Abstract
The T-cell response to pigeon cytochrome c peptide, residues 88-104 (pcytC), in B10.BR mice is mediated largely by cells bearing both V beta 3 and V alpha 11 variable regions of the T-cell antigen receptor. These cells are, therefore, reactive with the superantigen staphylococcal enterotoxin A (SEA). Recent reports have shown that in vivo exposure to superantigen can lead to deletion of superantigen-reactive T cells from the pool of mature T cells in the periphery. Here we show that upon cotreatment of animals with both SEA and pcytC, bulk deletion of the population of SEA-reactive cells is maintained, while the subpopulation of SEA-reactive T cells that also responds to pcytC is not deleted but instead proliferates in response to pcytC. These results are discussed with regard to mechanisms regulating the balance between T-cell tolerance and T-cell activation in vivo.
Full text
PDF
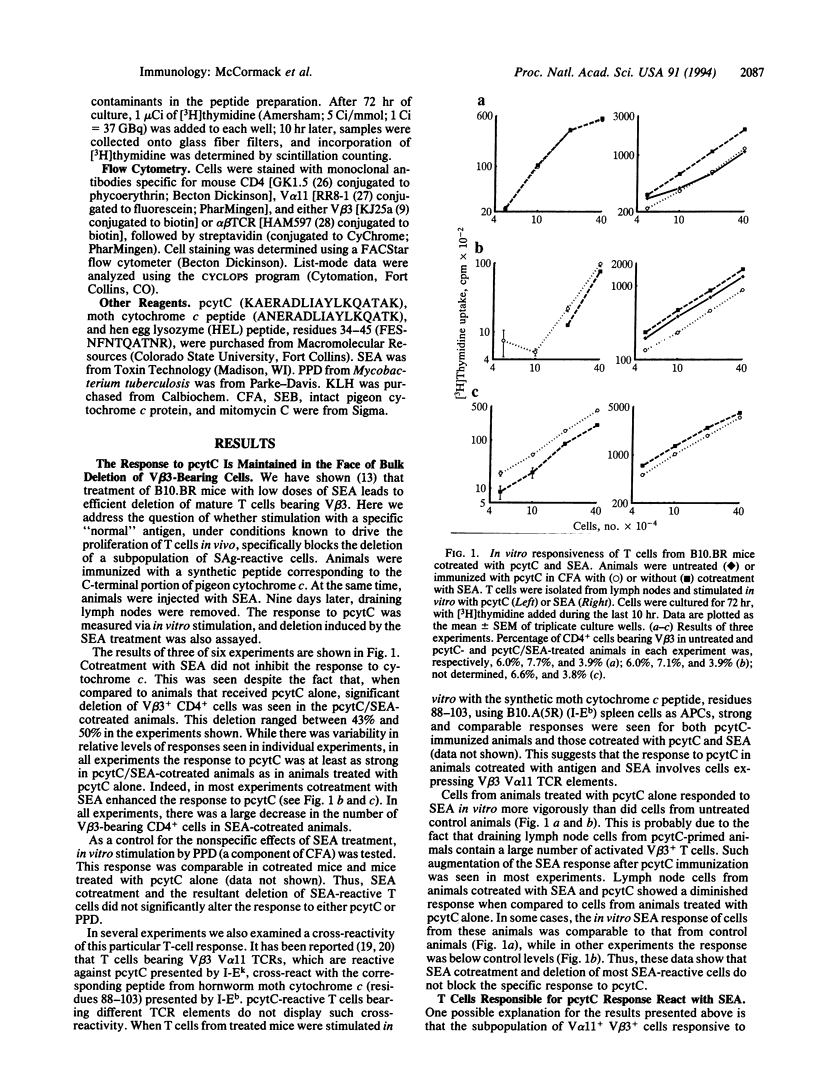


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
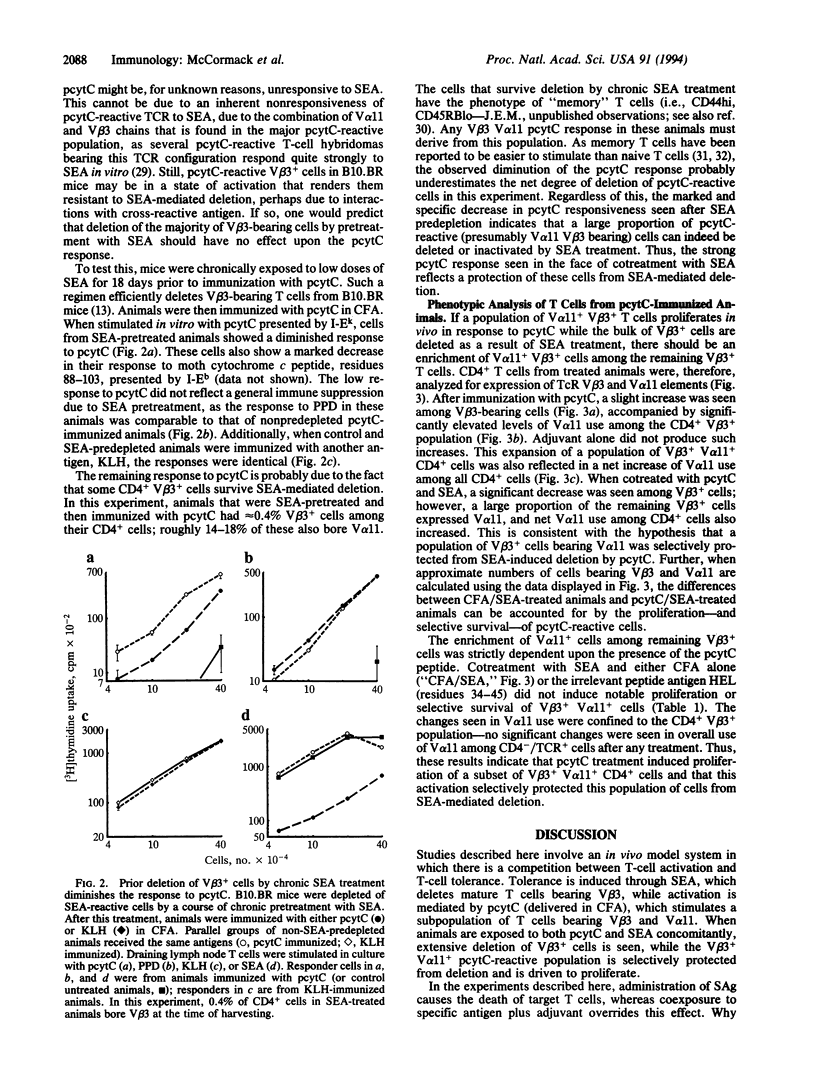
- Byrne J. A., Butler J. L., Cooper M. D. Differential activation requirements for virgin and memory T cells. J Immunol. 1988 Nov 15;141(10):3249–3257. [PubMed] [Google Scholar]
- Dellabona P., Peccoud J., Kappler J., Marrack P., Benoist C., Mathis D. Superantigens interact with MHC class II molecules outside of the antigen groove. Cell. 1990 Sep 21;62(6):1115–1121. doi: 10.1016/0092-8674(90)90388-u. [DOI] [PubMed] [Google Scholar]
- Eynon E. E., Parker D. C. Small B cells as antigen-presenting cells in the induction of tolerance to soluble protein antigens. J Exp Med. 1992 Jan 1;175(1):131–138. doi: 10.1084/jem.175.1.131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fink P. J., Matis L. A., McElligott D. L., Bookman M., Hedrick S. M. Correlations between T-cell specificity and the structure of the antigen receptor. Nature. 1986 May 15;321(6067):219–226. doi: 10.1038/321219a0. [DOI] [PubMed] [Google Scholar]
- Fleischer B., Schrezenmeier H. T cell stimulation by staphylococcal enterotoxins. Clonally variable response and requirement for major histocompatibility complex class II molecules on accessory or target cells. J Exp Med. 1988 May 1;167(5):1697–1707. doi: 10.1084/jem.167.5.1697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fry A. M., Matis L. A. Self-tolerance alters T-cell receptor expression in an antigen-specific MHC restricted immune response. Nature. 1988 Oct 27;335(6193):830–832. doi: 10.1038/335830a0. [DOI] [PubMed] [Google Scholar]
- Fuchs E. J., Matzinger P. B cells turn off virgin but not memory T cells. Science. 1992 Nov 13;258(5085):1156–1159. doi: 10.1126/science.1439825. [DOI] [PubMed] [Google Scholar]
