Abstract
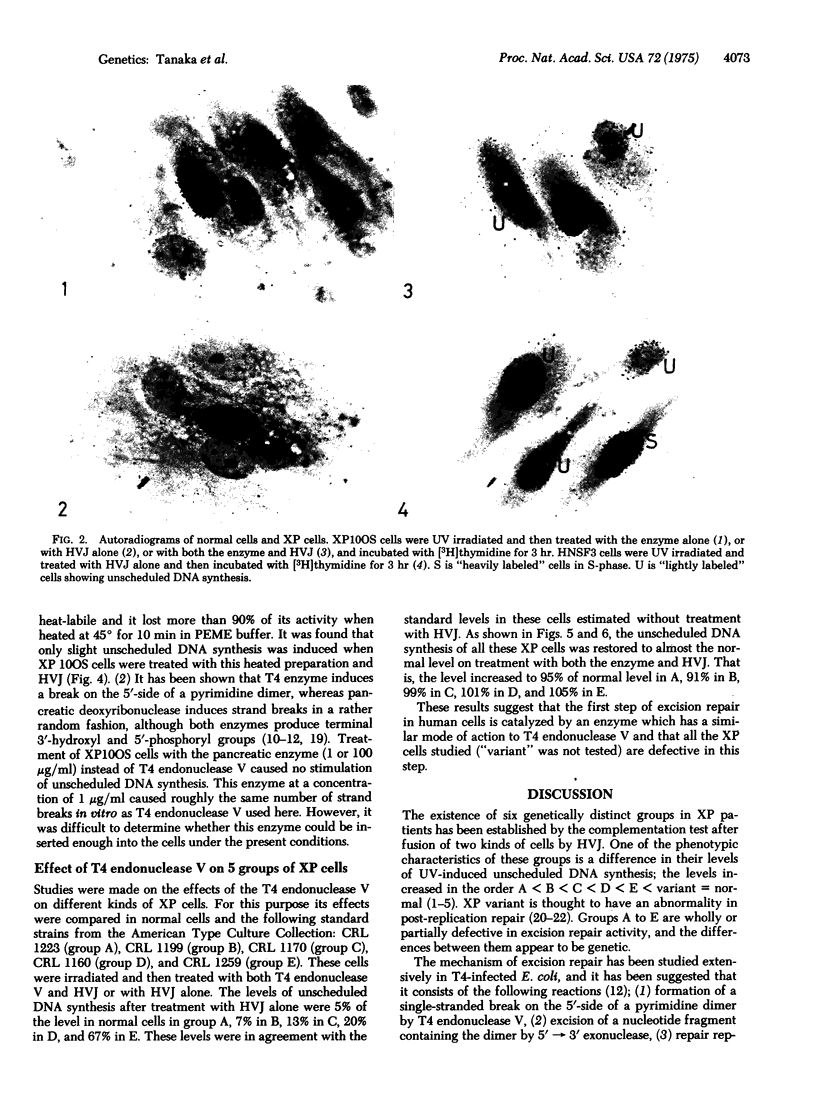
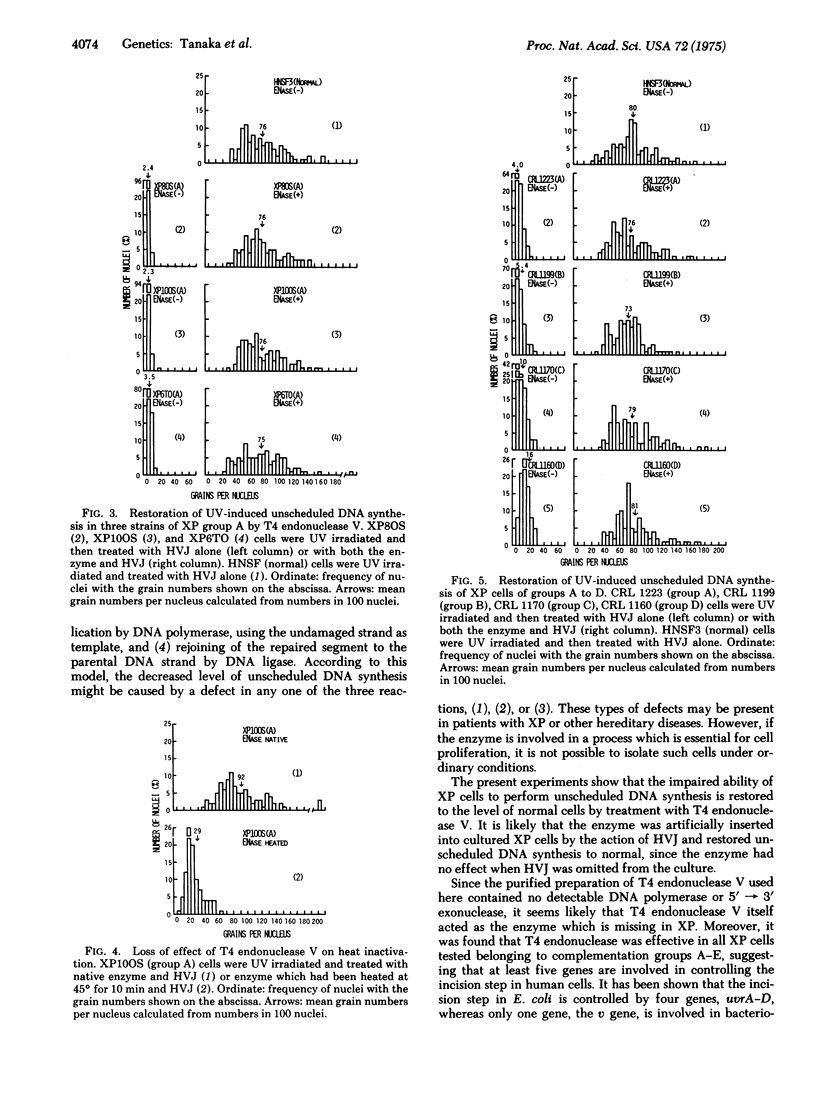
Ultraviolet (UV)-induced unscheduled DNA synthesis of xeroderma pigmentosum cells, belonging to complementation groups A, B, C, D, and E, was restored to the normal level by concomitant treatment of the cells with T4 endonuclease V and UV-inactivated HVJ (Sendai virus). The present results suggest that (1) T4 endonuclease molecules were inserted effectively into the cells by the interaction of HVJ with the cell membranes, (2) the enzyme was functional on human chromosomal DNA which had been damaged by UV irradiation in the viable cells, (3) all the studied groups of xeroderma pigmentosum ("variant" was not tested) were defective in the first step (incision) of excision repair.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bacchetti S., van der Plas A., Veldhuisen G. A UV-specific endonucleolytic activity present in human cell extracts. Biochem Biophys Res Commun. 1972 Aug 7;48(3):662–669. doi: 10.1016/0006-291x(72)90399-3. [DOI] [PubMed] [Google Scholar]
- Braun A., Grossman L. An endonuclease from Escherichia coli that acts preferentially on UV-irradiated DNA and is absent from the uvrA and uvrB mutants. Proc Natl Acad Sci U S A. 1974 May;71(5):1838–1842. doi: 10.1073/pnas.71.5.1838. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brent T. P. Repair enzyme suggested by mammalian endonuclease activity specific for ultraviolet-irradiated DNA. Nat New Biol. 1972 Oct 11;239(93):172–173. doi: 10.1038/newbio239172a0. [DOI] [PubMed] [Google Scholar]
- Buhl S. N., Regan J. D. Repair endonuclease-sensitive sites in daughter DNA of ultraviolet-irradiated human cells. Nature. 1973 Dec 21;246(5434):484–484. doi: 10.1038/246484a0. [DOI] [PubMed] [Google Scholar]
- Cleaver J. E. Defective repair replication of DNA in xeroderma pigmentosum. Nature. 1968 May 18;218(5142):652–656. doi: 10.1038/218652a0. [DOI] [PubMed] [Google Scholar]
- Cleaver J. E. Sedimentation of DNA from human fibroblasts irradiated with ultraviolet light: possible detection of excision breaks in normal and repair-deficient xeroderma pigmentosum cells. Radiat Res. 1974 Feb;57(2):207–227. [PubMed] [Google Scholar]
