Abstract
The severe fever with thrombocytopenia syndrome (SFTS), caused by a novel Phlebovirus in the Bunyaviridae family named SFTS virus (SFTSV), is an emerging hemorrhagic fever with a wide distribution and high case-fatality rate. Neither effective treatment nor vaccines are available to treat and prevent this disease to date. It was recently reported that SFTSV nonstructural protein in S segment (SFTSV/NSs) functioned as the interferon (IFN) antagonist targeting for suppressing host's innate immunity. This study was designed to investigate the potential of recombinant SFTSV (rSFTSV)/NSs protein for inducing anti-NSs antibodies by pre-exposure vaccination to block SFTSV/NSs in the SFTSV-infected C57BL/6J mice. All mice in the rSFTSV/NSs-vaccinated group, negative control group, and blank control group survived with no visible clinical abnormities throughout the experiment, except for their sacrifice for sampling at each observation point. However, unexpectedly, a negative effect on the bodyweight of rSFTSV/NSs-vaccinated mice was observed after 21 days postinoculation. Pre-exposure vaccination with rSFTSV/NSs did not accelerate virus removal in mice though high titer of anti-NSs antibodies and elevated IFN-γ were detected in sera. Before virus challenge, the rSFTSV/NSs-vaccinated mice and negative control mice had a larger amount of platelets (PLT) than the blank control mice, which indicated that Freund's adjuvants could stimulate PLT production. In the aspect of cytokines, the rSFTSV/NSs-vaccinated mice had a 5- to 10-fold increase in interleukin (IL)-2, IL-5, IL-6, IFN-γ, and tumor necrosis factor-α, which probably just had a negative effect on the bodyweight of mice. In general, therefore, previous vaccination with rSFTSV/NSs did not accelerate virus clearance in the SFTSV-infected mice.
Introduction
The severe fever with thrombocytopenia syndrome virus (SFTSV), a new member of the genus Phlebovirus in the Bunyaviridae family, was first isolated by Chinese scientists from the patients who presented primarily with fever, thrombocytopenia, leukocytopenia, gastrointestinal symptoms, and multiorgans dysfunction and reported in 2011 (14,22). SFTS cases have been reported in Henan, Hubei, Shandong, Jiangsu, Anhui, Liaoning, and other provinces, expanding from the initial 6 provinces in 2010 to at least 15 provinces/municipalities in 2013, presenting with a highly sporadic pattern in mainland China (11,14, 22, 23; Liu SL, 2011, Institute of infectious diseases prevention of Zhejiang provincial center for disease control and prevention. Practice and advances in the novel bunyavirus—training course on insect-borne infectious diseases and new imported infectious diseases technology.). In 2011, 571 SFTS cases were reported among whom 59 were fatal. In 2012, the reported cases in the nationwide increased to 1,476 and 70 cases were fatal (3; Liu SL, 2011, Institute of infectious diseases prevention of Zhejiang provincial center for disease control and prevention. Practice and advances in the novel bunyavirus—training course on insect-borne infectious diseases and new imported infectious diseases technology.). Broad distribution, high incidence, and high case fatality have made SFTSV-caused disease one of the most important zoonosis in China.
It was reported that porcine reproductive and respiratory syndrome virus, yellow fever virus (YFV), and rotavirus could express nonstructural proteins (nonstructural protein in S segment [NSs]) after infecting their mammalian host to interrupt the host's interferon (IFN) expression by binding with interferon regulating factor 3 (IRF3) and blocking its activation, finally to regulate and evade host's innate immune attack (1,2,21). Researchers have recently also found that SFTSV NSs had a similar effect on the viral multiplication, through the regulation of and escape from the host's innate immunity (15,16,20). Based on the physician's experience in clinical practice of SFTS treatment, it is unacceptable to give extrinsic IFN to SFTS patients due to the serious side effects of IFN administration, for example, severe fever, leukocytopenia, and thrombocytopenia, which are just the typical symptoms caused by SFTSV infection. Logically, we hypothesized that by blocking SFTSV NSs and its binding to host's IRF3, the IFN expression and innate immunity of host can be fully mobilized to get rid of the virus, therefore eventually to improve the disease outcome.
Previous studies have demonstrated inspiring results about the role of NS in eliciting protection against virus infection. In Schlesinger et al.'s study, four out of five rhesus monkeys acquiring subcutaneous immunization with YFV NS1 survived after being inoculated with a lethal dose of YFV, in contrast, all the four rhesus monkeys in control group died, which suggested protective role of immunity against YFV NS1 in protection to YFV infection and pathogenesis (17). In another study, immunization with recombinant vaccinia virus expressing authentic dengue virus nonstructural protein NS1 provided 63–67% or even 100% of protection against Dengue virus infection in mice (5). Vaccination with NS1 DNA vaccine of Japanese encephalitis virus yielded a survival rate of approximately 90% in mice, while only 40% in the control mice were vaccinated with the empty plasmid (10).
