Abstract
Whole-organism compound sensitivity assays are a valuable strategy in infectious diseases to identify active molecules. In schistosomiasis drug discovery, larval-stage Schistosoma allows the use of a certain degree of automation in the screening of compounds. Unfortunately, the throughput is limited, as drug activity is determined by manual assessment of Schistosoma viability by microscopy. To develop a simple and quantifiable surrogate marker for viability, we targeted glucose metabolism, which is central to Schistosoma survival. Lactate is the end product of glycolysis in human Schistosoma stages and can be detected in the supernatant. We assessed lactate as a surrogate marker for viability in Schistosoma drug screening assays. We thoroughly investigated parameters of lactate measurement and performed drug sensitivity assays by applying schistosomula and adult worms to establish a proof of concept. Lactate levels clearly reflected the viability of schistosomula and correlated with schistosomulum numbers. Compounds with reported potencies were tested, and activities were determined by lactate assay and by microscopy. We conclude that lactate is a sensitive and simple surrogate marker to be measured to determine Schistosoma viability in compound screening assays. Low numbers of schistosomula and the commercial availability of lactate assay reagents make the assay particularly attractive to throughput approaches. Furthermore, standardization of procedures and quantitative evaluation of compound activities facilitate interassay comparisons of potencies and, thus, concerted drug discovery approaches.
INTRODUCTION
Schistosomiasis ranks top among neglected tropical diseases and causes the loss of a considerable number of life years due to disability and premature death (1). Treatment and preventive chemotherapy rely solely on one drug, praziquantel (PZQ) (2). WHO estimates that 243 million people required PZQ in 2011, and an agreement with Merck KGaA signed early in 2013 will lead to a 10-fold increase in the supply of tablets from 20 million to 250 million per year (3, 4). This tremendous increase in PZQ deployment in sub-Saharan Africa fuels the ever-growing concern of the emergence and establishment of Schistosoma resistance to PZQ and particularly so since isolates with reduced susceptibility have been identified sporadically in sub-Saharan Africa (5–7). The need for new schistosomiasis chemotherapeutics is self-evident.
Drug development activities for helminth diseases still suffer from little effort in both funding and concerted approaches undertaken by the research community (8). Even though several new drug discovery techniques have been developed in the recent past, methodologies in pharmaceutical drug discovery for schistosomiasis lack elaboration, interlaboratory standardization, harmonization, and automation (9–13). This is in contrast to other poverty-related diseases, such as malaria, where tremendous attention and support in the last 10 years by The Global Fund, a nongovernmental funding organization, have allowed the development of elaborate drug discovery programs (14). Whole-organism screening of compounds has become a highly valuable and effective strategy to identify chemical lead structures for further clinical development, when performed rigorously and in high-throughput setups.
In schistosomiasis, whole-cell screening assays using schistosomula, the larval stage in humans, have become a valuable approach to identify active compounds justified to be tested against adult worms, implying experiments with mice. Even though the handling of schistosomula allows automation, at least to a certain degree, the viability of the worms, which determines the drug's activity, is commonly assessed by microscopy. To overcome the subjectivity and complexity of microscopy, several novel techniques to determine schistosomulum viability have recently been presented, although microscopy is still the standard reference methodology (12, 13, 15, 16). However, the phenotype and the degree of changes in the worms' morphology and motility heavily depend on the chemical nature of the drug, and in adult worms it has been shown that damaged tissues have the potential to regenerate (17, 18). Whether these observations always truly reflect parasite death can only be supposed.
Therefore, we investigated if the energy metabolic pathway essential to parasite survival is able to precisely reflect the viability of Schistosoma mansoni. Intravascular Schistosoma worms primarily rely on glycolysis for energy generation (19, 20). Adult parasites consume large amounts of glucose and every 5 h consume an amount of glucose equal to their dry weight (21). In both adult and larval-stage Schistosoma parasites, glucose is transported from the host bloodstream across the parasite outer surface, the tegument, via the specific parasite glucose transporters SGTP1 and SGTP4 (22). The breakdown of glucose via glycolysis mainly results in lactate, which is excreted via the aquaglyceroporin homologue SmAQP, located in the tegument of adult worms and schistosomula (23, 24). The amount of lactate excreted into the surrounding environment by the parasites is considerable, and the lactate can easily be analyzed and quantified by colorimetric or fluorometric assays (25).
We targeted glycolysis by detecting lactate as the major end product secreted by schistosomula and adult worms. We show that lactate quantification can serve as a novel objective and a precise readout for assessment of the viability of schistosomula and adult worms. Lactate was measured by a fluorometric assay which is simple to perform and may potentially be used for throughput analysis. We tested several compounds which have previously been subjected to drug screens for their activity against schistosomula and adult worms and found that their anti-Schistosoma activities were comparable when they were analyzed by lactate measurement and the “gold standard” assay, microscopy. The availability of commercial lactate assays for the quantification of lactate is an additional advantage and may facilitate implementation of this assay in projects screening drugs for activity against Schistosoma.
MATERIALS AND METHODS
S. mansoni life cycle stages.
The life cycle of S. mansoni (Puerto Rico PR-1 strain) is routinely maintained at the Institute of Tropical Medicine, University of Tübingen, Tübingen, Germany, in C57BL/6 mice and in Biomphalaria glabrata snails as the intermediate host. All animal experiments were conducted in accordance with German laws after approval by the Regional Administrative Authority of Tübingen, Germany (Anzeige from 24 August 2004).
Mice were obtained from Harlan Laboratories and were kept under standard laboratory conditions with food and water ad libitum and a 12-h and light and 12-h dark cycle. Each animal was infected percutaneously with 50 cercariae. After 8 weeks of infection, the mice were euthanized by CO2, and adult worms were obtained by perfusion, i.e., cutting the portal vein, injecting phosphate-buffered saline into the heart, and flushing the worms out of the vein into prewarmed RPMI medium containing 100 U/ml penicillin plus 100 μg/ml streptomycin and 5% fetal calf serum (FCS).
