Abstract
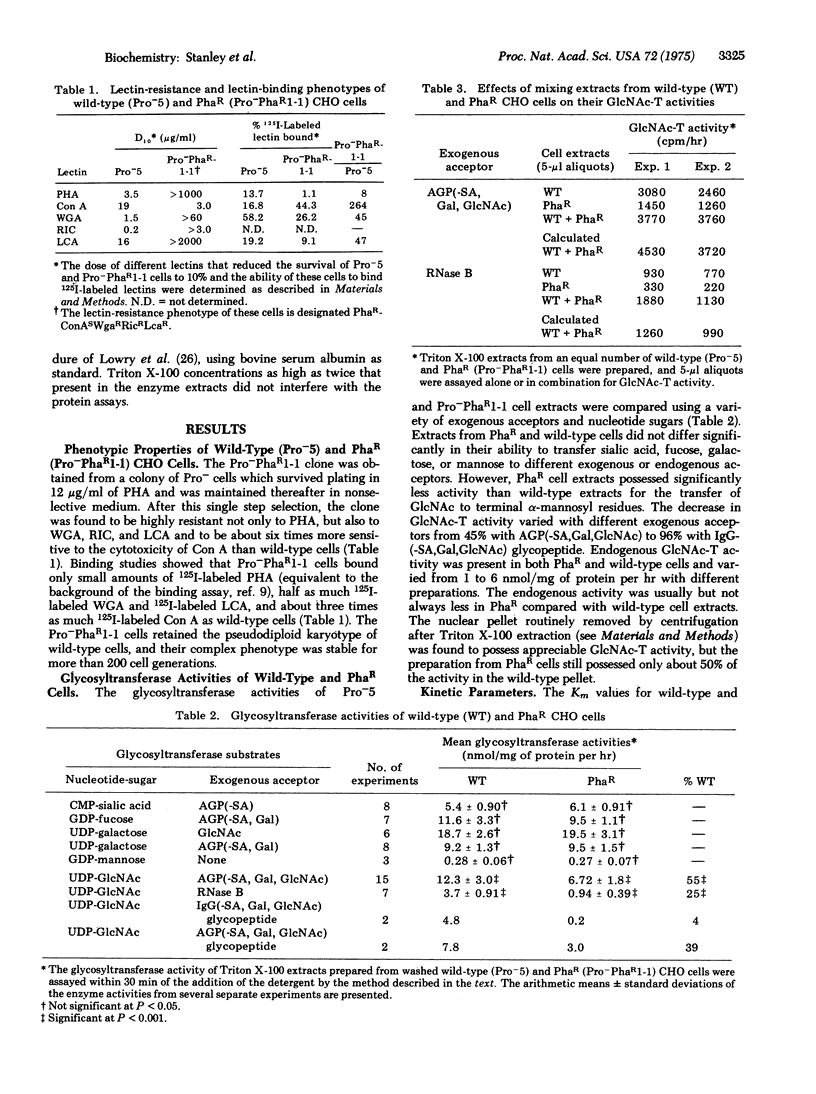
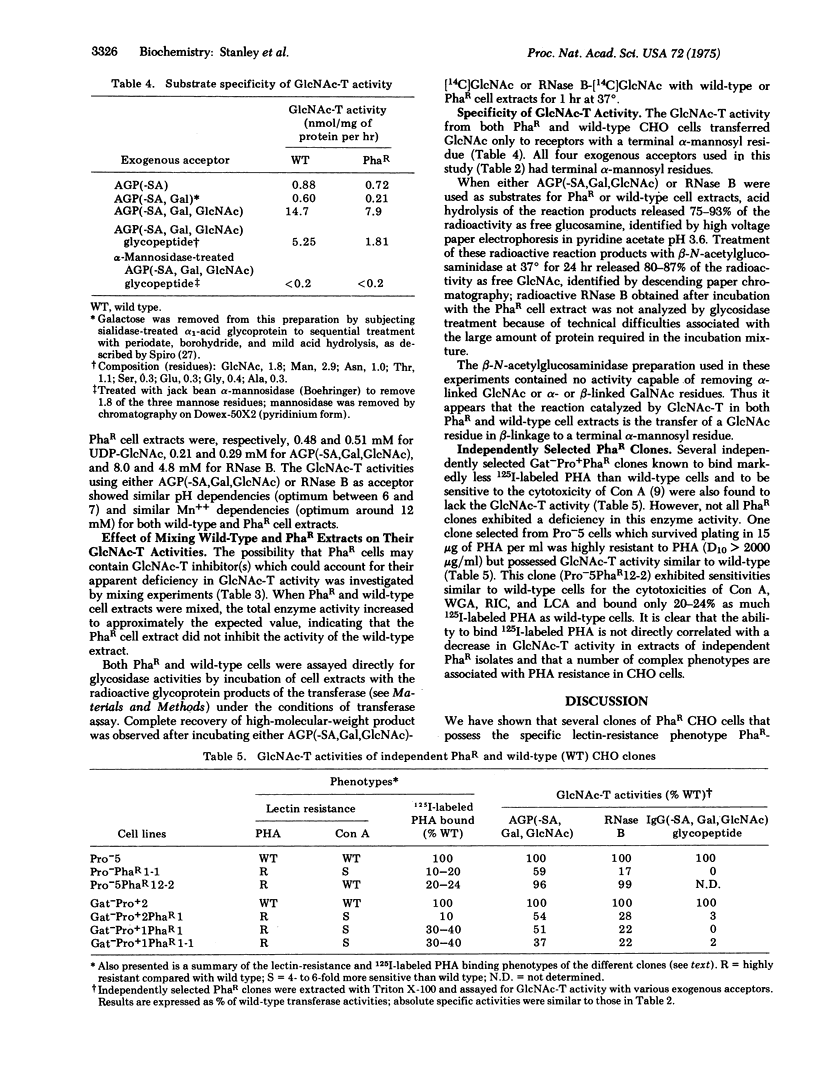
Several clones of Chinese hamster ovary cells resistant to the cytotoxicity of the phytohemagglutinin from Phaseolus vulgaris show decreased binding of 125I-labeled phytohemagglutinin and contain decreased levels of a UDP-N-acetylglucosamine--glycoprotein N-acetylglucosaminyltransferase (EC 2.4.1.51; UDP-2-acetamido-2-deoxy-D-glucose:glycoprotein 2-acetamido-2-deoxy-D-glucosyltransferase) activity when compared to wild-type cells. The decrease in transferase activity varies from 45% to 96%, depending on the exogenous acceptor used in the enzyme assay. No differences between lectin-resistant and wild-type cells were noted for several other glycosyltransferases. The absence of a particular N-acetylglucosaminyltransferase in the lectin-resistant cells apparently results in defective glycosylation of lectin-binding glycoproteins on the cell surface. A phytohemagglutinin-resistant clone which shows decreased binding of 125I-labeled phytohemagglutinin but does not exhibit the enzyme deficiency has also been isolated.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- COMB D. G., ROSEMAN S. The sialic acids. I. The structure and enzymatic synthesis of N-acetylneuraminic acid. J Biol Chem. 1960 Sep;235:2529–2537. [PubMed] [Google Scholar]
- Cassidy J. T., Jourdian G. W., Roseman S. The sialic acids. VI. Purification and properties of sialidase from Clostridium perfringens. J Biol Chem. 1965 Sep;240(9):3501–3506. [PubMed] [Google Scholar]
- Culp L. A., Black P. H. Contact-inhibited revertant cell lines isolated from simian virus 40-transformed cells. 3. Concanavalin A-selected revertant cells. J Virol. 1972 Apr;9(4):611–620. doi: 10.1128/jvi.9.4.611-620.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gottlieb C., Baenziger J., Kornfeld S. Deficient uridine diphosphate-N-acetylglucosamine:glycoprotein N-acetylglucosaminyltransferase activity in a clone of Chinese hamster ovary cells with altered surface glycoproteins. J Biol Chem. 1975 May 10;250(9):3303–3309. [PubMed] [Google Scholar]
