Abstract
Purpose
Epstein-Barr virus (EBV) is associated with several cancers in which the tumour cells express EBV antigens EBNA1 and LMP2. A therapeutic vaccine comprising a recombinant vaccinia virus, MVA-EL, was designed to boost immunity to these tumour antigens. A phase I trial was conducted to demonstrate the safety and immunogenicity of MVA-EL across a range of doses.
Experimental Design
Sixteen patients in the United Kingdom (UK) with EBV-positive nasopharyngeal carcinoma (NPC), received three intradermal vaccinations of MVA-EL at 3-weekly intervals at dose levels between 5×107 and 5×108 plaque forming units (pfu). Blood samples were taken at screening, after each vaccine cycle and during the post-vaccination period. T-cell responses were measured using IFNγ ELISpot assays with overlapping EBNA1/LMP2 peptide mixes or HLA-matched epitope peptides. Polychromatic flow cytometry was used to characterize functionally responsive T-cell populations.
Results
Vaccination was generally well-tolerated. Immunity increased after vaccination to at least one antigen in 8/14 patients (7/14, EBNA1; 6/14, LMP2), including recognition of epitopes that vary between EBV strains associated with different ethnic groups. Immunophenotypic analysis revealed that vaccination induced differentiation and functional diversification of responsive T-cell populations specific for EBNA1 and LMP2 within the CD4 and CD8 compartments respectively.
Conclusions
MVA-EL is safe and immunogenic across diverse ethnicities and thus suitable for use in trials against different EBV-positive cancers globally as well as in South East Asia where NPC is most common. The highest dose (5×108 pfu) is recommended for investigation in current phase IB and II trials.
Introduction
Approximately one fifth of human cancers are causally linked with infectious agents (1). Epstein-Barr virus (EBV) is harboured as a life-long asymptomatic infection in most adults but has potent growth transforming capability (2). It is aetiologically linked to several human tumours, including undifferentiated nasopharyngeal carcinoma (NPC), an epithelial malignancy common throughout South-East Asia that also occurs at lower incidence rates worldwide (1,2). The possibility of exploiting EBV-specific T-cell immunity as a novel treatment for NPC is attractive because disease-free survival rates at 2 and 5 years with current chemoradiotherapy regimens are still only 63% and 52%, respectively; furthermore, distant metastases account for over 40% of recurrences and confer a poor outlook (3-6). However, most NPCs express just two EBV latent cycle proteins: (i) the Epstein-Barr nuclear antigen 1 (EBNA1), a sequence-specific DNA binding protein involved in maintenance of the episomal virus genome; and (ii) latent membrane protein 2 (LMP2), a membrane signalling protein with growth-promoting activity in epithelial cells (2). In other EBV genome-positive tumours, such as all extranodal NK/T lymphomas (ENKTLs), up to 40% of Hodgkin lymphomas (HLs) and 5-10% of gastric carcinomas (GCs), EBNA1 and LMP2 are likewise among the limited range of expressed viral proteins, emphasizing the broader importance of these antigens as targets for immunotherapy (7-9).
In light of these considerations, we developed a therapeutic vaccine (MVA-EL) using the modified vaccinia Ankara (MVA) vector to encode a functionally inactive fusion protein of full-length LMP2 and the C-terminal half of EBNA1 (10). The latter region of EBNA1 contains almost all of the protein’s known CD4 and CD8 T-cell epitopes, but lacks the glycine-alanine repeat domain that partially impairs antigen presentation to CD8 T-cells (11-13). We recently reported the outcome of a phase I dose escalation trial conducted in a Hong Kong-based cohort of eighteen Chinese NPC patients vaccinated after initial chemoradiotherapy for their disease. In many cases, vaccination resulted in substantial expansions of circulating T-cells specific for EBNA1 and/or LMP2; moreover, the numbers of responders and size of responses across the cohort both suggested a positive association with vaccine dose (14).
Virus-induced T-cell responses are specifically directed against particular combinations of HLA molecules and bound viral peptides. Any global approach to T-cell-based targeting of virus-associated tumours must therefore deal with the twin challenges of HLA diversity and viral heterogeneity. Differences between human populations in terms of HLA allele distribution will alter epitope selection (15-17), while polymorphisms between circulating viral strains can affect epitope processing and presentation (18-23). In this regard, the MVA-EL vaccine was constructed using EBNA1 and LMP2 gene sequences cloned from typical Chinese-origin EBV strains. These proteins differ from their European-origin EBV counterparts at various residues, some of which lie within known T-cell epitopes (15, 24). To what extent the vaccine would be able to boost epitope-specific responses in European patients therefore remained an open question.
