Abstract
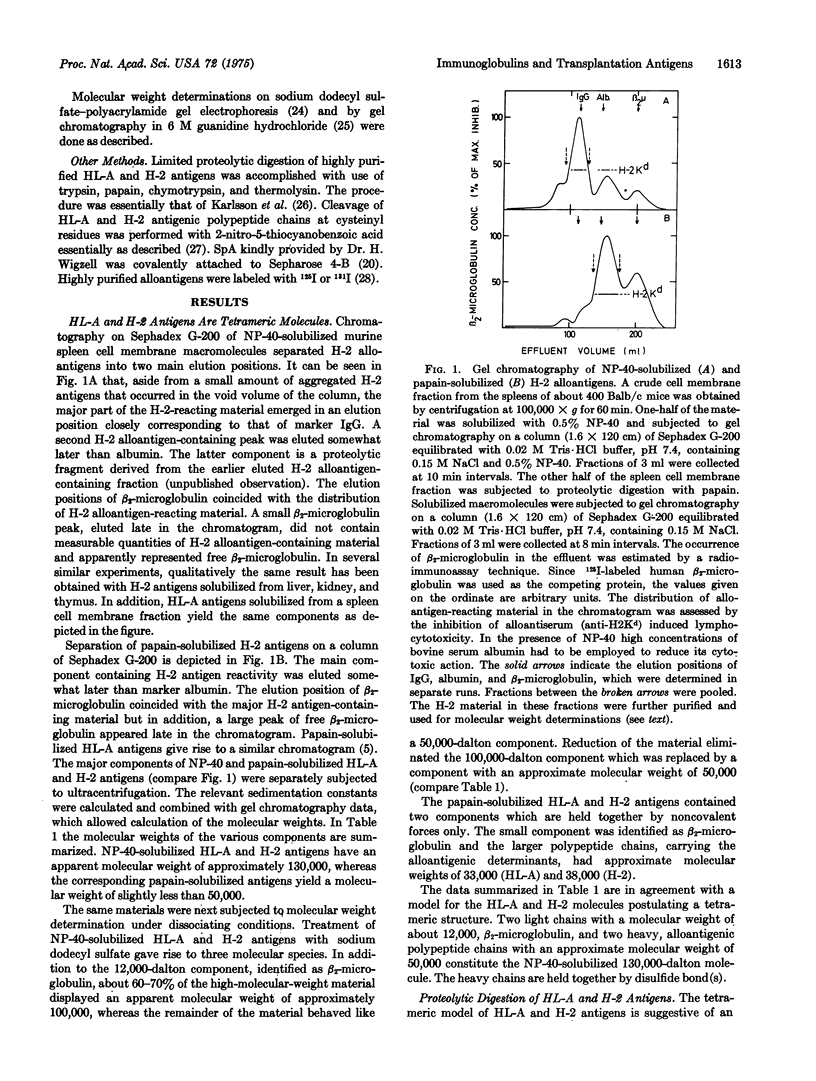
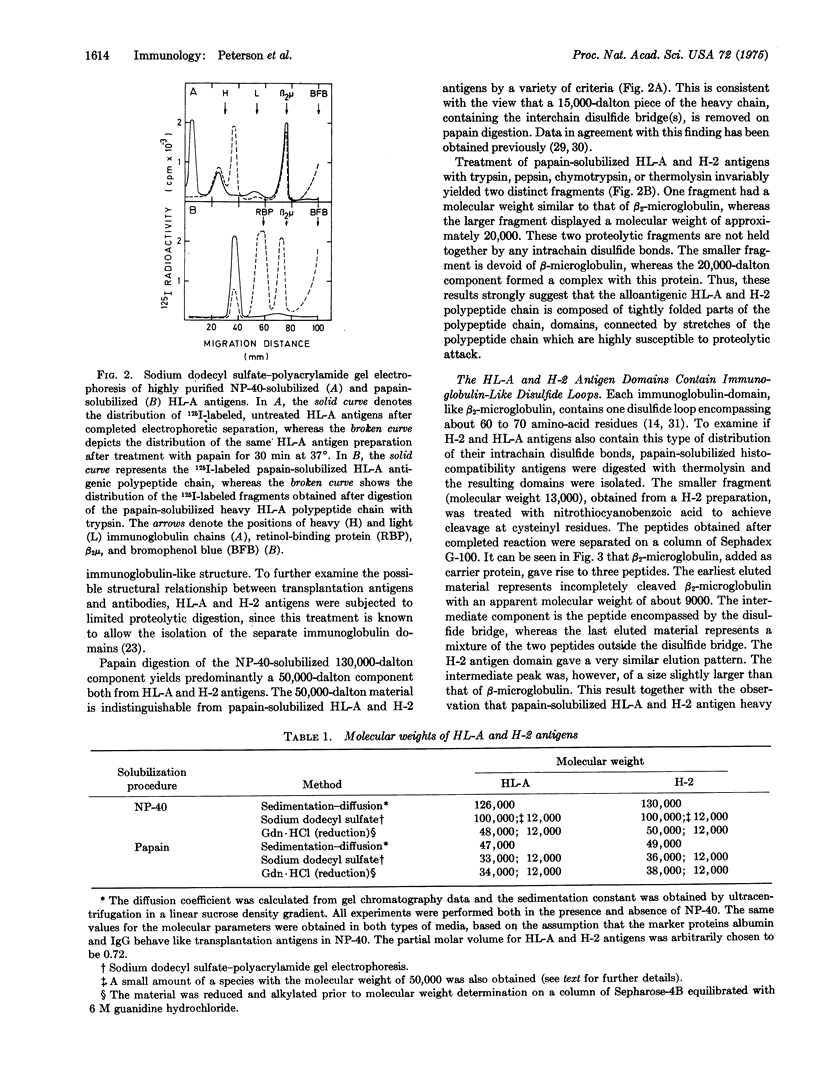
The major human and murine histocompatibility antigens are tetrameric molecules with an apparent molecular weight of about 130,000. They are composed of two types of polypeptide chains. The two light chains, previously identified as beta2-microglobulins, are bound to the two heavy, alloantigenic HL-A or H-2 polypeptide chains by noncovalent interactions only. The heavy chains are held together by disulfide bridge(s) located in the part of the molecule that is attached to the cell membrane. By limited proteolysis of the histocompatibility antigens evidence was obtained suggesting that the heavy chain may consist of three compact domains connected by more extended stretches of polypeptide chain. Each domain appeared to contain a single disulfide bride encompassing about 60 to 70 amino-acid residues. Staphylococcus aureus protein A is known to bind exclusively to the Fe region of immunoglobulin G. It was, however, observed that protein A interacts in a similar way with the H-2 antigen heavy chain. This observation, together withthe homology of the primary structure of beta2-microglobulin to immunoglobulin G, the tetrameric structure of the alloantigens, the ogranizations of the heavy polypeptide chain into compact domains, and the presence of a single, immunoglobulin-like disulfide loop in each domain, establishes a close similarity in structure between histocompatibility antigens and immunoglobulins. The similarity in structural features suggests a common evolutionary origin of the two types of molecules.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Cresswell P., Springer T., Strominger J. L., Turner M. J., Grey H. M., Kubo R. T. Immunological identity of the small subunit of HL-A antigens and beta2-microglobulin and its turnover on the cell membrane. Proc Natl Acad Sci U S A. 1974 May;71(5):2123–2127. doi: 10.1073/pnas.71.5.2123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cresswell P., Turner M. J., Strominger J. L. Papain-solubilized HL-A antigens from cultured human lymphocytes contain two peptide fragments. Proc Natl Acad Sci U S A. 1973 May;70(5):1603–1607. doi: 10.1073/pnas.70.5.1603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cuatrecasas P. Protein purification by affinity chromatography. Derivatizations of agarose and polyacrylamide beads. J Biol Chem. 1970 Jun;245(12):3059–3065. [PubMed] [Google Scholar]
