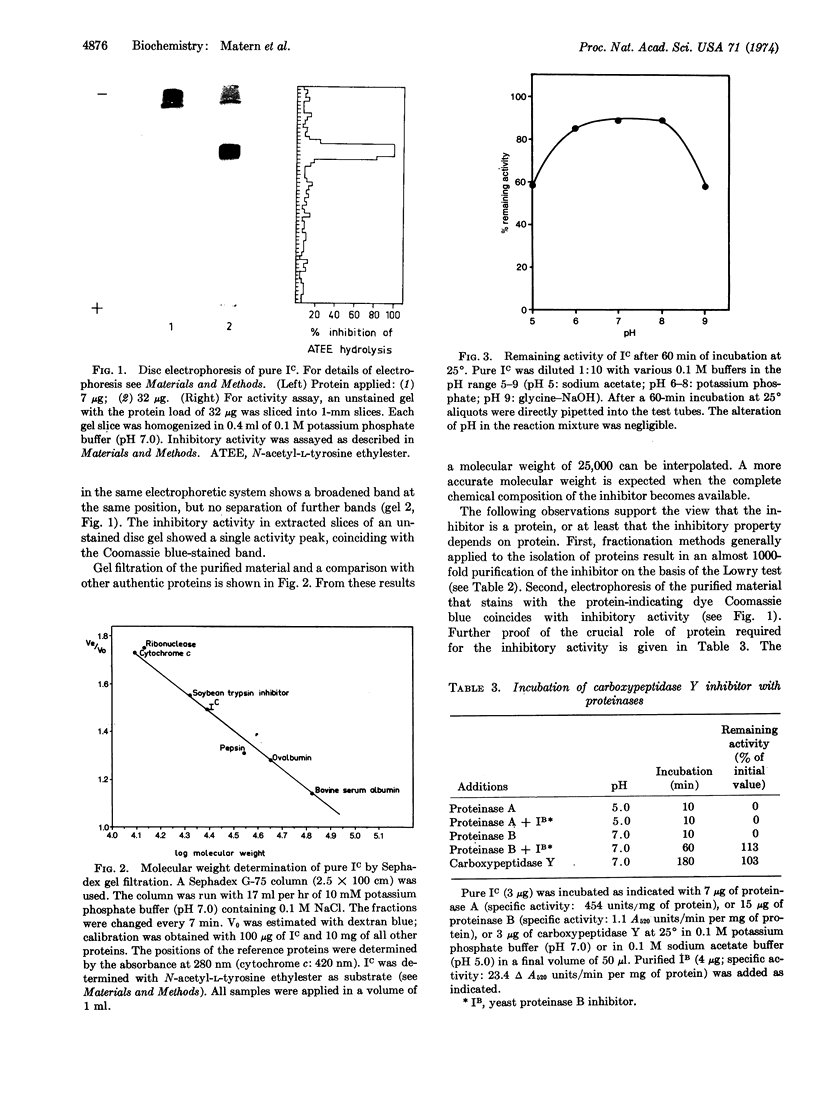
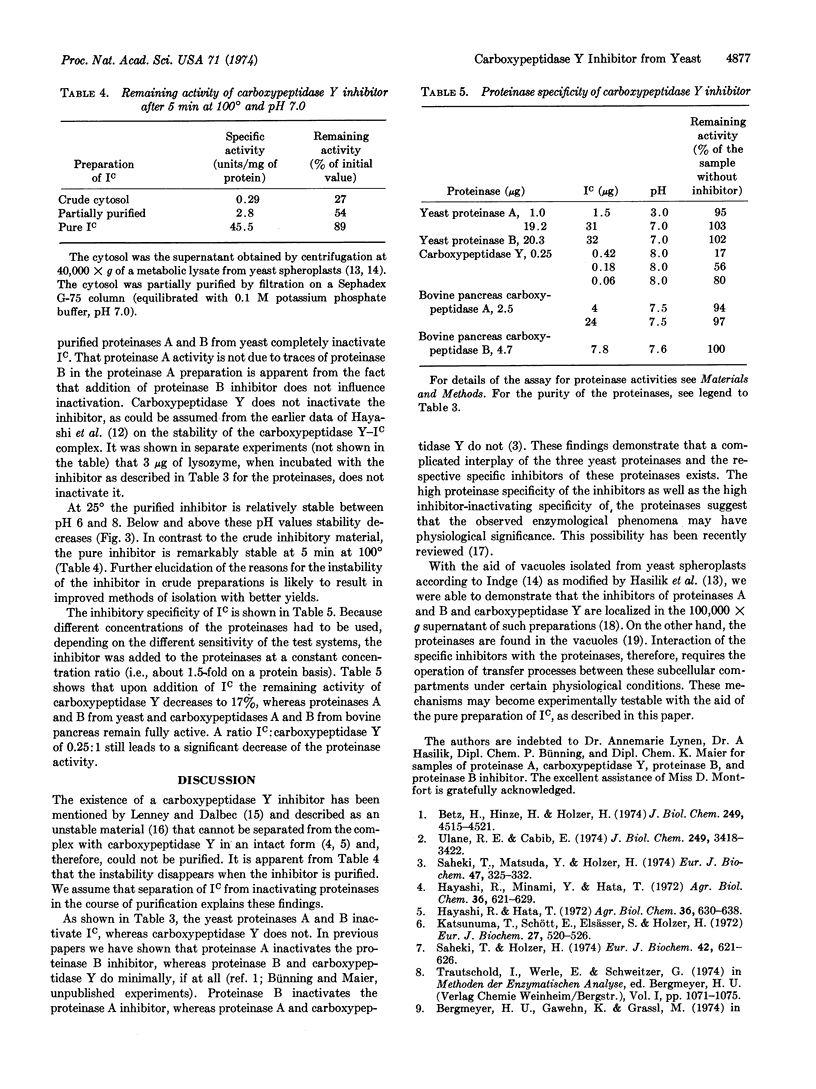
Abstract
Rapid acetone fractionation of crude yeast extract at low temperature separates carboxypeptidase Y inhibitor from the carboxypeptidase Y-inhibitor complex. On a protein basis the inhibitor has been purified 890-fold, resulting in homogeneity as determined by disc electrophoresis and filtration on Sephadex G-75. The molecular weight was calculated to be about 25,000. The inhibitor is heat-labile in crude extracts, whereas in the purified form it loses only 11% of its activity when it is heated at 100° for 5 min. The inhibitor is inactivated by the yeast proteinases A (EC 3.4.23.8) and B (EC 3.4.22.9), respectively, but not by carboxypeptidase Y (EC 3.4.12.8). The inhibitor inhibits carboxypeptidase Y at a 1.5:1.0 protein-based weight ratio by 80%. At the same concentration ratios, proteinases A and B from yeast, as well as bovine pancreas carboxypeptidases A and B, are not inhibited.
Keywords: proteinase inhibitor
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Betz H., Hinze H., Holzer H. Isolation and properties of two inhibitors of proteinase B from yeast. J Biol Chem. 1974 Jul 25;249(14):4515–4521. [PubMed] [Google Scholar]
- Hasilik A., Müller H., Holzer H. Compartmentation of the tryptophan-synthase-proteolyzing system in Saccharomyces cerevisiae. Eur J Biochem. 1974 Oct 1;48(1):117–117. doi: 10.1111/j.1432-1033.1974.tb03748.x. [DOI] [PubMed] [Google Scholar]
- Indge K. J. The isolation and properties of the yeast cell vacuole. J Gen Microbiol. 1968 May;51(3):441–446. doi: 10.1099/00221287-51-3-441. [DOI] [PubMed] [Google Scholar]