To address this issue, we undertook a dose escalation phase I trial of MVA-EL vaccination in the United Kingdom (UK). Eligible patients with EBV-positive (EBV+) tumours were either in remission or harboured low volume residual disease for which no standard therapy was indicated. The objectives were to determine the safety profile and immunogenicity of MVA-EL vaccination and to assess changes in circulating cell-free EBV genome measurements over time. Here, we report that the vaccine was immunogenic and well tolerated in UK patients, potentially inducing the release of cell-free EBV genomes suggestive of tumour-specific T-cell-mediated cytotoxic effects. Combined data from 27 patients across both the Hong Kong and UK trials further confirmed the immunogenicity of MVA-EL across HLA subtypes and antigenic variants of EBV, supporting the use of this vaccine in different populations and thus potentially against a wider range of malignancies.
Methods
Patients
Sixteen patients with histologically confirmed EBV+ malignancy, either in first remission or with persisting disease for which no standard therapy was available, were recruited for this study. All patients were in complete (or unconfirmed complete) remission at least 12 weeks after first-line treatment (either definitive chemoradiotherapy or palliative chemotherapy) and met pre-defined inclusion criteria (see Supplementary Information). Conduct was ICH-GCP compliant and all patients gave written informed consent.
Study design
This was a three centre, phase IA trial of MVA-EL vaccination against EBV+ malignancies, employing a 3+3 design across a pre-defined dose escalation schedule of sequential cohorts spanning five dose levels (Supplementary Figure 1). The primary objectives were to determine the safety and toxicity profile of MVA-EL and to describe changes in EBNA1/LMP2-specific functional T-cell response frequencies in peripheral blood before, during and after vaccination. Additional objectives were to assess changes in EBV genome levels and EBNA1-specific antibodies in plasma. Disease response assessment was not an objective, but patients known to have residual or recurrent disease were assessed by cross-sectional imaging at screening and at week 14. Toxicity was graded and dose-limiting toxicity (DLT) defined according to the National Cancer Institute Common Terminology Criteria for Adverse Events (CTCAE) v3.0 using a protocol-specific grading of local injection site reactions. Provided the maximum tolerated dose, i.e. the dose at which 2/6 patients suffered DLT, was not reached, the lowest dose for which there was clear evidence of cellular immunogenicity was to be recommended for further investigation.
Study procedures
Patients received three intradermal vaccinations of MVA-EL at 3-weekly intervals. Blood samples were collected before vaccination (screening) immediately before each vaccination (day 1 cycle 1, 2 or 3), 7 days after each vaccination (day 8, cycle 1, 2 or 3) and immediately post-trial (day 22 and day 29, cycle 3). Peripheral blood mononuclear cell (PBMC) and plasma samples were cryopreserved from each blood draw. T-cell responses were measured simultaneously for each individual patient across all time points via IFNγ ELISpot assays conducted against defined epitope peptides or pools of overlapping 15mer peptides spanning the primary sequence of each vaccine-encoded antigen (pepmixes; JPT Peptide Technologies). Negative controls comprised DMSO solvent alone or an actin pepmix to assess spontaneous IFNγ release. Assignment of specific recognition required that the number of spot forming cells (sfc) observed in response to viral antigen stimulation exceeded defined thresholds, namely ≥10sfc/well and ≥2-fold greater than the negative control. Mean spot counts for negative control wells were subtracted from those for the test wells to generate mean adjusted readings. Vaccine responses were defined as peptide epitope or pepmix recognition for which the adjusted counts were ≥10sfc/well and ≥2-fold higher post-vaccination compared to pre-vaccination baseline.
Antigen-specific T-cell phenotype was determined by stimulating PBMCs for 6hr with antigen pepmixes in the presence of brefeldin A, monensin and αCD107a-FITC antibody. Cells were then washed, stained with additional antibodies acquired on a custom-built 20-parameter FACS AriaII flow cytometer (BD Biosciences). Data were analysed with FlowJo software v9.7.5 and SPICE software v5.32 was used for combinatorial analysis and data presentation (25).
EBV DNA loads in plasma were assayed by qPCR. EBNA1 antibodies were measured before and after vaccination using standard assays. PCR and sequencing were used to genotype EBV strains present in patients. Further details of procedures are provided in Supplementary Information.
Statistical methods
Immunogenicity was not defined prospectively. Instead, interpretation of the immunological data was carried out in an exploratory manner using complete longitudinal data for each patient. Full details are provided in Supplementary Information.
