Abstract
Background:
The aim of this study was to analyse and compare the activity of factor V, VIII and fibrinogen level in fresh frozen plasma and frozen plasma frozen after 8 hrs but within 24 hours after phlebotomy.
Materials and Methods:
Fresh frozen plasma separated from whole blood within 8 hours was compared with plasma separated within 24 hours after phlebotomy in terms of coagulation factors V and VIII and level of fibrinogen by standard methods using semi automated coagulometer sysmex CA50.
Results:
Longer storage of whole blood before processing resulted in significant decrease (18.4%) in activity of factor VIII but the fall in activity of factor V (6.52%) or level of fibrinogen (1.81%) was not significant.
Discussion:
These data suggest that there is good retention of coagulation factors in both types of plasma. Although there is significant fall in activity of factor VIII, but it is an acute phase reactant and raised in most of the diseases so it is suggested that frozen plasma would be an acceptable product for most patients requiring fresh frozen plasma.
Keywords: Factor V, Factor VIII, fibrinogen, fresh frozen plasma, frozen plasma
Introduction
Plasma is the aqueous component of blood in which many different cellular elements and macromolecules are suspended, but it is the proteins that have been the focus of interest for transfusion medicine, including specifically albumin, coagulation factors, and immunoglobulins.[1] Plasma can be of different types such as fresh frozen plasma (FFP), plasma frozen within 24 h, single donor plasma, cryoprecipitate – reduced plasma, liquid plasma, recovered plasma, pathogen inactivated plasma, and thawed plasma. FFP is human donor plasma frozen within a short specified period after collection (often 8 h).[1] Plasma frozen at slightly later intervals (typically up to 24 h) after collection is referred to as frozen plasma (FP) or plasma frozen within 24 h after phlebotomy (FP24).[1] There is increasing international interest in producing blood components from whole blood that has been stored for 24 h since this not only offers operational flexibility and efficiency, but could indirectly enhance component safety, this would maximize the ability to use plasma from male donors for pooling buffy coat platelets and to produce plasma components to reduce the risk of transfusion-related acute lung injury (TRALI), since antibodies against human leukocyte antigens in the plasma of female donors are the principle cause of TRALI.[2] The purpose of this study was to analyze and compare the difference in labile coagulation factors and fibrinogen in FFP and FP.
Materials and Methods
Blood collection and processing
The study was conducted in the Department of Immunohematology and Blood Transfusion Medicine, Government Medical College, Jammu. This is a regional blood transfusion center and the study extended over a period of 1 year. Only those donors fulfilling the eligibility criteria were made to donate blood. Blood was collected by a clean, single venipuncture in either double bag or SAGM/Adsol quadruple bag. Flow of blood was rapid and constant. Total time taken to collect 450 ml of blood was never more than 8 min. FFP was prepared from whole blood in Heraeus Cryofuge 6000i centrifuge by centrifuging at either heavy spin (5000 ×g for 5 min) or light spin (2000 ×g for 3 min). Study design is shown in Figure 1.
Figure 1.