Snails acting as the intermediate host were of the Schistosoma-susceptible and albinotic M line and originated from the Institute of Zoology, Oregon State University (Corvallis, OR, USA). They were maintained at 27°C in charcoal-filtered water in tanks with 20 snails each with a 12-h light and 12-h dark cycle or, after infection with miracidia, in total darkness. Snails were individually infected with eight miracidia. After 8 weeks, the snails were exposed to light for 4 h and the cercariae were harvested without aspirating excretions or debris for schistosomulum transformation.
Schistosomulum in vitro cultures were kept under sterile conditions in schistosomulum culture medium (SCM; phenol-red free medium 199 [M199; catalog number 11043-023; Gibco], 5.5 mM d-glucose, 200 U/ml penicillin, 200 μg/ml streptomycin, 1% heat-inactivated FCS [iFCS], if not otherwise stated). Adult worms were cultured in vitro in adult worm culture medium (ACM; phenol-red free RPMI, 100 U/ml penicillin plus 100 μg/ml streptomycin, 5% iFCS). Incubator conditions for schistosomula and adult worms in in vitro cultures were set at 37°C and 5% CO2.
Preparation of S. mansoni schistosomula.
Cercariae of S. mansoni were mechanically transformed into schistosomula by vortexing following published procedures (11, 26). Briefly, the cercaria suspension was cooled for 15 min on ice and centrifuged at 350 × g for 10 min at 4°C. The cercaria pellet was resuspended in 10 ml ice-cold SCM and vortexed at high speed for 4 min to induce tail loss. After 5 min of incubation on ice, the parasite solution was poured into a 5-cm petri dish and cercaria bodies and tails were separated by gently swirling the plate (27). Cercaria bodies, which correspond to schistosomula, accumulated in the middle of the plate and were collected by pipetting. The swirling process was repeated, the schistosomula were transferred into a 15-ml tube, the volume was made up to 5 ml with SCM, and the mixture was kept on ice. Parasites were counted before and after transformation to determine the efficiency of the transformation procedures and the purity and numbers of schistosomula obtained after the separation step. Schistosomula were kept in 24-well plates in SCM (500 schistosomula/1 ml SCM/well) for 24 h to allow maturation before being further processed.
Reagents.
All compounds were purchased from Sigma-Aldrich. Reagents were dissolved in sterile dimethyl sulfoxide (DMSO) at the following concentrations: 50 mM mefloquine (MQ) hydrochloride (molecular weight [MW], 414), 14.7 mM auranofin (AU; MW, 678), 15.9 mM gambogic acid (GA; MW, 629), 67.3 mM salinomycin (SAL) monosodium (MW, 773), 50 mM praziquantel (PZQ; MW, 312), and 10.4 mM niclosamide (NI; MW, 327). Stock solutions were kept at −20°C and continuously protected from light. Dilutions for drug sensitivity assays were done with SCM or ACM.
Schistosomulum drug sensitivity assays.
Ninety-six-well flat-bottomed plates were predosed with compounds in a 3-fold serial dilution to obtain the following range of concentrations in a final volume of 25 μl/well: mefloquine, 0.07 to 50 μM; auranofin, 0.01 to 10 μM; gambogic acid, 0.07 to 50 μM; salinomycin, 1.23 to 900 μM; praziquantel, 0.69 to 500 μM; and niclosamide, 0.14 to 100 μM. Control wells were predosed with 1% DMSO in SCM only.
Mature schistosomula (24 h old) in SCM were added to predosed wells to obtain 100 schistosomula per well in a total volume of 225 μl/well, if not otherwise stated. The true number of schistosomula per well was confirmed by microscopy. The highest concentration of DMSO per well did not exceed 1%. Schistosomulum drug assay plates were kept at 37°C, and the viability of the schistosomula was assessed by lactate assay and in parallel by microscopy after the culture period indicated below (usually 48 h). To measure the relative levels of lactate secreted by schistosomula into the surrounding medium, 10 μl/well of the assay medium was carefully transferred into a fresh 96-well plate without aspirating any schistosomula and immediately stored at −80°C for further processing at a later time point. Where dead schistosomula were used as a negative control, parasites were killed by heating to 65°C for 10 min and allowed to chill before further use.
Microscopic assessment of schistosomulum viability.
All schistosomula in each well were investigated using an inverted microscope (Nikon eclipse Ti). On the basis of motility and morphology, the parasites were classified as viable (movement and normal appearance) or dead (no movement within 10 s and/or severe morphological changes of any kind compared to the morphology of untreated parasites, e.g., granularity, blebbing).
S. mansoni adult worm in vitro drug sensitivity assay.
For in vitro drug sensitivity assays, adult worms were set in 24-well plates at 1 worm per well in 1 ml ACM. Parasites were allowed to adapt to the culture condition for 24 h. PZQ was added to obtain a final concentration of 9.6 μM, 3.2 μM, or 1.07 μM. MQ was tested at 24 μM or 8 μM. The viability of the worms was assessed by microscopy every 24 h. After 72 h, the supernatant was collected from each well and immediately stored at −80°C. At a later time point, 0.5 μl of the samples was processed in the lactate assay.
Lactate assay.
Final lactate assay procedures were preceded by rigorous optimization and validation experiments to identify the appropriate volume of sample and of assay components, and the time required to incubate the assay mixture prior to taking the fluorometric reading was also determined.