Enlightened by results from the viral hemorrhage fever virus mentioned earlier, the current research was designed to investigate the potential protective capability of pre-exposure vaccination with prokaryotic-expressed recombinant SFTSV (rSFTSV)/NSs protein to promote virus clearance in SFTSV-infected C57BL/6J mice, which had been used as an animal model of SFTS in a previous study (7).
Materials and Methods
Cloning and expression of SFTSV/NSs
The strain Phlebovirus WCH/97/HN/China/2011 of SFTSV (9) isolated from the serum of an SFTS patient in 2011 was used. Virus propagation was carried out with Vero E6 cells cultured in RPMI 1640 medium containing 10% fetal calf serum (Gibco) (7). Virus titer was determined by a focus forming assay on Vero E6 cells (12). SFTSV RNA genome was isolated from SFTSV virus stocks using the TIANamp Virus DNA/RNA Kit [Tiangen Biotech (Beijing) Co., Ltd.] according to the manufacturer's instructions. The first-strand cDNA was synthesized using the SuperScript® III First-Strand Synthesis System (Invitrogen™) according to the manufacturer's instructions. A pair of primers containing the EcoRI and XhoI restriction sites (the underlined bases) (the forward primer: 5′-CCGGAATTCGAAACCA TGTCGCTGAGCA-3′ and the reverse primer: 5′-CCG CTCGAGTGCTGCTGCTTACAGAAACCT-3′) were designed to amplify the open reading frame of SFTSV/NSs, which yielded a DNA fragment of 985 bp. The 50 μL PCR mixture consisted of 4.0 μL of cDNA as the template, 0.8 μM primers, and TaKaRa Ex Taq® DNA polymerase [TaKaRa Biotech (Dalian) Co., Ltd.] according to the manufacturer's instructions. The thermal cycling conditions were as follows: an initial denaturation at 94°C for 5 min followed by 15 cycles at 94°C for 30 sec, 56°C for 30 sec, and 72°C for 1.5 min, and further 30 cycles at 94°C for 30 sec, 68°C for 30 sec, and 72°C for 1.5 min with a final extension at 72°C for 10 min, which was performed on an Applied Biosystems GeneAmp PCR System 9700. The amplified products were identified on a 2% ethidium bromide-stained agarose gel and recovered using the TIANgel Midi Purification Kit [Tiangen Biotech (Beijing) Co., Ltd.].
The purified PCR products were digested with EcoRI and XhoI (FastDigest enzymes; Thermo Fisher Scientific, Inc.) and then ligated into pET-28a(+) plasmid DNA (Novagen), which was digested with the same restriction enzymes. After ligation reaction using T4 DNA Ligase (Thermo Fisher Scientific, Inc.), the reaction mixture containing the rSFTSV/NSs-pET28a (+) plasmid was transformed into the competent cells Escherichia coli BL21 (DE3) (Novagen). The positive clones were screened by kanamycin resistance selection and colony PCR method, and were finally sequenced to confirm absence of any mutations at the amino-acid residue level.
The expression of recombinant protein SFTSV/NSs was induced at 37°C by the addition of 1 mM isopropyl-β-thiogalactopyranoside into the culture medium when its OD600 reached ∼0.6, and then the mixture was shaken at 250 rpm and 37°C for further 4 h. Bacteria cells were collected, the expression pattern of the target rSFTSV/NSs protein was determined with 6× His tag by SDS-PAGE, and the rSFTSV/NSs protein was finally isolated and purified by Ni2+-NTA agarose affinity using the Ni-NTA Sefinose™ Kit (Bio Basic, Inc.) according to the manufacturer's instructions (Fig. 1).
FIG. 1.
Isopropyl-β-thiogalactopyranoside-induced expression and affinity purification of recombinant SFTSV/NSs protein. M, protein molecular weight markers; UnI, un-induced sample; Ind, induced sample; InS, the soluble component of the induced sample after ultrasonic treatment; InI, the precipitate of the induced sample after ultrasonic treatment; FT, the flow-through liquid in the binding step; P, the purified SFTSV/NSs protein sample; SFTSV, severe fever with thrombocytopenia syndrome virus; SFTSV/NSs, SFTSV nonstructural protein in S segment.
Immunization and virus inoculation in mice
One hundred and five female C57BL/6J mice that were 3- to 4 weeks old were purchased from the Laboratory Animal Center of PLA Military Academy of Medical Sciences, and were randomly divided into three groups with 35 in each group. The following protocol was used for immunization and subsequent SFTSV inoculation. Briefly, 35 mice received abdominal subcutaneous immunization with 100 μg of the purified rSFTSV/NSs protein emulsified thoroughly with an equal volume of Freund's complete adjuvant (Sigma-Aldrich Co. LLC.) (day 1), followed by a boost with half dose of protein emulsified thoroughly with an equal volume of Freund's incomplete adjuvant (Sigma-Aldrich Co. LLC.) on day 14. Meanwhile, 35 mice as the negative controls received immunization with the corresponding volume of solvent (8 M urea solution) emulsified thoroughly with Freund's complete or incomplete adjuvant, and 35 mice as the blank controls received nothing.
