, Gorton R, Richardson AJ (2012) Predicting interactions among fishing, ocean warming, and ocean acidification in a marine system with whole-ecosystem models. Conserv Biol 26: 1145–1152. 10.1111/j.1523-1739.2012.01937.x [DOI] [PubMed] [Google Scholar]
- 345.CRIMS (2013) Saltwater Finfish and Shellfish Tenures (geospatial dataset, accessed 2013–12–19). In: Coastal Resource Information Management System, DataBC, Ministry of Forests, Lands and Natural Resource Operations, Province of British Columbia. https://apps.gov.bc.ca/pub/geometadata/ (finfish: UID = 4025; shellfish: UID = 4031). URL https://apps.gov.bc.ca/pub/geometadata/.
- 346.International Hydrographic Organization Data Centre (2014) IHO-IOC GEBCO Gazetteer of Undersea Feature Names. In: General Bathymetric Chart of the Oceans, www.gebco.net URL http://www.gebco.net/data_and_products/undersea_feature_names/.
- 347.R Core Team (2014) R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna, Austria. URL http://www.R-project.org/.
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Number of hits by Web of Science for OA-related studies on different groups of animals in March 2013 and March 2014.
(PDF)
The experimental details for all manipulation experiments referred to in this document.
(PDF)
Details (e.g. species, location, and carbon state) for OA-related in situ studies of marine organisms.
(PDF)
Marine animal use protocols in British Columbia, Canada.
(PDF)
Data Availability Statement
Fig. 1: Saltwater finfish and hatchery sites and commercial marine shellfish farms (referenced in Figure caption) are available at: https://apps.gov.bc.ca/pub/geometadata/. R code used to make the figure is available as a Supporting Information file titled S1 Code. Fig 2: Values of PCO2 and pH are calculated from published data (refs 27 and 40). We include these data in the R code used to make the figure in S2 Code. Fig. 3: We include the data used to make this figure—S1 Data—and the R code used to make this figure—S3 Code—as supporting information. Fig. 4: Landed values are available publicly from http://www.env.gov.bc.ca/omfd/reports/Seafood-YIR-2011.pdf (ref 34) and (for euphausiids) from http://www.pac.dfo-mpo.gc.ca/fm-gp/mplans/2013/krill-sm-2013-17-eng.pdf (ref 39). We include the data in the R code used to make this figure in S4 Code.