Abstract
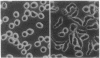
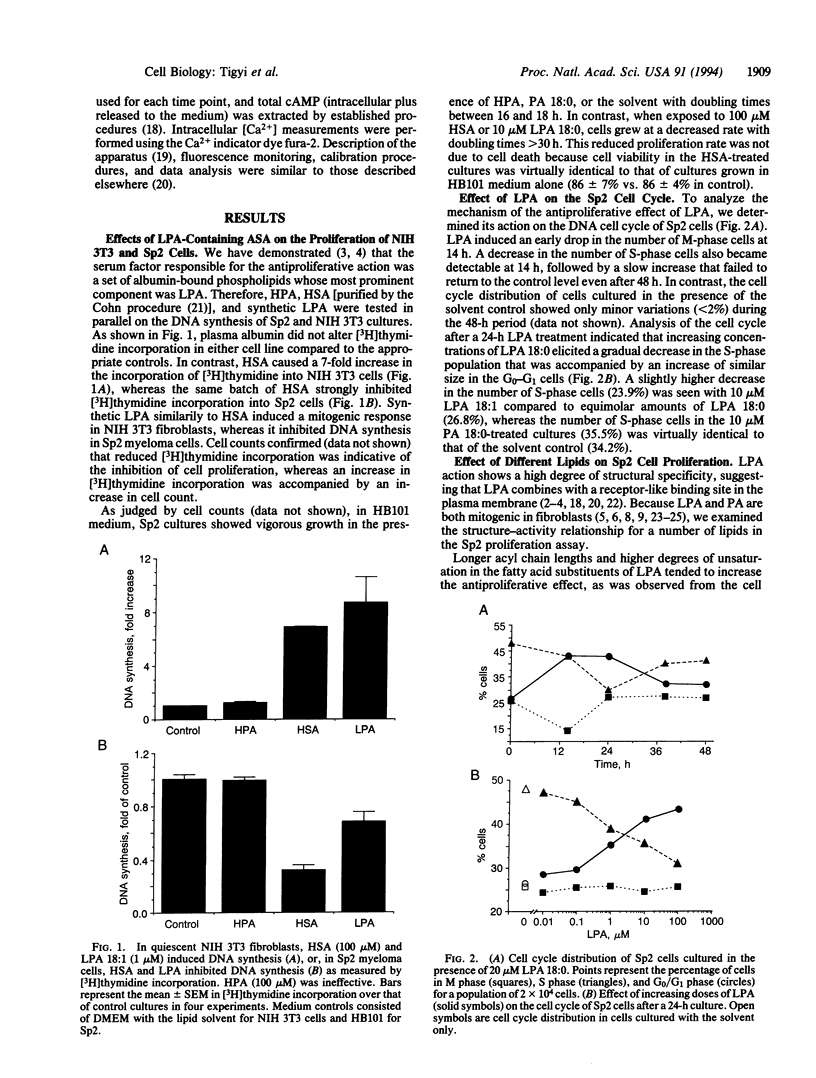
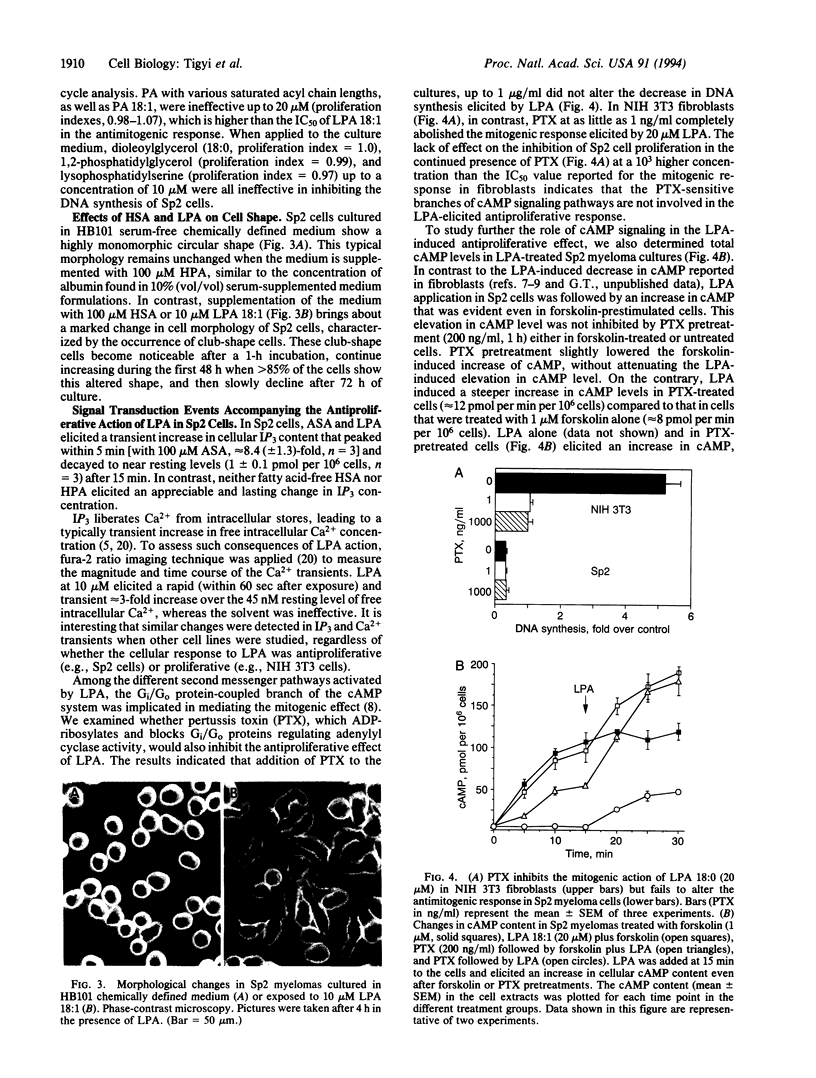
Lysophosphatidic acid (LPA) induces mitogenic responses in cultured fibroblasts through a pertussis toxin-sensitive signaling pathway. In contrast, we have shown that LPA inhibits the proliferation of Sp2/0-Ag14 myeloma cells. To resolve this apparent controversy, LPA-elicited responses in cell proliferation and the underlying second messenger mechanisms were compared in Sp2/0-Ag14 myeloma and NIH 3T3 fibroblast cells. The antimitogenic response was not elicited by micromolar concentrations of phosphatidic acid, phosphatidylglycerol, or diacylglycerol. In NIH 3T3 and Sp2 cells, LPA elicited an increase in inositol trisphosphate and a subsequent transient increase in free cytoplasmic Ca2+. Unlike the mitogenic response in NIH 3T3 cells, the antimitogenic effect was not affected by pertussis toxin; on the contrary, it was accompanied by an increase in cAMP. In Sp2 cells, cAMP analogs, forskolin, and isobutylmethylxanthine inhibited cell proliferation and enhanced LPA action in an additive manner, suggesting that an LPA-elicited increase in cAMP-mediated signaling was responsible for the antimitogenic response. In addition to the mitogenic response in fibroblasts and the antimitogenic response in tumor cell lines, there are some cell types (Jurkat T-cell lymphoma and primary astrocytes) in which LPA is ineffective in altering cell proliferation. The cell-type-specific dual action of LPA suggests that this endogenous lipid mediator when released from activated cells might play an important role as a regulator, rather than a ubiquitous inducer, of cell proliferation.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Botelho L. H., Rothermel J. D., Coombs R. V., Jastorff B. cAMP analog antagonists of cAMP action. Methods Enzymol. 1988;159:159–172. doi: 10.1016/0076-6879(88)59017-1. [DOI] [PubMed] [Google Scholar]
- Cho-Chung Y. S. Suppression of malignancy targeting cyclic AMP signal transducing proteins. Biochem Soc Trans. 1992 May;20(2):425–430. doi: 10.1042/bst0200425. [DOI] [PubMed] [Google Scholar]
- Dyer D., Tigyi G., Miledi R. The effect of active serum albumin on PC12 cells: I. Neurite retraction and activation of the phosphoinositide second messenger system. Brain Res Mol Brain Res. 1992 Aug;14(4):293–301. doi: 10.1016/0169-328x(92)90096-t. [DOI] [PubMed] [Google Scholar]
