Summary
Parental Posttraumatic Stress Disorder (PTSD), particularly maternal PTSD, confers risk for stress-related psychopathology among offspring. Altered hypothalamic-pituitary-adrenal (HPA) axis functioning is one mechanism proposed to explain transmission of this intergenerational risk. Investigation of this mechanism has been largely limited to general stress response (e.g., diurnal cortisol), rather than reactivity in response to an acute stressor. We examined cortisol reactivity in response to a laboratory stressor among offspring of mothers with a lifetime diagnosis of PTSD (n=36) and age- and gender- matched control offspring of mothers without PTSD (n=36). Youth (67% girls; mean age = 11.4, SD = 2.6) participated in a developmentally sensitive laboratory stressor and had salivary cortisol assessed five times (one pre-stress, one immediate post-stress, and three recovery measures, spaced 15 minutes apart). Results were consistent with the hypothesis that offspring of mothers with PTSD would exhibit a dysregulated, blunted cortisol reactivity profile and control offspring would display the expected adaptive peak in cortisol response to challenge profile. Findings were maintained after controlling for youth traumatic event history, physical anxiety symptoms, and depression, as well as maternal depression. This finding contributes to the existing literature indicating that attenuated HPA axis functioning, inclusive of hyposecretion of cortisol in response to acute stress, is robust among youth of mothers with PTSD. Future research is warranted in elucidating cortisol reactivity as a link between maternal PTSD and stress-related psychopathology vulnerability among offspring.
Keywords: traumatic stress, intergenerational transmission, salivary cortisol, psychopathology
1. Introduction
Posttraumatic Stress Disorder (PTSD) is a prevalent, costly, and debilitating disorder among adults (Kessler et al., 2005) that can impact multiple areas of an affected person’s day-to-day functioning, including one’s family (Galovski & Lyons, 2004). Offspring in particular are often impacted by parental PTSD (Samuelson & Cashman, 2008; Chemtob et al., 2010; Weems & Scheeringa, 2013), which may involve ongoing exposure to the parent’s symptoms of intrusion (e.g., traumatic nightmares), avoidance (e.g., of people or places that serve as trauma reminders), negative core thoughts and beliefs (e.g., distorted self-blame), and hyperarousal (e.g., exaggerated startle reflex) (APA, American Psychiatric Association, 2013). Increased rates of PTSD and other stress-related psychopathology are found among offspring of parents with PTSD (e.g., Scheeringa & Zeanah, 2001; Yehuda et al., 2008), which is partially attributed to shared trauma exposure or increased likelihood of trauma exposure (e.g., Lehmann, 2000). Roberts et al. (2012) found that children’s trauma exposure accounted for 74% of the increased risk of PTSD in their population-based sample of children of mothers with PTSD. Nonetheless, even in the absence of an offspring’s exposure to a parent’s index traumatic event, the literature supports well-established associations between parents with PTSD and related psychopathology among their offspring (Yehuda et al., 2001a; Leen-Feldner et al., 2013).
As such, the intergenerational transmission of offspring stress-related psychopathology from parents has been of keen empirical and clinical interest. Research that has specifically examined the role of parent gender in the intergenerational transmission of psychopathology from parental PTSD to offspring psychopathology has indicated that effects are particularly robust for mothers (Leen-Feldner et al., 2013); although links between paternal PTSD and offspring depression (Yehuda et al., 2008), as well as the notion of ‘dose effects’ (i.e., associations with offspring psychopathology are stronger when both parents have PTSD) (Yehuda et al., 2001a; Al-Turkait & Ohaeri, 2008), are supported by this small literature base. That maternal PTSD has been shown to lead to significantly elevated risk for offspring PTSD (Yehuda et al., 1998b), as well as a range of other psychopathology outcomes (i.e., multifinality; Cicchetti & Toth, 2009), such as anxiety symptoms and externalizing problems (e.g., Linares et al., 2001; Graham-Bermann et al., 2009), lends itself to questions regarding the mechanisms underlying this intergenerational transmission process. Among the biological (e.g., epigenetics; Yehuda & Bierer, 2009; see Schmidt et al., 2011 for a review), psychological, and environmental mechanisms that have been proposed (Leen-Feldner et al., 2013; Rijlaarsdam et al., 2014), a growing body of neouroendocrine research supports the postulation that altered hypothalamic-pituitary-adrenal (HPA) axis functioning plays a role in this transmission (Yehuda et al., 2002). The main aim of this study is to extend this literature by examining the cortisol reactivity of child offspring of mothers with PTSD.
Yehuda and colleagues have examined altered HPA axis functioning in the link between maternal PTSD and offspring vulnerability to psychopathology among offspring of Holocaust-exposed parents (Yehuda et al., 2000; Yehuda et al., 2001b; Yehuda et al., 2007a; 2007b) and among a sample of a sample of mothers exposed to 9/11 during their pregnancy (Yehuda et al., 2005). These studies have focused on general biological stress response (e.g., basal cortisol levels, diurnal HPA rhythms). This research team has also examined glucocorticoid sensitivity via dexamethasone suppression tests (Yehuda et al., 2007a), and more recently, via lysozyme suppression tests (Lehrner et al., 2014) among offspring of Holocaust survivors, supporting its association with parental PTSD—and maternal PTSD in particular (Lehrner et al., 2014). In general, the results of these seminal investigations show that the offspring of mothers with PTSD have an attenuated diurnal cortisol response and greater glucocorticoid sensitivity. Similarly, in one of the only studies to date examining HPA axis functioning among school-aged, traumatically-injured offspring of parents with posttraumatic stress, Nugent et al. (2007) found that children with low (initial) cortisol levels and with parents with high levels of posttraumatic stress were at highest risk for the later development of posttraumatic stress. With the exception of this body of literature, relatively few studies have focused specifically on mothers diagnosed with PTSD. There is a broader range of studies that have examined HPA axis functioning through the measurement of cortisol stress response involving offspring whose mothers had experienced stress while pregnant (see Glover et al., 2010 for a review) or who were depressed (Halligan et al., 2007; see Guerry & Hastings, 2011 for a review). However, given the literature that has emphasized specific links between maternal PTSD and later problems in offspring functioning (see Leen-Feldner et al., 2013 for review), beyond exposure to traumatic events (Li et al., 2010) and other psychiatric disorders (Yehuda et al., 1998b), children of mothers with PTSD are an important population to study in unveiling stress-related psychopathology transmission mechanisms.