- Gaur A., Fathman C. G., Steinman L., Brocke S. SEB induced anergy: modulation of immune response to T cell determinants of myoglobin and myelin basic protein. J Immunol. 1993 Apr 1;150(7):3062–3069. [PubMed] [Google Scholar]
- Harding F. A., McArthur J. G., Gross J. A., Raulet D. H., Allison J. P. CD28-mediated signalling co-stimulates murine T cells and prevents induction of anergy in T-cell clones. Nature. 1992 Apr 16;356(6370):607–609. doi: 10.1038/356607a0. [DOI] [PubMed] [Google Scholar]
- Herman A., Labrecque N., Thibodeau J., Marrack P., Kappler J. W., Sekaly R. P. Identification of the staphylococcal enterotoxin A superantigen binding site in the beta 1 domain of the human histocompatibility antigen HLA-DR. Proc Natl Acad Sci U S A. 1991 Nov 15;88(22):9954–9958. doi: 10.1073/pnas.88.22.9954. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jameson S. C., Nakajima P. B., Brooks J. L., Heath W., Kanagawa O., Gascoigne N. R. The T cell receptor V alpha 11 gene family. Analysis of allelic sequence polymorphism and demonstration of J alpha region-dependent recognition by allele-specific antibodies. J Immunol. 1991 Nov 1;147(9):3185–3193. [PubMed] [Google Scholar]
- Jenkins M. K., Burrell E., Ashwell J. D. Antigen presentation by resting B cells. Effectiveness at inducing T cell proliferation is determined by costimulatory signals, not T cell receptor occupancy. J Immunol. 1990 Mar 1;144(5):1585–1590. [PubMed] [Google Scholar]
- Jenkins M. K., Pardoll D. M., Mizuguchi J., Quill H., Schwartz R. H. T-cell unresponsiveness in vivo and in vitro: fine specificity of induction and molecular characterization of the unresponsive state. Immunol Rev. 1987 Feb;95:113–135. doi: 10.1111/j.1600-065x.1987.tb00502.x. [DOI] [PubMed] [Google Scholar]
- Jenkins M. K., Schwartz R. H. Antigen presentation by chemically modified splenocytes induces antigen-specific T cell unresponsiveness in vitro and in vivo. J Exp Med. 1987 Feb 1;165(2):302–319. doi: 10.1084/jem.165.2.302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jenkins M. K., Taylor P. S., Norton S. D., Urdahl K. B. CD28 delivers a costimulatory signal involved in antigen-specific IL-2 production by human T cells. J Immunol. 1991 Oct 15;147(8):2461–2466. [PubMed] [Google Scholar]
- Julius M. H., Simpson E., Herzenberg L. A. A rapid method for the isolation of functional thymus-derived murine lymphocytes. Eur J Immunol. 1973 Oct;3(10):645–649. doi: 10.1002/eji.1830031011. [DOI] [PubMed] [Google Scholar]