- Dyer D., Tigyi G., Miledi R. The effect of active serum albumin on PC12 cells: II. Intracellular Ca2+ transients and their role in neurite retraction. Brain Res Mol Brain Res. 1992 Aug;14(4):302–309. doi: 10.1016/0169-328x(92)90097-u. [DOI] [PubMed] [Google Scholar]
- Eichholtz T., Jalink K., Fahrenfort I., Moolenaar W. H. The bioactive phospholipid lysophosphatidic acid is released from activated platelets. Biochem J. 1993 May 1;291(Pt 3):677–680. doi: 10.1042/bj2910677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fernhout B. J., Dijcks F. A., Moolenaar W. H., Ruigt G. S. Lysophosphatidic acid induces inward currents in Xenopus laevis oocytes; evidence for an extracellular site of action. Eur J Pharmacol. 1992 Mar 24;213(2):313–315. doi: 10.1016/0014-2999(92)90698-4. [DOI] [PubMed] [Google Scholar]
- Fukami K., Takenawa T. Phosphatidic acid that accumulates in platelet-derived growth factor-stimulated Balb/c 3T3 cells is a potential mitogenic signal. J Biol Chem. 1992 Jun 5;267(16):10988–10993. [PubMed] [Google Scholar]
- Gerrard J. M., Robinson P. Identification of the molecular species of lysophosphatidic acid produced when platelets are stimulated by thrombin. Biochim Biophys Acta. 1989 Feb 20;1001(3):282–285. doi: 10.1016/0005-2760(89)90112-4. [DOI] [PubMed] [Google Scholar]
- Jalink K., van Corven E. J., Moolenaar W. H. Lysophosphatidic acid, but not phosphatidic acid, is a potent Ca2(+)-mobilizing stimulus for fibroblasts. Evidence for an extracellular site of action. J Biol Chem. 1990 Jul 25;265(21):12232–12239. [PubMed] [Google Scholar]
- Krabak M. J., Hui S. W. The mitogenic activities of phosphatidate are acyl-chain-length dependent and calcium independent in C3H/10T1/2 cells. Cell Regul. 1991 Jan;2(1):57–64. doi: 10.1091/mbc.2.1.57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lewis R. S., Cahalan M. D. Mitogen-induced oscillations of cytosolic Ca2+ and transmembrane Ca2+ current in human leukemic T cells. Cell Regul. 1989 Nov;1(1):99–112. doi: 10.1091/mbc.1.1.99. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lockwood A. H., Murphy S. K., Borislow S., Lazarus A., Pendergast M. Cellular signal transduction and the reversal of malignancy. J Cell Biochem. 1987 Apr;33(4):237–255. doi: 10.1002/jcb.240330403. [DOI] [PubMed] [Google Scholar]
- Moolenaar W. H., Jalink K., van Corven E. J. Lysophosphatidic acid: a bioactive phospholipid with growth factor-like properties. Rev Physiol Biochem Pharmacol. 1992;119:47–65. doi: 10.1007/3540551921_3. [DOI] [PubMed] [Google Scholar]
- Moolenaar W. H., Kruijer W., Tilly B. C., Verlaan I., Bierman A. J., de Laat S. W. Growth factor-like action of phosphatidic acid. Nature. 1986 Sep 11;323(6084):171–173. doi: 10.1038/323171a0. [DOI] [PubMed] [Google Scholar]