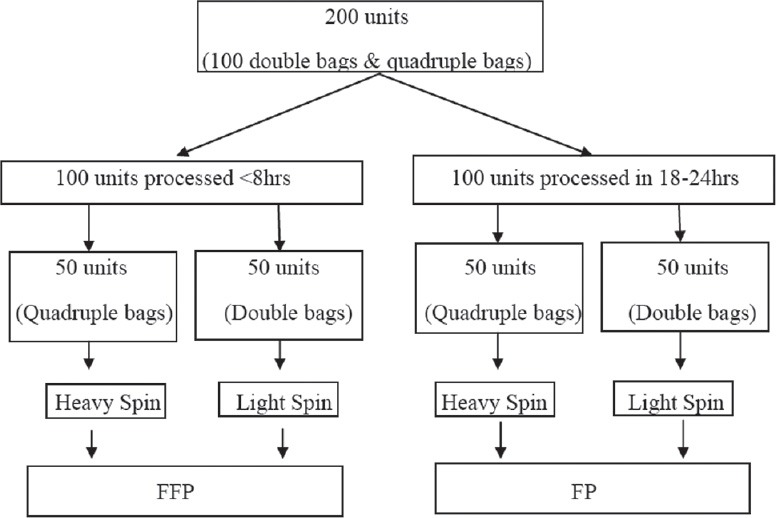
Algorithm to show the study design
Coagulation studies
Assessment of both FFP and FP were done. Following coagulation factors were studied: factor V, f VIII, fibrinogen on Sysmex CA-50 coagulometer. Tests were run as per the manufacturer's instructions that measured clot formation. Factor V was assessed using factor V deficient plasma and Thromborel S-PT reagent (Seimen Diagnostic, Marburg, Germany). For factor VIII assessment, factor VIII deficient plasma, calcium chloride and actin-aPTT reagent (seimen diagnostic, Marburg, Germany) was used. The fibrinogen assay was a modified Claus procedure using Thrombin reagent (Seimen Diagnostic, Marburg, Germany). The results were interpreted using a reference curve obtained with dilutions of standard human plasma mixed with deficient plasma. Positive and negative controls were used along with the test run.
Statistical analysis
Fresh frozen plasma and FP were compared using comparison of means. A P < 0.05 was considered as significant.
Results
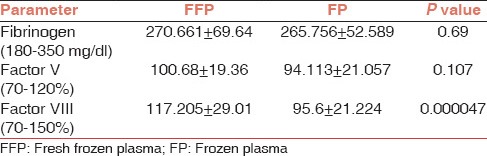
A total of 100 units of FFP were compared with 100 units of FP in terms of fibrinogen level which is a stable coagulation factor and activity of Factor V and VIII, which are labile factors. As expected, longer storage of whole blood before processing resulted in significant decrease (18.4%) in activity of factor VIII, but the fall in activity of Factor V (6.52%) or fibrinogen level (1.81%) was not significant [Table 1].
Table 1.
Comparison of clotting factors in FFP and FP
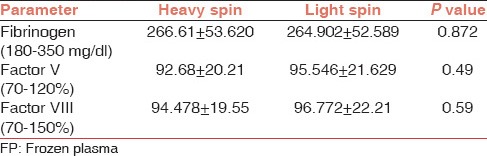
In this study, plasma prepared by heavy spin and light spin were also compared in terms of level of fibrinogen and activity of Factor V and VIII. Tables 2 and 3 show a comparison of coagulation factors centrifuged at heavy spin and light spin. No significant differences were noted between product types.
Table 2.
Comparisons of clotting factors in FFP prepared by centrifuging at heavy spin and light spin

Table 3.
Comparisons of clotting factors in FP prepared by centrifuging at heavy spin and light spin
Discussion
This study assessed the effect of storing whole blood overnight at 4°C on the coagulation factors, that is, on the level of fibrinogen and activity of factor V and f VIII. Factor V and f VIII are known labile factors, whereas fibrinogen is a stable factor. Factor VIII level is also a quality control parameter required for FFP as per Directorate General Of Health Services (DGHS).[3] In our study, the most significantly affected factor was factor VIII, which showed a loss of 18.4%, when FFP was compared with FP as also reported by Alakech et al.,[4] Sheffield et al.,[5] Alhumaidan et al.,[6] Kakaiya et al.,[7] Smith et al.,[8] Cardigan et al.,[9] and Agus et al.[10] Loss of 6.52% was seen in the activity of Factor V, which was not significant. The result was comparable to the studies done by Alhumaidan et al.,[6] Sheffield et al.,[5] and Smith et al., however Cardigan et al.[9] showed a loss of 15% which was significant. Compared to FFP, fall in fibrinogen levels to 1.81% was observed in FP although the loss was not significant as also reported by Alhumaidan et al.,[6] Sheffield et al.[5] where the fall was insignificant, whereas Cardigan et al.[9] showed a loss of 12%, which was significant where slight differences in testing methods between the studies might have accounted for loss of fibrinogen.