- Cunningham B. A., Wang J. L., Berggård I., Peterson P. A. The complete amino acid sequence of beta 2-microglobulin. Biochemistry. 1973 Nov 20;12(24):4811–4822. doi: 10.1021/bi00748a001. [DOI] [PubMed] [Google Scholar]
- Edelman G. M., Gall W. E. The antibody problem. Annu Rev Biochem. 1969;38:415–466. doi: 10.1146/annurev.bi.38.070169.002215. [DOI] [PubMed] [Google Scholar]
- Evrin P. E., Peterson P. A., Wide L., Berggård I. Radioimmunoassay of 2 -microglobulin in human biological fluids. Scand J Clin Lab Invest. 1971 Dec;28(4):439–443. doi: 10.3109/00365517109095721. [DOI] [PubMed] [Google Scholar]
- Forsgren A., Sjöquist J. "Protein A" from S. aureus. I. Pseudo-immune reaction with human gamma-globulin. J Immunol. 1966 Dec;97(6):822–827. [PubMed] [Google Scholar]
- Forsgren A., Sjöquist J. "Protein A" from Staphylococcus aureus. 3. Reaction with rabbit gamma-globulin. J Immunol. 1967 Jul;99(1):19–24. [PubMed] [Google Scholar]
- Gally J. A., Edelman G. M. The genetic control of immunoglobulin synthesis. Annu Rev Genet. 1972;6:1–46. doi: 10.1146/annurev.ge.06.120172.000245. [DOI] [PubMed] [Google Scholar]
- Grey H. M., Kubo R. T., Colon S. M., Poulik M. D., Cresswell P., Springer T., Turner M., Strominger J. L. The small subunit of HL-A antigens is beta 2-microglobulin. J Exp Med. 1973 Dec 1;138(6):1608–1612. doi: 10.1084/jem.138.6.1608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- HUNTER W. M., GREENWOOD F. C. Preparation of iodine-131 labelled human growth hormone of high specific activity. Nature. 1962 May 5;194:495–496. doi: 10.1038/194495a0. [DOI] [PubMed] [Google Scholar]
- Jacobson G. R., Schaffer M. H., Stark G. R., Vanaman T. C. Specific chemical cleavage in high yield at the amino peptide bonds of cysteine and cystine residues. J Biol Chem. 1973 Oct 10;248(19):6583–6591. [PubMed] [Google Scholar]
