Abstract
The native structure of the Azoarcus group I ribozyme is stabilized by the cooperative formation of tertiary interactions between double helical domains. Thus, even single mutations that break this network of tertiary interactions reduce ribozyme activity in physiological Mg2+ concentrations. Here, we report that molecular crowding comparable to that in the cell compensates for destabilizing mutations in the Azoarcus ribozyme. Small angle X-ray scattering, native polyacrylamide gel electrophoresis and activity assays were used to compare folding free energies in dilute and crowded solutions containing 18% PEG1000. Crowder molecules allowed the wild-type and mutant ribozymes to fold at similarly low Mg2+ concentrations and stabilized the active structure of the mutant ribozymes under physiological conditions. This compensation helps explains why ribozyme mutations are often less deleterious in the cell than in the test tube. Nevertheless, crowding did not rescue the high fraction of folded but less active structures formed by double and triple mutants. We conclude that crowding broadens the fitness landscape by stabilizing compact RNA structures without improving the specificity of self-assembly.
INTRODUCTION
Many non-coding RNAs must fold into specific three-dimensional (3D) shapes to function in the cell (1–3). These 3D structures are defined by tertiary interaction motifs that hold double helices together and that contribute to the overall stability of the folded RNA (4–9). Using the group I ribozyme from the Azoarcus bacterium as a model system, we previously showed that tertiary interactions in different regions of the RNA form cooperatively during assembly of the core helices early in the folding process (10). This cooperative interaction network favors native-like intermediates and suppresses alternative, non-native conformations.
Because single mutations can disrupt this cooperative tertiary interaction network, they can substantially destabilize the native structure of the RNA and reduce its in vitro activity, as observed in a number of ribozymes (4,11–14). Consequently, many RNAs appear intolerant to mutation, raising the question of how non-coding RNAs evolve new functions or adapt to different genetic contexts. Nevertheless, previous studies on group I ribozymes showed that mutations in tertiary interaction motifs impaired the ribozyme much less in the cell than in the test tube (12,15–16). These observations suggested that the intracellular environment allows less stable sequences to fold, either by stabilizing the native tertiary structure or by reducing misfolding.
Here, we show that molecular crowding comparable to that in the cell overcomes the destabilizing effects of single, double or triple mutations that disrupt specific tertiary interactions in the Azoarcus group I ribozyme. In the crowded interior of a cell, macromolecules occupy ∼30% of the available volume and thus restrict the space available to an RNA molecule (17). Such excluded volume effects lower the translational and configurational entropy of the unfolded RNA more than they lower the entropy of the folded RNA, driving the folding equilibrium toward the native state.
We previously used small angle X-ray scattering (SAXS) to show that synthetic crowders such as polyethylene glycol (PEG) or Ficoll stabilize the folded structure of the wild-type (WT) ribozyme through excluded volume effects (18,19). By contrast, small co-solutes such as sucrose and ethylene glycol do not stabilize this ribozyme's tertiary structure (18,19). Here, we show that these excluded volume effects are sufficient to offset the loss of specific RNA tertiary interactions, so that mutant and WT ribozymes fold in similar Mg2+ concentrations in a crowded solution. Molecular crowding also increases the ribozyme activity. Nevertheless, the double and triple mutants turnover more slowly than the WT RNA, because the mutants form both active and inactive structures. We conclude that crowding drives RNAs into compact structures in physiological Mg2+ concentrations, but cannot improve the specificity of folding when tertiary interactions are lost. We discuss how specific and non-specific stabilization of RNA tertiary structure is likely to influence the natural selection of non-coding RNAs.
MATERIALS AND METHODS
RNA preparation
The Azoarcus group I L-9 and L-3 ribozymes (WT and mutants) were prepared by run-off T7 transcription and purified by polyacrylamide gel electrophoresis (PAGE) as previously described (10,20). The 9-nt RNA substrate 5′ rCAUAUCGCC was obtained from Integrated DNA Technologies. Final samples contained 0.4 mg/ml (6 μM) ribozyme in 20 mM Tris–HCl (pH 7.5), plus the stated concentrations of MgCl2 and PEG 1000 at 37°C.
Small angle X-ray scattering
SAXS measurements were conducted at BioCAT (ID18; Advanced Photon Source, Argonne National Laboratory, IL, USA) at wavelength = 1.033 Å (12 keV) over the momentum transfer range Q = 0.006–0.34 Å−1. The SAXS data were corrected for background scattering and indirectly transformed to yield the pair distribution function, P(r), and the radius of gyration, Rg (21). RNA was refolded as previously described (10,18) except that diluted RNA in 20 mM Tris–HCl was incubated at 50°C for 5 min before and after the addition of PEG1000. Each sample (100 μl) was individually adjusted to the desired MgCl2 concentration, then incubated 10 min at 50°C and 30 min at 37°C. This method of sample preparation may account for slightly smaller differences between the WT and particular mutant ribozymes than previously observed when the RNA was prefolded at 50°C and titrated in MgCl2 at 37°C (10).
Native PAGE
RNA samples were prepared as for SAXS, except samples were doped with a small amount of 32P-labeled RNA and contained 7% (v/v) glycerol and <0.1% (w/v) xylene cyanol. After equilibration at 37°C, samples were immediately loaded on a native 6% polyacrylamide gel as previously described (22).