- Cleaver J. E. Xeroderma pigmentosum: variants with normal DNA repair and normal sensitivity to ultraviolet light. J Invest Dermatol. 1972 Mar;58(3):124–128. doi: 10.1111/1523-1747.ep12538913. [DOI] [PubMed] [Google Scholar]
- Day R. S. Xeroderma pigmentosum variants have decreased repair of ultraviolet-damaged DNA. Nature. 1975 Feb 27;253(5494):748–749. doi: 10.1038/253748a0. [DOI] [PubMed] [Google Scholar]
- De Weerd-Kastelein E. A., Keijzer W., Bootsma D. Genetic heterogeneity of xeroderma pigmentosum demonstrated by somatic cell hybridization. Nat New Biol. 1972 Jul 19;238(81):80–83. doi: 10.1038/newbio238080a0. [DOI] [PubMed] [Google Scholar]
- Duker N. J., Teebor G. W. Different ultraviolet DNA endonuclease activity in human cells. Nature. 1975 May 1;255(5503):82–84. doi: 10.1038/255082a0. [DOI] [PubMed] [Google Scholar]
- Enders J. F., Holloway A., Grogan E. A. Replication of poliovirus I in chick embryo and hamster cells exposed to sendai virus. Proc Natl Acad Sci U S A. 1967 Mar;57(3):637–644. doi: 10.1073/pnas.57.3.637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Epstein J. H., Fukuyama K., Reed W. B., Epstein W. L. Defect in DNA synthesis in skin of patients with xeroderma pigmentosum demonstrated in vivo. Science. 1970 Jun 19;168(3938):1477–1478. doi: 10.1126/science.168.3938.1477. [DOI] [PubMed] [Google Scholar]
- Friedberg E. C., King J. J. Dark repair of ultraviolet-irradiated deoxyribonucleic acid by bacteriophage T4: purification and characterization of a dimer-specific phage-induced endonuclease. J Bacteriol. 1971 May;106(2):500–507. doi: 10.1128/jb.106.2.500-507.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goulian M., Lucas Z. J., Kornberg A. Enzymatic synthesis of deoxyribonucleic acid. XXV. Purification and properties of deoxyribonucleic acid polymerase induced by infection with phage T4. J Biol Chem. 1968 Feb 10;243(3):627–638. [PubMed] [Google Scholar]
- Kraemer K. H., Coon H. G., Petinga R. A., Barrett S. F., Rahe A. E., Robbins J. H. National Cancer Institute, National Institutes of Health, Bethesda, Maryland 20014, USA. Proc Natl Acad Sci U S A. 1975 Jan;72(1):59–63. doi: 10.1073/pnas.72.1.59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lehmann A. R., Kirk-Bell S., Arlett C. F., Paterson M. C., Lohman P. H., de Weerd-Kastelein E. A., Bootsma D. Xeroderma pigmentosum cells with normal levels of excision repair