Abstract
D-Alanine carboxypeptidase from Bacillus stearothermophilus is a membrane-bound enzyme which is inhibited by covalent interaction with penicillin G. The penicilloyl enzyme spontaneously reactivates and simultaneously releases a penicillin G degradation product; 0.2 mumol of the latter was isolated after incubation of 4.2 mumol of [8-14C]penicillin G with 10 g of membrane protein. It was identified as phenylacetylglycine by chromatographic techniques, infrared spectroscopy, and mass spectrometry. A mechanism for the degradation is proposed in which the remaining part of penicillin G would be released as 5,5-dimethyl-delta2-thiazoline-4-carboxylic acid. The implications of this finding are discussed.
Full text
PDF
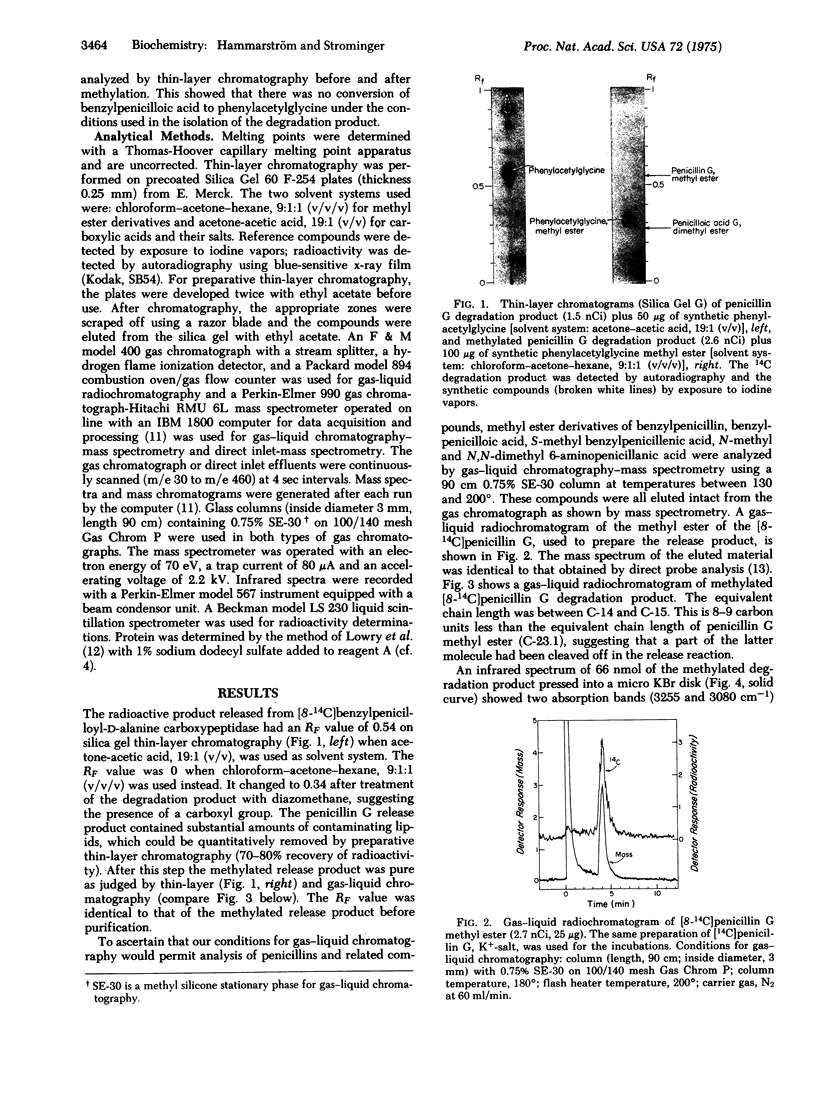
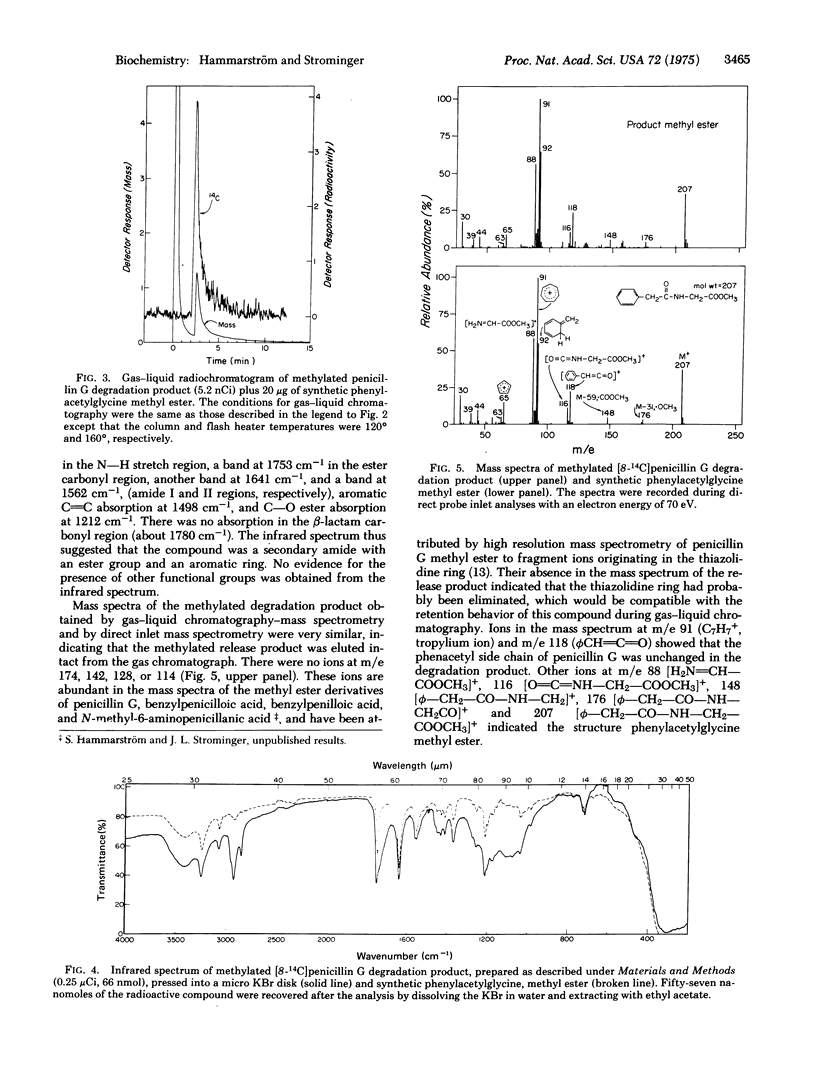


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bell M. R., Carlson J. A., Oesterlin R. Degradation of penicillin G methyl ester with trifluoracetic acid. J Org Chem. 1972 Aug 25;37(17):2733–2735. doi: 10.1021/jo00982a023. [DOI] [PubMed] [Google Scholar]
- Blumberg P. M., Strominger J. L. Five penicillin-binding components occur in Bacillus subtilis membranes. J Biol Chem. 1972 Dec 25;247(24):8107–8113. [PubMed] [Google Scholar]
- Blumberg P. M., Strominger J. L. Interaction of penicillin with the bacterial cell: penicillin-binding proteins and penicillin-sensitive enzymes. Bacteriol Rev. 1974 Sep;38(3):291–335. doi: 10.1128/br.38.3.291-335.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blumberg P. M., Yocum R. R., Willoughby E., Strominger J. L. Binding of (14C)penicillin G to the membrane-bound and the purified D-alanine carboxypeptidases from Bacillus stearothermophilus and Bacillus subtilis and its release. J Biol Chem. 1974 Nov 10;249(21):6828–6835. [PubMed] [Google Scholar]