Ribozyme activity assay
Single-turnover cleavage assays were carried out at 37°C as previously described (23). L-3 ribozyme (6 μM) was refolded in the desired [MgCl2] as for SAXS, but with 5 mM GTP. Reactions were started with the addition of ∼10 nM 32P-labeled RNA substrate and quenched after 25 s or 3 min by an equal volume of 30 mM ethylenediaminetetraacetic acid, 10 M urea and 0.1X Tris-borate EDTA on ice. Both the ribozyme and substrate were equilibrated with the same concentration of PEG1000 at 37°C before mixing to eliminate any change in viscosity. Quenched reactions were analyzed by denaturing 20% PAGE and the fraction of product over time fit to a stretched exponential rate equation (see SI Methods). The slower rate of cleavage in 18% PEG can be accounted for by the change in the viscosity of the solution (19).
Free energy calculations
Previous work showed that Rg behaves as an order parameter of the solution and can be used to measure the fraction of folded and unfolded RNA (24). Footprinting results confirmed that the tertiary folding of the Azoarcus ribozyme is reasonably approximated by a two or three-state equilibrium (25,26). The change in Rg as a function of Mg2+ concentration C was fit to a three-state model (27):
 |
(1) |
in which Rg,U and Rg,Ic are directly determined from the SAXS data in the lowest and highest Mg2+ concentrations and the other parameters are obtained from fits to the data. The fitted parameter values were used to simulate the population of IC (see SI Methods). The populations of IC and N (from activity assays) were fit to two-state transitions, ƒ(C) = Cn/(Cmn+Cn), to give the midpoint Cm and slope n of each folding transition with and without PEG1000. For each mutant, the free energies of the IU→IC and IC→N folding transitions, ΔG(C) = -nRT ln(Cref/Cm), were evaluated at a reference concentration of Mg2+ Cref, which is equal to half-saturation of the corresponding WT folding transition in dilute buffer or 18% PEG1000, respectively. The confidence intervals of fitted parameters were estimated by a bootstrapping method as previously described (10). The value and reported error of Cm, n and ΔG were obtained from the mean and standard deviation of the bootstrapping results. The free energy perturbations due to the mutations were calculated from ΔΔG = ΔGmut – ΔGWT in each folding condition.
RESULTS
Folding of the Azoarcus ribozyme
For our experiments, we used the group I ribozyme from the purple bacterium Azoarcus sp. BH72, which has an unusually stable secondary and tertiary structure (Figure 1a and b) (28). The tertiary structure of the ribozyme folds in three macroscopic phase transitions (Figure 1c). In 0.2 mM MgCl2, the ribozyme forms an extended intermediate (IU) that is more compact and flexible than the unfolded RNA in no MgCl2 (27,29). In 1 mM MgCl2, the double helices assemble into a compact, native-like intermediate (IC) that is readily detected by solution scattering (30) and native PAGE (22). Further rearrangement of the tertiary interactions in ≥3 mM MgCl2 results in the active, native state (N) that can be detected in biochemical assays (31,32). We used SAXS, native PAGE and ribozyme activity to distinguish the effects of crowding and mutations on each stage of folding (Figure 1d).
Figure 1.
Molecular crowding stabilizes the folded Azoarcus ribozyme. (a,b) Tertiary and secondary structure (PDB 1U6B). Red arrows (a) and circles (b) indicate point mutations that disrupt tertiary interactions: L2 tetraloop, A25U; TH (triple helix), G125A; L9 tetraloop, A190U. (c) As [Mg2+] rises, the population shifts from an unfolded state (U) with secondary but no tertiary structure, to the extended intermediate (IU), the native-like compact intermediate (IC) and finally the native structure (N). (d) Landscape illustrating the Mg2+-dependence of folding in dilute (blue) and crowded (red) solutions. Both RNAs fold at lower [Mg2+] in a crowded solution, but the L9 mutation changes the relative stabilities of IU and IC. Midpoints of the U→IU and IU→IC transitions were calculated from three-state fits to SAXS data. Midpoints of the IC→N transition were obtained from the relative ribozyme activity (25 s). The degree of foldedness represented by the y-axis is arbitrary.
For this study, we measured the destabilization of the RNA structure owing to single base substitutions that disrupt a conserved triple helix (TH) in the center of the ribozyme core, or that disrupt docking of two peripheral L2 and L9 tetraloops with their respective helical receptor in P8/P8a and P5/P5a (Figure 1a and b). These comprise three of the main tertiary interaction motifs that stabilize the Azoarcus ribozyme. As previously demonstrated (10), ribozymes carrying single, double or triple combinations of these mutations were able to fold into a compact tertiary structure (Supplementary Figure S1) and were catalytically active in 15 mM MgCl2 (Supplementary Figure S2). Because these mutations do not prevent the ribozyme from folding in high Mg2+, we could evaluate how each mutation perturbed the equilibria between the U, IC and N conformational ensembles.
Crowding compensates for missing RNA tertiary interactions
To test how much molecular crowding similar to that in the cell's interior can compensate for the loss of RNA tertiary interactions, we compared the folding of mutant and WT Azoarcus ribozymes in the presence or absence of PEG with an average 1000 Da molecular weight. PEG stabilizes the folded ribozyme primarily by excluded volume effects (18,20), and is advantageous for SAXS experiments because of its low X-ray scattering contrast with water.