Plasma prepared by centrifuging at heavy spin and light spin were comparable in terms of fibrinogen level and activity of factor V and VIII as no statistically significant difference was found between them, however Serrano et al.[11] found significant fall in factor VIII and elevation in fibrinogen levels in plasma prepared by buffy coat method. Sheffield et al.[5] showed improved quality of FP – buffy coat over FP – platelet rich plasma, specifically a minor elevation in FP – buffy coat factor V levels although this effect was limited to 2007 data set.
Plasma for transfusion is most often used where there is abnormal coagulation screening tests, either therapeutically in the face of bleeding, or prophylactically in nonbleeding patients prior to invasive procedures or surgery.[12] The common acquired coagulopathies are liver disease,[13,14,15] vitamin K antagonist coagulopathy,[16,17] disseminated intravascular coagulation (DIC),[18] and dilutional coagulopathy.[19] Clotting factor replacement in the acquired coagulopathies is prescribed either prophylactically before an invasive procedure or therapeutically in the context of active bleeding.[13,18] Two of these coagulopathies are associated with normal or high level of factor VIII (liver disease, vitamin K antagonist coagulopathy) and thus factor VIII replacement is not a consideration.[14,17] Acute DIC and dilutional coagulopathy may be associated with transient decrease in factor VIII levels.[18,19] However, factor VIII is an acute – phase protein which, if decreased, will rebound rapidly.[6] Hence, the loss of coagulation factors observed is unlikely to be clinically significant and that FP can be used for the same indications as FFP. Furthermore, method of preparation of plasma whether it is prepared by centrifugation at heavy spin or light spin also does not affect the quality of plasma, which is true for both FFP and FP. Knowledge of quality of blood components will be effective for the efficient management of blood bank's inventory and data generated by this study will help us in modifying the guidelines of blood transfusion followed at our center for the betterment of patients.
Footnotes
Source of Support: Institutional
Conflicting Interest: None declared.
References
- 1.Stanworth SJ, Tinmouth AT. Plasma transfusion and use of albumin. In: Simon TL, editor. Rossi's Principles of Transfusion Medicine. 4th ed. Oxford, West Sussex, New Jersey: Blackwell Publishing; 2009. pp. 287–97. [Google Scholar]
- 2.Cardigan R, Van der Meer PF, Pergande C, Cookson P, Baumann-Baretti B, Cancelas JA, et al. Coagulation factor content of plasma produced from whole blood stored for 24 hours at ambient temperature: Results from an international multicenter BEST Collaborative study. Transfusion. 2011;51(Suppl 1):50S–7. doi: 10.1111/j.1537-2995.2010.02963.x. [DOI] [PubMed] [Google Scholar]
- 3.Saran RK, editor. Technical Manual. 2nd ed. New Delhi: Directorate General of Health Services; 2003. Quality assurance in blood transfusion; p. 354. [Google Scholar]
- 4.Alakech B, Miller B, Berry TH, Ambruso DR. Coagulation profile for cryoprecipitate produced from 24-hour stored whole blood. Lab Med. 2009;40:540–3. [Google Scholar]
- 5.Sheffield WP, Bhakta V, Jenkins C, Devine DV. Conversion to the buffy coat method and quality of frozen plasma derived from whole blood donations in Canada. Transfusion. 2010;50:1043–9. doi: 10.1111/j.1537-2995.2009.02555.x. [DOI] [PubMed] [Google Scholar]