The relatively consistent finding across offspring-maternal PTSD studies to date indicating a blunted effect for cortisol suggests that the mechanism transmitted across generations is related to the tendency for low cortisol levels (Yehuda et al., 2002). Indeed, hyposecretion of cortisol, which represents insufficient activation of the HPA-axis in response to stress, has been found to be a risk factor for PTSD among adults (Mason et al., 1986; Yehuda et al., 1993; Mouthaan et al., 2014) and youth (Nugent et al., 2007). Theories that speak to this link between altered HPA axis functioning and PTSD suggest that low levels of cortisol, or hypocortisolism, may negatively impact the metabolic, immuno- and neurodefensive processes necessary for coping adaptively with acute stressors (Yehuda, 2002). That is, instead of the potentially adaptive pattern of HPA-axis activation (i.e. increasing cortisol in response to a perceived stressor, thereby allowing the body to be physiologically prepared to meet that challenge), levels of cortisol in individuals with PTSD may instead be blunted (Heim, Ehlert, & Hellhammer, 2000). This hypocortisolism may increase a person’s vulnerability in the face of a traumatic event or major life stressor. That is, underactivation of the HPA axis may result in a higher propensity towards a heightened state of arousal and increased difficulty modulating intrusive images and thoughts, etc.—in other words, PTSD. The mechanism through which hypocortisolism develops is not yet fully understood, but is most likely to result from prolonged, chronic stress exposure that leads to changes in the biosynthesis of cortisol and the related HPA axis receptors (Heim et al., 2000).
There is robust evidence that maternal trauma (and subsequent PTSD) have intergenerational effects. Yehuda et al. (2005) documented in utero transmission of hypocortisolism in children whose mother was traumatically impacted by the events of September 11th, particularly during the third trimester of pregnancy. These effects are also potentially explained through genetic and epigenetic programming (Pitman et al., 2006; Pratchett & Yehuda, 2014). Animal models also highlight the importance of intergenerational transmission of differences in the expression of glucocorticoid receptor genes (Francis, Diorio, Liu & Meaney, 1999). If a mother with PTSD transmits a lower cortisol tendency, this altered aspect of the HPA axis system may increase the offspring’s susceptibility to PTSD when facing traumatic events, as it promotes the likelihood of a non-adaptive physiological response to such stressors. The parenting environment could also play a role in the development of hypocortisolism in children of mothers with PTSD through maladaptive modeling of ineffective coping techniques and/or increased stress exposure (e.g., Sturge-Apple, Davies, Cicchetti, & Manning, 2012).
In advancing the science underlying the intergenerational transmission of stress-related psychopathology from mothers to offspring, enhancing understanding of the offsprings’ adaptive versus maladaptive reactions to acute stress is vital. Thus, beyond diurnal patterns of general cortisol response and glucocorticoid sensitivity, there is merit in capturing how cortisol levels of PTSD-negative offspring of mothers with PTSD respond when confronted with an acute stressor. Given the value of such laboratory challenge assessments, gold standard paradigms have been validated and widely used to measure cortisol stress reactivity to acute stressors (e.g., Stroud et al., 2002; Nock & Mendes, 2008). A few offspring studies, including with infants (Tollenaar et al., 2011) and young adults (Entringer et al., 2009), have linked altered cortisol stress reactivity in response to a lab-based stressor with having mothers who endured prenatal stress. No study to date has examined cortisol reactivity in response to an acute stressor specifically among youth offspring whose mothers have been diagnosed with PTSD—but who have not yet themselves developed the disorder.
To address this gap, we utilized an offspring case control design in the context of a well-established acute stressor paradigm to investigate the intergenerational transmission of altered stress reactivity among mothers with PTSD and their offspring. Specifically, we compared cortisol reactivity in provocation to a stressor among offspring of mothers without PTSD versus offspring of mothers with PTSD—a population known to be at heightened risk for PTSD but who had not yet developed the disorder themselves. The goal of the current study is to inform this line of research, which seeks to better understand mechanisms (in this case, transmitted altered reactivity) by which offspring of mothers with PTSD are more vulnerable to eventual stress-related psychopathology and PTSD in particular. Based on the plethora of studies that have reported lower basal cortisol levels as noted above (see Yehuda & Bierer, 2008; Leen-Feldner et al., 2013 for reviews), we hypothesized that offspring of mothers with a diagnosis of PTSD would exhibit a blunted cortisol reactivity profile, whereas control offspring would display the expected adaptive peak in cortisol response to the laboratory stressor. As an exploratory aim of the study, we also examined whether offspring of mothers with PTSD would be more likely to report psychopathology symptoms that have been associated with PTSD and stress-related psychopathology, particularly among offspring, including physical symptoms of anxiety, externalizing behaviors, and suicidal ideation (Leen-Feldner et al., 2013).
2. Method
2.1 Participants and procedures
Participants for this study were 72 mother-offspring dyads. They were a subset of mothers and offspring who were selected from a larger study investigating the development of emotional distress disorders (see Barrocas et al., 2012). Briefly, children and adolescents for the larger parent study were recruited by letters sent home to families with a child in 3rd, 6th, or 9th grades of public schools. Interested parents called the laboratory and responded to a brief phone screen that established that both the parent and child were fluent in English, and the child did not carry an autism spectrum or psychotic disorder and had an IQ > 70. Participants were 366 youth ranging in age from 9–15 (M = 11.4, SD = 2.27). The sample was approximately evenly divided by sex (boys: 44%, girls: 56%) and grade (32% 3rd grade, 32% 6th grade, 36% 9th grade). Ethnicity was as follows: Caucasian: 64%, African American: 8%, Latino: 7%, Asian/Pacific Islander: 5%, Other/Mixed Race: 16%.
For the current offspring case-control design study that focused on cortisol reactivity patterns in offspring of mothers with a diagnosis of PTSD versus controls, we selected all of the child participants of those mothers, from the larger parent study, who met criteria for current or lifetime DSM-IV (APA, American Psychiatric Association, 1994) PTSD (n=36) and a matched control group of age and gender-matched offspring (n=36) of mothers from the larger study with no PTSD diagnosis. Importantly, for this study on cortisol reactivity patterns among offspring, none of the children, from either maternal PTSD or control groups, had a PTSD diagnosis. All of the children were biological offspring of the mothers. Any children with a PTSD diagnosis or nonbiological child of a mother were excluded from the current study.
Mothers’ average age was 42.83 (range 28–53, SD = 5.83). The youth ranged in age from 9–15 (M = 11.4, SD = 2.60); 67% were girls. Ethnicity of the families was as follows: Caucasian: 67%, African-American: 5%, Latino: 7%, Asian/Pacific Islander: 4%, Other/Mixed Race: 17%.
The mother and her child visited the laboratory. Mothers provided informed written consent for herself and for her child’s participation; youth provided written assent. In the first hour of the visit, participants completed questionnaires and diagnostic interviews. The youth then participated in a psychosocial challenge.
