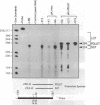
Abstract
Despite the identification and characterization of cell surface receptors for the extracellular matrix, it is unknown how their relative expression and cytoskeletal association regulate cell migration. Previous studies have identified beta-1,4-galactosyltransferase (GalTase; EC 2.4.1.38) on the surface of migrating cells, where it mediates cell migration on basal lamina matrices by associating with the cytoskeleton and binding to N-linked oligosaccharides in the E8 domain of laminin. In this study, the function of GalTase during cell migration was examined directly by analyzing the migration rate of stably transfected cell lines in which the relative level of surface GalTase and its ability to associate with the cytoskeleton were altered. We show here that the cytoskeleton contains a limiting, saturable, number of binding sites for surface GalTase. Furthermore, the rate of cell migration was inversely related to the ability of surface GalTase to associate with the cytoskeleton. Elevating surface GalTase in excess of the number of cytoskeleton-binding sites reduced the rate of cell migration, whereas decreasing the amount of surface GalTase available to bind the cytoskeleton increased migration rates. These results show that the rate of cell migration on basal lamina is directly dependent upon the expression of surface GalTase and the ability of this protein to associate with a limiting number of cytoskeleton-binding sites.
Full text
PDF

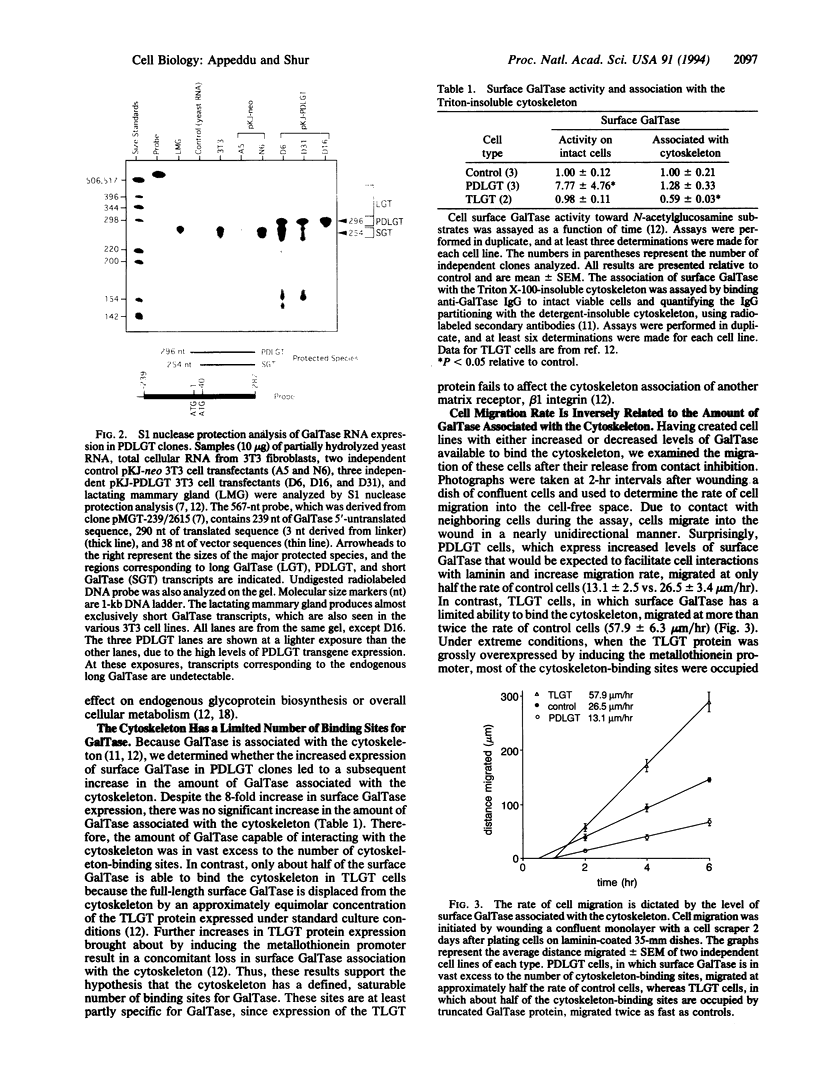
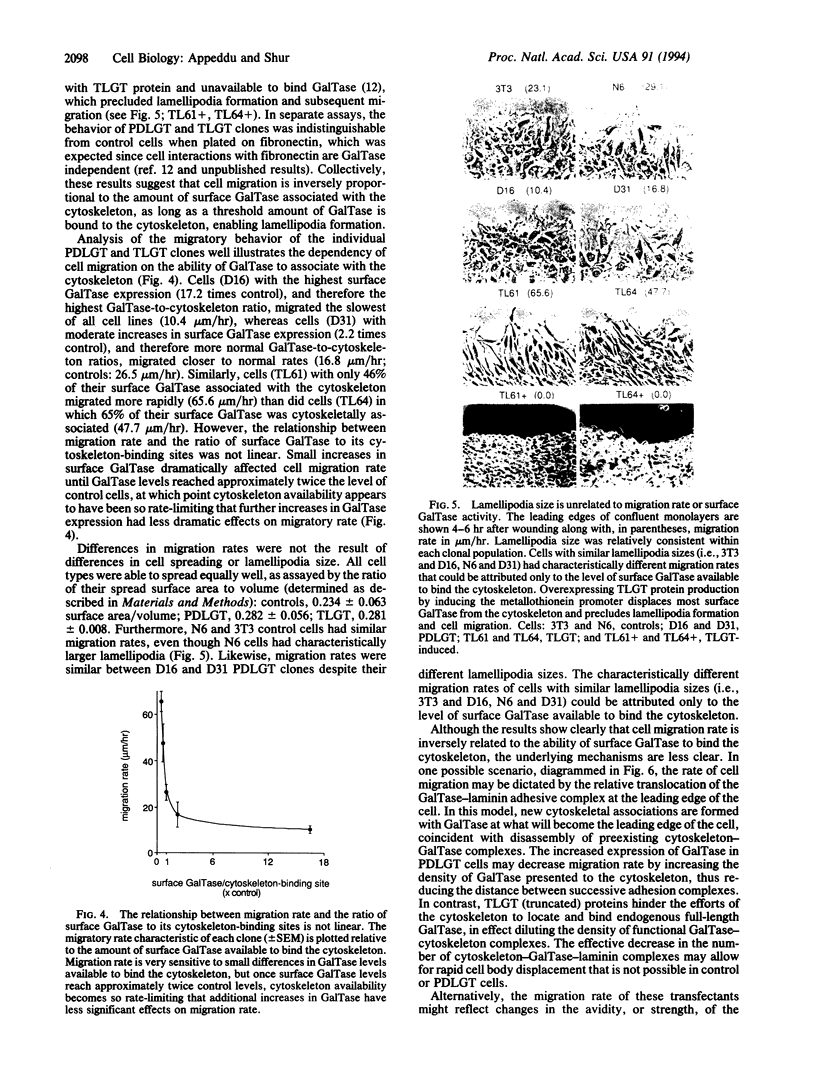

Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Abercrombie M., Heaysman J. E., Pegrum S. M. The locomotion of fibroblasts in culture. I. Movements of the leading edge. Exp Cell Res. 1970 Mar;59(3):393–398. doi: 10.1016/0014-4827(70)90646-4. [DOI] [PubMed] [Google Scholar]
- Akiyama S. K., Nagata K., Yamada K. M. Cell surface receptors for extracellular matrix components. Biochim Biophys Acta. 1990 Feb 28;1031(1):91–110. doi: 10.1016/0304-4157(90)90004-v. [DOI] [PubMed] [Google Scholar]
- Bayna E. M., Shaper J. H., Shur B. D. Temporally specific involvement of cell surface beta-1,4 galactosyltransferase during mouse embryo morula compaction. Cell. 1988 Apr 8;53(1):145–157. doi: 10.1016/0092-8674(88)90496-5. [DOI] [PubMed] [Google Scholar]
