Abstract
A heat stable protein essentail for microtubule assembly has been isolated. This protein, which we designate tau (tau), is present in association with tubulin purified from porcine brain by repeated cycles of polymerization. Tau is separated from tubulin by ion exchange chromatography on phosphocellulose. In the absence of tau, tubulin exists entirely as a 6S dimer of two polypeptide chains (alpha and beta tubulin) with a molecular weight of 120,000, which will not assemble into microtubules in vitro. Addition of tau completely restores tubule-forming capacity. Under nonpolymerizing conditions, tau converts 6S dimers to 36S rings-structures which have been implicated as intermediates in tubule formation. Hence, tau appears to act on the 6S tubulin dimer, activating it for polymerization. The unique ability of tau to restore the normal features of in vitro microtubule assembly makes it likely that tau is a major regulator of microtubule formation in cells.
Full text
PDF

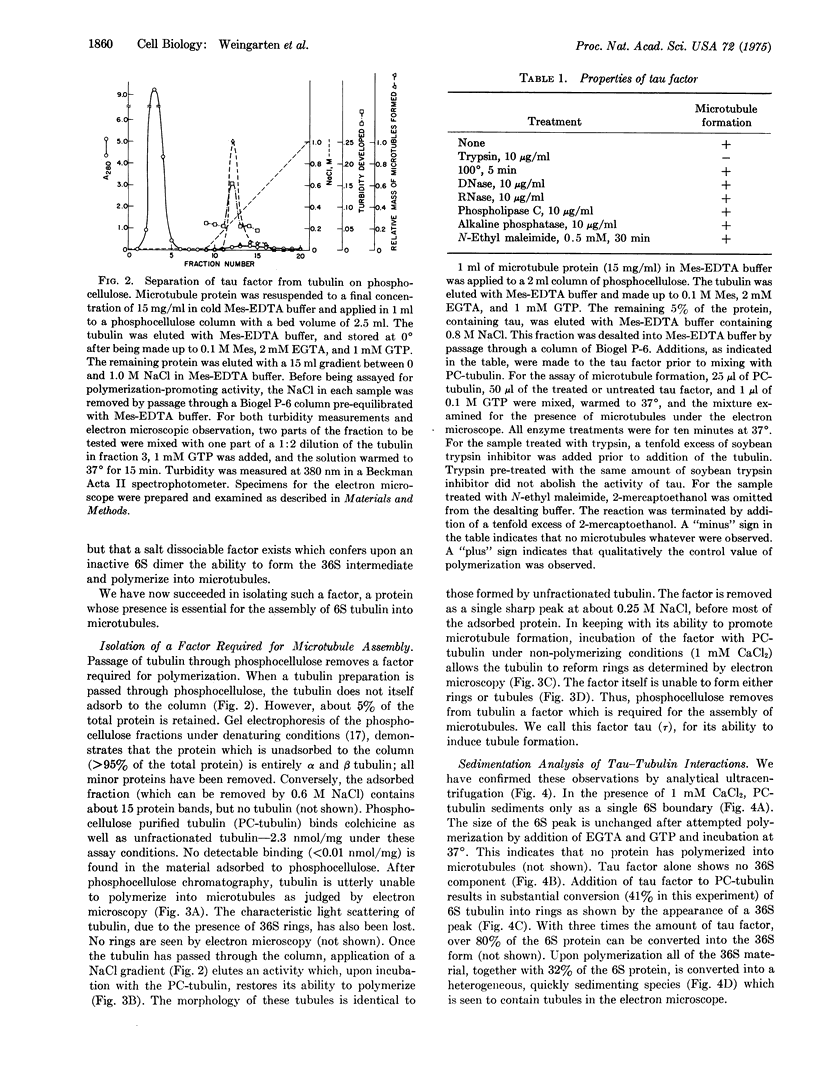


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Borisy G. G., Olmsted J. B., Marcum J. M., Allen C. Microtubule assembly in vitro. Fed Proc. 1974 Feb;33(2):167–174. [PubMed] [Google Scholar]
- Borisy G. G., Olmsted J. B. Nucleated assembly of microtubules in porcine brain extracts. Science. 1972 Sep 29;177(4055):1196–1197. doi: 10.1126/science.177.4055.1196. [DOI] [PubMed] [Google Scholar]
- Eipper B. A. Properties of rat brain tubulin. J Biol Chem. 1974 Mar 10;249(5):1407–1416. [PubMed] [Google Scholar]
- Erickson H. P. Assembly of microtubules from preformed, ring-shaped protofilaments and 6-S tubulin. J Supramol Struct. 1974;2(2-4):393–411. doi: 10.1002/jss.400020228. [DOI] [PubMed] [Google Scholar]
- Forrest G. L., Klevecz R. R. Synthesis and degradation of microtubule protein in synchronized Chinese hamster cells. J Biol Chem. 1972 May 25;247(10):3147–3152. [PubMed] [Google Scholar]
- Kirschner M. W., Williams R. C. The mechanism of microtubule assembly in vitro. J Supramol Struct. 1974;2(2-4):412–428. doi: 10.1002/jss.400020229. [DOI] [PubMed] [Google Scholar]
- Kirschner M. W., Williams R. C., Weingarten M., Gerhart J. C. Microtubules from mammalian brain: some properties of their depolymerization products and a proposed mechanism of assembly and disassembly. Proc Natl Acad Sci U S A. 1974 Apr;71(4):1159–1163. doi: 10.1073/pnas.71.4.1159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Olmsted J. B., Borisy G. G. Microtubules. Annu Rev Biochem. 1973;42:507–540. doi: 10.1146/annurev.bi.42.070173.002451. [DOI] [PubMed] [Google Scholar]
- Raff R. A., Greenhouse G., Gross K. W., Gross P. R. Synthesis and storage of microtubule proteins by sea urchin embryos. J Cell Biol. 1971 Aug;50(2):516–527. doi: 10.1083/jcb.50.2.516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shelanski M. L., Gaskin F., Cantor C. R. Microtubule assembly in the absence of added nucleotides. Proc Natl Acad Sci U S A. 1973 Mar;70(3):765–768. doi: 10.1073/pnas.70.3.765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weber K., Pollack R., Bibring T. Antibody against tuberlin: the specific visualization of cytoplasmic microtubules in tissue culture cells. Proc Natl Acad Sci U S A. 1975 Feb;72(2):459–463. doi: 10.1073/pnas.72.2.459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weingarten M. D., Suter M. M., Littman D. R., Kirschner M. W. Properties of the depolymerization products of microtubules from mammalian brain. Biochemistry. 1974 Dec 31;13(27):5529–5537. doi: 10.1021/bi00724a012. [DOI] [PubMed] [Google Scholar]
- Weisenberg R. C., Borisy G. G., Taylor E. W. The colchicine-binding protein of mammalian brain and its relation to microtubules. Biochemistry. 1968 Dec;7(12):4466–4479. doi: 10.1021/bi00852a043. [DOI] [PubMed] [Google Scholar]
- Weisenberg R. C. Microtubule formation in vitro in solutions containing low calcium concentrations. Science. 1972 Sep 22;177(4054):1104–1105. doi: 10.1126/science.177.4054.1104. [DOI] [PubMed] [Google Scholar]
- Wilson L. Properties of colchicine binding protein from chick embryo brain. Interactions with vinca alkaloids and podophyllotoxin. Biochemistry. 1970 Dec 8;9(25):4999–5007. doi: 10.1021/bi00827a026. [DOI] [PubMed] [Google Scholar]