2.2 Measures
2.2.1. Parental mood and anxiety disorder and traumatic event history
The Structured Clinical Interview for DSM-IV (APA, American Psychiatric Association, 1994) Axis I Disorders (SCID-NP) was used to assess mothers’ lifetime history and current level of depressive and anxiety disorders, including PTSD. A listing of traumatic events (Criterion A for PTSD) is included in the SCID; participants were asked whether or not they had experienced each listed event (e.g., physical assault, sexual assault, witnessed violence). Highly trained clinical psychology Ph.D. graduate students interviewed the youth. A random selection of 20% of all interviews from the larger study were rated by an independent interviewer (2nd author, or advanced Ph.D. clinical psychology student). Reliability in this study was excellent (κ = .91). The SCID was used to select mothers with and without a lifetime PTSD diagnosis and assess traumatic event history among the selected sample. Regarding the Criterion A traumatic event for first onset PTSD diagnosis (M age of first onset = 18.1; SD = 9.1), 36% of mothers described death to family member or close friend; 28% reported rape/sexual abuse; 18% reported witnessing family violence; and 18% reported being the victim of a car accident.
2.2.2. Offspring anxiety disorder
The Anxiety Disorders Interview Schedule for DSM–IV (ADIS; (ADIS; Silverman & Nelles, 1988) was used to ascertain offspring’s diagnostic status for anxiety disorders, according to DSM–IV (APA, American Psychiatric Association, 1994) criteria. The ADIS is a semistructured interview designed specifically for the diagnosis of anxiety, including PTSD, in children and adolescents. Like the SCID, the ADIS assesses exposure to traumatic events prior to administering questions about PTSD symptoms. It is the most frequently used diagnostic interview with youth and has demonstrated strong evidence of reliability and validity. The ADIS was used to ascertain and confirm that no offspring met criteria for PTSD for this investigation. Traumatic event history, which did not statistically differ between the two offspring groups (X2 = .46, p = .489), was as follows: 12% of offspring described death to family member or close friend; 6% reported physical abuse; 12% reported rape/sexual abuse, 26% reported witnessing family violence; 34% reported witnessing community violence; 3% reported being in a natural disaster or fire; and 18% reported being a victim of a car accident. As with the SCID, highly trained clinical psychology Ph.D. graduate students interviewed the youth. A random selection of 20% of all interviews from the larger study were rated by an independent interviewer (2nd author, or advanced Ph.D. clinical psychology student). Interrater reliability was good (κ = .78).
2.2.3. Child depressive symptoms and suicidality
Youths’ depressive symptoms were assessed using the Children’s Depression Inventory (Kovacs, 2003), a well-established, reliable, and valid measure of depressive symptoms in youth (Klein et al., 2005). Suicidal ideation was measured using suicidality items from the Self-Injurious Thoughts and Behaviors Interview (SITBI; Nock et al., 2007), a valid structured clinical interview that assesses suicidal and non-suicidal thoughts and engagement.
2.2.4. Child physical anxiety symptoms
Children’s physical anxiety symptoms were measured with Multidimensional Anxiety Scale for Children (2nd edition) (March et al., 1997), physical anxiety subscale. Items include symptoms that resemble PTSD, including tense/restless and somatic/autonomic symptoms.
2.2.5. Externalizing behaviors
Externalizing behavior was measured using the Parent-Report form of the Child Behavior Checklist (CBCL; Achenbach & Rescorla, 2001), a reliable and widely used method of identifying problem behavior in children.
2.2.4. Laboratory stress paradigm and cortisol assessment
The developmentally sensitive stress paradigm for youth was modeled after other reliable, valid distress-inducing laboratory challenges (Stroud et al., 2002; Nock & Mendes, 2008) and included two components. First, youth completed a modified Wisconsin Card Sort Task (Nock & Mendes, 2008). The experimenter informed the child that s/he had correctly matched the key cards after the first three trials (to engage the child) but then told the child the next seven responses were incorrect (to induce distress). Then, the child was told the 11th trial was suitable while the next nine responses were wrong. The second half of the challenge included a stressful math performance task, modified from a validated math achievement stress task (Stroud et al., 2002) to be developmentally appropriate for youth. The participants had to complete a series of difficult math problems (on average 2 grade levels above the child’s grade) on a white board, while time pressure was imposed, and the child had to explain his/her reasoning and answers to a panel (2–3 trained individuals) of impassive judges. This set of laboratory challenge tasks was used, because they were expected to be developmentally appropriate across the ages of the participants, they have been used previously in other stress-inducing laboratory challenge paradigms, and they involved the essential elements (e.g., threat of social rejection and evaluation, anticipatory and processive stress) known to activate the HPA axis in children and adolescents (Lupien et al., 2009). Prior research using these stress elicitation paradigms demonstrates their validity for relevant outcomes (Stroud et al., 2002; Nock & Mendes, 2008; Hankin, et al., 2010).
Five saliva samples were assessed across the psychosocial challenge. The first was collected an hour after laboratory arrival (baseline), the second was after the challenge (reactivity), and the remaining three (recovery) were at 15-minute intervals after the challenge. Initial cortisol samples were collected at 17:00 (range 16:00–18:30). We followed standard procedures that we have used previously (Hankin et al., 2010; Badanes et al., 2011; Hayden et al., 2014).
Saliva samples were obtained via synthetic salivette collection devices (Sarstedt, Nuembrecht, Germany). Saliva was extracted by centrifuging for 4 minutes at 2500 RPM. Vials and salivettes were frozen at −20° C until data collection was complete. Samples were then defrosted and batched for assay in groups of 36 and were assigned to batches; all samples from the same child were analyzed in the same batch. As is standard protocol, all assays were run in duplicate and the average of the two duplicate assays was used. Samples were sent to the Biochemical Laboratory, Psychobiology, University of Trier, Germany to be assayed. Cortisol levels were determined by employing a competitive solid phase time-resolved fluorescence immunoassay with fluorometric end point detection (DELFIA; Höferl et al., 2005). For samples retained in the analyses described next, the mean interassay coefficients of variation (CV) for controls were 6.6% to 8.5%. For duplicates of the samples used in this study, the intraassay CV was 5%.
3. Results
3.1 Data Analytic Plan
We first examined descriptive statistics regarding maternal and offspring psychopathology associations as a function of maternal PTSD group, reported below. Then, we analyzed the main study question with a 2 (maternal PTSD group) × 5 (cortisol time point) repeated measures ANOVA. Cortisol distributions were not normally distributed, so ln transformations were used in analyses, and Greenhouse-Geisser corrections were applied because sphericity was apparent. In this repeated measures ANOVA, we first controlled for child pubertal status, because prior research indicates that puberty affects youths’ cortisol reactivity (e.g., Gunnar et al, 2009; Stroud et al., 2009; Hankin et al., 2010). We then examined whether offspring of mothers with a diagnosis of PTSD have hyporeactive cortisol profiles to a laboratory stressor. We included covariates of youth traumatic event history, physical anxiety symptoms, and depression and maternal depression, as these factors have been shown in prior research to affect youths’ cortisol reactivity profiles (e.g., Brennan et al., 2008; Lopez-Duran et al., 2009).