- Begovac P. C., Hall D. E., Shur B. D. Laminin fragment E8 mediates PC12 cell neurite outgrowth by binding to cell surface beta 1,4 galactosyltransferase. J Cell Biol. 1991 May;113(3):637–644. doi: 10.1083/jcb.113.3.637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Begovac P. C., Shur B. D. Cell surface galactosyltransferase mediates the initiation of neurite outgrowth from PC12 cells on laminin. J Cell Biol. 1990 Feb;110(2):461–470. doi: 10.1083/jcb.110.2.461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Couchman J. R., Rees D. A. The behaviour of fibroblasts migrating from chick heart explants: changes in adhesion, locomotion and growth, and in the distribution of actomyosin and fibronectin. J Cell Sci. 1979 Oct;39:149–165. doi: 10.1242/jcs.39.1.149. [DOI] [PubMed] [Google Scholar]
- Duband J. L., Dufour S., Yamada S. S., Yamada K. M., Thiery J. P. Neural crest cell locomotion induced by antibodies to beta 1 integrins. A tool for studying the roles of substratum molecular avidity and density in migration. J Cell Sci. 1991 Apr;98(Pt 4):517–532. doi: 10.1242/jcs.98.4.517. [DOI] [PubMed] [Google Scholar]
- Eckstein D. J., Shur B. D. Cell surface beta-1,4-galactosyltransferase is associated with the detergent-insoluble cytoskeleton on migrating mesenchymal cells. Exp Cell Res. 1992 Jul;201(1):83–90. doi: 10.1016/0014-4827(92)90350-h. [DOI] [PubMed] [Google Scholar]
- Eckstein D. J., Shur B. D. Laminin induces the stable expression of surface galactosyltransferase on lamellipodia of migrating cells. J Cell Biol. 1989 Jun;108(6):2507–2517. doi: 10.1083/jcb.108.6.2507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Evans S. C., Lopez L. C., Shur B. D. Dominant negative mutation in cell surface beta 1,4-galactosyltransferase inhibits cell-cell and cell-matrix interactions. J Cell Biol. 1993 Feb;120(4):1045–1057. doi: 10.1083/jcb.120.4.1045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hay E. D. Role of cell-matrix contacts in cell migration and epithelial-mesenchymal transformation. Cell Differ Dev. 1990 Dec 2;32(3):367–375. doi: 10.1016/0922-3371(90)90052-x. [DOI] [PubMed] [Google Scholar]
- Hynes R. O. Integrins: versatility, modulation, and signaling in cell adhesion. Cell. 1992 Apr 3;69(1):11–25. doi: 10.1016/0092-8674(92)90115-s. [DOI] [PubMed] [Google Scholar]