Three mice in each group were bled from the tail on day 21 to assay the antisera titer against SFTSV/NSs by the standard enzyme-linked immunosorbent assay (ELISA) method (8). Briefly, 96-well ELISA plates (Corning) were coated with 100 μL of purified rSFTSV/NSs protein (5 μg/mL in 0.05 M carbonate buffer solution) at 4°C overnight. ELISA plates were washed with washing solution (WANTAI BioPharm), and blocked with 5% bovine serum albumin prepared with washing solution. Sera diluted from 1:20 to 1:2,560 in twofold steps were used as the primary antibodies, and the peroxidase-conjugated goat anti-mouse IgG (H+ L) (ZSGB-BIO) was used as the detection antibody. Color development was performed using A+B TMB color liquid (Wantai BioPharm), and the OD values were measured at 450 nm in an ELISA reader. DH82 cells inoculated on coverslips and cultured for 3 days after being infected with SFTSV were used to perform immunofluorescence assay to check the specificity of recognition of anti-NSs antibodies and NSs in cells according to the protocol described elsewhere (20). Meanwhile, cell culture medium was collected and used to detect the potential NSs secreted out of cells by ELISA; in-parallel cell lysate was used to detect the NSs in cells.
After that, the mice were challenged on day 26 by an intraperitoneal injection with 3×107 ffu of SFTSV.
Effect evaluation
Before challenge, seven mice from each group were sacrificed and their fresh blood, livers, spleens, and kidneys were collected as prechallenge samples. The rest of mice were observed for 28 days for signs of disease, and six or seven mice from each group were sacrificed for their blood, livers, spleens, and kidneys to be collected on 3, 14, 21, and 28 days postinoculation (DPI). The number of deaths from SFTSV infection was recorded daily. SFTSV loads, anti-SFTSV/NSs antibodies titer, hematological and biochemical parameters, cytokines concentration, and histopathological evaluations were examined to assess the protection effect of vaccination with SFTSV/NSs protein against SFTSV infection. Viral RNA from serum and organs (liver, spleen, and kidney) was extracted using the TIANamp Virus DNA/RNA Kit and RNAprep Pure Tissue Kit [Tiangen Biotech (Beijing) Co., Ltd.] according to the manufacturer's protocols, respectively. Virus loads were determined by TaqMan quantitative real-time RT-PCR method using One-Step PrimeScript™ RT-PCR Kit [TaKaRa Biotechnology (Dalian) Co., Ltd.] according to the method previously described (25), in which a serial dilution of recombinant plasmid with tested fragment inserted from 108 to 104 copies/mL in 10-fold steps was used as the standard.
Anti-SFTSV/NSs sera collected from immunized mice and diluted from 1:20 to 1:2,560 in twofold steps was analyzed by ELISA as described earlier. Positive samples were those whose A450 values exceeded twice the average A450 value of the 1:20 dilutions of the negative control sera from mice immunized with solvent (8 M urea) mixed with Freund's adjuvant (5). The blood cells and biochemical parameters included leukocyte count (WBC), red blood cell count (RBC), hemoglobin (HGB), lymphocyte count (LYM) and percentage (LYM%), neutrophil count (NEUT), and percentage (NEUT%); platelet count (PLT), alanine transaminase (ALT), aspartate aminotransferase (AST), lactate dehydrogenase (LDH), creatine kinase (CK), and blood urea nitrogen (BUN) were tested for assessing the function of heart, liver, and kidney. A multiplex-biometric immunoassay containing fluorescent microspheres conjugating with monoclonal antibodies specific for target cytokines was performed to test serum cytokine levels following the manufacturer's instructions (Bio-Plex Pro Mouse Cytokine Group I 7-Plex Assay; Bio-Rad) to identify differential cytokine responses among the three groups. The cytokines tested are as follows: interleukin (IL)-2, IL-4, IL-5, IL-6, IL-10, IFN-γ, and tumor necrosis factor (TNF)-α. In addition, the platelet-derived growth factor-BB (PDGF-BB) level was detected using the Mouse Platelet-Derived Growth Factor BB (PDGF-BB) ELISA Kit (R&D Systems, Inc.) according to the manufacturer's instruction.
Pathological lesions of liver, spleen, and kidney were examined by hematoxylin-eosin (H&E) staining of 5-μm-thick paraffin-embedded tissue sections.
Ethics statement
All experimental procedures with animals were in strict accordance with the recommendations in the Guide for the Care and Use of Laboratory Animals and approved by the Committee on the Ethics of Animal Experiments of the Academy of Military Medical Sciences.
Data analysis
Continuous variables were summarized as means±standard deviations (SD). The difference of the observation targets and the laboratory indexes among the three groups described earlier were analyzed by one-way analysis of variance (one-way ANOVA). p-Value of <0.05 was considered statistically significant. All analyses were performed using the IBM SPSS version 20.0.0 (IBM). Figures were generated by Microsoft® Excel 2013 (Microsoft Corporation) or GraphPad Prism 6.02 (GraphPad Software, Inc.).