3.2 Descriptives
Mothers with a diagnosis of PTSD were more likely to have had a lifetime diagnosis of depressive disorder compared to control mothers: (χ2 = 12.51, p < .001), as 72% of PTSD mothers had a comorbid depressive disorder relative to 21% of control mothers.1 We examined whether offspring stress-related psychopathology symptoms, including whether: (a) physiological symptoms of anxiety (i.e., those which resemble PTSD symptoms, such as tense/restless symptoms); (b) externalizing behaviors; and (c) suicidal ideation significantly differed between the offspring groups (with mothers with and without PTSD). There was a significant maternal PTSD group difference in offspring across all three outcomes. Specifically, maternal PTSD offspring [M = 9.30 (SD = 6.20)] reported significantly more physical anxiety symptoms than offspring whose mothers did not have PTSD [M = 6.79 (SD = 5.43], t (70) = 2.02, p < .05. Similarly, maternal PTSD offspring [M = 12.72 (SD = 9.42)] reported significantly more externalizing behaviors than offspring whose mothers did not have PTSD [M = 8.66 (SD = 8.65], t (70) = 2.10, p < .05. Last, maternal PTSD offspring [M = .24 (SD = .43)] reported significantly more suicidal ideation than offspring whose mothers did not have PTSD [M = .03 (SD = .17], t (70) = 3.49, p < .001.
3.3 Cortisol Reactivity
Preliminary analyses showed that child gender did not predict cortisol reactivity, either as main effect [F(2, 70) = 1.08, p = .34] or in interaction with cortisol values across the laboratory visit [F(1.87, 116.34) = 2.26, p = .11]. Likewise, child age did not significantly affect cortisol reactivity as main effect [F(1, 70) = .009, p = .92] or interacting with cortisol levels over time [F(3.63, 126.92)= 2.25, p = .08]. Finally, neither age nor gender moderated the primary analysis of maternal PTSD × cortisol reactivity.
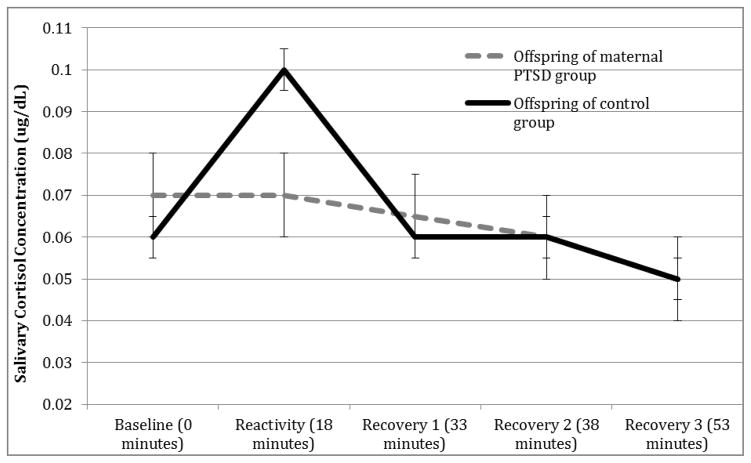
To examine the primary hypothesis, we used a 2 (maternal PTSD group) × 5 (cortisol time point) repeated measures ANOVA. The 2-way interaction was significant [F(1.62, 141.53) = 4.39, p = .02; eta squared = .05]. Planned follow-up analyses to decompose this significant interaction involved repeated measures ANOVA within maternal PTSD groups. For the control group, the expected effect was observed with a rise from baseline to reactivity followed by recovery to baseline levels: [F(1.45, 50.98) = 13.79, p < .001)] with a significant quartic component [F(1, 35) = 9.79, p = .004]. For the offspring of maternal PTSD group, there was no significant cortisol change over time: [F(1.45, 50.98) = 1.87, p = .17)]. Consistent with our primary hypothesis, group differences emerged only for the reactivity component, t(1,70) = 2.39, p = .02.
Finally, these significant differences were maintained even after controlling for maternal clinical depression [i.e., primary 2×5 repeated measures ANOVA, F(1.82, 127.41) = 6.49, p = .01], youths’ traumatic event history [F(1.83, 130.32) = 3.89, p = .026], depressive symptoms [F(1.82, 118.14) = 5.55, p = .02], and youths’ physical anxiety symptoms [F(1.85, 121.84) = 6.32, p = .004]. As depicted in the Figure, the control offspring exhibited the anticipated peaked cortisol reactivity pattern to the stress challenge, whereas the offspring of mothers with PTSD displayed a flat, hyporeactive cortisol configuration.
Figure.
Between Group Differences in Offspring Salivary Cortisol Before, During, and After Laboratory Stressor Tasks
4. Discussion
The current study is the first to investigate stressor-provoked cortisol levels of youth offspring of mothers diagnosed with PTSD in comparison to stressor-provoked cortisol levels of youth offspring of mothers without PTSD. This study showed that offspring of mothers with PTSD demonstrated an attenuated response to the acute stressor, whereas offspring of mothers without PTSD demonstrated an expected sharp peak in cortisol immediately following the stressor. Results were maintained even after controlling for offspring traumatic event history and maternal and child depression. Importantly, none of the children in the current sample had a diagnosis of PTSD. Thus, the blunted cortisol response in reaction to the stressor among the maternal PTSD group cannot be attributed to the youth themselves having PTSD.
Other areas of stress-related psychopathology were examined and compared among the offspring in the two groups. Offspring of mothers with PTSD reported significantly higher physical symptoms of anxiety (e.g., tense, restlessness), externalizing behaviors, and suicidality in comparison to offspring of mothers without PTSD. These higher levels of stress-related psychopathology among maternal PTSD offspring are in line with previous studies demonstrating that this population has heightened vulnerability to a host of problems across the lifespan, including PTSD (Leen-Feldner et al., 2013).
Although results from the current study are not able to speak to the role of blunted stress reactivity (low levels of cortisol) in the intergenerational transmission of PTSD per se in this relatively young sample, increased levels of other related mental health symptoms suggest the possible link between transmission of altered stress reactivity and susceptibility to stress-related psychopathology. Building upon a relatively small but consistent psychoneuroendocrine transmission literature in which non-provoked low cortisol stress response has been implicated (see Leen-Feldner et al., 2013 for review), our findings suggest promising next steps in this line of research that seek to understand when offspring stress reactivity is altered in the context of maternal traumatic stress—and how this ultimately relates to psychopathology and impairment in functioning across the generations.