We used SAXS to measure the effect of molecular crowding on the initial stage of folding in which the double helices assemble into a compact structure. As expected, all of the RNAs went through a cooperative folding transition from the unfolded ensemble (U) in no Mg2+ (Rg ∼72 Å) to the compact IC intermediate in 1 mM Mg2+ (Rg = 32 Å; dark blue, Figure 2a). We also detected a decrease in Rg at very low Mg2+ that corresponds to formation of the extended intermediate, IU. The change in Rg2 was fit to a previously validated three-state model for folding (27) to obtain the equilibrium populations of U, IU and IC at each Mg2+ concentration and the free energy gaps between these states (Supplementary Table S1).
Figure 2.
Molecular crowding overcomes destabilizing RNA mutations. Folding transitions from the unfolded state (U) to the compact native-like intermediate (IC) were measured by SAXS at different [Mg2+]. Filled symbols indicate experimental data on the WT ribozyme; lines show the best fit of the data to a 3-state partition function (Equation 1). Light lines indicate fitted transitions for one of the seven possible single, double or triple L2, TH and L9 mutants; see data in Supplementary Figure S3. No PEG, blue/light blue; 18% PEG1000, red/pink. All experiments were done with 0.4 mg/ml RNA equilibrated in 20 mM Tris–HCl, pH 7.5 plus the desired [MgCl2] at 37°C. (b) Normalized P(r) of the folded RNAs in 2 mM MgCl2 in water or in 18% PEG1000. Colors as in (a).
In dilute buffer, the mutant ribozymes required more Mg2+ to fold than the WT ribozyme, consistent with a less stable tertiary structure (compare dark and light blue curves in Figure 2a; see Supplementary Figure S3 for further data). The mutants also formed less compact structures than the WT ribozyme in 2–5 mM MgCl2, as indicated by a greater Rg and more asymmetric pair-distance distribution function P(r) (Figure 2b). When 18% PEG1000 was added to the solution, all of the RNAs were stabilized. They folded at lower Mg2+ concentrations (red; Figure 2a) and had more symmetric P(r) functions (Figure 2b) in 2 mM MgCl2, as previously observed for the WT Azoarcus ribozyme (20).
Importantly, PEG stabilized the mutant ribozymes more than the WT ribozyme, so that [Mg2+]1/2 for folding the WT ribozyme was near the average for the group (compare red and pink lines in Figure 2a). This result showed that the stabilization arising from a crowded environment compensates the loss of RNA tertiary interactions, equalizing the folding of the WT and mutant RNAs. This compensation was also apparent from the similar P(r) functions of the WT and mutant RNAs folded in 18% PEG (Figure 2b).
Crowding does not change the folding pathway
We next asked whether the Azoarcus ribozyme forms different intermediate structures in the crowded and dilute solutions, and whether this accounts for the stabilization by PEG. We used native PAGE to separate native-like and non-native intermediates, which have different electrophoretic mobility because of their different hydrodynamic shapes (see SI text). RNAs were folded with or without 18% PEG in the same conditions used for SAXS experiments (0.4 mg/ml RNA) and loaded on a native 8% polyacrylamide gel containing 3 mM MgCl2 at 4°C.
Only the folded (IC + N) and the unfolded (U) conformations were observed for the WT ribozyme (22). We measured the folded and unfolded populations using a model that accounts for the fraction of RNA which folds as the sample is loaded into the gel (Supplementary Figure S1b). The increase in the folded population with Mg2+ concentration coincided with the main folding transition measured by SAXS (Supplementary Figure S3a). This transition shifted to a lower Mg2+ concentration in 18% PEG, in agreement with PEG's stabilization of compact structures.
In contrast to the WT ribozyme, the mutant ribozymes either migrated more slowly in the gel or formed additional bands corresponding to the extended IU intermediate and other non-native structures structures (Figure 3; see Supplementary Figure S1). For each ribozyme sequence, PEG increased the population of folded RNA but did not change the types of intermediates formed (Supplementary Figure S3b–h). We also observed that 18% PEG favored the IU state over the less compact U state, in low Mg2+ (Figure 3b). This is consistent with the idea that crowding stabilizes the folded RNA by restricting the volume (and conformational space) available to the unfolded chains (18). These results also agreed with the suggestion that ions, crowders and other co-solutes change the relative stabilities of different conformations but not the folding pathways encoded by the RNA sequence (33–35).
Figure 3.
Crowding stabilizes native and non-native intermediates. Folding of the L9 mutant was analyzed by native PAGE in (a) dilute solution and (b) 18% PEG1000. Top, native PAGE was performed in the same experimental condition as SAXS. Bands were assigned as described in SI Methods. The two high mobility bands have the same response to [Mg2+] and were grouped. Bottom, populations of each conformer in solution calculated from native PAGE (solid lines) and the change in Rg (dashed lines). Some of the unfolded RNA in solution folds to IU or IC when the sample contacts the MgCl2 in the gel running buffer. U, unfolded RNA (red); IU, extended folding intermediates (green); IC, compact intermediates (blue). Fitted parameters are the standard deviation from 2–3 trials. Data for L9 and other ribozymes are shown in Supplementary Figure S3.