Results
SFTSV/NSs vaccination caused a negative effect on bodyweight of mice
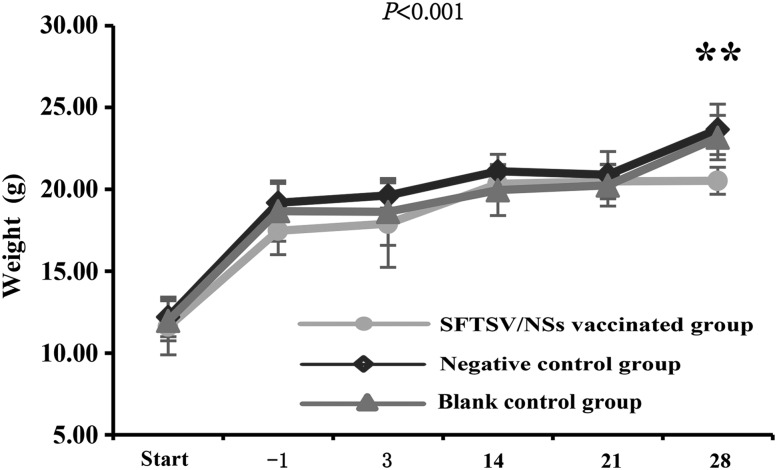
All mice of the three groups survived throughout the experiment with no visible medical abnormalities except being sacrificed for sample collection at each observation point. However, the SFTSV/NSs-vaccinated mice had significantly lower weight than those of the negative controls and the blank controls on 28 DPI (p<0.01) (Fig. 2).
FIG. 2.
Dynamic change of the bodyweight of mice. The sample sizes at each time point are 6 for the SFTSV/NSs-vaccinated group, and are 7 for both the negative control group and the blank group. The values are calculated and expressed as means±SD. p-Values were the result of multivariate ANOVA among the three groups. **p<0.01, which were the result of one-way ANOVA among the three groups at each observation point. SD, standard deviation. ANOVA, analysis of variance.
High titer of anti-SFTSV/NSs antibodies was induced
Sera obtained from mice at five observation points (day 7 after the second vaccination, 3, 14, 21, and 28 DPI with SFTSV to mice) were analyzed to determine rSFTSV/NSs-specific antibodies by ELISA method. It was shown that antibodies with a titer higher than 2,560 were induced in rSFTSV/NSs-vaccinated mice before challenge (Fig. 3A), which were detectable throughout the observation period after virus challenge (Fig. 3B–E). In contrast, no rSFTSV/NSs-specific antibodies were generated in the negative control group and blank control group throughout the observation period after virus inoculation (Fig. 3B–E), which suggested that antibodies against SFTSV/NSs were not independently induced in SFTSV-infected mice.
FIG. 3.
Analysis of mouse anti-SFTSV/NSs protein antibodies in sera by ELISA method. (A) Preinoculation, three mice from each group were exsanguinated from the tail and sera were isolated on day 7 after the second immunization (one boost). (B) 3 DPI; (C) 2 weeks postinoculation; (D) 3 weeks postinoculation; (E) 4 weeks postinoculation. For (B–E), 2 mL of individual sera specimens in each group were mixed, and their average sera titer was assayed by ELISA method. The sample size at each time point is 6 for the SFTSV/NSs-vaccinated group, 7 for both the negative control group (received immunizations with the corresponding volume of solvent (8 M urea solution) mixed and emulsified thoroughly with Freund's complete or incomplete adjuvant) and the blank group (received no treatment). DPI, days postinoculation; ELISA, enzyme-linked immunosorbent assay.
Pre-exposure vaccination with SFTSV/NSs did not promote virus clearance
The SFTSV quantification was determined on 3, 14, 21, and 28 DPI. As displayed in Figure 4, the mean virus loads initially evaluated on 3 DPI were relatively the highest, followed by a sharp decrease on 14 DPI, but maintained at a moderate level till the last observation on 28 DPI. At each observation point, relatively higher virus loads were detected in serum samples, followed by liver, spleen, and kidney on 3 DPI, by spleen, liver, and kidney on 14 DPI, and by spleen, kidney, and liver on 21 and 28 DPI. Generally, the three groups of mice displayed highly comparable virus loads at each time points throughout the observation period. However, the negative control mice had higher virus loads in serum samples than the other two groups on 3 DPI (p=0.003, Fig. 4A); the blank control mice had higher virus loads in spleen than the other two groups on 21 DPI (p=0.007, Fig. 4C); and the SFTSV/NSs-vaccinated mice had higher virus loads in spleen than the other two groups on 28 DPI (p=0.028, Fig. 4C). This indicated no biological significance in the fluctuation of virus loads among the three groups and suggested that vaccination with rSFTSV/NSs before virus exposure did not promote clearance of SFTSV in mice.
FIG. 4.
Quantification of SFTSV loads in serum, liver, spleen, and kidney of C57BL/6J mice by TaqMan One-step Real-time RT-PCR method. (A) serum samples, scaled by copies/mL; (B) liver samples, scaled by copies/mg; (C) spleen samples, scaled by copies/mg; (D) kidney samples, scaled by copies/mg. The sample size at each time point is 6 for the SFTSV/NSs-vaccinated group, and are both 7 for the negative control group and the blank group. The solid line in each group marks the position of the mean values of the index numbers.