Our results are consistent with previous diurnal cortisol studies demonstrating attenuated cortisol responses with adult (e.g., Yehuda et al., 2002), adolescent (Van Den Bergh et al., 2007), and infant offspring samples (e.g., Yehuda et al., 2005). This consistency in findings across differing cortisol response methodology (e.g., diurnal levels vs. levels following a dexamethasone suppression test vs. levels in response to an acute stressor) suggest that attenuated HPA axis functioning among offspring of mothers with PTSD is robust. We have extended these prior results by measuring cortisol levels in response to an acute stressor using a gold standard paradigm shown to induce adaptive cortisol responses (i.e., showing a sharp peak following the stressor) in child populations (Stroud et al., 2002; Nock & Mendes, 2008). Such acute stressor paradigms are designed to provide an ecologically valid snapshot of the HPA axis response when an individual is faced with acute stress, which is particularly relevant in considering transmission of trauma-related psychopathology, such as PTSD. That is, consideration of how a person responds in the face of a potentially traumatic event—including at a neuroendocrine level (e.g., acute cortisol measures)—may have implications for the mental health sequelae that follows a flight/flight/freeze response to the environmental stressor. Indeed, recent research has highlighted acute cortisol levels as a potential biomarker for PTSD risk (Mouthhaan et al., 2014)— although it has been noted that the positive and negative predictive value of acute cortisol levels for PTSD remains to be seen. That stress-provoked acute cortisol levels in the current study were low among maternal PTSD offspring contributes to the notion of a, ‘unique risk profile marked by reduced cortisol levels’ (Leen-Feldner et al., 2013, p. 1123) among offspring of parents with PTSD. However, in contrast to prior studies, this risk profile can now be extended to include low cortisol reactivity in response to an environmental stressor (i.e., a situation where an elevated cortisol response would be considered most adaptive). Importantly, in the absence of longitudinal outcomes for offspring PTSD tied to earlier low cortisol reactivity, questions remain whether acute low cortisol reactivity levels hold predictive value in terms of response in future traumatic event exposures and eventual PTSD and related psychopathology.
Among the offspring in our sample, none had been diagnosed with PTSD. As reviewed earlier, altered HPA axis functioning has been implicated as a risk factor and functional outcome for PTSD, and intergenerational transmission of low cortisol has similarly been implicated in the transmission of PTSD from mother to child. Extending from this, our design permits specific examination of acute stress reactivity as a function of maternal PTSD-- rather than a function of the offspring’s PTSD. Given the high rates of PTSD among offspring of parents with PTSD, as well as the traumatic event history among this group (as a preamble to the effects of future and cumulative trauma; Macdonald et al., 2010), it is highly likely that some of the youth in this group will go on to develop PTSD. Current results suggest offspring of mothers with PTSD have altered HPA axis functioning in the form of low cortisol reactivity. Thus, when these youth are exposed to future traumatic events and their body initiates a normal stress response, they may have difficulty ending this stress-response in the absence of sufficient cortisol levels to do so (e.g., Yehuda, 2002), which in turn may lead to extended periods of arousal and a heightened vulnerability to onset of PTSD. Indeed, vulnerability stress models purport that offspring of mothers with PTSD are susceptible to onset of PTSD due to their genetic/biological makeup, which then interacts with stress in the environment to create overarching vulnerability.
In addition to their relevance to vulnerability stress models, results from the current study support other biologically–driven intergenerational transmission theories, which have suggested that stress reactivity plays a noteworthy role in linking maternal PTSD and stress-related psychopathology vulnerability among offspring. Importantly, this altered stress reactivity may result from a range of direct (e.g., fostering environments that lead to early and chronic exposure to stress and traumatic events for the offspring; Rijlaarsdam et al., 2014) and indirect influences (e.g., stressful in-utero environment; e.g., Yehuda et al., 2005; inherited epigenetic signatures; Yehuda & Bierer, 2009; genetic vulnerabilities; Zoladz & Diamond, 2013), which may vary by individual. For example, Yehuda (2002) has proposed that altered stress reactivity may result from a pre-existing biological (e.g., genetic) risk for a tendency for reduced adrenal output or result from the environmental exposure to severe and possibly chronic traumatic stress, and that the biological demand of this environmental severe stress can lead to an enhanced negative feedback cortisol inhibition on the pituitary. Trickett and colleagues (2010) have found support for the latter theory in a longitudinally-followed sample of female sexual abuse victims, among whom a hypersecretion of cortisol in childhood and earlier adolescence was initially found, followed by evidence of hyposecretion (when body can no longer uphold biological demand) in later adolescence. Thus, mothers may be conferring this risk environmentally (i.e., by perpetuating an environment of severe stress, to which the offspring’s body reacts) or biologically (i.e., offspring inheriting the tendency for reduced adrenal output). Developmental psychopathology research, which calls for longitudinal designs with community samples and multiple units of analysis, is necessary to more fully inform these theoretical models and the implications of the intergenerational transmission of altered HPA axis regulation, and low cortisol in particular, among mothers with PTSD and their offspring.
Several strengths of our study are worth noting. First, where some previous investigations have focused solely on self-reported, continuous measures of posttraumatic stress, mothers for the current investigation were diagnosed with PTSD using structured clinical interviewing and reliability testing. Second, as noted above, our study was conducted with children with mothers with PTSD—but without PTSD themselves. That is, our target population was youth who have been empirically-demonstrated to be vulnerable to the onset of later PTSD-- but have not yet developed the disorder. Our selection of this population is a strength in that it prohibits calling into question whether the blunted cortisol response results could be attributed to the offspring’s own PTSD. Third, also as noted above, a well-established acute stressor paradigm was applied in the current study to create an ecologically valid snapshot of stress provoked acute cortisol response in the maternal PTSD offspring population, which had not been previously reported in the literature with this population.
Interpretation of the current results is limited by the focus on mothers only. As reviewed in the Introduction, a growing body of research suggests that maternal PTSD plays a more significant role than paternal PTSD in an offspring’s vulnerability to PTSD-- however, paternal PTSD is a more potent risk factor for offspring depression, and dose effects (where risk for offspring psychopathology is heightened when both parents have PTSD) may be relevant in certain samples as well (Yehuda et al., 2008). Another limitation to note is that the assessment of other aspects of altered HPA axis functioning, such as glucocorticoid sensitivity, was not included. Finally, the lack of longitudinal data as to whether the offspring of the maternal PTSD group eventually developed PTSD themselves prohibits more extensive conclusions regarding the intergenerational transmission of PTSD.
The current study offers support for the intergenerational transmission of low stress-provoked cortisol from mothers diagnosed with PTSD to youth offspring. Additional research is warranted to better understand how and when HPA axis functioning is altered in this transmission. Better elucidating the timing of maternal PTSD onset and the circumstances under which low cortisol reactivity is conferred will further inform the study of the neuroendocrinology of PTSD. The answer to such ‘when’ and ‘how’ empirical questions in this area also establish important future avenues for prevention-focused research—such as whether environmental manipulations (e.g., intervention) can be used to alter HPA axis functioning in adaptive directions.
Highlights.
We examine cortisol reactivity among offspring of mothers with and without PTSD
We measure salivary cortisol across five time-points in response to a lab stressor
Offspring of mothers with PTSD exhibit flat cortisol reactivity to stressor
Offspring of mothers without PTSD display peaked cortisol reactivity
Low cortisol reactivity is implicated in intergenerational risk transmission
Acknowledgments
This work was supported by R01MH077195 (PI: Hankin) from the National Institute of Mental Health (NIMH). The preparation of this manuscript was supported by R01DA031285 (PI: Danielson) from the National Institute on Drug Abuse (NIDA) and P50AA010761 (PI: Becker) from the National Institute on Alcohol Abuse and Alcoholism (NIAAA). Views expressed herein are those of the authors and do not necessarily represent the official views of NIMH, NIDA, or NIAAA. We thank Kathryn Soltis for her assistance in the preparation of this manuscript.