Molecular crowding stabilizes the native state
The results above showed that crowder molecules such as PEG offset the destabilizing effects of our ribozyme mutations, so that the mutant and WT RNAs form compact structures in comparable Mg2+ concentrations. To determine whether the folded RNAs are active, we measured the amount of native ribozyme at different Mg2+ concentrations using single-turnover RNA cleavage assays (36,37). In 15 mM MgCl2, almost half the substrate was cleaved within 13–25 s at 37°C (Supplementary Figures S4 and S5). This is close to the maximal activity of the Azoarcus ribozyme (Supplementary Figure S2) and reflects the internal equilibrium between substrate cleavage and ligation (38). The amount of product formed in 25 s increased sharply between 1 and 3 mM Mg2+ as the native ribozyme became more populated (Figure 4a).
Figure 4.
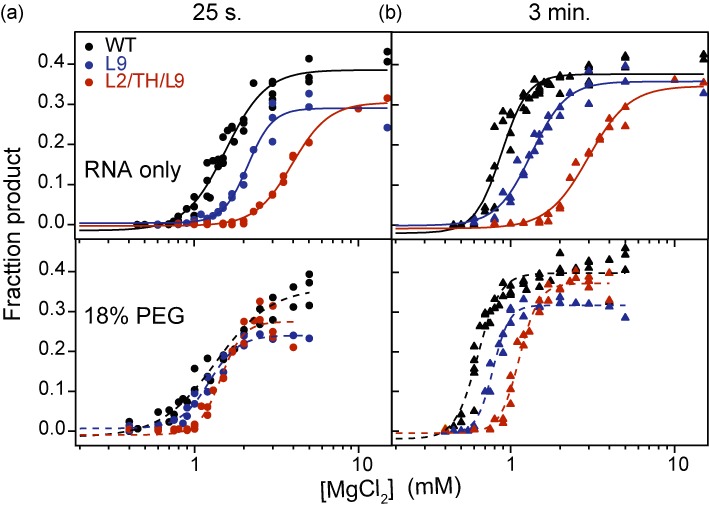
Crowding increases ribozyme activity. Formation of the native state (N) was measured in single-turnover activity assays (Supplementary Figure S4). The fraction of substrate cleaved versus [Mg2+] in three or more trials were fit to a two-state function. See Supplementary Figure S6 for further data on other mutants. (a) The product formed in 25 s indicates the fraction of native ribozyme at each [Mg2+]. The mutants and WT fold similarly in 18% PEG, but the mutants have lower maximum activity. (b) Product formed in 3 min indicates the total population competent to react. PEG only partially compensates for destabilizing mutations in this assay, indicating a greater fraction of misfolded RNAs.
The native states of the mutant ribozymes were less stable than the native WT ribozyme and required higher Mg2+ concentration to form, as expected (Figure 4a and Supplementary Figure S6). The addition of 18% PEG shifted the titration curves for the mutants to lower Mg2+, however, so that even the least active mutants (TH/L9 and L2/TH/L9) had the same [Mg2+]1/2 for forming the catalytically active native state as the WT ribozyme (Figure 4a and Supplementary Figure S6a). Therefore, a crowded environment not only stabilizes the native ribozyme (19,39–40) but the WT and mutant ribozymes experience a similar free energy difference between the IC and N states.
Crowding does not rescue misfolded ribozymes
Although PEG stabilized the compact (IC) and native (N) states, certain ribozymes such as those containing the L9 mutation had a lower maximum activity than the WT ribozyme (Supplementary Figure S2). This reduced catalytic activity was consistent with the presence of an additional non-native conformer (Figure 3b) and some aggregation of the L9 mutant above 3 mM Mg2+. PEG did not improve the maximum activity of such mutants, suggesting that molecular crowding cannot fully compensate for the loss of sequence-specific RNA interactions.
At low Mg2+ concentrations, the initial phase of the substrate cleavage reaction requires up to 3 min (Supplementary Figure S7) (19). This slow cleavage rate is presumably because near-native ribozyme–substrate complexes only occasionally fluctuate into the reactive structure when the Mg2+ concentration is too low to fully stabilize the ribozyme active site. The possibility of sampling suboptimal structures is supported by the observation that individual Tetrahymena group I ribozymes can react at different rates (41).
In the conditions tested here, the total fraction of ribozyme–substrate complexes capable of reaching the native state formed product within 3 min (Supplementary Figure S7). When this fraction of ‘refoldable’ RNA was compared at different [MgCl2], the mutant ribozymes appeared less stable than the WT ribozyme in either dilute or crowded solutions (Figure 4b and Supplementary Figure S6b). In other words, PEG was less able to improve the activity of incorrectly folded RNA molecules that required more time to react. This result agreed with the inability of PEG to raise the maximum activity of mutants that readily form alternative structures, such as L9 or the least stable L2/TH/L9 triple mutant (Supplementary Figure S2 and S3). From these results, we concluded that a crowded environment stabilizes the native ribozyme structure in low Mg2+, but cannot overcome the loss of folding specificity that arises when tertiary interaction motifs are mutated.