Regulatory status
The trial was approved by the Medicines and Healthcare Products Regulatory Agency (MHRA) in 2004 (21106/0006/001 and 21106/0225/001) and by the UK Gene Therapy Advisory Committee (GTAC) in the same year (GTAC066). Sponsorship was provided by Cancer Research UK (Protocol PH1/101) and the trial was registered at clinicaltrials.gov (NCT01147991).
Results
Patients, treatments and outcomes
Sixteen patients with confirmed EBV+ NPC were enrolled from three UK clinical centres between 2005 and 2010. The median age of this cohort was 48 years (range, 28-68 years). All patients had previously undergone either radical locoregional radiotherapy (n=4) or chemoradiotherapy (n=12), in ten cases with adjuvant chemotherapy (Table 1). Eleven patients were described as White and five as South East Asian. To genotype their resident EBV strains we PCR amplified a region of the viral genome used to discriminate between Chinese and European EBV strains (18,19) and that can be reliably amplified from the very low numbers of EBV-infected B cells present in small PBMC samples. Six EBV strain variants were identified in the eight patients who yielded an amplification product. The association between strain variants and ethnic origin (five White and three Asian) were consistent with previously described patterns (Table 1).
Table 1. Diagnosis, staging and previous treatment.
| Trial no. | Age | Sex | Ethnicity | EBV genotype1 | Diagnosis | Stage | Tumour EBV status | Radiotherapy | Chemotherapy | Weeks from last treatment | Previous treatment outcomes |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 0101 | 46 | M | White | AVF/S1L2 IVT/L2 | NPC | T3N0M0 | + | 1. 2003 nasopharynx: 66 Gy/33 + cisplatin 100mg/m(2) week 1, 3 and 6 [R] | 1. 2004 cisplatin 70 mg/m(2) plus 5FU 1000mg/m(2) d1-4 q21/7, 2 cycles [R] | 91 | 1. CR |
| 0102 | 39 | M | White | AVF/wt IVT/wt | NPC | T1N2M0 | + | 1. 2004: external beam dose NK + cisplatin [R]; 2. 2006: nasopharynx brachytherapy 60.1 Gy | 1. 2004: Cisplatin and 5FU | 29 | 1. CR; 2. CR |
| 0103 | 28 | F | South Asian | N.D. | NPC | T2N2cM0 | + | 1. 2004: nasopharynx 65 Gy/30 + cisplatin [R]; 2. 2004: neck 60Gy/30 [R]; 3. 2004: vertebral column 20Gy/5 [P] | 1. 2004: cisplatin and 5FU; 2006: 4. cisplatin and 5FU | 12 | 1. CR; 2. CR; 3. PR; 4. PR |
| 0104 | 41 | F | White | AVF/S1L2 IVT/L2R9 | NPC | Not Known | + | 1. 1999: nasopharynx, neck 64Gy/32 [R]; 2. 2004: nasopharynx brachytherapy 60Gy [R]; 3. 2005: right thyroid and neck 65Gy/30 + cisplatin then carboplatin [R]. | 73 | 1. CR; 2. CR 3. SD | |
| 0105 | 48 | F | White | N.D. | NPC | T1N1M0 | + | 1. 1996 nasopharynx dose NK [R]; 2. 2004: nasopharynx brachytherapy 58.6Gy [R] | 3. 2006: cisplatin then carboplatin, + 5FU | 25 | 1. CR; 2. PR; 3. SD |
| 0106 | 49 | F | White | N.D. | NPC | T3N1M1 | + | 1. 2003: nasopharynx and neck 60Gy/30 [R]; 2. 2004: nasopharynx brachytherapy 42Gy [R]; 3. 2006: stereotactic right parapharyngeal 60Gy/30 [R] | 1. 2003: cisplatin and 5FU [R] | 50 | 1. not-evaluable; 2. CR; 3. SD |
| 0207 | 39 | F | South Asian | AVF/S1L2 IVT/N9 | NPC | T3N2bM0 | + | 1.2006: nasopharynx and neck, dose NK | 1. 2006: NK [R] | 83 | 1.CR |
| 0208 | 55 | M | White | AVF/wt IVT/L2 | NPC | T2bN1M0 | + | 1. 2008: nasopharynx 66Gy/33 + carboplatin 100 mg on weeks 1-5 [R] | 28 | 1. CR | |