- 6.Alhumaidan H, Cheves T, Holme S, Sweeney J. Stability of coagulation factors in plasma prepared after a 24-hour room temperature hold. Transfusion. 2010;50:1934–42. doi: 10.1111/j.1537-2995.2010.02648.x. [DOI] [PubMed] [Google Scholar]
- 7.Kakaiya RM, Morse EE, Panek S. Labile coagulation factors in thawed fresh frozen plasma prepared by two methods. Vox Sang. 1984;46:44–6. doi: 10.1111/j.1423-0410.1984.tb00046.x. [DOI] [PubMed] [Google Scholar]
- 8.Smith JF, Ness PM, Moroff G, Luban NL. Retention of coagulation factors in plasma frozen after extended holding at 1-6 degrees C. Vox Sang. 2000;78:28–30. doi: 10.1159/000031145. [DOI] [PubMed] [Google Scholar]
- 9.Cardigan R, Lawrie AS, Mackie IJ, Williamson LM. The quality of fresh-frozen plasma produced from whole blood stored at 4 degrees C overnight. Transfusion. 2005;45:1342–8. doi: 10.1111/j.1537-2995.2005.00219.x. [DOI] [PubMed] [Google Scholar]
- 10.Agus N, Yilmaz N, Colak A, Liv F. Levels of factor VIII and factor IX in fresh-frozen plasma produced from whole blood stored at 4°C overnight in Turkey. Blood Transfus. 2012;10:191–3. doi: 10.2450/2011.0050-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Serrano K, Scammell K, Weiss S, Culibrk B, Levin E, Gyöngyössy-Issa M, et al. Plasma and cryoprecipitate manufactured from whole blood held overnight at room temperature meet quality standards. Transfusion. 2010;50:344–53. doi: 10.1111/j.1537-2995.2009.02441.x. [DOI] [PubMed] [Google Scholar]
- 12.Stanworth SJ. The evidence-based use of FFP and cryoprecipitate for abnormalities of coagulation tests and clinical coagulopathy. Hematology Am Soc Hematol Educ Program. 2007:179–86. doi: 10.1182/asheducation-2007.1.179. [DOI] [PubMed] [Google Scholar]
- 13.Youssef WI, Salazar F, Dasarathy S, Beddow T, Mullen KD. Role of fresh frozen plasma infusion in correction of coagulopathy of chronic liver disease: A dual phase study. Am J Gastroenterol. 2003;98:1391–4. doi: 10.1111/j.1572-0241.2003.07467.x. [DOI] [PubMed] [Google Scholar]
- 14.Spector I, Corn M, Ticktin HE. Effect of plasma transfusions on the prothrombin time and clotting factors in liver disease. N Engl J Med. 1966;275:1032–7. doi: 10.1056/NEJM196611102751902. [DOI] [PubMed] [Google Scholar]
- 15.Ragni MV, Lewis JH, Spero JA, Hasiba U. Bleeding and coagulation abnormalities in alcoholic cirrhotic liver disease. Alcohol Clin Exp Res. 1982;6:267–74. doi: 10.1111/j.1530-0277.1982.tb04973.x. [DOI] [PubMed] [Google Scholar]
- 16.Ozgonenel B, O’Malley B, Krishen P, Eisenbrey AB. Warfarin reversal emerging as the major indication for fresh frozen plasma use at a tertiary care hospital. Am J Hematol. 2007;82:1091–4. doi: 10.1002/ajh.20902. [DOI] [PubMed] [Google Scholar]
- 17.Preston FE, Laidlaw ST, Sampson B, Kitchen S. Rapid reversal of oral anticoagulation with warfarin by a prothrombin complex concentrate (Beriplex): Efficacy and safety in 42 patients. Br J Haematol. 2002;116:619–24. doi: 10.1046/j.0007-1048.2001.03295.x. [DOI] [PubMed] [Google Scholar]
- 18.Saba HI, Morelli GA. The pathogenesis and management of disseminated intravascular coagulation. Clin Adv Hematol Oncol. 2006;4:919–26. [PubMed] [Google Scholar]
- 19.Murray DJ, Pennell BJ, Weinstein SL, Olson JD. Packed red cells in acute blood loss: Dilutional coagulopathy as a cause of surgical bleeding. Anesth Analg. 1995;80:336–42. doi: 10.1097/00000539-199502000-00022. [DOI] [PubMed] [Google Scholar]