Free energy landscape for folding in crowded solutions
To quantify the thermodynamic compensation for destabilizing RNA mutations, we compared the free energy change for forming the compact IC intermediate or the native (active) state in dilute solution and in 18% PEG. Individual Mg2+ titrations were fit to two or three-state folding models to obtain the populations of U, IU and IC (SAXS) and N (activity). These populations were used to calculate the standard free energy change (ΔG) associated with the U to IC and IC to N folding transitions (SI Methods) (10). The energy perturbations caused by the mutations, ΔΔG = ΔGmut – ΔGWT, were evaluated at the MgCl2 concentration corresponding to 50% saturation of the WT folding transition in each solution condition. This choice of reference condition avoids extrapolation to Mg2+ concentrations where the assumptions underlying the thermodynamic model are less valid (42).
All of the mutations destabilized the compact IC intermediate in dilute buffer, resulting in positive values for ΔΔGU→IC (blue bars, Figure 5a; Supplementary Table S1). These energy perturbations followed the same order as reported previously (10), but with a smaller magnitude. In 18% PEG, however, several mutants had a similar folding free energy as the WT ribozyme, and L2 and L2/TH even appeared more stable than the WT RNA (red bars; Figure 5a).
Figure 5.
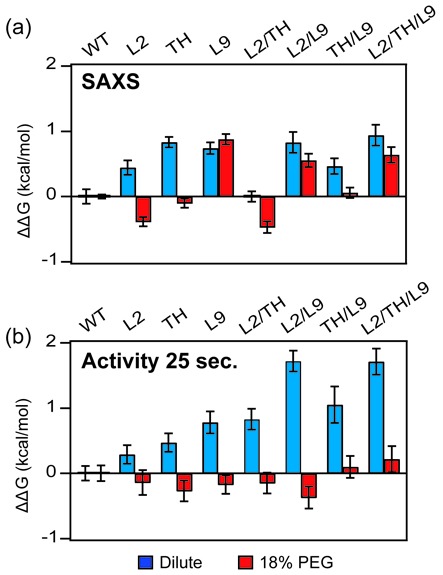
Crowding stabilizes the folded ribozymes. Relative folding free energies ΔΔG = ΔGmut – ΔGWT for (a) U→IC transition measured by SAXS and (b) the IC→N transition measured by activity (25 s). The folding free energies were evaluated at [Mg2+]1/2 for the WT ribozyme in each solution condition: 0.47 mM MgCl2 (SAXS, dilute), 0.28 mM (SAXS, 18% PEG), 1.56 mM (activity, dilute) and 1.34 mM MgCl2 (activity, 18% PEG). Error bars depict the standard deviation calculated from 10 000 resampling of fit residuals. The free energy obtained from activity assay is normalized to maximum activity of each RNA (Supplementary Figure S2). The [Mg2+]1/2 values of the WT and single mutant ribozymes reported here are lower than previously reported (10), owing to the 25 s quench used here, versus <20 s quench used previously. The longer reaction interval allows a portion of semi-native intermediates to refold, boosting the relative activity of the WT ribozyme and making the relative effects of multiple mutations more apparent (Supplementary Figure S7).
PEG also lowered the standard free energy change of the Ic→N transition calculated from the amount of product formed after 25 s. In dilute solution, the native state became progressively less stable (ΔΔGIC→N > 0) as more tertiary interaction motifs were mutated. By contrast, the folding free energies of all ribozymes were very similar in 18% PEG (Figure 5b), again demonstrating that crowded environments can compensate for the loss of tertiary interactions outside the active site. As discussed above, crowding does not rescue near-native or non-native structures that require more time to fluctuate into the active state and react (Supplementary Figure S8).
DISCUSSION
Molecular crowding favors the compact conformation of the Azoarcus ribozyme, allowing WT and mutant RNAs to fold into native-like structures in low physiological Mg2+ concentrations. The similar folding free energies of the mutants tested here suggests that non-coding RNA structures can tolerate sequence changes during evolution better than predicted by in vitro folding assays. For example, some destabilizing mutations in the td group I ribozyme harmed intracellular self-splicing much less than expected (12). Recent work on the CPEB3 ribozyme suggests that stabilization from molecular crowding is particularly important for marginally stable RNA structures (40), but is observed for a variety of RNA structures (39,43–44).
We previously found that the cooperative formation of tertiary interactions during helix assembly makes folding more accurate because only native-like structures enjoy the additional drop in free energy that arises when multiple tertiary interactions form simultaneously (10). This cooperativity is most important when solvent conditions (such as low [Mg2+]) barely stabilize the native state (45,46). By contrast, strongly stabilizing conditions diminish the selectivity of folding.
By non-selectively tilting the free energy landscape toward compact structures, molecular crowding allows the WT and mutant ribozymes an equal chance of accessing the native state without improving the precision of self-assembly. Instead, a crowded environment produces a broad basin of attraction that favors a variety of compact structures. Our SAXS, native PAGE and activity measurements all indicate that the mutated ribozymes sample folded conformations that are inactive in substrate cleavage assays, and this less-active fraction increases with the number of mutations with or without PEG (Supplementary Figure S6). Thus, our results show that molecular crowding does not overcome the loss of folding specificity that arises when one or more tertiary interaction motifs are lost.