The Role of the Funding Sources
Funding Sources for the research have been described in the Acknowledgements attachment. The funding sources had no involvement in the study procedures or manuscript preparation.
Footnotes
Repeated Measures ANOVA analyses were completed for offspring with regard to maternal depression (MDD) (comparing cortisol reactivity between offspring of mothers with and without MDD). No effect on offspring cortisol reactivity was found between the groups where the 2-way interaction was not significant [F(1.78, 103.27)=.85, p=.42; eta squared = .01], and the quadratic within subjects contrast was not significant, F (1, 70) = 1.37, p = .25, eta squared = .023.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Contributor Information
Carla Kmett Danielson, Department of Psychiatry & Behavioral Sciences, 67 President Street, Medical University of South Carolina, Charleston, South Carolina, 29425 United States
Benjamin L. Hankin, Department of Psychology, 2155 South Race Street, University of Denver, Denver, Colorado, 80208 United States
Lisa S. Badanes, Department of Psychology, Plaza Building 220-P, Metropolitan State University of Denver, Denver, Colorado, 80204 United States
References
- Achenbach TM, Rescorla LA. Manual for the ASEBA School-Age Forms and Profiles. University of Vermont Research Center for Children, Youth, and Families; Burlington, VT: 2001. [Google Scholar]
- Al-Turkait FA, Ohaeri JU. Child and adolescent psychiatry and mental health. Child Adolesc Psychiat Ment Health. 2008;2(12):12. doi: 10.1186/1753-2000-2-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- American Psychiatric Association. Diagnostic and statistical manual of mental disorders. 4. American Psychiatric Association; Washington, D. C: 1994. [Google Scholar]
- American Psychiatric Association. Diagnostic and statistical manual of mental disorders. 5. American Psychiatric Association; Washington, D. C: 2013. [Google Scholar]
- Badanes LS, Watamura SE, Hankin BL. Hypocortisolism as a potential marker of allostatic load in children: associations with family risk and internalizing disorders. Dev Psychopathol. 2011;23(03):881–896. doi: 10.1017/S095457941100037X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barrocas A, Hankin BL, Young JF, Abela JRZ. Rates of non-suicidal self-injury in youth: age, gender, and behavioral methods in a general community sample. Pediatrics. 2012;130:39–45. doi: 10.1542/peds.2011-2094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brennan PA, Pargas R, Walker EF, Green P, Jeffrey Newport D, Stowe Z. Maternal depression and infant cortisol: influences of timing, comorbidity and treatment. J Child Psychol Psychiatry. 2008;49(10):1099–1107. doi: 10.1111/j.1469-7610.2008.01914.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chemtob CM, Nomura Y, Rajendran K, Yehuda R, Schwartz D, Abramovitz R. Impact of maternal posttraumatic stress disorder and depression following exposure to the September 11 attacks on preschool children’s behavior. Child Dev. 2010;81(4):1129–1141. doi: 10.1111/j.1467-8624.2010.01458.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cicchetti D, Toth SL. The past achievements and future promises of developmental psychopathology: the coming of age of a discipline. J Child Psychol Psychiatry. 2009;50:16–25. doi: 10.1111/j.1469-7610.2008.01979.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Entringer S, Kumsta R, Hellhammer DH, Wadwa PD, Wust S. Prenatal exposure to maternal psychosocial stress and HPA axis regulation in young adults. Horm Behav. 2009;55:292–298. doi: 10.1016/j.yhbeh.2008.11.006. [DOI] [PubMed] [Google Scholar]
- Francis D, Diorio J, Liu D, Meaney MJ. Nongenomic transmission across generations of maternal behavior and stress responses in the rat. Science. 1999;268:1155–1158. doi: 10.1126/science.286.5442.1155. [DOI] [PubMed] [Google Scholar]
- Galovski T, Lyons JA. Psychological sequelae of combat violence: a review of the impact of PTSD on the veteran’s family and possible interventions. Aggress Violent Beh. 2004;9(5):477–501. [Google Scholar]
- Glover V, O’Connor TG, O’Donnell K. Prenatal stress and the programming of the HPA axis. Neurosci Biobehav R. 2010;35(1):17–22. doi: 10.1016/j.neubiorev.2009.11.008. [DOI] [PubMed] [Google Scholar]
- Graham-Bermann SA, Gruber G, Howell KH, Girz L. Factors discriminating among profiles of resilience and psychopathology in children exposed to intimate partner violence (IPV) Child Abuse Neglect. 2009;33(9):648–660. doi: 10.1016/j.chiabu.2009.01.002. [DOI] [PubMed] [Google Scholar]
- Guerry JD, Hastings PD. In search of HPA axis dysregulation in child and adolescent depression. Clin Child Fam Psych. 2011;14(2):135–160. doi: 10.1007/s10567-011-0084-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gunnar MR, Wewerka S, Frenn K, Long JD, Griggs C. Developmental changes in hypothalamus–pituitary–adrenal activity over the transition to adolescence: normative changes and associations with puberty. Dev Psychopathol. 2009;21(01):69–85. doi: 10.1017/S0954579409000054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Halligan SL, Herbert J, Goodyer I, Murray L. Disturbances in morning cortisol secretion in association with maternal postnatal depression predict subsequent depressive symptomatology. Biol Psychiat. 2007;62:40–46. doi: 10.1016/j.biopsych.2006.09.011. [DOI] [PubMed] [Google Scholar]
- Hankin BL, Badanes LS, Abela JRZ, Watamura SE. Hypothalamic pituitary adrenal axis dysregulation in dysphoric children and adolescents: cortisol reactivity to psychological distress from preschool through middle adolescence. Biol Psychiat. 2010;68:484–490. doi: 10.1016/j.biopsych.2010.04.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hayden EP, Hankin BL, Mackrell SV, Sheikh HI, Jordan PL, Dozois DJ, Singh SM, Badanes LS. Parental depression and child cognitive vulnerability predict children’s cortisol reactivity. Dev Psychopathol. 2014;26(4pt2):1445–1460. doi: 10.1017/S0954579414001138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heim C, Ehlert U, Hellhammer DH. The potential role of hypocortisolism in the pathophysiology of stress-related bodily disorders. Psychoneuroendocrinology. 2000;25:1–35. doi: 10.1016/s0306-4530(99)00035-9. [DOI] [PubMed] [Google Scholar]