Lactate levels in the supernatants of schistosomulum cultures (4 μl) or of in vitro adult worms (0.5 μl) were measured with a fluorometric l-lactate assay kit (catalog no. ab65330; Abcam) using 96-well, black-sided, optical clear-bottom plates (catalog no. 3340; Corning) following the manufacturer′s specifications, with minor modifications. We reduced the volumes in the lactate reaction mix of both the enzyme mix and the probe to 0.5 μl each and increased the volume of the assay buffer to 49 μl. After 40 min of incubation at room temperature, the plate was read by a fluorometer (Fluoroskan Ascent; Thermo Scientific) by applying an excitation and emission filter pair of 530 nm and 590 nm. The fluorometer detected relative fluorescence light units (RFUs) over a dynamic range of more than 6 decades with the lowest reading set at 0.001. If samples were diluted with water beforehand to fit within the linear range of the assay, RFU values were adjusted by the respective dilution factor. All measurement series included SCM to determine the background lactate level, which was then subtracted from the readings of the respective measurements. To allow interassay comparison of RFU values, a sample of 0.01 μM l-lactate (Abcam) was included in each measurement series as a reference. RFU readings were normalized to the lactate reference value (normalized RFUs) to correct for variability among lactate assays and fluorometer performance.
Statistical analysis.
Statistical analysis was done with either JMP (version 5.0.1; SAS Institute), Prism (version 6; GraphPad Software), or R software. Differences between two samples were analyzed by an unpaired, two-tailed t test. A one-way analysis of variance with Tukey's multiple-comparison test was applied to analyze group differences. Dose-response curves were determined by nonlinear regression analysis of RFU values and log concentrations, and the 50% inhibitory concentration (IC50) and the 99% inhibitory concentration (IC99) were calculated (28).
RESULTS
Lactate levels reflect schistosomulum viability.
Schistosomula catabolize glucose via glycolysis, with lactate being the major end product (23). Lactate is expelled into the surrounding medium via an aquaporin protein, which has been identified to be an important lactate transporter in the tegument of adult worms and is also present in schistosomula (24, 29). We investigated if lactate levels in the supernatant of schistosomulum in vitro cultures reflect the viability of the larvae. Alive and heat-killed schistosomula (n = 200) were cultured in medium for 72 h, and living parasites produced 4.3 times more lactate (P = 0.0007) than dead parasites when RFU levels were comparable to the background signal of medium only (Fig. 1A). Microscopy confirmed the viability of the schistosomula (Fig. 1B). On the basis of a parallel lactate standard measurement, it was calculated that 200 schistosomula produced, on average, 47.9 μM lactate within 72 h of in vitro culture.
FIG 1.
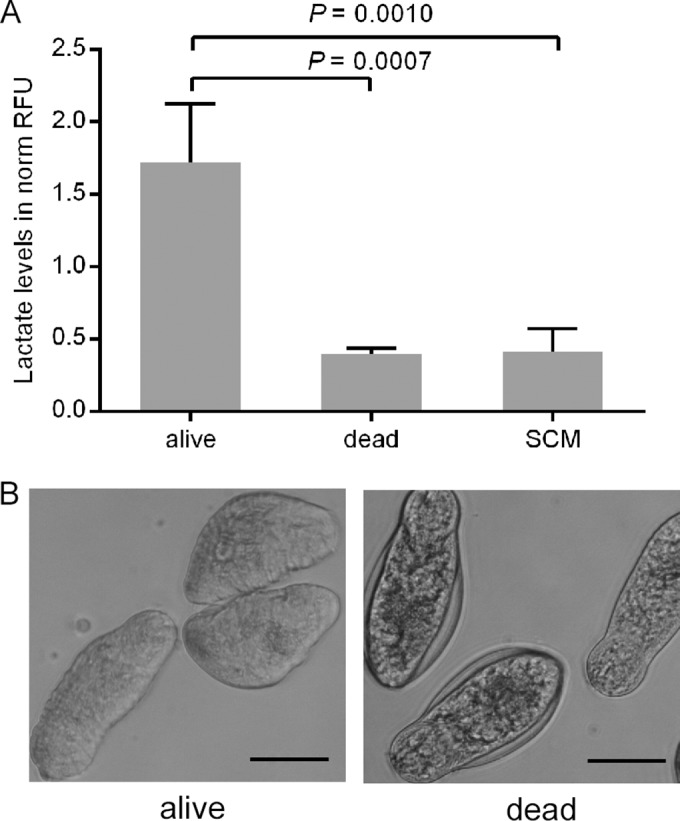
Lactate levels of alive and dead schistosomula. (A) Mechanically transformed alive (alive, n = 200) and heat-killed (dead, n = 200) schistosomula were cultured in vitro for 72 h, and relative lactate levels in the supernatant were measured. The culture medium (SCM) without schistosomula indicates the background level of lactate. Lactate levels are expressed as normalized RFUs (norm RFU). The mean and standard deviation from four independently performed experiments are shown. (B) The morphology of alive and heat-killed parasites after 72 h of culture is depicted. Bars, 50 μm.
Optimizing procedures to accurately and precisely measure schistosomulum viability.
Schistosomulum fitness depends on serum (in our case, iFCS) in in vitro culture. On the other hand, we observed that the serum in the medium increases background RFU values (not shown). Therefore, we identified the minimum iFCS concentration in SCM where the maximum number of schistosomula survived at a given time point. Schistosomula were cultured in medium containing 5%, 2.5%, 1%, or 0% iFCS, and viability was assessed microscopically for a period of 8 days. No difference in viability was observed by microscopy at day 3 for any of the iFCS concentrations (see Fig. S1 in the supplemental material). At day 8, viability in 5% iFCS, 2.5% iFCS, and 1% iFCS did not differ significantly (P > 0.05); however, these three iFCS concentrations resulted in viabilities significantly different from those under culture conditions without iFCS (0% iFCS). No iFCS in the medium was detrimental to schistosomula, and less than 40% survived 8 days of culture. We continued using medium with 1% iFCS, as this concentration showed the best performance with respect to viability and the minimization of background lactate levels.
To ensure that the readings for the samples were within the linear range of the lactate assay, lactate of defined concentrations was measured. Lactate and RFU values showed a linear correlation up to 40 μM l-lactate (corresponding here to 280 RFUs), but the assay became unreliable at higher lactate concentrations, probably due to the consumption of assay components (see Fig. S2 in the supplemental material). Thus, in further assays readings were kept below 280 RFUs, and where necessary, samples were appropriately diluted before measurement.