One prediction of these results is that non-coding RNAs that act catalytically in the cell will be more sensitive to destabilizing mutations than non-coding RNAs that turnover only once, such as the Azoarcus group I ribozyme. Estimates of intracellular splicing rates suggest that a precursor RNA has 1–2 min to fold and react before it is degraded (47–50), so that even suboptimal sequences may succeed by this measure. By contrast, sequence changes that destabilize multiple turnover ribozymes such as RNase P (51) must presumably be accompanied by the evolution of other RNA or RNA–protein interactions that specifically reinforce the optimal RNA structure. Since other polymers stabilize the folded RNA as well as PEG does, generic excluded volume effects might have even boosted the functions of RNA catalysts in a pre-biotic world.
SUPPLEMENTARY DATA
Supplementary Data are available at NAR Online.
Acknowledgments
The authors thank Rita Graceffa and Srinivas Chakravarthy (APS ID18 BioCAT) for assistance with SAXS experiments. Use of the Advanced Photon Source, Argonne National Laboratory was supported by the U.S. Department of Energy, Office of Science, Office of Basic Energy Sciences, under Contracts Nos. DE-AC02-06CH11357.
Footnotes
Present addresses:
Hui-Ting Lee, Department of Bioengineering, University of Illinois, Urbana Champaign, Urbana IL 61801, USA.
Duncan Kilburn, Circulomics Inc., 810 Wyman Park Dr., Unit G01, Baltimore, MD 21211, USA.
Reza Behrouzi, Department of Cell Biology, Harvard Medical School, Boston, MA 02115-5730, USA.
FUNDING
National Institute of General Medical Sciences [R01 GM60819]; National Institute of Standards and Technology [70NANB10H257]. The open access publication charge for this paper has been waived by Oxford University Press—NAR Editorial Board members are entitled to one free paper per year in recognition of their work on behalf of the journal.
Conflict of interest statement. None declared.
REFERENCES
- 1.Holbrook S.R. Structural principles from large RNAs. Annu. Rev. Biophys. 2008;37:445–464. doi: 10.1146/annurev.biophys.36.040306.132755. [DOI] [PubMed] [Google Scholar]
- 2.Butcher S.E., Pyle A.M. The molecular interactions that stabilize RNA tertiary structure: RNA motifs, patterns, and networks. Acc. Chem. Res. 2011;44:1302–1311. doi: 10.1021/ar200098t. [DOI] [PubMed] [Google Scholar]
- 3.Cruz J.A., Westhof E. The dynamic landscapes of RNA architecture. Cell. 2009;136:604–609. doi: 10.1016/j.cell.2009.02.003. [DOI] [PubMed] [Google Scholar]
- 4.Baird N.J., Srividya N., Krasilnikov A.S., Mondragon A., Sosnick T.R., Pan T. Structural basis for altering the stability of homologous RNAs from a mesophilic and a thermophilic bacterium. RNA. 2006;12:598–606. doi: 10.1261/rna.2186506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Jaeger L., Michel F., Westhof E. Involvement of a GNRA tetraloop in long-range RNA tertiary interactions. J. Mol. Biol. 1994;236:1271–1276. doi: 10.1016/0022-2836(94)90055-8. [DOI] [PubMed] [Google Scholar]
- 6.Silverman S.K., Cech T.R. Energetics and cooperativity of tertiary hydrogen bonds in RNA structure. Biochemistry. 1999;38:8691–8702. doi: 10.1021/bi9906118. [DOI] [PubMed] [Google Scholar]
- 7.Doherty E.A., Herschlag D., Doudna J.A. Assembly of an exceptionally stable RNA tertiary interface in a group I ribozyme. Biochemistry. 1999;38:2982–2990. doi: 10.1021/bi982113p. [DOI] [PubMed] [Google Scholar]
- 8.Costa M., Michel F. Frequent use of the same tertiary motif by self-folding RNAs. EMBO J. 1995;14:1276–1285. doi: 10.1002/j.1460-2075.1995.tb07111.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Shi X., Bisaria N., Benz-Moy T.L., Bonilla S., Pavlichin D.S., Herschlag D. Roles of long-range tertiary interactions in limiting dynamics of the Tetrahymena group I ribozyme. J. Am. Chem. Soc. 2014;136:6643–6648. doi: 10.1021/ja413033d. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Behrouzi R., Roh J.H., Kilburn D., Briber R.M., Woodson S.A. Cooperative tertiary interaction network guides RNA folding. Cell. 2012;149:348–357. doi: 10.1016/j.cell.2012.01.057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Wilson T.J., Zhao Z.Y., Maxwell K., Kontogiannis L., Lilley D.M. Importance of specific nucleotides in the folding of the natural form of the hairpin ribozyme. Biochemistry. 2001;40:2291–2302. doi: 10.1021/bi002644p. [DOI] [PubMed] [Google Scholar]