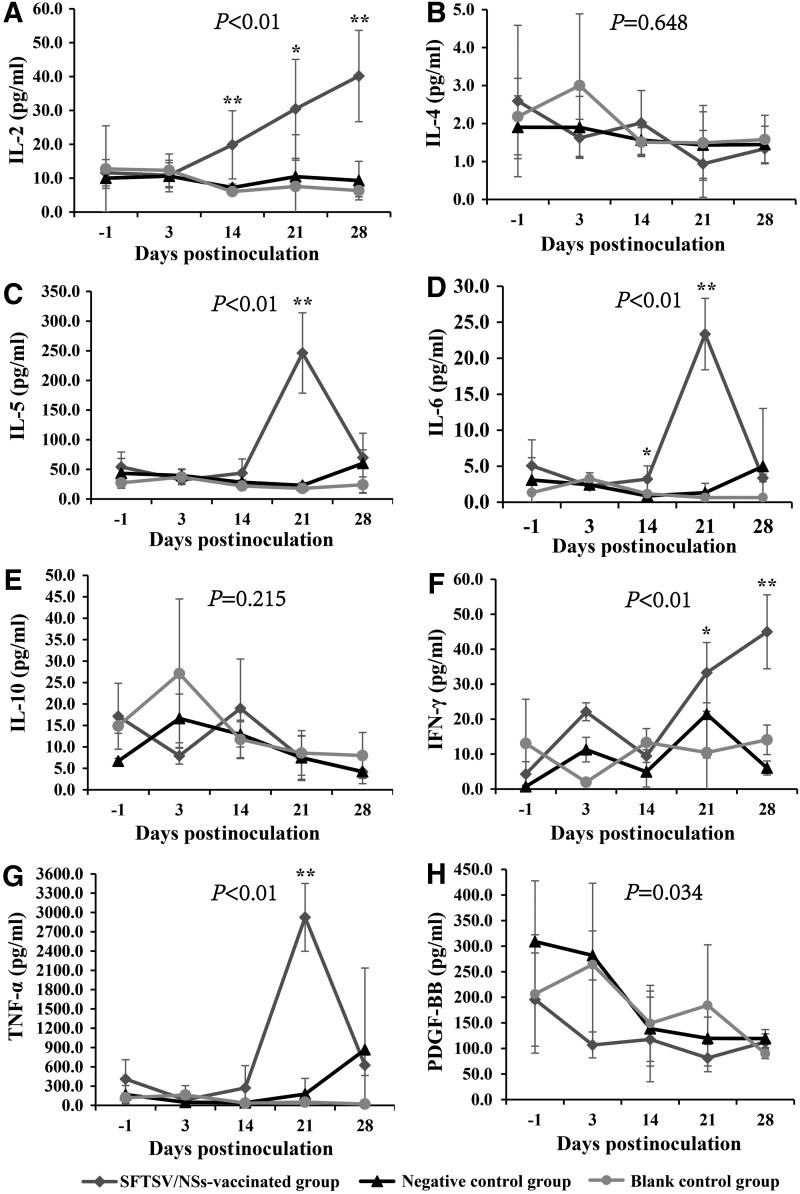
Enhanced cytokines response was detected in SFTSV/NSs-vaccinated mice after virus challenge
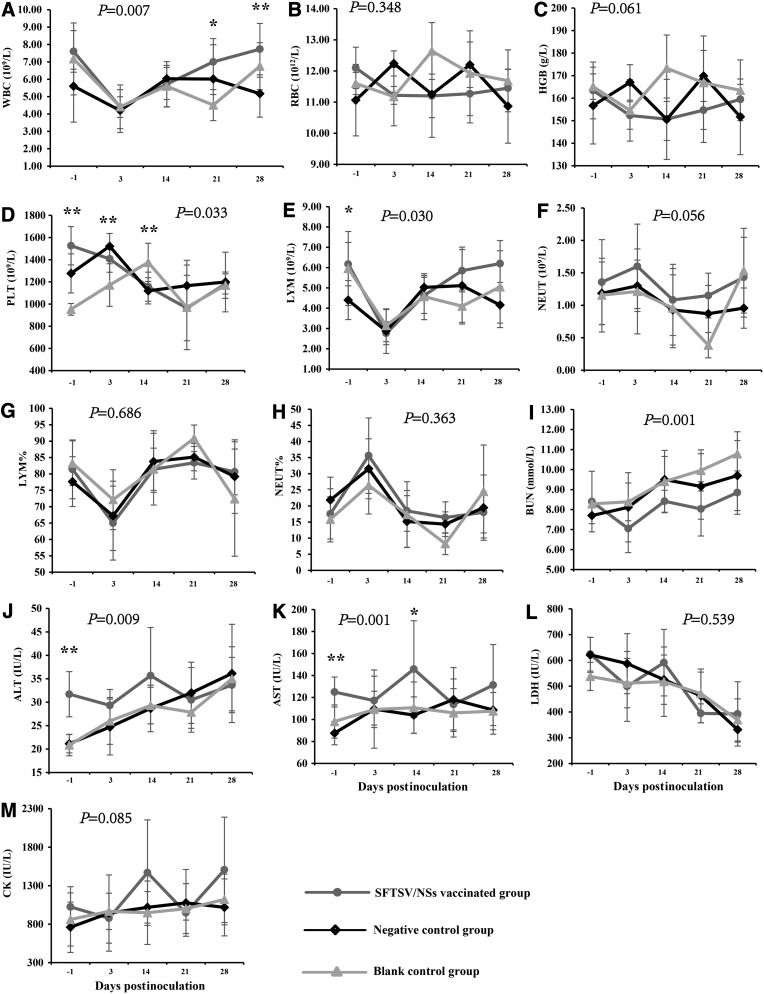
The dynamic changes of hematological and biochemical parameters (WBC, RBC, HGB, LYM and LYM%, NEUT and NEUT%, PLT; ALT, AST, LDH, CK, BUN) and cytokine concentration (IL-2, IL-4, IL-5, IL-6, IL-10, IFN-γ, TNF-α, and PDGF-BB) were delineated on the day before challenge (−1), the day 3, 14, 21, and 28 DPI. From the results, SFTSV inoculation caused similar dynamic changes (with no significant difference) in RBC (Fig. 5B), HGB (Fig. 5C), NEUT (Fig. 5F), LYM% (Fig. 5G), NEUT% (Fig. 5H), LDH (Fig. 5L), and CK (Fig. 5M) among the three groups of mice. However, SFTSV inoculation caused WBC count decrease before the 3 DPI, and then peaked up to levels with significant difference among the three groups on the day 21 and 28 after challenge (Fig. 5A). Before challenge, both the rSFTSV/NSs-vaccinated mice and the negative control mice had a higher level of PLT than the blank controls (Fig. 5D), which suggested that Freund's adjuvant had a stimulating effect on PLT production. Then, SFTSV inoculation made PLT in the rSFTSV/NSs-vaccinated mice decrease greatly, but PLT in the negative control mice and blank control mice first increased and then decreased. Unexpectedly, in addition, just vaccination with rSFTSV/NSs before challenge had made a slightly increased level of ALT (Fig. 5J) and AST (Fig. 5K).
FIG. 5.
Dynamic change of blood cells and biochemical parameters of C57BL/6J mice. The tested parameters include WBC (A), RBC (B), HGB (C), PLT (D), LYM (E), NEUT (F), LYM% (G), NEUT% (H), BUN (I) ALT (J) AST (K), LDH (L) AND CK (M). The sample sizes at each time point are 6 for the SFTSV/NSs-vaccinated group, and are 7 for both the negative control group and the blank group. The values for the above indexes are calculated and expressed as means±SD. p-Values above each subfigure were the result of multivariate ANOVA among the three groups. *p<0.05 and **p<0.01, which were the result of one-way ANOVA among the three groups at each observation