- Gottlieb C., Skinner A. M., Kornfeld S. Isolation of a clone of Chinese hamster ovary cells deficient in plant lectin-binding sites. Proc Natl Acad Sci U S A. 1974 Apr;71(4):1078–1082. doi: 10.1073/pnas.71.4.1078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guérin C., Zachowski A., Prigent B., Paraf A., Dunia I., Diawara M. A., Benedetti E. L. Correlation between the mobility of inner plasma membrane structure and agglutination by concanavalin A in two cell lines of MOPC 173 plasmocytoma cells. Proc Natl Acad Sci U S A. 1974 Jan;71(1):114–117. doi: 10.1073/pnas.71.1.114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Howard I. K., Sage H. J., Stein M. D., Young N. M., Leon M. A., Dyckes D. F. Studies on a phytohemagglutinin from the lentil. II. Multiple forms of Lens culinaris hemagglutinin. J Biol Chem. 1971 Mar 25;246(6):1590–1595. [PubMed] [Google Scholar]
- Hudgin R. L., Schachter H. Porcine sugar nucleotide: glycoprotein glycosyltransferases. 3. Blood serum and liver N-acetylglucosaminyltransferase. Can J Biochem. 1971 Jul;49(7):847–852. doi: 10.1139/o71-119. [DOI] [PubMed] [Google Scholar]
- Hudgin R. L., Schachter H. Porcine sugar nucleotide: glycoprotein glycosyltransferases. I. Blood serum and liver sialyltransferase. Can J Biochem. 1971 Jul;49(7):829–837. doi: 10.1139/o71-117. [DOI] [PubMed] [Google Scholar]
- Hudgin R. L., Schachter H. Porcine sugar nucleotide: glycoprotein glycosyltransferases. II. Blood serum and liver galactosyltransferase. Can J Biochem. 1971 Jul;49(7):838–846. doi: 10.1139/o71-118. [DOI] [PubMed] [Google Scholar]
- Hyman R., Lacorbiere M., Stavarek S., Nicolson G. Derivation of lymphoma variants with reduced sensitivity to plant lectins. J Natl Cancer Inst. 1974 Mar;52(3):963–969. doi: 10.1093/jnci/52.3.963. [DOI] [PubMed] [Google Scholar]
- Jabbal I., Schachter H. Pork liver guanosine diphosphate-L-fucose glycoprotein fucosyltransferases. J Biol Chem. 1971 Aug 25;246(16):5154–5161. [PubMed] [Google Scholar]
- Juliano R. L., Stanley P. Altered cell surface glycoproteins in phytohemagglutinin-resistant mutants of Chinese hamster ovary cells. Biochim Biophys Acta. 1975 May 6;389(2):401–406. doi: 10.1016/0005-2736(75)90332-6. [DOI] [PubMed] [Google Scholar]