- 12.Brion P., Michel F., Schroeder R., Westhof E. Analysis of the cooperative thermal unfolding of the td intron of bacteriophage T4. Nucleic Acids Res. 1999;27:2494–2502. doi: 10.1093/nar/27.12.2494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.McDowell S.E., Jun J.M., Walter N.G. Long-range tertiary interactions in single hammerhead ribozymes bias motional sampling toward catalytically active conformations. RNA. 2010;16:2414–2426. doi: 10.1261/rna.1829110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Tanner M.A., Anderson E.M., Gutell R.R., Cech T.R. Mutagenesis and comparative sequence analysis of a base triple joining the two domains of group I ribozymes. RNA. 1997;3:1037–1051. [PMC free article] [PubMed] [Google Scholar]
- 15.Nikolcheva T., Woodson S.A. Facilitation of group I splicing in vivo: misfolding of the Tetrahymena IVS and the role of ribosomal RNA exons. J. Mol. Biol. 1999;292:557–567. doi: 10.1006/jmbi.1999.3083. [DOI] [PubMed] [Google Scholar]
- 16.Donahue C.P., Yadava R.S., Nesbitt S.M., Fedor M.J. The kinetic mechanism of the hairpin ribozyme in vivo: influence of RNA helix stability on intracellular cleavage kinetics. J. Mol. Biol. 2000;295:693–707. doi: 10.1006/jmbi.1999.3380. [DOI] [PubMed] [Google Scholar]
- 17.Ellis R.J. Macromolecular crowding: obvious but underappreciated. Trends Biochem. Sci. 2001;26:597–604. doi: 10.1016/s0968-0004(01)01938-7. [DOI] [PubMed] [Google Scholar]
- 18.Kilburn D., Roh J.H., Behrouzi R., Briber R.M., Woodson S.A. Crowders perturb the entropy of RNA energy landscapes to favor folding. J. Am. Chem. Soc. 2013;135:10055–10063. doi: 10.1021/ja4030098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Desai R., Kilburn D., Lee H.T., Woodson S.A. Increased ribozyme activity in crowded solutions. J. Biol. Chem. 2014;289:2972–2977. doi: 10.1074/jbc.M113.527861. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kilburn D., Roh J.H., Guo L., Briber R.M., Woodson S.A. Molecular crowding stabilizes folded RNA structure by the excluded volume effect. J. Am. Chem. Soc. 2010;132:8690–8696. doi: 10.1021/ja101500g. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Svergun D.I. Determination of the regularization parameter in indirect- transform methods using perceptual criteria. J. Appl. Crystallogr. 1992;25:495–503. [Google Scholar]
- 22.Rangan P., Masquida B., Westhof E., Woodson S.A. Assembly of core helices and rapid tertiary folding of a small bacterial group I ribozyme. Proc. Natl. Acad. Sci. U.S.A. 2003;100:1574–1579. doi: 10.1073/pnas.0337743100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Chauhan S., Behrouzi R., Rangan P., Woodson S.A. Structural rearrangements linked to global folding pathways of the Azoarcus group I ribozyme. J. Mol. Biol. 2009;386:1167–1178. doi: 10.1016/j.jmb.2008.12.075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Fang X., Littrell K., Yang X.J., Henderson S.J., Siefert S., Thiyagarajan P., Pan T., Sosnick T.R. Mg2+-dependent compaction and folding of yeast tRNAPhe and the catalytic domain of the B. subtilis RNase P RNA determined by small-angle X-ray scattering. Biochemistry. 2000;39:11107–11113. doi: 10.1021/bi000724n. [DOI] [PubMed] [Google Scholar]
- 25.Chauhan S., Caliskan G., Briber R.M., Perez-Salas U., Rangan P., Thirumalai D., Woodson S.A. RNA tertiary interactions mediate native collapse of a bacterial group I ribozyme. J. Mol. Biol. 2005;353:1199–1209. doi: 10.1016/j.jmb.2005.09.015. [DOI] [PubMed] [Google Scholar]
- 26.Chauhan S., Woodson S.A. Tertiary interactions determine the accuracy of RNA folding. J. Am. Chem. Soc. 2008;130:1296–1303. doi: 10.1021/ja076166i. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Moghaddam S., Caliskan G., Chauhan S., Hyeon C., Briber R.M., Thirumalai D., Woodson S.A. Metal ion dependence of cooperative collapse transitions in RNA. J. Mol. Biol. 2009;393:753–764. doi: 10.1016/j.jmb.2009.08.044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Tanner M., Cech T. Activity and thermostability of the small self-splicing group I intron in the pre-tRNA(lle) of the purple bacterium Azoarcus. RNA. 1996;2:74–83. [PMC free article] [PubMed] [Google Scholar]
- 29.Roh J.H., Guo L., Kilburn J.D., Briber R.M., Irving T., Woodson S.A. Multistage collapse of a bacterial ribozyme observed by time-resolved small-angle X-ray scattering. J. Am. Chem. Soc. 2010;132:10148–10154. doi: 10.1021/ja103867p. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Perez-Salas U.A., Rangan P., Krueger S., Briber R.M., Thirumalai D., Woodson S.A. Compaction of a bacterial group I ribozyme coincides with the assembly of core helices. Biochemistry. 2004;43:1746–1753. doi: 10.1021/bi035642o. [DOI] [PubMed] [Google Scholar]
- 31.Rangan P., Woodson S.A. Structural requirement for Mg2+ binding in the group I intron core. J. Mol. Biol. 2003;329:229–238. doi: 10.1016/s0022-2836(03)00430-3. [DOI] [PubMed] [Google Scholar]