An in-depth investigation of the relationship between schistosomulum numbers and cumulative lactate levels over time was performed. The numbers of schistosomula and the incubation time highly correlated with lactate levels (expressed as normalized RFUs) (Fig. 2B). At any time point, the assay significantly (P < 0.001) discriminated the mean lactate levels (expressed as normalized RFUs) produced by 100 schistosomula from those produced by 50 and 200 schistosomula (Fig. 2A). In the further compound sensitivity assays, we used 100 schistosomula per well as the starting material to allow for potential throughput. The 48-h time point of culture was chosen both to address the maximum fitness of the schistosomula and to allow sufficient time for the drug to exert its antischistosomal activity.
FIG 2.
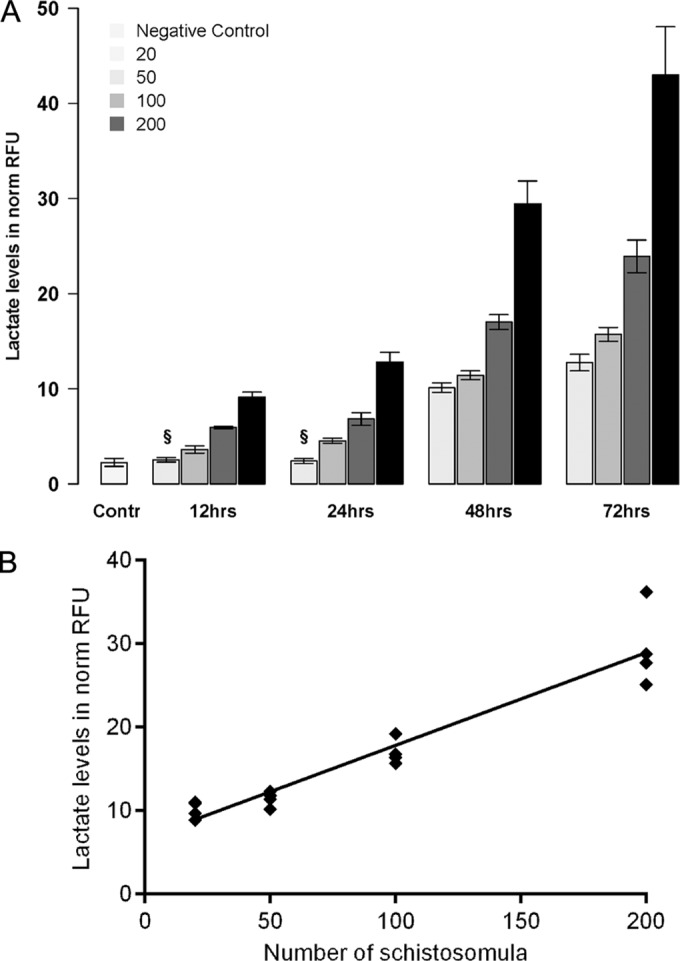
Different numbers of schistosomula were cultured, and lactate levels were assessed over time. (A) Supernatant was collected after 12 h, 24 h, 48 h, and 72 h, and accumulated lactate levels (expressed as normalized RFUs) are shown for 20, 50, 100, and 200 schistosomula. The data for the negative control (Contr) indicate the background lactate levels in SCM. The mean and standard deviation from four independently performed experiments are presented. §, at 12 h and 24 h, the lactate levels produced by 20 schistosomula did not differ significantly from the background levels. (B) The linear relationship (R2 = 0.91) between lactate levels and schistosomulum numbers is shown for the 48-h time point.
Schistosomula rely on glucose consumption to survive. The relationship between glucose consumption, lactate production, and viability was investigated. Schistosomula were produced and cultured in DMEM without supplemented d-glucose (M199 contains 5.5 mM glucose and is not available with 0 mM glucose) or in medium additionally supplemented with 5 mM, 10 mM, or 20 mM glucose. All schistosomula cultured without supplemented glucose were dead after 18 h, and no lactate was secreted (Fig. 3).
FIG 3.
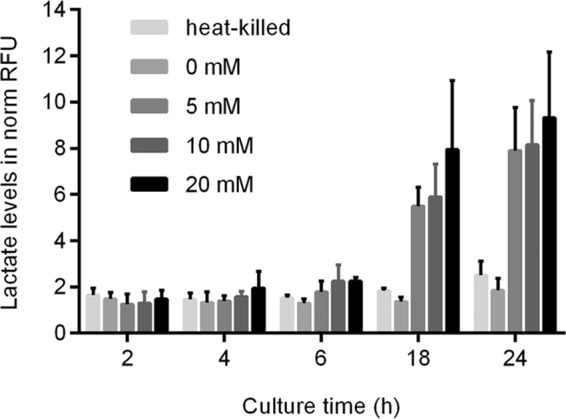
The influence of glucose consumption was assessed for lactate production and percent viability. Immediately after transformation, schistosomula were cultured in glucose-free medium (DMEM; 0 mM glucose) or in DMEM supplemented with 5 mM, 10 mM, or 20 mM glucose. Heat-killed schistosomula served as controls. Sampling was performed at the indicated time points, and lactate levels (expressed in normalized RFUs) were measured. Mean values and standard deviations of four measurements are shown.
Drug assay with control drugs.