- Kappler J. W., Staerz U., White J., Marrack P. C. Self-tolerance eliminates T cells specific for Mls-modified products of the major histocompatibility complex. Nature. 1988 Mar 3;332(6159):35–40. doi: 10.1038/332035a0. [DOI] [PubMed] [Google Scholar]
- Kawabe Y., Ochi A. Programmed cell death and extrathymic reduction of Vbeta8+ CD4+ T cells in mice tolerant to Staphylococcus aureus enterotoxin B. Nature. 1991 Jan 17;349(6306):245–248. doi: 10.1038/349245a0. [DOI] [PubMed] [Google Scholar]
- Kubo R. T., Born W., Kappler J. W., Marrack P., Pigeon M. Characterization of a monoclonal antibody which detects all murine alpha beta T cell receptors. J Immunol. 1989 Apr 15;142(8):2736–2742. [PubMed] [Google Scholar]
- Ledbetter J. A., Herzenberg L. A. Xenogeneic monoclonal antibodies to mouse lymphoid differentiation antigens. Immunol Rev. 1979;47:63–90. doi: 10.1111/j.1600-065x.1979.tb00289.x. [DOI] [PubMed] [Google Scholar]
- Lee W. T., Vitetta E. S. Memory T cells are anergic to the superantigen staphylococcal enterotoxin B. J Exp Med. 1992 Aug 1;176(2):575–579. doi: 10.1084/jem.176.2.575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu Z., Sun Y. K., Xi Y. P., Hong B., Harris P. E., Reed E. F., Suciu-Foca N. Limited usage of T cell receptor V beta genes by allopeptide-specific T cells. J Immunol. 1993 Apr 15;150(8 Pt 1):3180–3186. [PubMed] [Google Scholar]
- MacDonald H. R., Baschieri S., Lees R. K. Clonal expansion precedes anergy and death of V beta 8+ peripheral T cells responding to staphylococcal enterotoxin B in vivo. Eur J Immunol. 1991 Aug;21(8):1963–1966. doi: 10.1002/eji.1830210827. [DOI] [PubMed] [Google Scholar]
- MacDonald H. R., Schneider R., Lees R. K., Howe R. C., Acha-Orbea H., Festenstein H., Zinkernagel R. M., Hengartner H. T-cell receptor V beta use predicts reactivity and tolerance to Mlsa-encoded antigens. Nature. 1988 Mar 3;332(6159):40–45. doi: 10.1038/332040a0. [DOI] [PubMed] [Google Scholar]
- McCormack J. E., Callahan J. E., Kappler J., Marrack P. C. Profound deletion of mature T cells in vivo by chronic exposure to exogenous superantigen. J Immunol. 1993 May 1;150(9):3785–3792. [PubMed] [Google Scholar]
- Mollick J. A., Cook R. G., Rich R. R. Class II MHC molecules are specific receptors for staphylococcus enterotoxin A. Science. 1989 May 19;244(4906):817–820. doi: 10.1126/science.2658055. [DOI] [PubMed] [Google Scholar]
- Perkins D. L., Wang Y., Ho S. S., Wiens G. R., Seidman J. G., Rimm I. J. Superantigen-induced peripheral tolerance inhibits T cell responses to immunogenic peptides in TCR (beta-chain) transgenic mice. J Immunol. 1993 May 15;150(10):4284–4291. [PubMed] [Google Scholar]
- Pullen A. M., Marrack P., Kappler J. W. The T-cell repertoire is heavily influenced by tolerance to polymorphic self-antigens. Nature. 1988 Oct 27;335(6193):796–801. doi: 10.1038/335796a0. [DOI] [PubMed] [Google Scholar]