- Kean E. L. Nuclear cytidine 5'-monophosphosialic acid synthetase. J Biol Chem. 1970 May 10;245(9):2301–2308. [PubMed] [Google Scholar]
- Kornfeld R., Keller J., Baenziger J., Kornfeld S. The structure of the glycopeptide of human gamma G myeloma proteins. J Biol Chem. 1971 May 25;246(10):3259–3268. [PubMed] [Google Scholar]
- Kornfeld R., Kornfeld S. The structure of a phytohemagglutinin receptor site from human erythrocytes. J Biol Chem. 1970 May 25;245(10):2536–2545. [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Lis H., Sharon N. The biochemistry of plant lectins (phytohemagglutinins). Annu Rev Biochem. 1973;42(0):541–574. doi: 10.1146/annurev.bi.42.070173.002545. [DOI] [PubMed] [Google Scholar]
- Nicolson G. L. The interactions of lectins with animal cell surfaces. Int Rev Cytol. 1974;39:89–190. doi: 10.1016/s0074-7696(08)60939-0. [DOI] [PubMed] [Google Scholar]
- SPIRO R. G. PERIODATE OXIDATION OF THE GLYCOPROTEIN FETUIN. J Biol Chem. 1964 Feb;239:567–573. [PubMed] [Google Scholar]
- Schachter H., Jabbal I., Hudgin R. L., Pinteric L., McGuire E. J., Roseman S. Intracellular localization of liver sugar nucleotide glycoprotein glycosyltransferases in a Golgi-rich fraction. J Biol Chem. 1970 Mar 10;245(5):1090–1100. [PubMed] [Google Scholar]
- Stanley P., Caillibot V., Siminovitch L. Stable alterations at the cell membrane of Chinese hamster ovary cells resistant to the cytotoxicity of phytohemagglutinin. Somatic Cell Genet. 1975 Jan;1(1):3–26. doi: 10.1007/BF01538729. [DOI] [PubMed] [Google Scholar]
- Stanners C. P., Eliceiri G. L., Green H. Two types of ribosome in mouse-hamster hybrid cells. Nat New Biol. 1971 Mar 10;230(10):52–54. doi: 10.1038/newbio230052a0. [DOI] [PubMed] [Google Scholar]
- Toyoshima S., Fukuda M., Osawa T. Chemical nature of the receptor site for various phytomitogens. Biochemistry. 1972 Oct 10;11(21):4000–4005. doi: 10.1021/bi00771a025. [DOI] [PubMed] [Google Scholar]
- Wright J. A. Evidence for pleiotropic changes in lines of Chinese hamster ovary cells resistant to concanavalin A and phytohemagglutinin-P. J Cell Biol. 1973 Mar;56(3):666–675. doi: 10.1083/jcb.56.3.666. [DOI] [PMC free article] [PubMed] [Google Scholar]