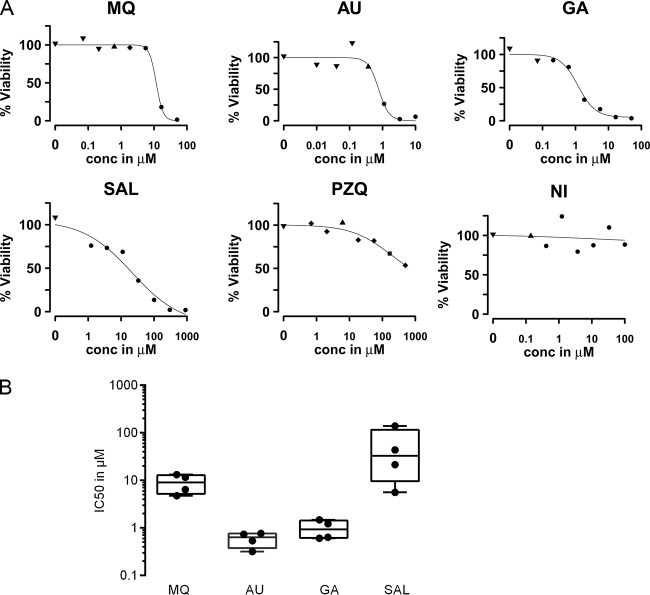
In vitro testing of the activities of the compounds against the schistosomula was performed with drugs that had previously been investigated in S. mansoni. For a detailed work flow, see Fig. S3 in the supplemental material. Due to the wealth of data available, the standard compounds mefloquine (MQ) and praziquantel (PZQ) were chosen to allow comparison of lactate assay data with microscopic viability data. MQ was previously reported to be active against schistosomula, in contrast to PZQ, which is recognized for its moderate potency against the larval stage (30, 31). In addition, auranofin (AU), gambogic acid (GA), salinomycin (SAL), and niclosamide (NI) were tested, as data on their activity against schistosomula are scarce.
The lactate levels produced by schistosomula depended on the drug's concentration and allowed modeling of dose-response relationships. In the same experiment, each well was assessed manually by microscopy to determine schistosomulum viability (in percent) in the presence of each drug concentration. MQ, AU, GA, and SAL showed a sigmoidal concentration-dependent effect on lactate levels, which allowed calculation of inhibitory concentrations (IC50s and IC99s) (Fig. 4A and B). The potencies of MQ, AU, and GA were generally in accordance with the microscopy data (Table 1). For SAL, observational data differed from the activity determined by lactate levels, which indicated a rather weak response against schistosomula. Dose-response relationships for PZQ and NI did not allow calculation of the IC50 or IC99, as either the effect on lactate production was very weak (PZQ) or no effect was observed (NI). Microscopy confirmed previously reported data showing that PZQ does not inhibit motility in Schistosoma larval stages even at high concentrations, even though the tegument was drastically affected. However, in contrast to results based on lactate levels, microscopy data demonstrated the high level of activity of NI, which caused immobility and drastic changes to morphology, including high levels of granularity and blebbing. The narrow range of IC50s of MQ, AU, GA, and SAL from four independently performed experiments indicated the robustness and reproducibility of the compound activity screens based on lactate quantification (Fig. 4B). To monitor the consistency of parasite numbers, the actual numbers of parasites per well were determined by manual counting in this proof-of-concept work. The median coefficient of variation calculated for all of the drug assays performed was 12.9%.
FIG 4.
Dose-response curves, based on lactate levels, including percent viability of schistosomulum sensitivity assays. Schistosomula were cultured with 3-fold dilutions of either mefloquine (MQ), auranofin (AU), gambogic acid (GA), salinomycin (SAL), praziquantel (PZQ), or niclosamide (NI). Unaffected controls were schistosomula not treated with any compound. After 48 h, viability was assessed by lactate assay and microscopy. (A) The dose-response curves (based on lactate levels and expressed as percent inhibition, with the maximum RFU value being set equal to 100%) from one representative experiment per drug are plotted. The different symbols for the data points in the dose-response curves represent percent viability determined by microscopy: ▼, 100% to 91% viability; ▲, 90% to 81% viability; ⧫, 80% to 71% viability; ■, 70% to 21% viability; ●, 20% to 0% viability. (B) Box plots (median [●], quartiles [bars in the boxes], and maximum and minimum values [top and bottom lines of the boxes, respectively]) show IC50 values (μM) from four independently performed drug sensitivity assays.
TABLE 1.
Activities against schistosomula determined by lactate assay and microscopy and previously reported activitiesa
| Compound | Activity (μM) by lactate assay |
MLC (μM) by microscopy | Previously reported activity |
||||
|---|---|---|---|---|---|---|---|
| IC50 | IC50 range | IC99 | IC99 range | MLC (μM) | Authors, yr (reference) | ||
| MQ | 9.02 | (4.76–13.2) | 13.0 | (6.09–19.7) | 5.55 | 12.1 | Manneck et al., 2010 (32) |
| AU | 0.64 | (0.32–0.77) | 2.43 | (1.80–4.01) | 1.10 | 1b | Abdullah et al., 2009 (27) |
| GA | 0.93 | (0.61–1.49) | 12.2 | (0.91–75.3) | 0.62 | 1 | Abdullah et al., 2009 (27) |
| SAL | 32.6 | (5.59–140.1) | 6,692 | (17.3–54,487) | 4.29 | 1 | Abdullah et al., 2009 (27) |
| PZQ | NA | NA | NA | NA | NA | >320 | Manneck et al., 2011 (11) |
| NI | NA | NA | NA | NA | 0.21 | 1 | Abdullah et al., 2009 (27) |
MLC, minimum lethal concentration; NA, not applicable.
The IC50 was 0.82 μM by FDA-PI staining (13).
Lactate assay to measure viability of adult S. mansoni worms ex vivo.
To investigate if the lactate assay is principally applicable to determination of the viability of S. mansoni adult worms ex vivo, we tested PZQ and MQ in an in vitro drug sensitivity assay with adult worms (males only) in a simplified setup. PZQ was tested at 9.6 μM, 3.2 μM, and 1.07 μM, and MQ was tested at 24 μM and 8 μM (Fig. 5). After 72 h, a strong increase in lactate production could be detected in untreated adult worms. The different concentrations of PZQ (9.6 μM, 3.2 μM, and 1.07 μM) and MQ (24 μM and 8 μM) tested resulted in a dose-dependent decrease in lactate levels, although the few concentrations tested did not allow the generation of proper dose-response curves. Parasites treated with PZQ at 1.07 μM gave a reduced lactate signal, and death was confirmed by microscopy. MQ at 24 μM killed all adult worms, as determined by the lactate assay and by microscopy.