| 0209 | 43 | F | White | N.D. | NPC | T1N0M0 | LMP1 IHC + | 1. 2007: nasopharynx 65Gy/30 + carboplatin [R] | 1. 2007: cisplatin + 5FU | 1. CR | |
| 0310 | 68 | M | White | AVF/wt IVT/wt | NPC | T2bN1M0 | EBER ISH + | 1. 2008: nasopharynx 66 Gy/33 + cisplatin [R] | 28 | 1. CR | |
| 0311 | 59 | M | White | N.D. | NPC | T3 N2 | EBER ISH + | 1. 2008: nasopharynx and neck 40Gy/25 and 26Gy/13 + cisplatin [R] | 1. 2008: cisplatin then carboplatin, +5FU | 18 | 1. CR |
| 0212 | 63 | M | White | N.D. | NPC | T2 N0 M0 | EBER ISH + | 1. 2008: nasopharynx 65Gy/30 + 5FU then cisplatin [R] | 29 | 1. CR | |
| 0313 | 49 | M | South Asian | N.D. | NPC | T4 N3 N0 | EBER ISH + | 2. 2007: nasopharynx and neck 66Gy/33 + cisplatin [R] | 1. 2007: cisplatin + 5FU | 69 | 1.not evaluable 2.PR |
| 0414 | 49 | M | South Asian | AVF/P1 IVT/L2 | NPC | T3 N0 M0 | EBER ISH + | 1. 2008: nasopharynx and neck 60Gy/33 + cisplatin [R] | 1. 2008: cisplatin + 5FU [R] | 17 | 1. PR |
| 0415 | 38 | M | White | N.D. | NPC | T2 N2 M0 | EBER ISH + | 1. 2008: neck and nasopharynx 66Gy/33 + cisplatin [R] | 68 | 1. CR | |
| 0516 | 52 | M | South Asian | AVF/S1L2 IVT/N9 | NPC | T2N2M0 | EBER ISH + | 2. 2009: neck & nasopharynx 70Gy/33 + cisplatin [R] | 1. 2009: docetaxol + cisplatin + 5FU [R] | 24 | 1.not evaluable 2. CR |
EBV genotypes as defined in (Midgley et al. J. Virol. 2003, 77(21):11507-11516). wt indicates amino acid sequence is identical to B95.8 type 1 EBV reference sequence.
[R] – radical intent; [P] – palliaive intent; CR – complete response; PR – partial response; SD – stable disease; NK – not known.
Patients received three vaccinations at 3-weekly intervals in dose escalating cohorts as summarized in Supplementary Figure 1. Dose level 1 was expanded after the first patient on the trial experienced a grade 2 injection site reaction (ISR) to the first vaccine cycle. All participants experienced ISR at grade 1 or, in seven cases, at grade 2; these were non-ulcerative, self-limiting lesions that did not worsen across cycles. Nine patients experienced systemic toxicity, in five cases at grade 2 with regional pain and lymphadenopathy (2 patients), fatigue (1 patient) and ‘flu-like symptoms (3 patients). One of these patients (UK0212) withdrew from the trial after the first vaccine dose.
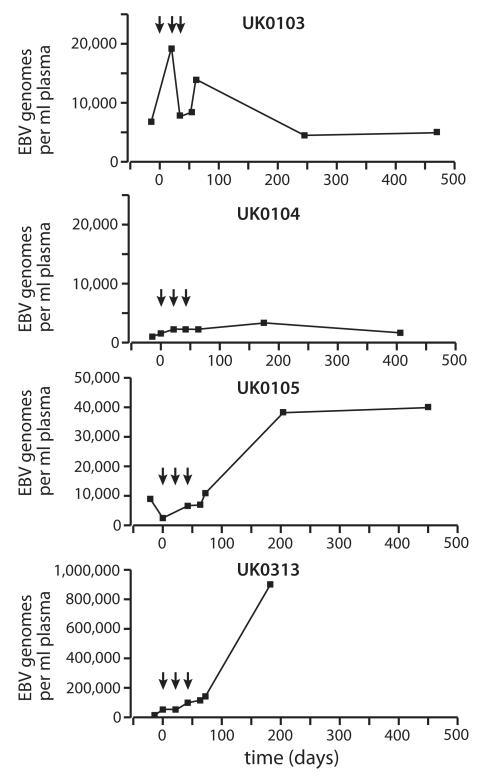
At trial entry, ten patients were in complete remission following primary treatment or, for two patients, second-line therapy (UK0102, UK0516). Six had residual disease following primary treatment (UK0414) or more than one line of treatment (UK0103, UK0104, UK0105, UK0106, UK0313). Of these, plasma samples from four patients (UK0103, UK0104, UK0105, UK0313) contained detectable EBV DNA either before or 4 weeks after vaccination, triggering analysis of the complete sample set (Figure 1). For UK0105 and UK0313, sustained rises in EBV levels were associated with disease progression. Patient UK0104 experienced increasing EBV loads before vaccination that appeared to stabilize initially, although relapse was clinically apparent 9 months after the first dose of MVA-EL. In UK0103, EBV load rose and fell transiently across the vaccination course and then stabilized at a lower level compared to baseline. Clinical relapse was diagnosed 25 months after the first vaccination.