- Immich H., Jansen H. H., Pisani K. Die Beziehungen zwischen Arcus lipoides der Hornhaut und Arteriosklerose. Vergleichende Untersuchungen anhand von 500 Sektionsfällen. Klin Wochenschr. 1967 Oct 15;45(20):1017–1020. doi: 10.1007/BF01727809. [DOI] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Lawrence P. J., Strominger J. L. Biosynthesis of the peptidoglycan of bacterial cell walls. XV. The binding of radioactive penicillin to the particulate enzyme preparation of Bacillus subtilis and its reversal with hydroxylamine or thiols. J Biol Chem. 1970 Jul 25;245(14):3653–3659. [PubMed] [Google Scholar]
- Strominger J. L., Willoughby E., Kamiryo T., Blumberg P. M., Yocum R. R. Penicillin-sensitive enzymes and penicillin-binding components in bacterial cells. Ann N Y Acad Sci. 1974 May 10;235(0):210–224. doi: 10.1111/j.1749-6632.1974.tb43267.x. [DOI] [PubMed] [Google Scholar]
- Tipper D. J., Strominger J. L. Mechanism of action of penicillins: a proposal based on their structural similarity to acyl-D-alanyl-D-alanine. Proc Natl Acad Sci U S A. 1965 Oct;54(4):1133–1141. doi: 10.1073/pnas.54.4.1133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Umbreit J. N., Strominger J. L. D-alanine carboxypeptidase from Bacillus subtilis membranes. I. Purification and characterization. J Biol Chem. 1973 Oct 10;248(19):6759–6766. [PubMed] [Google Scholar]
- Umbreit J. N., Strominger J. L. D-alanine carboxypeptidase from Bacillus subtilis membranes. II. Interaction with penicillins and cephalosporins. J Biol Chem. 1973 Oct 10;248(19):6767–6771. [PubMed] [Google Scholar]
- Yocum R. R., Blumberg P. M., Strominger J. L. Purification and characterization of the thermophilic D-alanine carboxypeptidase from membranes of Bacillus stearothermophilus. J Biol Chem. 1974 Aug 10;249(15):4863–4871. [PubMed] [Google Scholar]




