Abstract
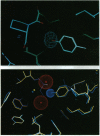
Parallel measurements of the thermodynamics (free-energy, enthalpy, entropy and heat-capacity changes) of ligand binding to FK506 binding protein (FKBP-12) in H2O and D2O have been performed in an effort to probe the energetic contributions of single protein-ligand hydrogen bonds formed in the binding reactions. Changing tyrosine-82 to phenylalanine in FKBP-12 abolishes protein-ligand hydrogen bond interactions in the FKBP-12 complexes with tacrolimus or rapamycin and leads to a large apparent enthalpic stabilization of binding in both H2O and D2O. High-resolution crystallographic analysis reveals that two water molecules bound to the tyrosine-82 hydroxyl group in unliganded FKBP-12 are displaced upon formation of the protein-ligand complexes. A thermodynamic analysis is presented that suggests that the removal of polar atoms from water contributes a highly unfavorable enthalpy change to the formation of C=O...HO hydrogen bonds as they occur in the processes of protein folding and ligand binding. Despite the less favorable enthalpy change, the entropic advantage of displacing two water molecules upon binding leads to a slightly more favorable free-energy change of binding in the reactions with wild-type FKBP-12.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aldape R. A., Futer O., DeCenzo M. T., Jarrett B. P., Murcko M. A., Livingston D. J. Charged surface residues of FKBP12 participate in formation of the FKBP12-FK506-calcineurin complex. J Biol Chem. 1992 Aug 15;267(23):16029–16032. [PubMed] [Google Scholar]
- Baker E. N., Hubbard R. E. Hydrogen bonding in globular proteins. Prog Biophys Mol Biol. 1984;44(2):97–179. doi: 10.1016/0079-6107(84)90007-5. [DOI] [PubMed] [Google Scholar]
- Baldwin R. L. Temperature dependence of the hydrophobic interaction in protein folding. Proc Natl Acad Sci U S A. 1986 Nov;83(21):8069–8072. doi: 10.1073/pnas.83.21.8069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Connelly P. R., Thomson J. A., Fitzgibbon M. J., Bruzzese F. J. Probing hydration contributions to the thermodynamics of ligand binding by proteins. Enthalpy and heat capacity changes of tacrolimus and rapamycin binding to FK506 binding protein in D2O and H2O. Biochemistry. 1993 Jun 1;32(21):5583–5590. doi: 10.1021/bi00072a013. [DOI] [PubMed] [Google Scholar]
- Connelly P. R., Thomson J. A. Heat capacity changes and hydrophobic interactions in the binding of FK506 and rapamycin to the FK506 binding protein. Proc Natl Acad Sci U S A. 1992 Jun 1;89(11):4781–4785. doi: 10.1073/pnas.89.11.4781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee B. Estimation of the maximum change in stability of globular proteins upon mutation of a hydrophobic residue to another of smaller size. Protein Sci. 1993 May;2(5):733–738. doi: 10.1002/pro.5560020505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matthews B. W. Structural and genetic analysis of protein stability. Annu Rev Biochem. 1993;62:139–160. doi: 10.1146/annurev.bi.62.070193.001035. [DOI] [PubMed] [Google Scholar]
- Michnick S. W., Rosen M. K., Wandless T. J., Karplus M., Schreiber S. L. Solution structure of FKBP, a rotamase enzyme and receptor for FK506 and rapamycin. Science. 1991 May 10;252(5007):836–839. doi: 10.1126/science.1709301. [DOI] [PubMed] [Google Scholar]
- Moore J. M., Peattie D. A., Fitzgibbon M. J., Thomson J. A. Solution structure of the major binding protein for the immunosuppressant FK506. Nature. 1991 May 16;351(6323):248–250. doi: 10.1038/351248a0. [DOI] [PubMed] [Google Scholar]
- Murphy K. P., Gill S. J. Solid model compounds and the thermodynamics of protein unfolding. J Mol Biol. 1991 Dec 5;222(3):699–709. doi: 10.1016/0022-2836(91)90506-2. [DOI] [PubMed] [Google Scholar]
- Murphy K. P., Privalov P. L., Gill S. J. Common features of protein unfolding and dissolution of hydrophobic compounds. Science. 1990 Feb 2;247(4942):559–561. doi: 10.1126/science.2300815. [DOI] [PubMed] [Google Scholar]
- PAULING L., COREY R. B., BRANSON H. R. The structure of proteins; two hydrogen-bonded helical configurations of the polypeptide chain. Proc Natl Acad Sci U S A. 1951 Apr;37(4):205–211. doi: 10.1073/pnas.37.4.205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pace C. N. Contribution of the hydrophobic effect to globular protein stability. J Mol Biol. 1992 Jul 5;226(1):29–35. doi: 10.1016/0022-2836(92)90121-y. [DOI] [PubMed] [Google Scholar]
- Park S. T., Aldape R. A., Futer O., DeCenzo M. T., Livingston D. J. PPIase catalysis by human FK506-binding protein proceeds through a conformational twist mechanism. J Biol Chem. 1992 Feb 15;267(5):3316–3324. [PubMed] [Google Scholar]
- Pauling L., Corey R. B. Configurations of Polypeptide Chains With Favored Orientations Around Single Bonds: Two New Pleated Sheets. Proc Natl Acad Sci U S A. 1951 Nov;37(11):729–740. doi: 10.1073/pnas.37.11.729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shirley B. A., Stanssens P., Hahn U., Pace C. N. Contribution of hydrogen bonding to the conformational stability of ribonuclease T1. Biochemistry. 1992 Jan 28;31(3):725–732. doi: 10.1021/bi00118a013. [DOI] [PubMed] [Google Scholar]
- Spolar R. S., Livingstone J. R., Record M. T., Jr Use of liquid hydrocarbon and amide transfer data to estimate contributions to thermodynamic functions of protein folding from the removal of nonpolar and polar surface from water. Biochemistry. 1992 Apr 28;31(16):3947–3955. doi: 10.1021/bi00131a009. [DOI] [PubMed] [Google Scholar]
- Van Duyne G. D., Standaert R. F., Karplus P. A., Schreiber S. L., Clardy J. Atomic structure of FKBP-FK506, an immunophilin-immunosuppressant complex. Science. 1991 May 10;252(5007):839–842. doi: 10.1126/science.1709302. [DOI] [PubMed] [Google Scholar]
- Van Duyne G. D., Standaert R. F., Karplus P. A., Schreiber S. L., Clardy J. Atomic structures of the human immunophilin FKBP-12 complexes with FK506 and rapamycin. J Mol Biol. 1993 Jan 5;229(1):105–124. doi: 10.1006/jmbi.1993.1012. [DOI] [PubMed] [Google Scholar]
- Yang A. S., Sharp K. A., Honig B. Analysis of the heat capacity dependence of protein folding. J Mol Biol. 1992 Oct 5;227(3):889–900. doi: 10.1016/0022-2836(92)90229-d. [DOI] [PubMed] [Google Scholar]