Figure 1. Time course of EBV DNA in plasma.
Four patients had detectable EBV DNA in their pre-vaccination plasma. Following vaccination (indicated by arrows), EBV load increased transiently in one patient (UK0103) and then stabilized at a lower level compared to baseline. In a second patient (UK0104), a rising EBV load before vaccination appeared to stabilize after vaccination. Sustained increases in EBV load associated with disease progression were observed in two further patients (UK0105, UK0313).
Immune responses to vaccination
Total lymphocyte counts remained stable across the vaccination course (pre-cycle 1, mean 0.94×109 cells/L; week 11, mean 1.04×109/L). For most patients, CD3+CD4+ and CD3+CD8+ T-cell subset frequencies were typical of those in healthy controls; however, two patients showed atypical patterns that nonetheless also remained stable (Supplementary Figure 2A). The proportions of circulating CD4+Foxp3+ regulatory T-cells ranged within 3-10% of the total CD4+ T-cell compartment, comparable to values reported for healthy individuals (26). These frequencies did not change significantly with vaccination (Supplementary Figure 2B).
To evaluate T-cell responses against known EBNA1 or LMP2 epitopes restricted by HLA class I or class II molecules, we tested PBMCs from sequential blood samples in IFNγ ELISpot assays. In particular, we looked for evidence of responses against polymorphic epitopes based on B95.8 type I EBV, which is prototypic for prevalent viral strains in Europe but differs at key epitopic sites compared to the equivalent MVA-EL sequences derived from Chinese strains (Supplementary Table 1).
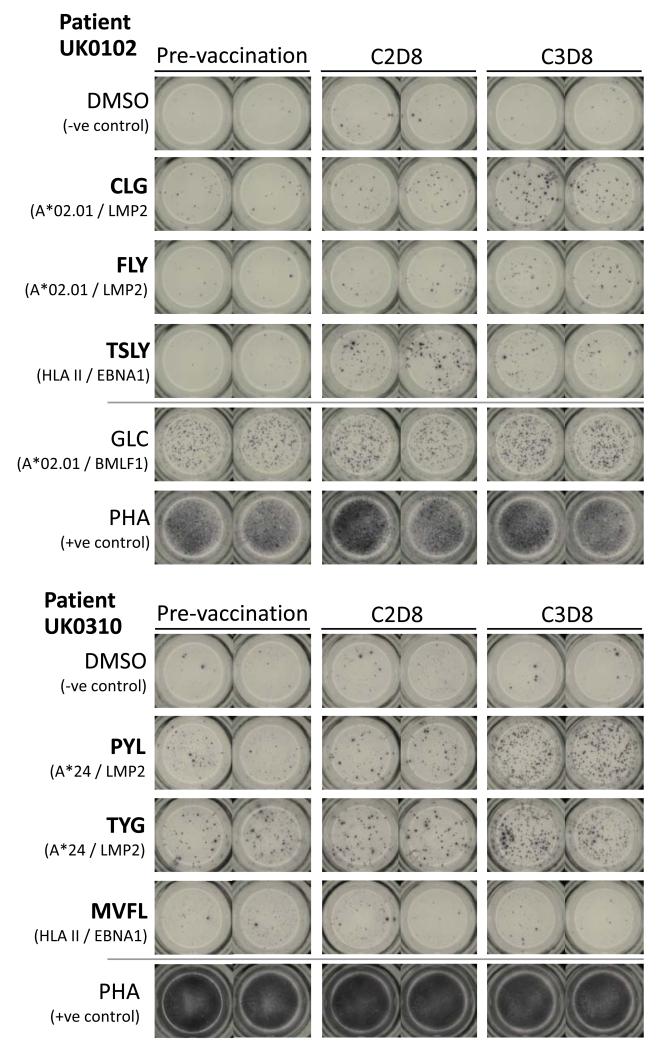
Results from two patients are illustrated in Figure 2. For UK0102, candidate targets included the HLA-A*02.01-restricted CLG and FLY epitopes from LMP2 and the HLA-DRB1*0101-restricted TSLY epitope from EBNA1. The CLG and TSLY target peptides used in the assay varied compared to the vaccine-derived epitopes, whereas the FLY epitope was conserved. Weak pre-vaccination recognition was observed against CLG, but reactivity to all three epitopes increased after vaccination. In contrast, there was no corresponding increase in reactivity to a non-vaccine control HLA-A*02.01-restricted epitope, GLC, derived from the immunodominant EBV lytic cycle protein BMLF1. For UK0310, candidate targets included the LMP2-derived epitopes, PYL and TYG, restricted by HLA-A*24. The PYL epitope is conserved between Chinese and European strains, whereas TYG differs at a single residue. For both targets, pre-existing T-cell recognition was amplified by MVA-EL vaccination.