FIG 5.
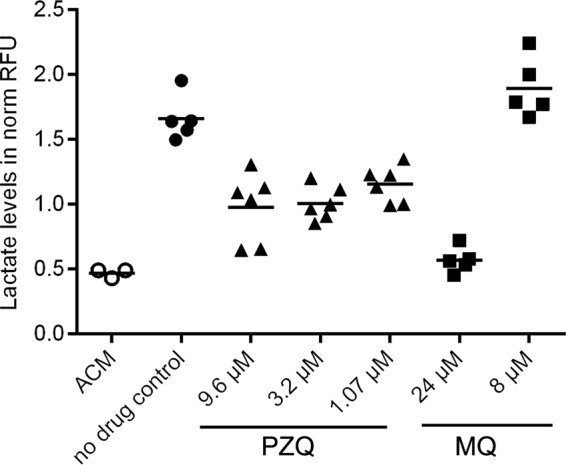
Lactate assay to determine the in vitro sensitivity of S. mansoni adult worms (all males) toward PZQ (▲) and MQ (■). Adult worms were cultured for 72 h in ACM (●) without drug (no drug control) or with PZQ (9.6 μM, 3.2 μM, or 1.07 μM) or MQ (24 μM or 8.0 μM). The data for ACM only (○) show background lactate levels. Normalized RFU values are shown for each single experiment. The bars indicate the means.
DISCUSSION
Even though efforts to develop objective and sensitive measures of Schistosoma viability—the readout in Schistosoma phenotypic screens—are ongoing, the lack of quantitative, robust, and relatively simple and quick-to-perform assays is still evident (11–13, 16). This might be due to the complex nature of determining the viability of invertebrate multicellular parasites in vitro. So far, Schistosoma death is determined by observation of morphology as well as motility, and the changes detected are ranked. This procedure is highly subjective and hampers high-throughput screening of compounds. An additional concern is the reliability of morphological/motility changes reflecting parasite viability. It is known that adult schistosomes can recover damaged tegument after sublethal PZQ doses, and recent investigations have identified somatic stem cells in Schistosoma which show potential for tissue regeneration (17, 18).
To overcome these shortcomings, we established a novel readout for compound sensitivity assays based on targeting of the energy metabolism, which is essential for Schistosoma survival and should thus reflect parasite viability. S. mansoni larval stages and adult worms rely on glycolysis, and lactate is the catabolic end product of glucose metabolism. Both human stages of Schistosoma expel lactate into the surrounding medium, which can be measured by a fluorometric lactate assay in which the levels of the fluorometric probe reflect lactate levels. Potential lead structures are identified by their potency in inhibiting the lactate production of the worms, which informs on parasite viability.
To provide an accurate and robust assay for application in schistosomulum screens, the parameters of the lactate assay were thoroughly assessed and validated. The assay could clearly differentiate between alive and dead schistosomula. Measurement resolution was identified to be 100 schistosomula as early as 48 h of in vitro culture, and lactate levels in the well were distinct from those in wells with 50 schistosomula. Correlation analysis of lactate levels and schistosomulum numbers revealed a strong linear relationship, which is an important requirement for the indirect measurement of parasite viability (where various percentages of live and dead schistosomula are found in the same sample when the drug is less than 100% efficient). Based on these results, we chose 100 schistosomula and 48 h of culture as the best assay conditions. The low starting number of schistosomula is highly favorable for a throughput setup. Other assays applied 5- to 10-fold more schistosomula (11, 13). Additionally, monitoring of drug activity over time is very convenient to perform, as the assay allows interim sampling by taking the culture supernatant at any time point, while the worms remain untouched, provided that parasite viability is not substantially affected by the assay conditions themselves (e.g., depletion of nutrients, accumulation of excretion products such as lactate and others).
For assay validation, we measured the activities of six compounds against the schistosomulum stage by the lactate assay and compared them to the viability findings obtained by microscopic evaluation. Lactate levels showed a clear dose-response relationship for compounds with previously reported activities against schistosomula. The data generated for MQ and PZQ by lactate measurement confirmed the previously reported MQ activity and the low potency of PZQ against larval-stage Schistosoma. This indicates that the lactate assay is a valid methodology to determine the viability of schistosomula and thus can serve as a novel readout in schistosomulum drug sensitivity assays. This is further supported by the accordance of the activities of AU and GA and, additionally for AU, with previously reported results obtained by an assay based on fluorescence measurement of propidium iodide (PI) and fluorescein diacetate (FDA) (12, 27). The lactate-based screen of SAL and NI activity did not reflect the findings of the microscopic assessment of viability, which judged both compounds to be highly active.
This disagreement was also reported in 2010 by Peak et al., who evaluated PI and FDA staining for determination of the antischistosomal activities of compounds (13). It remains unclear which readout truly reflects the viability of the schistosomula, as the ultimate evidence could be deduced only from monitoring the development of schistosomula reimplanted into host animals. This observation further underlines the fact that assessment of the viability of metazoan organisms is a difficult undertaking and any surrogate marker needs to be carefully investigated. Additionally, lactate measurement was identified to be a promising readout in the adult Schistosoma worms in in vitro drug assays, even though the test setup was only basic. Thus, an extensive test series is required, including test series with males, females, and pairs.