- Karlsson F. A., Peterson P. A., Berggard I. Properties of halves of immunoglobulin light chains. Proc Natl Acad Sci U S A. 1969 Dec;64(4):1257–1263. doi: 10.1073/pnas.64.4.1257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karlsson F. A., Peterson P. A., Berggård I. A structural feature of human immunoglobulin light chains. Two compact domains connected by a small switch region. J Biol Chem. 1972 Feb 25;247(4):1065–1073. [PubMed] [Google Scholar]
- Nakamuro K., Tanigaki N., Pressman D. Multiple common properties of human beta2-microglobulin and the common portion fragment derived from HL-A antigen molecules. Proc Natl Acad Sci U S A. 1973 Oct;70(10):2863–2865. doi: 10.1073/pnas.70.10.2863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neville D. M., Jr Molecular weight determination of protein-dodecyl sulfate complexes by gel electrophoresis in a discontinuous buffer system. J Biol Chem. 1971 Oct 25;246(20):6328–6334. [PubMed] [Google Scholar]
- Nilsson S. F., Peterson P. A. Evidence for multiple thyroxine-binding sites in human prealbumin. J Biol Chem. 1971 Oct 10;246(19):6098–6105. [PubMed] [Google Scholar]
- Ostberg L., Rask L., Wigzell H., Peterson P. A. Thymus leukaemia antigen contains beta2-microglobulin. Nature. 1975 Feb 27;253(5494):735–737. doi: 10.1038/253735a0. [DOI] [PubMed] [Google Scholar]
- Peterson P. A., Cunningham B. A., Berggård I., Edelman G. M. 2 -Microglobulin--a free immunoglobulin domain. Proc Natl Acad Sci U S A. 1972 Jul;69(7):1697–1701. doi: 10.1073/pnas.69.7.1697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peterson P. A., Evrin P. E., Berggård I. Differentiation of glomerular, tubular, and normal proteinuria: determinations of urinary excretion of beta-2-macroglobulin, albumin, and total protein. J Clin Invest. 1969 Jul;48(7):1189–1198. doi: 10.1172/JCI106083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peterson P. A., Rask L., Lindblom J. B. Highly purified papain-solubilized HL-A antigens contain beta2-microglobulin. Proc Natl Acad Sci U S A. 1974 Jan;71(1):35–39. doi: 10.1073/pnas.71.1.35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rask L., Lindblom J. B., Peterson P. A. Subunit structure of H-2 alloantigens. Nature. 1974 Jun 28;249(460):833–834. doi: 10.1038/249833a0. [DOI] [PubMed] [Google Scholar]