point. ALT, alanine transaminase; AST, aspartate aminotransferase; CK, creatine kinase; BUN, blood urea nitrogen; HGB, hemoglobin; LDH, lactate dehydrogenase; LYM%, lymphocyte count percentage; LYM, lymphocyte count; NEUT%, neutrophil count percentage; NEUT, neutrophil count; PLT, platelet count; RBC, red blood cell count; WBC, leukocyte count.
Serial serum samples from the three groups of mice were subjected to cytokine immunoassay. Among the eight cytokines detected, IL-2 and IFN-γ increased significantly to a higher level on 3 DPI and 14 DPI in the SFTSV/NSs-vaccinated group, while was maintained at a low level in other two groups throughout the observation (Fig. 6A, F). On 21 days postinfection, a sharp increase of IL-5, IL-6, and TNF-α that was 5- to 10-fold was demonstrated in the SFTSV/NSs-vaccinated group, in comparison the other two groups (Fig. 6C, D, G). The PDGF-BB kept decreasing after infection in three groups of mice, but was even lower in mice of the SFTSV/NSs-vaccinated group at all the observation points (Fig. 6H).
FIG. 6.
Dynamic profile of sera cytokines of C57BL/6J mice. The tested parameters include IL-2 (A), IL-4 (B), IL-5 (C), IL-6 (D), IL-(10) (E), IFN-γ (F), TNF-α (G) and PDGF-BB (H). The sample sizes at the serial observation points are 5/5/5/5/2 for the SFTSV/NSs-vaccinated group, 5/4/5/5/5 for the negative control group, and 3/3/5/5/5 for the blank control group. The values for the above indexes are calculated and expressed as means±SD. p-Values above each subfigure were the result of multivariate ANOVA among the three groups. *p<0.05 and **p<0.01, which were the results of one-way ANOVA among the three groups at each observation point. IL, interleukin; TNF, tumor necrosis factor; PDGF-BB, platelet-derived growth factor-BB.
No obvious difference in pathological lesions was observed
By H&E staining, none of the tested tissues demonstrated obvious pathological change for three groups of mice throughout the observation (Fig. 7), as well as there was no distinct difference in the change of livers, spleens, and kidneys among the three groups of mice after SFTSV inoculation.
FIG. 7.