The lactate assay is based on fluorometric quantification of NADH, the amount of which is proportional to the amount of lactate and which is generated on the basis of the conversion of lactate to pyruvate in the presence of NAD+ and lactate dehydrogenase. We cannot rule out the possibility that some drugs may interfere with the lactate assay and make interpretation of the results difficult. For example, it is conceivable that some compounds might lead to increased NADH levels in the culture supernatant due to drug-induced parasite damage. We minimized any potential impact of the medium and FCS (which is part of the sample analyzed) on the outcome of the lactate assay by reducing their volumes, by heat inactivating FCS, and by equalizing the assay conditions to precisely determine drug concentration-dependent lactate levels.
The use of whole-organism screens applying larval-stage Schistosoma is a valuable strategy to narrow and prioritize the molecules in arrays to be tested for their activities against schistosomiasis in animal models. However, it has to be considered that channeling of compounds through screening of activity against schistosomula might eliminate molecules with potential activity against later parasite stages, e.g., praziquantel, if it had not already been discovered.
We provide a simple work flow which should allow other laboratories to easily apply our assay setup to test the activities of drugs against schistosomula. We recommend the inclusion of two positive controls (medium only and compound solvent) to check the overall viability of the schistosomulum preparation over the culture period. In addition, we ask for the inclusion of quality controls by testing compounds with known performance under the specified assay conditions—mefloquine and/or auranofin—to control assay quality and interassay variability and to allow comparisons of drug activity determined over time and by different staff and sites executing the assays.
We conclude that lactate measurement is a promising new approach to assess the viability of larval-stage Schistosoma in drug sensitivity testing. The assay is sensitive, simple, and quick to perform and does not require highly specialized equipment. The low starting number of schistosomula in the assay and the possibility of interim sampling render the methodology particularly attractive for throughput screening to identify hit compounds which warrant further tests of activity in assays involving animals. Additionally, commercial products which enable the easy and rapid implementation of the lactate assay in drug discovery programs are already available. This approach facilitates the standardization and harmonization of procedures and allows comparison of compound activities generated in drug screening collaborations at different laboratories and at different times.
Supplementary Material
ACKNOWLEDGMENT
We are grateful to Steffen Borrmann, who critically reviewed the manuscript.
Footnotes
Supplemental material for this article may be found at http://dx.doi.org/10.1128/AAC.03809-14.
REFERENCES
- 1.Hotez PJ, Fenwick A, Savioli L, Molyneux DH. 2009. Rescuing the bottom billion through control of neglected tropical diseases. Lancet 373:1570–1575. doi: 10.1016/S0140-6736(09)60233-6. [DOI] [PubMed] [Google Scholar]
- 2.Doenhoff MJ, Pica-Mattoccia L. 2006. Praziquantel for the treatment of schistosomiasis: its use for control in areas with endemic disease and prospects for drug resistance. Expert Rev Anti Infect Ther 4:199–210. doi: 10.1586/14787210.4.2.199. [DOI] [PubMed] [Google Scholar]
- 3.WHO. 2013. Schistosomiasis: number of people treated in 2011. Wkly Epidemiol Rec 88:81–88. [PubMed] [Google Scholar]
- 4.WHO. 2013. New agreement expands access to schistosomiasis treatment for millions. WHO, Geneva, Switzerland: http://www.who.int/neglected_diseases/schistosomiasis_Merck_2013/en/index.html Accessed 2 October 2013. [Google Scholar]
- 5.Fallon PG, Sturrock RF, Niang AC, Doenhoff MJ. 1995. Short report: diminished susceptibility to praziquantel in a Senegal isolate of Schistosoma mansoni. Am J Trop Med Hyg 53:61–62. [PubMed] [Google Scholar]
- 6.De Clercq D, Vercruysse J, Picquet M, Shaw DJ, Diop M, Ly A, Gryseels B. 1999. The epidemiology of a recent focus of mixed Schistosoma haematobium and Schistosoma mansoni infections around the ‘Lac de Guiers’ in the Senegal River Basin, Senegal. Trop Med Int Health 4:544–550. doi: 10.1046/j.1365-3156.1999.00444.x. [DOI] [PubMed] [Google Scholar]
- 7.Wang W, Wang L, Liang Y-S. 2012. Susceptibility or resistance of praziquantel in human schistosomiasis: a review. Parasitol Res 111:1871–1877. doi: 10.1007/s00436-012-3151-z. [DOI] [PubMed] [Google Scholar]
- 8.Keiser J. 2010. In vitro and in vivo trematode models for chemotherapeutic studies. Parasitology 137:589–603. doi: 10.1017/S0031182009991739. [DOI] [PubMed] [Google Scholar]
- 9.Lee H, Moody-Davis A, Saha U, Suzuki BM, Asarnow D, Chen S, Arkin M, Caffrey CR, Singh R. 2012. Quantification and clustering of phenotypic screening data using time-series analysis for chemotherapy of schistosomiasis. BMC Genomics 13(Suppl 1):S4. doi: 10.1186/1471-2164-13-S1-S4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Magalhães LG, Machado CB, Morais ER, de Carvalho Moreira EB, Soares CS, da Silva SH, Da Silva-Filho AA, Rodrigues V. 2009. In vitro schistosomicidal activity of curcumin against Schistosoma mansoni adult worms. Parasitol Res 104:1197–1201. doi: 10.1007/s00436-008-1311-y. [DOI] [PubMed] [Google Scholar]
