Abstract
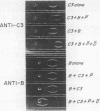
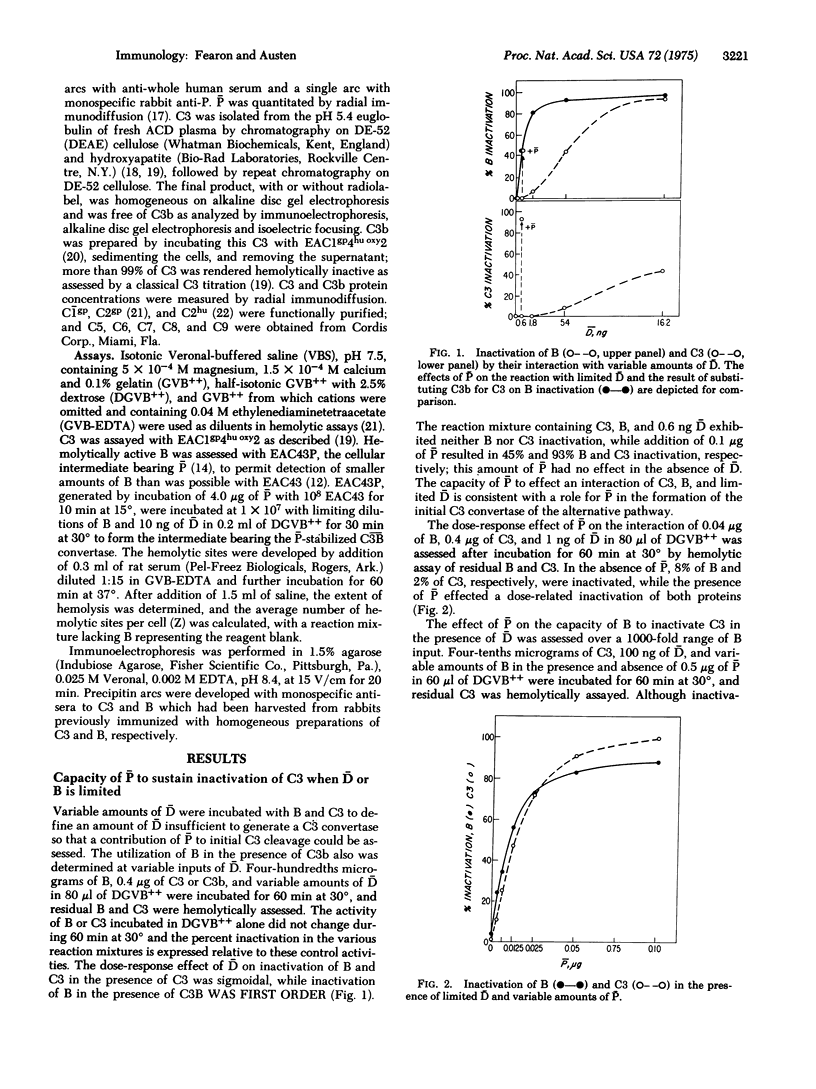
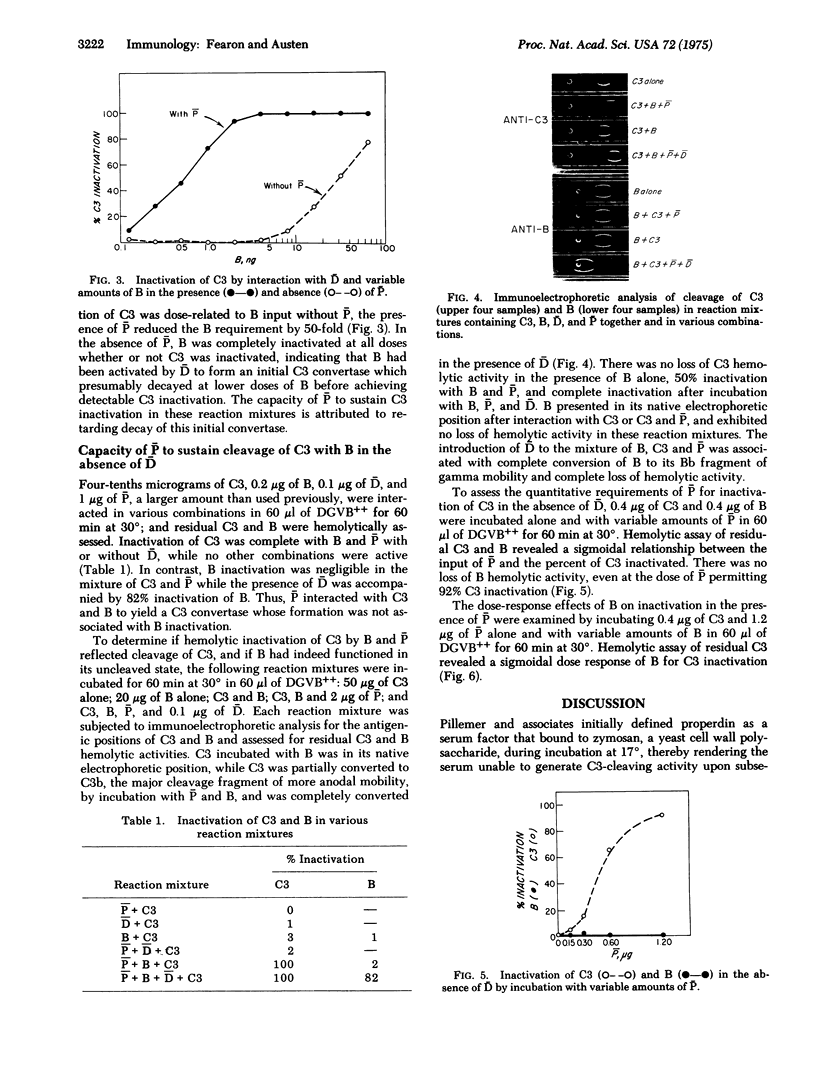
Activation of the classical complement (C) system involves conversion of C1 to its active state with subsequent cleavage of C4 and -d C2 so as to form the classical C3 convertase, C42 (a bar indicates the activated form of a protein), which sequentially cleaves C3 and C5 to initiate the cytolytic event associated with the complete reaction. An alternative, pr properdin-dependent, pathway to complement activation generates a C3 convertase, C3B, that is formed by cleavage of B with D in the presence of a C3b, the major cleavage fragment of C3. C3b is capable of binding activated properidin (P) with resultant stabilization of C3B, which otherwise rapidly decays by loss of B activity. Initial cleavage of C3, a prerequisite for formation of C3B, is demonstrated to occur through the interaction of native C3 and B in the presence of either D or P alone, or together. The effect of P on the interaction of D, B, and C3 is attributed to stabilization of C3B as has been shown for C3B. Larger amounts of P and B with C3 in the absence of D form a C3 convertase that is designated (P)C3B to indicate that demonstrable cleavage of B does not occur although the active site is available. The generation of this initial convertase, as assessed by C3 inactivation, is dose-related to P and B inputs. The presence of both P and D greatly augments initial cleavage of C3 with D fully uncovering the active site of B and P stabilizing that site.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Alper C. A., Goodkofsky I., Lepow I. H. The relationship of glycine-rich -glycoprotein to factor B in the properdin system and to the cobra factor-binding protein of huan serum. J Exp Med. 1973 Feb 1;137(2):424–437. doi: 10.1084/jem.137.2.424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- BLUM L., PILLEMER L., LEPOW I. H. The properdin system and immunity. XIII. Assay and properties of a heat-labile serum factor (factor B) in the properdin system. Z Immun exp ther. 1959 Oct-Nov;118:349–357. [PubMed] [Google Scholar]