Dynamic profile of pathological changes in C57BL/6J mice after inoculation with SFTSV. Representative hematoxylin-eosin-stained liver, spleen, and kidney tissue paraffin-embedded sections from mice of the three groups (SFTSV/NSs-vaccinated group, negative control group, and blank control group) at the five observation points (Preinoculation (A), 3 (B), 14 (C), 21 (D), and 28 (E) DPI) (Preinoculation, 3, 14, 21, and 28 DPI) are shown here. The white arrows in the subfigures of liver tissue indicate the central veins of hepatic lobule, and the black arrows in the subfigures of kidney tissue indicate glomerulus. In the spleen tissue sections, RP indicates the red pulp and WP indicates the white pulp. All the images are 400× in amplification.
Discussion
Similar to most of viral nonstructural proteins (4,6), SFTSV/NSs was reported to be involved in suppressing the IFN expression and regulating innate immune response after SFTSV enters the host (15). It was also thought to be involved in the replication of SFTSV, as it was found to have an interaction with nucleoprotein and RNA in the infected cells (20). It was frustrating, however, that we did not observe the expected effects as pre-exposure vaccination with rSFTSV/NSs did not elicit efficient virus clearance in SFTSV-infected mice. Therefore, pre-exposure vaccination with rSFTSV/NSs cannot accelerate virus clearance in SFTSV-infected mice.
Although C57BL/6J mice had been used as an animal model to mimic SFTS in Jin et al.'s article (7), all tested immunocompetent C57BL/6J mice survived without medical signs or weight loss observed. In this study, mice were confirmed to be infected with SFTSV load combined with the three groups reaching the maximum mean virus loads of 105.46 copies/mL, 103.84 copies/mg, 103.03 copies/mg, and 102.77 copies/mg on the 3 DPI in average in sera, livers, spleens, and kidneys, respectively. Thereafter, virus titers declined progressively, but infectivity could still be detected till 28 DPI, which implied that SFTSV could replicate in mice. However, no abnormal manifestations were demonstrated in any groups of the mice although an increased virus amount was inoculated when compared with those used in Jin et al.'s experiment (7). This result implies that C57BL/6J mouse might not be an ideal animal model to mimic human SFTS.
On the other hand, we indeed observed an obviously different immunological effect in the rSFTSV/NSs-vaccinated mice after SFTSV inoculation. All the IL-2, IL-5, IL-6, and TNF-α in rSFTSV/NSs-vaccinated group of mice had a sharp increase after virus challenge, suggesting that these increased cytokines were mainly induced by secondary immune response and caused a negative effect on the bodyweight of mice as they were just not observed in the negative control mice and blank control mice. The decreasing PDGF-BB level in all the three groups of mice after virus challenge indicated decreased PDGF-BB to be beneficial to virus removal, which was consistent with that PDGF-BB pathway was found to be required for virus infection (18,19). In addition, elevated IFN-γ was detected in the rSFTSV/NSs-vaccinated mice after virus inoculation. However, unexpectedly, the elevated IFN-γ failed to accelerate virus removal. The possible reasons to interpret this may be the distinct difference between SFTSV/NSs and nonstructural proteins of most viruses. The nonstructural proteins of YFV, Japanese encephalitis virus, and dengue virus were usually expressed on the surface of and/or secreted extracellular from infected cells (5,10,17,24), which could induce antiviral immunity. However, SFTSV/NSs was expressed as cytoplasmic inclusion bodies in infected cells (15,20), which was further verified by locating NSs in SFTSV-infected DH82 cells cultured for 3 days but not secreted to their culture medium by ELISA and cell immunofluorescence test in this study (Supplementary Fig. S1; Supplementary Data are available online at www.liebertpub.com/vim). This is consistent with the results that no rSFTSV/NSs-specific antibodies were detected in the negative control mice and the blank control mice. So, according to the theory of intracellular antibody (intrabody) (13), which express and locate inside a cell and could be competent to achieve the effect of binding and blocking intracellular molecules, it is an option for further investigation to design an intrabody specific against intracellular SFTSV/NSs.
Supplementary Material
Acknowledgments
This work was supported by the National Natural Science Foundation of China (81222037 and 81130086), China Mega-Project for Infectious Diseases (2013ZX10004-202), and the 55th-class General Financial Grant from the China Postdoctoral Science Foundation (2014 M550570).
Authors' Contributions
Conceived and designed the experiments: R.L., W.L., and J.Y.W. Performed the experiments: R.L., D.D.H., and J.Y.B. Analyzed the data: R.L., W.L. Contributed reagents/materials/analysis tools: R.L., J.Y.B., L.Z., Q.B.L., X.A.Z., and W.C.C. Wrote the paper: R.L., W.L.
Author Disclosure Statement
No competing financial interests exist.