- Rask L., Ostberg L., Lindblom B., Fernstedt Y., Peterson P. A. The subunit structure of transplantation antigens. Transplant Rev. 1974;21(0):85–105. doi: 10.1111/j.1600-065x.1974.tb01547.x. [DOI] [PubMed] [Google Scholar]
- Schwartz B. D., Kato K., Cullen S. E., Nathenson S. G. H-2 histocompatibility alloantigens. Some biochemical properties of the molecules solubilized by NP-40 detergent. Biochemistry. 1973 May 22;12(11):2157–2164. doi: 10.1021/bi00735a023. [DOI] [PubMed] [Google Scholar]
- Schwartz B. D., Nathenson S. G. Isolation of H-2 alloantigens solubilized by the detergent NP-40. J Immunol. 1971 Nov;107(5):1363–1367. [PubMed] [Google Scholar]
- Silver J., Hood L. Detergent-solubilised H-2 alloantigen is associated with a small molecular weight polypeptide. Nature. 1974 Jun 21;249(459):764–765. doi: 10.1038/249764a0. [DOI] [PubMed] [Google Scholar]
- Smithies O., Poulik M. D. Initiation of protein synthesis at an unusual position in an immunoglobulin gene? Science. 1972 Jan 14;175(4018):187–189. doi: 10.1126/science.175.4018.187. [DOI] [PubMed] [Google Scholar]
- Springer T. A., Strominger J. L., Mann D. Partial purification of detergent-soluble HL-A antigen and its cleavage by papain. Proc Natl Acad Sci U S A. 1974 Apr;71(4):1539–1543. doi: 10.1073/pnas.71.4.1539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Strominger J. L., Cresswell P., Grey H., Humphreys R. E., Mann D., McCuneJ, Parham P., Robb R., Sanderson A. R., Springer T. A. The immunoglobulin-like structure of human histocompatibility antigens. Transplant Rev. 1974;21(0):126–143. doi: 10.1111/j.1600-065x.1974.tb01549.x. [DOI] [PubMed] [Google Scholar]
- Tanigaki N., Katagiri M., Nakamuro K., Kreiter V. P., Pressman D. Common antigenic structures of HL-A antigens. II. Small fragments derived from papain-solubilized HL-A antigen molecules. Immunology. 1974 Jan;26(1):155–168. [PMC free article] [PubMed] [Google Scholar]
- Thorsby E. The human major histocompatibility system. Transplant Rev. 1974;18:51–129. [PubMed] [Google Scholar]
- WIGZELL H. QUANTITATIVE TITRATIONS OF MOUSE H-2 ANTIBODIES USING CR-51-LABELLED TARGET CELLS. Transplantation. 1965 May;3:423–431. doi: 10.1097/00007890-196505000-00011. [DOI] [PubMed] [Google Scholar]