Figure 2. MVA-EL stimulates EBNA1 and LMP2 epitope-specific T-cell responses.
Representative IFNγ ELISpot data from two patients showing the quantification of T-cell responses to defined EBNA1 or LMP2 epitope peptides. PBMCs from three different time points (pre-vaccination, cycle 2 day 8 and cycle 3 day 8) were stimulated in parallel with the indicated epitope peptides at a cell input number of 300,000 PBMCs/well. Negative or positive control wells comprised the same number of PBMCs stimulated with DMSO solvent or phytohemagglutinin (PHA), respectively. Vaccine-encoded epitopes are highlighted in bold.
Overall, given the patients’ HLA types, 12 vaccine recipients could be screened for responses against potentially relevant EBNA1 or LMP2 epitopes (Supplementary Table 2). Epitope-specific responses were detected in 4/9 White patients tested. In addition to UK0102 and UK0310 illustrated above, patients UK0105 and UK0209 (both HLA-A*02.01+) responded to the FLY epitope; UK0105 also responded to a second HLA-A*02.01-restricted LMP2 epitope (LLW) and an HLA class II-restricted EBNA1 epitope (VFLQ). These epitopes are all conserved between typical European EBV strains and the vaccine insert. Epitope-specific responses were also observed in 2/3 South East Asian patients. In one case (UK0103), responses were directed against a pool of peptides containing two HLA class II-restricted EBNA1 peptides, each differing by one residue from the corresponding vaccine-expressed epitopes. Responses against CLG and LLW, as well as an HLA-B*40-restricted LMP2 epitope (IED), were detected in UK0414. Again, the CLG and IED test peptides differed from the corresponding vaccine-encoded sequences.
Next, we used surplus PBMCs from nine patients to examine T-cell responses against whole antigens, represented in pools of 15mer peptides with consecutive 10 residue overlaps (pepmixes) spanning the EBNA1 and LMP2 protein sequences derived from EBV strain B95.8. The self-antigen actin was used as a negative control for spontaneous IFNγ release. In addition, we included pepmixes for EBNA3A, an EBV latent protein not encoded by the vaccine, and influenza matrix and nucleoproteins (FLU). Recognition of these antigens was expected to be present in at least some patients and to fluctuate, but variations over time would not be predicted to have a consistent relationship to vaccination.
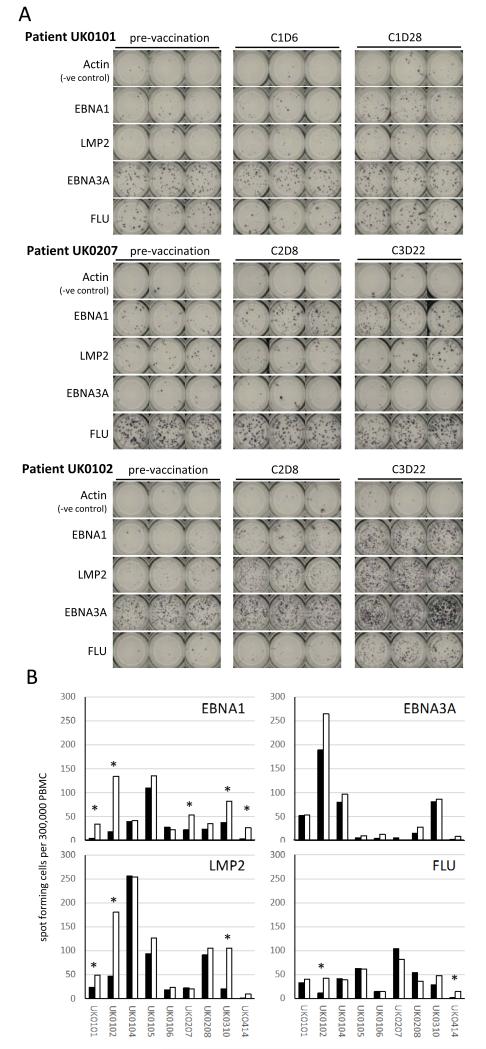
Longitudinal pepmix data from three patients are shown in Figure 3A. In each case, as in most NPC patients, pre-existing levels of EBNA1 and LMP2 recognition were low or even undetectable. Patient UK0101, who did not complete the vaccine course, nevertheless showed evidence of small EBNA1 and LMP2 responses 28 days after a single vaccine cycle, while the EBNA3A response remained static. Patient UK0207 showed an EBNA1 response as early as day 8 after the second vaccine cycle and an LMP2 response appeared after the third cycle, whereas control antigen responses again did not change. Patient UK0102 also mounted responses to both EBNA1 and LMP2, both of which were detectable after the second cycle and increased dramatically after the third. In this patient, FLU reactivity also increased markedly at the third time point. An increase in EBNA3A reactivity was observed in addition, although this increase was relatively small compared to baseline.