References
- 1.Barro M, and Patton JT. Rotavirus nonstructural protein 1 subverts innate immune response by inducing degradation of IFN regulatory factor 3. Proc Natl Acad Sci U S A 2005;102:4114–4119 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Beura LK, Sarkar SN, Kwon B, et al. Porcine reproductive and respiratory syndrome virus nonstructural protein 1beta modulates host innate immune response by antagonizing IRF3 activation. J Virol 2010;84:1574–1584 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Ding F, Zhang W, Wang L, et al. Epidemiologic features of severe fever with thrombocytopenia syndrome in China, 2011–2012. Clin Infect Dis 2013;56:1682–1683 [DOI] [PubMed] [Google Scholar]
- 4.Eifan S, Schnettler E, Dietrich I, et al. Non-structural proteins of arthropod-borne bunyaviruses: roles and functions. Viruses 2013;5:2447–2468 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Falgout B, Bray M, Schlesinger JJ, et al. Immunization of mice with recombinant vaccinia virus expressing authentic dengue virus nonstructural protein NS1 protects against lethal dengue virus encephalitis. J Virol 1990;64:4356–4363 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Hollidge BS, Weiss SR, and Soldan SS. The role of interferon antagonist, non-structural proteins in the pathogenesis and emergence of arboviruses. Viruses 2011;3:629–658 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Jin C, Liang M, Ning J, et al. Pathogenesis of emerging severe fever with thrombocytopenia syndrome virus in C57/BL6 mouse model. Proc Natl Acad Sci U S A 2012;109:10053–10058 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.John MW. The ELISA Guidebook, 2nd edition. New York: Humana Press, 2009 [Google Scholar]
- 9.Lam TT, Liu W, Bowden TA, et al. Evolutionary and molecular analysis of the emergent severe fever with thrombocytopenia syndrome virus. Epidemics 2013;5:1–10 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Lin YL, Chen LK, Liao CL, et al. DNA immunization with Japanese encephalitis virus nonstructural protein NS1 elicits protective immunity in mice. J Virol 1998;72:191–200 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Liu S, Chai C, Wang C, et al. Systematic review of severe fever with thrombocytopenia syndrome: virology, epidemiology, and clinical characteristics. Rev Med Virol 2014;24:90–102 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Liu Y, Wu B, Paessler S, et al. The pathogenesis of severe fever with thrombocytopenia syndrome virus infection in alpha/beta interferon knockout mice: insights into the pathologic mechanisms of a new viral hemorrhagic fever. J Virol 2014;88:1781–1786 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Marasco WA. Intrabodies: turning the humoral immune system outside in for intracellular immunization. Gene Ther 1997;4:11–15 [DOI] [PubMed] [Google Scholar]
- 14.Ministry of Health, People's Republic of China. Guideline for prevention and treatment of severe fever with thrombocytopenia syndrome (2010 version). Chin J Clin Infect Dis 2011;4:193–194 [Google Scholar]
- 15.Ning YJ, Wang M, Deng M, et al. Viral suppression of innate immunity via spatial isolation of TBK1/IKKɛ from mitochondrial antiviral platform. J Mol Cell Biol 2014;6:324–337 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Qu B, Qi X, Wu X, et al. Suppression of the interferon and NF-kappaB responses by severe fever with thrombocytopenia syndrome virus. J Virol 2012;86:8388–8401 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Schlesinger JJ, Brandriss MW, Cropp CB, et al. Protection against yellow fever in monkeys by immunization with yellow fever virus nonstructural protein NS1. J Virol 1986;60:1153–1155 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Soroceanu L, Akhavan A, and Cobbs CS. Platelet-derived growth factor-alpha receptor activation is required for human cytomegalovirus infection. Nature 2008;455:391–395 [DOI] [PubMed] [Google Scholar]
- 19.Tikkanen JM, Krebs R, Bruggeman C, et al. Platelet-derived growth factor regulates cytomegalovirus infection-enhanced obliterative bronchiolitis in rat tracheal allografts. Transplantation 2004;77:655–658 [DOI] [PubMed] [Google Scholar]
- 20.Wu X, Qi X, Liang M, et al. Roles of viroplasm-like structures formed by nonstructural protein NSs in infection with severe fever with thrombocytopenia syndrome virus. FASEB J 2014;28:2504–2516 [DOI] [PubMed] [Google Scholar]
- 21.Youn S, Cho H, Fremont DH, et al. A short N-terminal peptide motif on flavivirus nonstructural protein NS1 modulates cellular targeting and immune recognition. J Virol 2010;84:9516–9532 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Yu XJ, Liang MF, Zhang SY, et al. Fever with thrombocytopenia associated with a novel bunyavirus in China. N Engl J Med 2011;364:1523–1532 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Zhang X, Liu Y, Zhao L, et al. An emerging hemorrhagic fever in China caused by a novel bunyavirus SFTSV. Science China. Life Sci 2013;56:697–700 [DOI] [PubMed] [Google Scholar]
- 24.Zhang YM, Hayes EP, McCarty TC, et al. Immunization of mice with dengue structural proteins and nonstructural protein NS1 expressed by baculovirus recombinant induces resistance to dengue virus encephalitis. J Virol 1988;62:3027–3031 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Zhang YZ, He YW, Dai YA, et al. Hemorrhagic fever caused by a novel Bunyavirus in China: pathogenesis and correlates of fatal outcome. Clin Infect Dis 2012;54:527–533 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.