- Murakami-Murofushi K., Shioda M., Kaji K., Yoshida S., Murofushi H. Inhibition of eukaryotic DNA polymerase alpha with a novel lysophosphatidic acid (PHYLPA) isolated from myxoamoebae of Physarum polycephalum. J Biol Chem. 1992 Oct 25;267(30):21512–21517. [PubMed] [Google Scholar]
- Nishizuka Y. Intracellular signaling by hydrolysis of phospholipids and activation of protein kinase C. Science. 1992 Oct 23;258(5082):607–614. doi: 10.1126/science.1411571. [DOI] [PubMed] [Google Scholar]
- Ostman A., Thyberg J., Westermark B., Heldin C. H. PDGF-AA and PDGF-BB biosynthesis: proprotein processing in the Golgi complex and lysosomal degradation of PDGF-BB retained intracellularly. J Cell Biol. 1992 Aug;118(3):509–519. doi: 10.1083/jcb.118.3.509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pardee A. B. G1 events and regulation of cell proliferation. Science. 1989 Nov 3;246(4930):603–608. doi: 10.1126/science.2683075. [DOI] [PubMed] [Google Scholar]
- Plevin R., MacNulty E. E., Palmer S., Wakelam M. J. Differences in the regulation of endothelin-1- and lysophosphatidic-acid-stimulated Ins(1,4,5)P3 formation in rat-1 fibroblasts. Biochem J. 1991 Dec 15;280(Pt 3):609–615. doi: 10.1042/bj2800609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ridley A. J., Hall A. The small GTP-binding protein rho regulates the assembly of focal adhesions and actin stress fibers in response to growth factors. Cell. 1992 Aug 7;70(3):389–399. doi: 10.1016/0092-8674(92)90163-7. [DOI] [PubMed] [Google Scholar]
- Seuwen K., Pouysségur J. G protein-controlled signal transduction pathways and the regulation of cell proliferation. Adv Cancer Res. 1992;58:75–94. doi: 10.1016/s0065-230x(08)60291-2. [DOI] [PubMed] [Google Scholar]
- Taylor I. W. A rapid single step staining technique for DNA analysis by flow microfluorimetry. J Histochem Cytochem. 1980 Sep;28(9):1021–1024. doi: 10.1177/28.9.6157714. [DOI] [PubMed] [Google Scholar]
- Tigyi G., Dyer D., Matute C., Miledi R. A serum factor that activates the phosphatidylinositol phosphate signaling system in Xenopus oocytes. Proc Natl Acad Sci U S A. 1990 Feb;87(4):1521–1525. doi: 10.1073/pnas.87.4.1521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tigyi G., Henschen A., Miledi R. A factor that activates oscillatory chloride currents in Xenopus oocytes copurifies with a subfraction of serum albumin. J Biol Chem. 1991 Nov 5;266(31):20602–20609. [PubMed] [Google Scholar]