- Rellahan B. L., Jones L. A., Kruisbeek A. M., Fry A. M., Matis L. A. In vivo induction of anergy in peripheral V beta 8+ T cells by staphylococcal enterotoxin B. J Exp Med. 1990 Oct 1;172(4):1091–1100. doi: 10.1084/jem.172.4.1091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Röcken M., Urban J. F., Shevach E. M. Infection breaks T-cell tolerance. Nature. 1992 Sep 3;359(6390):79–82. doi: 10.1038/359079a0. [DOI] [PubMed] [Google Scholar]
- Sanders M. E., Makgoba M. W., June C. H., Young H. A., Shaw S. Enhanced responsiveness of human memory T cells to CD2 and CD3 receptor-mediated activation. Eur J Immunol. 1989 May;19(5):803–808. doi: 10.1002/eji.1830190504. [DOI] [PubMed] [Google Scholar]
- Scholl P. R., Sekaly R. P., Diez A., Glimcher L. H., Geha R. S. Binding of toxic shock syndrome toxin-1 to murine major histocompatibility complex class II molecules. Eur J Immunol. 1990 Sep;20(9):1911–1916. doi: 10.1002/eji.1830200907. [DOI] [PubMed] [Google Scholar]
- Sellins K. S., Danska J. S., Paragas V., Fathman C. G. Limited T cell receptor beta-chain usage in the sperm whale myoglobin 110-121/E alpha dA beta d response by H-2d congenic mouse strains. J Immunol. 1992 Oct 1;149(7):2323–2327. [PubMed] [Google Scholar]
- Sorger S. B., Hedrick S. M., Fink P. J., Bookman M. A., Matis L. A. Generation of diversity in T cell receptor repertoire specific for pigeon cytochrome c. J Exp Med. 1987 Feb 1;165(2):279–301. doi: 10.1084/jem.165.2.279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vitetta E. S., Berton M. T., Burger C., Kepron M., Lee W. T., Yin X. M. Memory B and T cells. Annu Rev Immunol. 1991;9:193–217. doi: 10.1146/annurev.iy.09.040191.001205. [DOI] [PubMed] [Google Scholar]
- Webb S., Morris C., Sprent J. Extrathymic tolerance of mature T cells: clonal elimination as a consequence of immunity. Cell. 1990 Dec 21;63(6):1249–1256. doi: 10.1016/0092-8674(90)90420-j. [DOI] [PubMed] [Google Scholar]
- White J., Herman A., Pullen A. M., Kubo R., Kappler J. W., Marrack P. The V beta-specific superantigen staphylococcal enterotoxin B: stimulation of mature T cells and clonal deletion in neonatal mice. Cell. 1989 Jan 13;56(1):27–35. doi: 10.1016/0092-8674(89)90980-x. [DOI] [PubMed] [Google Scholar]
- White J., Pullen A., Choi K., Marrack P., Kappler J. W. Antigen recognition properties of mutant V beta 3+ T cell receptors are consistent with an immunoglobulin-like structure for the receptor. J Exp Med. 1993 Jan 1;177(1):119–125. doi: 10.1084/jem.177.1.119. [DOI] [PMC free article] [PubMed] [Google Scholar]