- Höferl M, Krist S, Buchbauer G. Adaptation of DELFIA cortisol kit for determination of salivary cortisol concentration. Archiv der Pharmazie. 2005;338:493–497. doi: 10.1002/ardp.200500116. [DOI] [PubMed] [Google Scholar]
- Kessler RC, Chiu WT, Demler O, Walters EE. Prevalence, severity, and comorbidity of 12-month DSM-IV disorders in the National Comorbidity Survey Replication. Arch Gen Psychiat. 2005;62(6):617–627. doi: 10.1001/archpsyc.62.6.617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klein DN, Dougherty LR, Olino TM. Toward guidelines for evidence-based assessment of depression in children and adolescents. J Clin Child Adolesc. 2005;34(3):412–432. doi: 10.1207/s15374424jccp3403_3. [DOI] [PubMed] [Google Scholar]
- Kovacs M. The Children’s Depression Inventory (CDI): Technical Manual. Multi-Health Systems; North Tonawanda, NY: 2003. [Google Scholar]
- Leen-Feldner EW, Feldner MT, Knapp A, Bunaciu L, Blumenthal H, Amstadter AB. Offspring psychological and biological correlates of parental posttraumatic stress: review of the literature and research agenda. Clin Psychol Rev. 2013;33(8):1106–1133. doi: 10.1016/j.cpr.2013.09.001. [DOI] [PubMed] [Google Scholar]
- Lehmann P. Posttraumatic stress disorder (PTSD) and child witness to mother-assault: a summary and review. Child Youth Serv Rev. 2000;22:275–306. [Google Scholar]
- Lehrner A, Bierer LM, Passarelli V, Pratchett LC, Flory JD, Bader HN, Yehuda R. Maternal PTSD associates with greater glucocorticoid sensitivity in offspring of Holocaust survivors. Psychoneuroendocrino. 2014;40:213–220. doi: 10.1016/j.psyneuen.2013.11.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li X, Huang X, Tan H, Liu A, Zhou J, Yang T. A study on the relationship between posttraumatic stress disorder in flood victims parents and children in Hunan, China. Aust NZ J Psychiat. 2010;44:543–550. doi: 10.3109/00048671003601400. [DOI] [PubMed] [Google Scholar]
- Linares LO, Heeren T, Bronfman E, Zuckerman B, Augustyn M, Tronick E. A mediational model for the impact of exposure to community violence on early child behavior problems. Child Dev. 2001;72(2):639–652. doi: 10.1111/1467-8624.00302. [DOI] [PubMed] [Google Scholar]
- Lopez-Duran NL, Kovacs M, George CJ. Hypothalamic–pituitary–adrenal axis dysregulation in depressed children and adolescents: a meta-analysis. Psychoneuroendocrino. 2009;34(9):1272–1283. doi: 10.1016/j.psyneuen.2009.03.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lupien SJ, McEwen BS, Gunnar MR, Heim C. Effects of stress throughout the lifespan on the brain, behaviour and cognition. Nat Rev Neurosci. 2009;10:434–445. doi: 10.1038/nrn2639. [DOI] [PubMed] [Google Scholar]
- Macdonald A, Danielson CK, Resnick HS, Saunders BE, Kilpatrick DG. PTSD and comorbid disorders in a representative sample of adolescents: the risk associated with multiple exposures to potentially traumatic events. Child Abuse Neglect. 2010;34:773–783. doi: 10.1016/j.chiabu.2010.03.006. [DOI] [PubMed] [Google Scholar]
- March JS, Parker JD, Sullivan K, Stallings P, Conners CK. The Multidimensional Anxiety Scale for Children (MASC): factor structure, reliability, and validity. J Am Acad Child Psy. 1997;36(4):554–565. doi: 10.1097/00004583-199704000-00019. [DOI] [PubMed] [Google Scholar]
- Mason JU, Giller E, Kosten T, Ostroff R, Podd L. Urinary free cortisol levels in PTSD-patients. J Nerv Ment Dis. 1986;174:145–155. doi: 10.1097/00005053-198603000-00003. [DOI] [PubMed] [Google Scholar]
- Mouthaan J, Sijbrandij M, Luitse JSK, Goslings JC, Gersons BPR, Olff M. The role of acute cortisol and DHEAS in predicting acute and chronic PTSD symptoms. Psychoneuroendocrino. 2014;45:179–186. doi: 10.1016/j.psyneuen.2014.04.001. [DOI] [PubMed] [Google Scholar]
- Nock MK, Holmberg EB, Photos VI, Michel BD. Self-Injurious Thoughts and Behaviors Interview: development, reliability, and validity in an adolescent sample. Psychol Assess. 2007;19(3):309–317. doi: 10.1037/1040-3590.19.3.309. [DOI] [PubMed] [Google Scholar]
- Nock MK, Mendes WB. Physiological arousal, distress tolerance, and social problem-solving deficits among adolescent self-injurers. J Consult Clin Psych. 2008;76(1):28. doi: 10.1037/0022-006X.76.1.28. [DOI] [PubMed] [Google Scholar]
- Nugent NR, Ostrowski S, Christopher NC, Delanty D. Parental posttraumatic stress symptoms as a moderator of child’s acute biological response and subsequent posttraumatic stress symptoms in pediatric injury patients. J Pediatr Psychol. 2007;32:309–318. doi: 10.1093/jpepsy/jsl005. [DOI] [PubMed] [Google Scholar]
- Pitman RK, Gilbertson MW, Gurvits TV, May FS, Lasko NB, Metzger LJ, Shenton ME, Yehuda R, Orr SP. Clarifying the origin of biological abnormalities in PTSD through the study of identical twins discordant for combat exposure. Annals of the New tork Academy of Sciences. 2006;1071:242–254. doi: 10.1196/annals.1364.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pratchett LC, Yehuda R. Developmental trauma from a biophysical perspective. In: Pat-Horenczyk R, Irom D, Vogel JM, editors. Helping Child Cope with Trauma: Individual, Family, and Community Perspectives. Routledge; New York, NY: 2014. pp. 1–11. [Google Scholar]
- Roberts AL, Galea S, Austin SB, Cerda M, Wright RJ, Rich-Edwards JW, Koenen KC. Posttraumatic stress disorder across two generations: concordance and mechanisms in a population-based sample. Biol Psychiatry. 2012;72(6):505–511. doi: 10.1016/j.biopsych.2012.03.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rijlaarsdam J, Stevens GW, Jansen PW, Ringoot AP, Jaddoe VW, Hofman A, Tiemeier H. Maternal childhood maltreatment and offspring emotional and behavioral problems maternal and paternal mechanisms of risk transmission. Child Maltreatment. 2014;19(2):67–78. doi: 10.1177/1077559514527639. [DOI] [PubMed] [Google Scholar]
- Samuelson KW, Cashman C. Effects of intimate partner violence and maternal posttraumatic stress symptoms on children’s emotional and behavioral functioning. J of Emotional Abuse. 2008;8(1–2):139–153. [Google Scholar]