- Tigyi G., Miledi R. Lysophosphatidates bound to serum albumin activate membrane currents in Xenopus oocytes and neurite retraction in PC12 pheochromocytoma cells. J Biol Chem. 1992 Oct 25;267(30):21360–21367. [PubMed] [Google Scholar]
- Tokumura A., Fukuzawa K., Tsukatani H. Effects of synthetic and natural lysophosphatidic acids on the arterial blood pressure of different animal species. Lipids. 1978 Aug;13(8):572–574. doi: 10.1007/BF02533598. [DOI] [PubMed] [Google Scholar]
- Tokumura A., Harada K., Fukuzawa K., Tsukatani H. Involvement of lysophospholipase D in the production of lysophosphatidic acid in rat plasma. Biochim Biophys Acta. 1986 Jan 3;875(1):31–38. [PubMed] [Google Scholar]
- Tsai M. H., Yu C. L., Wei F. S., Stacey D. W. The effect of GTPase activating protein upon ras is inhibited by mitogenically responsive lipids. Science. 1989 Jan 27;243(4890):522–526. doi: 10.1126/science.2536192. [DOI] [PubMed] [Google Scholar]
- Yoshida S., Tamiya-Koizumi K., Kojima K. Interaction of DNA polymerases with phospholipids. Biochim Biophys Acta. 1989 Jan 23;1007(1):61–66. doi: 10.1016/0167-4781(89)90130-9. [DOI] [PubMed] [Google Scholar]
- Yu C. L., Tsai M. H., Stacey D. W. Cellular ras activity and phospholipid metabolism. Cell. 1988 Jan 15;52(1):63–71. doi: 10.1016/0092-8674(88)90531-4. [DOI] [PubMed] [Google Scholar]
- van Corven E. J., Groenink A., Jalink K., Eichholtz T., Moolenaar W. H. Lysophosphatidate-induced cell proliferation: identification and dissection of signaling pathways mediated by G proteins. Cell. 1989 Oct 6;59(1):45–54. doi: 10.1016/0092-8674(89)90868-4. [DOI] [PubMed] [Google Scholar]
- van Corven E. J., Hordijk P. L., Medema R. H., Bos J. L., Moolenaar W. H. Pertussis toxin-sensitive activation of p21ras by G protein-coupled receptor agonists in fibroblasts. Proc Natl Acad Sci U S A. 1993 Feb 15;90(4):1257–1261. doi: 10.1073/pnas.90.4.1257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Corven E. J., van Rijswijk A., Jalink K., van der Bend R. L., van Blitterswijk W. J., Moolenaar W. H. Mitogenic action of lysophosphatidic acid and phosphatidic acid on fibroblasts. Dependence on acyl-chain length and inhibition by suramin. Biochem J. 1992 Jan 1;281(Pt 1):163–169. doi: 10.1042/bj2810163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van der Bend R. L., Brunner J., Jalink K., van Corven E. J., Moolenaar W. H., van Blitterswijk W. J. Identification of a putative membrane receptor for the bioactive phospholipid, lysophosphatidic acid. EMBO J. 1992 Jul;11(7):2495–2501. doi: 10.1002/j.1460-2075.1992.tb05314.x. [DOI] [PMC free article] [PubMed] [Google Scholar]