- 11.Manneck T, Braissant O, Haggenmüller Y, Keiser J. 2011. Isothermal microcalorimetry to study drugs against Schistosoma mansoni. J Clin Microbiol 49:1217–1225. doi: 10.1128/JCM.02382-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Mansour NR, Bickle QD. 2010. Comparison of microscopy and Alamar blue reduction in a larval based assay for schistosome drug screening. PLoS Negl Trop Dis 4:e795. doi: 10.1371/journal.pntd.0000795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Peak E, Chalmers IW, Hoffmann KF. 2010. Development and validation of a quantitative, high-throughput, fluorescent-based bioassay to detect schistosoma viability. PLoS Negl Trop Dis 4:e759. doi: 10.1371/journal.pntd.0000759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.McCarthy M. 2007. The Global Fund: 5 years on. Lancet 370:307–308. doi: 10.1016/S0140-6736(07)61144-1. [DOI] [PubMed] [Google Scholar]
- 15.Manneck T, Braissant O, Ellis W, Keiser J. 2011. Schistosoma mansoni: antischistosomal activity of the four optical isomers and the two racemates of mefloquine on schistosomula and adult worms in vitro and in vivo. Exp Parasitol 127:260–269. doi: 10.1016/j.exppara.2010.08.011. [DOI] [PubMed] [Google Scholar]
- 16.Paveley RA, Mansour NR, Hallyburton I, Bleicher LS, Benn AE, Mikic I, Guidi A, Gilbert IH, Hopkins AL, Bickle QD. 2012. Whole organism high-content screening by label-free, image-based Bayesian classification for parasitic diseases. PLoS Negl Trop Dis 6:e1762. doi: 10.1371/journal.pntd.0001762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Collins JJ, Wang B, Lambrus BG, Tharp ME, Iyer H, Newmark PA. 2013. Adult somatic stem cells in the human parasite Schistosoma mansoni. Nature 494:476–479. doi: 10.1038/nature11924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Shaw MK, Erasmus DA. 1987. Schistosoma mansoni: structural damage and tegumental repair after in vivo treatment with praziquantel. Parasitology 94(Pt 2):243–254. doi: 10.1017/S0031182000053920. [DOI] [PubMed] [Google Scholar]
- 19.Shapiro TA, Talalay P. 1982. Schistosoma mansoni: mechanisms in regulation of glycolysis. Exp Parasitol 54:379–390. doi: 10.1016/0014-4894(82)90047-9. [DOI] [PubMed] [Google Scholar]
- 20.Thompson DP, Morrison DD, Pax RA, Bennett JL. 1984. Changes in glucose metabolism and cyanide sensitivity in Schistosoma mansoni during development. Mol Biochem Parasitol 13:39–51. doi: 10.1016/0166-6851(84)90100-2. [DOI] [PubMed] [Google Scholar]
- 21.Bueding E. 1950. Carbohydrate metabolism of Schistosoma mansoni. J Gen Physiol 33:475–495. doi: 10.1085/jgp.33.5.475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Krautz-Peterson G, Simoes M, Faghiri Z, Ndegwa D, Oliveira G, Shoemaker CB, Skelly PJ. 2010. Suppressing glucose transporter gene expression in schistosomes impairs parasite feeding and decreases survival in the mammalian host. PLoS Pathog 6:e1000932. doi: 10.1371/journal.ppat.1000932. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Horemans AM, Tielens AG, van den Bergh SG. 1992. The reversible effect of glucose on the energy metabolism of Schistosoma mansoni cercariae and schistosomula. Mol Biochem Parasitol 51:73–79. doi: 10.1016/0166-6851(92)90202-U. [DOI] [PubMed] [Google Scholar]
- 24.Faghiri Z, Camargo SMR, Huggel K, Forster IC, Ndegwa D, Verrey F, Skelly PJ. 2010. The tegument of the human parasitic worm Schistosoma mansoni as an excretory organ: the surface aquaporin SmAQP is a lactate transporter. PLoS One 5:e10451. doi: 10.1371/journal.pone.0010451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Massie CE, Lynch A, Ramos-Montoya A, Boren J, Stark R, Fazli L, Warren A, Scott H, Madhu B, Sharma N, Bon H, Zecchini V, Smith D-M, Denicola GM, Mathews N, Osborne M, Hadfield J, Macarthur S, Adryan B, Lyons SK, Brindle KM, Griffiths J, Gleave ME, Rennie PS, Neal DE, Mills IG. 2011. The androgen receptor fuels prostate cancer by regulating central metabolism and biosynthesis. EMBO J 30:2719–2733. doi: 10.1038/emboj.2011.158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Ramalho-Pinto FJ, Gazzinelli G, Howells RE, Mota-Santos TA, Figueiredo EA, Pellegrino J. 1974. Schistosoma mansoni: defined system for stepwise transformation of cercaria to schistosomule in vitro. Exp Parasitol 36:360–372. doi: 10.1016/0014-4894(74)90076-9. [DOI] [PubMed] [Google Scholar]
- 27.Abdulla M-H, Ruelas DS, Wolff B, Snedecor J, Lim K-C, Xu F, Renslo AR, Williams J, McKerrow JH, Caffrey CR. 2009. Drug discovery for schistosomiasis: hit and lead compounds identified in a library of known drugs by medium-throughput phenotypic screening. PLoS Negl Trop Dis 3:e478. doi: 10.1371/journal.pntd.0000478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.R Development Core Team. 2008. R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. [Google Scholar]
- 29.Faghiri Z, Skelly PJ. 2009. The role of tegumental aquaporin from the human parasitic worm, Schistosoma mansoni, in osmoregulation and drug uptake. FASEB J 23:2780–2789. doi: 10.1096/fj.09-130757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Sabah AA, Fletcher C, Webbe G, Doenhoff MJ. 1986. Schistosoma mansoni: chemotherapy of infections of different ages. Exp Parasitol 61:294–303. doi: 10.1016/0014-4894(86)90184-0. [DOI] [PubMed] [Google Scholar]
- 31.Doenhoff MJ, Cioli D, Utzinger J. 2008. Praziquantel: mechanisms of action, resistance and new derivatives for schistosomiasis. Curr Opin Infect Dis 21:659–667. doi: 10.1097/QCO.0b013e328318978f. [DOI] [PubMed] [Google Scholar]
- 32.Manneck T, Haggenmüller Y, Keiser J. 2010. Morphological effects and tegumental alterations induced by mefloquine on schistosomula and adult flukes of Schistosoma mansoni. Parasitology 137:85–98. doi: 10.1017/S0031182009990965. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.