- 32.Rangan P., Masquida B., Westhof E., Woodson S.A. Architecture and folding mechanism of the Azoarcus Group I Pre-tRNA. J. Mol. Biol. 2004;339:41–51. doi: 10.1016/j.jmb.2004.03.059. [DOI] [PubMed] [Google Scholar]
- 33.Baird N.J., Gong H., Zaheer S.S., Freed K.F., Pan T., Sosnick T.R. Extended structures in RNA folding intermediates are due to nonnative interactions rather than electrostatic repulsion. J. Mol. Biol. 2010;397:1298–1306. doi: 10.1016/j.jmb.2010.02.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Laederach A., Shcherbakova I., Jonikas M.A., Altman R.B., Brenowitz M. Distinct contribution of electrostatics, initial conformational ensemble, and macromolecular stability in RNA folding. Proc. Natl. Acad. Sci. U.S.A. 2007;104:7045–7050. doi: 10.1073/pnas.0608765104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Rook M.S., Treiber D.K., Williamson J.R. Fast folding mutants of the Tetrahymena group I ribozyme reveal a rugged folding energy landscape. J. Mol. Biol. 1998;281:609–620. doi: 10.1006/jmbi.1998.1960. [DOI] [PubMed] [Google Scholar]
- 36.Herschlag D., Cech T.R. Catalysis of RNA cleavage by the Tetrahymena thermophila ribozyme. 1. Kinetic description of the reaction of an RNA substrate complementary to the active site. Biochemistry. 1990;29:10159–10171. doi: 10.1021/bi00496a003. [DOI] [PubMed] [Google Scholar]
- 37.Sinan S., Yuan X., Russell R. The Azoarcus group I intron ribozyme misfolds and is accelerated for refolding by ATP-dependent RNA chaperone proteins. J. Biol. Chem. 2011;286:37304–37312. doi: 10.1074/jbc.M111.287706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Wan Y., Mitchell D., 3rd, Russell R. Catalytic activity as a probe of native RNA folding. Methods Enzymol. 2009;468:195–218. doi: 10.1016/S0076-6879(09)68010-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Nakano S., Karimata H.T., Kitagawa Y., Sugimoto N. Facilitation of RNA enzyme activity in the molecular crowding media of cosolutes. J. Am. Chem. Soc. 2009;131:16881–16888. doi: 10.1021/ja9066628. [DOI] [PubMed] [Google Scholar]
- 40.Strulson C.A., Yennawar N.H., Rambo R.P., Bevilacqua P.C. Molecular crowding favors reactivity of a human ribozyme under physiological ionic conditions. Biochemistry. 2013;52:8187–8197. doi: 10.1021/bi400816s. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Solomatin S.V., Greenfeld M., Chu S., Herschlag D. Multiple native states reveal persistent ruggedness of an RNA folding landscape. Nature. 2010;463:681–684. doi: 10.1038/nature08717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Leipply D., Draper D.E. Dependence of RNA tertiary structural stability on Mg2 +concentration: interpretation of the Hill equation and coefficient. Biochemistry. 2010;49:1843–1853. doi: 10.1021/bi902036j. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Paudel B.P., Rueda D. Molecular Crowding Accelerates Ribozyme Docking and Catalysis. J. Am. Chem. Soc. 2014;136:16700–16703. doi: 10.1021/ja5073146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Dupuis N.F., Holmstrom E.D., Nesbitt D.J. Molecular-crowding effects on single-molecule RNA folding/unfolding thermodynamics and kinetics. Proc. Natl. Acad. Sci. U.S.A. 2014;111:8464–8469. doi: 10.1073/pnas.1316039111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Rook M.S., Treiber D.K., Williamson J.R. An optimal Mg(2+) concentration for kinetic folding of the tetrahymena ribozyme. Proc. Natl. Acad. Sci. U.S.A. 1999;96:12471–12476. doi: 10.1073/pnas.96.22.12471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Thirumalai D., Woodson S.A. Maximizing RNA folding rates: a balancing act. RNA. 2000;6:790–794. doi: 10.1017/s1355838200000522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Koduvayur S.P., Woodson S.A. Intracellular folding of the Tetrahymena group I intron depends on exon sequence and promoter choice. RNA. 2004;10:1526–1532. doi: 10.1261/rna.7880404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Roberts G.C., Gooding C., Mak H.Y., Proudfoot N.J., Smith C.W. Co-transcriptional commitment to alternative splice site selection. Nucleic Acids Res. 1998;26:5568–5572. doi: 10.1093/nar/26.24.5568. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Donahue C.P., Fedor M.J. Kinetics of hairpin ribozyme cleavage in yeast. RNA. 1997;3:961–973. [PMC free article] [PubMed] [Google Scholar]
- 50.Hilleren P.J., Parker R. Cytoplasmic degradation of splice-defective pre-mRNAs and intermediates. Mol. cell. 2003;12:1453–1465. doi: 10.1016/s1097-2765(03)00488-x. [DOI] [PubMed] [Google Scholar]
- 51.Walker S.C., Engelke D.R. Ribonuclease P: the evolution of an ancient RNA enzyme. Crit. Rev. Biochem. Mol. Biol. 2006;41:77–102. doi: 10.1080/10409230600602634. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