- Scheeringa MS, Zeanah CH. A relational perspective on PTSD in early childhood. J Trauma Stress. 2001;14:799–815. doi: 10.1023/A:1013002507972. [DOI] [PubMed] [Google Scholar]
- Schmidt U, Holsboer F, Rein T. Epigenetic aspects of posttraumatic stress disorder. Dis Markers. 2011;30(2):77–87. doi: 10.3233/DMA-2011-0749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Silverman WK, Nelles WB. The anxiety disorders interview schedule for children. J Am Acad Child Psy. 1988;27(6):772–778. doi: 10.1097/00004583-198811000-00019. [DOI] [PubMed] [Google Scholar]
- Stroud LR, Foster E, Papandonatos GD, Handwerger K, Granger DA, Kivlighan KT, Niaura R. Stress response and the adolescent transition: performance versus peer rejection stressors. Dev Psychopathol. 2009;21(01):47–68. doi: 10.1017/S0954579409000042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stroud LR, Salovey P, Epel ES. Sex differences in stress response: social rejection versus achievement stress. Biol Psychiat. 2002;52:318–327. doi: 10.1016/s0006-3223(02)01333-1. [DOI] [PubMed] [Google Scholar]
- Sturge-Apple ML, Davies PT, Cicchetti D, Manning LG. Interparental violence, maternal emotional unavailability, and children’s cortisol functioning in family contexts. Developmental Psychology. 2012;48:237–249. doi: 10.1037/a0025419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tollenaar MS, Beijers R, Jansen J, Riksen-Walraven JM, de Weerth C. Maternal prenatal stress and cortisol reactivity to stressors in human infants. Stress. 2011;14:53–65. doi: 10.3109/10253890.2010.499485. [DOI] [PubMed] [Google Scholar]
- Trickett PK, Noll JG, Susman EJ, Shenk CE, Putnam FW. Attenuation of cortisol across development for victims of sexual abuse. Dev Psychopathol. 2010;22(1):165–175. doi: 10.1017/S0954579409990332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van den Bergh BR, Van Calster B, Smits T, Van Huffel S, Lagae L. Antenatal maternal anxiety is related to HPA-axis dysregulation and self-reported depressive symptoms in adolescence: a prospective study on the fetal origins of depressed mood. Neuropsychopharmacol. 2007;33(3):536–545. doi: 10.1038/sj.npp.1301450. [DOI] [PubMed] [Google Scholar]
- Weems CF, Scheeringa MS. Maternal depression and treatment gains following a cognitive behavioral intervention for posttraumatic stress in preschool children. J Anxiety Disord. 2013;27(1):140–146. doi: 10.1016/j.janxdis.2012.11.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yehuda R. Current status of cortisol findings in post-traumatic stress disorder. Psychiat Clin N Am. 2002;25:341–368. doi: 10.1016/s0193-953x(02)00002-3. [DOI] [PubMed] [Google Scholar]
- Yehuda R, Bell A, Bierer LM, Schmeidler J. Maternal, not paternal, PTSD is related to increased risk for PTSD in offspring of Holocaust survivors. J Psychiatr Res. 2008;42:1104–1111. doi: 10.1016/j.jpsychires.2008.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yehuda R, Bierer LM. Transgenerational transmission of cortisol and PTSD risk. Prog Brain Res. 2008;167:121–135. doi: 10.1016/S0079-6123(07)67009-5. [DOI] [PubMed] [Google Scholar]
- Yehuda R, Bierer LM. The relevance of epigenetics to PTSD: implications for the DSM V. J Trauma Stress. 2009;22(5):427–434. doi: 10.1002/jts.20448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yehuda R, Bierer LM, Schmeidler J, Aferiat DH, Breslau I, Dolan S. Low cortisol and risk for PTSD in adult offspring of holocaust survivors. Am J Psychiat. 2000;157(8):1252–1259. doi: 10.1176/appi.ajp.157.8.1252. [DOI] [PubMed] [Google Scholar]
- Yehuda R, Blair W, Labinsky E, Bierer LM. Effects of parental PTSD on the cortisol response to dexamethasone administration in their adult offspring. Am J Psychiat. 2007a;164:163–166. doi: 10.1176/ajp.2007.164.1.163. [DOI] [PubMed] [Google Scholar]
- Yehuda R, Boisoneau D, Mason JW, Giller EL. Glucocorticoid receptor number and cortisol excretion in mood, anxiety, and psychotic disorders. Biol Psychiat. 1993;34(1):18–25. doi: 10.1016/0006-3223(93)90252-9. [DOI] [PubMed] [Google Scholar]
- Yehuda R, Engel SM, Brand SR, Seckl J, Marcus SM, Berkowitz GS. Transgenerational effects of posttraumatic stress disorder in babies of mothers exposed to the World Trade Center attacks during pregnancy. J Clin Endocr Metab. 2005;90:4115–4118. doi: 10.1210/jc.2005-0550. [DOI] [PubMed] [Google Scholar]
- Yehuda R, Halligan SL, Bierer LM. Relationship of parental trauma exposure and PTSD to PTSD, depressive and anxiety disorders in offspring. J Psychiat Res. 2001a;35(5):261–270. doi: 10.1016/s0022-3956(01)00032-2. [DOI] [PubMed] [Google Scholar]
- Yehuda R, Halligan SL, Bierer LM. Cortisol levels in adult offspring of Holocaust survivors: relation to PTSD symptom severity in the parent and child. Psychoneuroendocrino. 2002;27:171–180. doi: 10.1016/s0306-4530(01)00043-9. [DOI] [PubMed] [Google Scholar]
- Yehuda R, Halligan SL, Grossman R. Childhood trauma and risk for PTSD: relationship to intergenerational effects of trauma, parental PTSD, and cortisol excretion. Dev Psychopathol. 2001b;13(3):733–753. doi: 10.1017/s0954579401003170. [DOI] [PubMed] [Google Scholar]
- Yehuda R, Schmeidler J, Wainberg M, Binder-Brynes K, Duvdevani T. Vulnerability to posttraumatic stress disorder in adult offspring of Holocaust survivors. Am J Psychiat. 1998a;155:1163–1171. doi: 10.1176/ajp.155.9.1163. [DOI] [PubMed] [Google Scholar]
- Yehuda R, Schmeidler J, Giller EL, Jr, Siever LJ, Binder-Brynes K. Vulnerability to posttraumatic stress disorder in adult offspring of Holocaust survivors. Am J Psychiat. 1998b;155:1163–1171. doi: 10.1176/ajp.155.9.1163. [DOI] [PubMed] [Google Scholar]
- Yehuda R, Teicher MH, Seckl JR, Grossman RA, Morris A, Bierer LM. Parental PTSD as a vulnerability factor for low cortisol trait in offspring of holocaust survivors. Arch Gen Psychiat. 2007b;64(9):1040–1048. doi: 10.1001/archpsyc.64.9.1040. [DOI] [PubMed] [Google Scholar]
- Zoladz PR, Diamond DM. Current status on behavioral and biological markers of PTSD: a search for clarity in a conflicting literature. Neurosci Biobehav R. 2013;37(5):860–895. doi: 10.1016/j.neubiorev.2013.03.024. [DOI] [PubMed] [Google Scholar]