have a defect in DNA synthesis after UV-irradiation. Proc Natl Acad Sci U S A. 1975 Jan;72(1):219–223. doi: 10.1073/pnas.72.1.219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- OKADA Y., TADOKORO J. Analysis of giant polynuclear cell formation caused by HVJ virus from Ehrlich's ascites tumor cells. II. Quantitative analysis of giant polynuclear cell formation. Exp Cell Res. 1962 Feb;26:108–118. doi: 10.1016/0014-4827(62)90206-9. [DOI] [PubMed] [Google Scholar]
- Okada Y., Murayama F. Requirement of calcium ions for the cell fusion reaction of animal cells by HVJ. Exp Cell Res. 1966 Nov-Dec;44(2):527–551. doi: 10.1016/0014-4827(66)90458-7. [DOI] [PubMed] [Google Scholar]
- Oshima S., Sekiguchi M. Induction of a new enzyme activity to excise pyrimidine dimers in Escherichia coli infected with bacteriophage T4. Biochem Biophys Res Commun. 1972 Jun 9;47(5):1126–1132. doi: 10.1016/0006-291x(72)90951-5. [DOI] [PubMed] [Google Scholar]
- Paterson M. C., Lohman P. H., Sluyter M. L. Use of UV endonuclease from Micrococcus luteus to monitor the progress of DNA repair in UV-irradiated human cells. Mutat Res. 1973 Aug;19(2):245–256. doi: 10.1016/0027-5107(73)90083-3. [DOI] [PubMed] [Google Scholar]
- Setlow R. B., Regan J. D., German J., Carrier W. L. Evidence that xeroderma pigmentosum cells do not perform the first step in the repair of ultraviolet damage to their DNA. Proc Natl Acad Sci U S A. 1969 Nov;64(3):1035–1041. doi: 10.1073/pnas.64.3.1035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takagi Y., Sekiguchi M., Okubo S., Nakayama H., Shimada K., Yasuda S., Nishimoto T., Yoshihara H. Nucleases specific for ultraviolet light-irradiated DNA and their possible role in dark repair. Cold Spring Harb Symp Quant Biol. 1968;33:219–227. doi: 10.1101/sqb.1968.033.01.025. [DOI] [PubMed] [Google Scholar]
- Taketo A., Yasuda S., Sekiguchi M. Initial step of excision repair in Escherichia coli: replacement of defective function of uvr mutants by T4 endonuclease V. J Mol Biol. 1972 Sep 14;70(1):1–14. doi: 10.1016/0022-2836(72)90160-x. [DOI] [PubMed] [Google Scholar]
- Yasuda S., Sekiguchi M. T4 endonuclease involved in repair of DNA. Proc Natl Acad Sci U S A. 1970 Dec;67(4):1839–1845. doi: 10.1073/pnas.67.4.1839. [DOI] [PMC free article] [PubMed] [Google Scholar]