- Kintner C. Regulation of embryonic cell adhesion by the cadherin cytoplasmic domain. Cell. 1992 Apr 17;69(2):225–236. doi: 10.1016/0092-8674(92)90404-z. [DOI] [PubMed] [Google Scholar]
- Lopez L. C., Bayna E. M., Litoff D., Shaper N. L., Shaper J. H., Shur B. D. Receptor function of mouse sperm surface galactosyltransferase during fertilization. J Cell Biol. 1985 Oct;101(4):1501–1510. doi: 10.1083/jcb.101.4.1501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lopez L. C., Youakim A., Evans S. C., Shur B. D. Evidence for a molecular distinction between Golgi and cell surface forms of beta 1,4-galactosyltransferase. J Biol Chem. 1991 Aug 25;266(24):15984–15991. [PubMed] [Google Scholar]
- Luna E. J., Hitt A. L. Cytoskeleton--plasma membrane interactions. Science. 1992 Nov 6;258(5084):955–964. doi: 10.1126/science.1439807. [DOI] [PubMed] [Google Scholar]
- Macek M. B., Lopez L. C., Shur B. D. Aggregation of beta-1,4-galactosyltransferase on mouse sperm induces the acrosome reaction. Dev Biol. 1991 Oct;147(2):440–444. doi: 10.1016/0012-1606(91)90301-i. [DOI] [PubMed] [Google Scholar]
- Mecham R. P. Receptors for laminin on mammalian cells. FASEB J. 1991 Aug;5(11):2538–2546. doi: 10.1096/fasebj.5.11.1651264. [DOI] [PubMed] [Google Scholar]
- Rosen S. D. Lymphocyte homing: progress and prospects. Curr Opin Cell Biol. 1989 Oct;1(5):913–919. doi: 10.1016/0955-0674(89)90058-6. [DOI] [PubMed] [Google Scholar]
- Shur B. D. Glycosyltransferases as cell adhesion molecules. Curr Opin Cell Biol. 1993 Oct;5(5):854–863. doi: 10.1016/0955-0674(93)90035-o. [DOI] [PubMed] [Google Scholar]
- Solowska J., Guan J. L., Marcantonio E. E., Trevithick J. E., Buck C. A., Hynes R. O. Expression of normal and mutant avian integrin subunits in rodent cells. J Cell Biol. 1989 Aug;109(2):853–861. doi: 10.1083/jcb.109.2.853. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wehland J., Osborn M., Weber K. Phalloidin-induced actin polymerization in the cytoplasm of cultured cells interferes with cell locomotion and growth. Proc Natl Acad Sci U S A. 1977 Dec;74(12):5613–5617. doi: 10.1073/pnas.74.12.5613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Youakim A., Shur B. D. Effects of overexpression of beta 1,4-galactosyltransferase on glycoprotein biosynthesis in F9 embryonal carcinoma cells. Glycobiology. 1993 Apr;3(2):155–163. doi: 10.1093/glycob/3.2.155. [DOI] [PubMed] [Google Scholar]