Figure 3. Antigen-specific T-cell responses stimulated by MVA-EL.
A. Representative IFNγ ELISpot data from three patients showing the quantification of T-cell responses to EBNA1 or LMP2 pepmixes. PBMCs from three different time points before and after vaccination as indicated were stimulated in triplicate with EBNA1 or LMP2 pepmixes at a cell input number of 300,000 PBMCs/well. Actin pepmix served as the negative control; EBNA3A and FLU pepmixes were included as additional controls. B. Graphs summarizing the results of whole antigen IFNγ ELISpot assays across nine patients. Spot-forming cells per well are shown for vaccine (left panels) and non-vaccine (right panels) pepmixes, using samples taken before (filled boxes) and after (open bars) vaccination. Asterisks indicate increases in T-cell frequencies above the cut-off.
Vaccination-induced changes in antigen reactivity are shown for all patients tested in Figure 3B. Pre-vaccination recognition of both vaccine and non-vaccine targets was commonly observed. Responses associated with vaccination, defined as ≥2-fold amplification and an increase of ≥10 spots/well over baseline, were observed in 5/9 patients for EBNA1 and 3/9 patients for LMP2. In two patients, a 2-fold amplification was also detected for FLU but not for EBNA3A.
Combining data from all ELISpot assays, using either defined epitope peptides or pepmixes as targets, there was evidence for low-level pre-vaccination recognition of EBNA1 and LMP2 in 11/15 (73%) and 10/15 (66%) patients, respectively. Vaccination led to a >2-fold increase in EBNA1 and LMP2 reactivity in 7/14 (50%) and 6/14 (43%) patients, respectively. Overall, 8/14 patients responded to one or both vaccine antigens (Supplementary Table 2). There was no obvious relationship between variation in the resident EBV strain and vaccine immunogenicity. Responses were observed for UK0101, UK0102, UK0207, UK0310 and UK0414, who carried four different EBV strains based on virus genotyping analysis (Table 1 and Supplementary Table 2). A small increase in anti-EBNA1 antibody titres was also noted after vaccination (Supplementary Figure 3).
Multivariate analysis of immune function
IFNγ ELISpot assays are sensitive but measure only a single T-cell function. To extend our analysis of vaccine immunogenicity, we selected one patient (UK0102) based on the presence of clear EBNA1/LMP2 responses and the availability of residual PBMCs to evaluate EBV-specific T-cell lineage, phenotype and function over the course of vaccination. PBMCs were stimulated with EBNA1, LMP2 or EBNA3A pepmixes and then co-stained for: (i) CD3, CD4 and CD8 (lineage); (ii) CD27 and CD45RO (phenotype); and (iii) IFNγ, IL-2, TNFα, MIP-1β and CD107a (function). The latter marker indicates degranulation and can act as a surrogate measure of cytotoxic activity (27).