- Katsunuma T., Schött E., Elsässer S., Holzer H. Purification and properties of tryptophan-synthase-inactivating enzymes from yeast. Eur J Biochem. 1972 Jun 9;27(3):520–526. doi: 10.1111/j.1432-1033.1972.tb01868.x. [DOI] [PubMed] [Google Scholar]
- Lenney J. F., Dalbec J. M. Yeast proteinase B: identification of the inactive form as an enzyme-inhibitor complex. Arch Biochem Biophys. 1969 Jan;129(1):407–409. doi: 10.1016/0003-9861(69)90194-5. [DOI] [PubMed] [Google Scholar]
- Matern H., Betz H., Holzer H. Compartmentation of inhibitors of proteinases A and B and carboxypeptidase Y in yeast. Biochem Biophys Res Commun. 1974 Oct 8;60(3):1051–1057. doi: 10.1016/0006-291x(74)90419-7. [DOI] [PubMed] [Google Scholar]
- Matile P., Wiemken A. The vacuole as the lysosome of the yeast cell. Arch Mikrobiol. 1967 Feb 20;56(2):148–155. doi: 10.1007/BF00408765. [DOI] [PubMed] [Google Scholar]
- Saheki T., Holzer H. Comparisons of the tryptophan synthase inactivating enzymes with proteinases from yeast. Eur J Biochem. 1974 Mar 1;42(2):621–626. doi: 10.1111/j.1432-1033.1974.tb03377.x. [DOI] [PubMed] [Google Scholar]
- Saheki T., Matsuda Y., Holzer H. Urification and characterization of macromolecular inhibitors of proteinase A from yeast. Eur J Biochem. 1974 Sep 1;47(2):325–332. doi: 10.1111/j.1432-1033.1974.tb03697.x. [DOI] [PubMed] [Google Scholar]
- Ulane R. E., Cabib E. The activating system of chitin synthetase from Saccharomyces cerevisiae. Purification and properties of an inhibitor of the activating factor. J Biol Chem. 1974 Jun 10;249(11):3418–3422. [PubMed] [Google Scholar]