- Cooper N. R. Formation and function of a complex of the C3 proactivator with a protein from cobra venom. J Exp Med. 1973 Feb 1;137(2):451–460. doi: 10.1084/jem.137.2.451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ensky J., Hinz C. F., Jr, Todd E. W., Wedgwood R. J., Boyer J. T., Lepow I. H. Properties of highly purified human properdin. J Immunol. 1968 Jan;100(1):142–158. [PubMed] [Google Scholar]
- Fearon D. T., Austen K. F., Ruddy S. Formation of a hemolytically active cellular intermediate by the interaction between properdin factors B and D and the activated third component of complement. J Exp Med. 1973 Dec 1;138(6):1305–1313. doi: 10.1084/jem.138.6.1305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fearon D. T., Austen K. F., Ruddy S. Properdin factor D. II. Activation to D by properdin. J Exp Med. 1974 Aug 1;140(2):426–436. doi: 10.1084/jem.140.2.426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fearon D. T., Austen K. F., Ruddy S. Properdin factor D: characterization of its active site and isolation of the precursor form. J Exp Med. 1974 Feb 1;139(2):355–366. doi: 10.1084/jem.139.2.355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Götze O., Müller-Eberhard H. J. The C3-activator system: an alternate pathway of complement activation. J Exp Med. 1971 Sep 1;134(3 Pt 2):90s–108s. [PubMed] [Google Scholar]
- Götze O., Müller-Eberhard H. J. The role of properdin in the alternate pathway of complement activation. J Exp Med. 1974 Jan 1;139(1):44–57. doi: 10.1084/jem.139.1.44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hunsicker L. G., Ruddy S., Austen K. F. Alternate complement pathway: factors involved in cobra venom factor (CoVF) activation of the third component of complement (C3). J Immunol. 1973 Jan;110(1):128–138. [PubMed] [Google Scholar]
- Kolb W. P., Müller-Eberhard H. J. The membrane attack mechanism of complement. Verification of a stable C5-9 complex in free solution. J Exp Med. 1973 Aug 1;138(2):438–451. doi: 10.1084/jem.138.2.438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LEPOW I. H., NAFF G. B., TODD E. W., PENSKY J., HINZ C. F. Chromatographic resolution of the first component of human complement into three activities. J Exp Med. 1963 Jun 1;117:983–1008. doi: 10.1084/jem.117.6.983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- MUELLER-EBERHARD H. J., LEPOW I. H. C'1 ESTERASE EFFECT ON ACTIVITY AND PHYSICOCHEMICAL PROPERTIES OF THE FOURTH COMPONENT OF COMPLEMENT. J Exp Med. 1965 May 1;121:819–833. doi: 10.1084/jem.121.5.819. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Müller-Eberhard H. J., Götze O. C3 proactivator convertase and its mode of action. J Exp Med. 1972 Apr 1;135(4):1003–1008. doi: 10.1084/jem.135.4.1003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Müller-Eberhard H. J., Polley M. J., Calcott M. A. Formation and functional significance of a molecular complex derived from the second and the fourth component of human complement. J Exp Med. 1967 Feb 1;125(2):359–380. doi: 10.1084/jem.125.2.359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- NILSSON U. R., MUELLER-EBERHARD H. J. ISOLATION OF BETA IF-GLOBULIN FROM HUMAN SERUM AND ITS CHARACTERIZATION AS THE FIFTH COMPONENT OF COMPLEMENT. J Exp Med. 1965 Aug 1;122:277–298. doi: 10.1084/jem.122.2.277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- PILLEMER L., BLUM L., LEPOW I. H., ROSS O. A., TODD E. W., WARDLAW A. C. The properdin system and immunity. I. Demonstration and isolation of a new serum protein, properdin, and its role in immune phenomena. Science. 1954 Aug 20;120(3112):279–285. doi: 10.1126/science.120.3112.279. [DOI] [PubMed] [Google Scholar]
- Rothfield N., Ross H. A., Minta J. O., Lepow I. H. Glomerular and dermal depostion of properdin in systemic lupus erythematosus. N Engl J Med. 1972 Oct 5;287(14):681–685. doi: 10.1056/NEJM197210052871402. [DOI] [PubMed] [Google Scholar]
- Ruddy S., Austen K. F. A stoichiometric assay for the fourth component of complement in whole human serum using EAC'la-gp and functionally pure human second component. J Immunol. 1967 Dec;99(6):1162–1172. [PubMed] [Google Scholar]
- Ruddy S., Austen K. F. C3 inactivator of man. I. Hemolytic measurement by the inactivation of cell-bound C3. J Immunol. 1969 Mar;102(3):533–543. [PubMed] [Google Scholar]
- Tenenhouse H. S., Deutsch H. F. Some physical-chemical properties of chicken gamma-globulins and their pepsin and papain digestion products. Immunochemistry. 1966 Jan;3(1):11–20. doi: 10.1016/0019-2791(66)90277-1. [DOI] [PubMed] [Google Scholar]