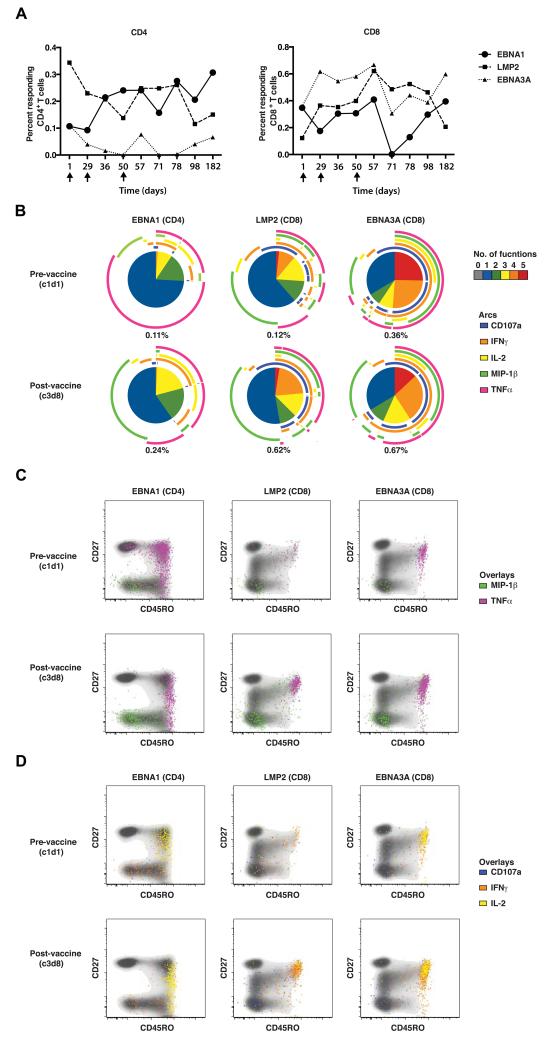
The chimeric protein encoded by MVA-EL contains a predominance of HLA class II-restricted epitopes in EBNA1 targeted by CD4 T-cells and HLA class I-restricted epitopes in LMP2 targeted by CD8 T-cells (Supplementary Table 1). We first asked whether vaccination amplifies both CD4 and CD8 T-cell responses detected with any of the five functional parameters. As predicted, the frequencies of EBNA1-specific CD4 T-cells and LMP2-specific CD8 T-cells increased with vaccination. Notably, the EBNA1-specific effectors were maintained at higher levels for more than a year, while the LMP2-specific response declined slowly over time (Figure 4A).
Figure 4. Immunophenotypic analysis of T-cell responses in patient UK0102 before, during and after vaccination with MVA-EL.
A. Graphs showing for patient UK0102 the total sizes of the indicated antigen-specific CD4 (left panel) and CD8 (right panel) T-cell responses before, during and after three cycles of vaccination with MVA-EL. PBMCs were stimulated with pepmixes and functionally responsive T-cells were enumerated by polychromatic flow cytometry on the basis of cytokine production (IFNγ, IL-2, MIP-1β and TNFα) and surface mobilization of CD107a, a marker of degranulation. Combination gates were created with FlowJo software to determine the total magnitude of each response, counting each cell only once irrespective of discrete functional profiles. B. Functional profiles of antigen-specific T-cells before (upper panels) and after (lower panels) three cycles of MVA-EL for patient UK0102. Pie charts display the numbers of functions expressed by EBNA1-specific CD4 T-cells (left), LMP2-specific CD8 T-cells (middle) and EBNA3A-specific CD8 T-cells (right) as indicated in the key. The outer arcs indicate the combinations of functions corresponding to each pie segment. The total size of the response is indicated in each case. C and D. Phenotypic profiles of functionally responsive CD4 and CD8 T-cells before and after vaccination for patient UK0102. The overall layout corresponds with panel B. The coloured dots depict functional antigen-specific T-cells, as indicated in the key, superimposed on cloud plots of the total CD4 and CD8 T-cell populations as appropriate.
Next, we asked whether vaccination also enhanced the quality of the T-cell response, determined as the number of effector functions elicited by antigen encounter. For this analysis, we compared data obtained from time points before and after three cycles of MVA-EL (Figure 4B). At the later time point, the frequencies of EBNA1-specific CD4 T-cells and LMP2-specific CD8 T-cells had increased by 120% and 420%, respectively. Moreover, for both vaccine-enhanced responses, the proportion of T-cells expressing only a single function decreased after MVA-EL administration, while the proportion of polyfunctional T-cells increased concomitantly. Pre-vaccination CD8 T-cell populations targeting the immunodominant EBNA3A protein were more polyfunctional compared to those specific for the subdominant LMP2 antigen, in line with previous reports (28, 29). However, there were no marked expansions of EBNA3A-specific CD8 T-cells after vaccination and no detectable shifts towards greater polyfunctionality.
Prior to vaccination, the majority of EBNA1-specific CD4 T-cells secreted TNFα alone or in combination with IL-2 or IFNγ; monofunctional MIP-1β-producing cells were also present. After vaccination, there was a substantial increase in polyfunctional CD4 T-cells secreting TNFα with IFNγ and/or IL-2. The proportion of single-function TNFα-producing cells decreased and the proportion of monofunctional MIP-1β-producing cells increased. The LMP2-specific CD8 T-cell population also contained a substantial proportion of monofunctional MIP-1β-producing cells that increased slightly after vaccination. Polyfunctionality was enhanced post-vaccination with more TNFα-producing cells making IFNγ in the presence or absence of MIP-1β, and a substantial increase in CD107a mobilization indicative of antigen-induced degranulation.
Figures 4C and 4D show how these functionally distinct T-cells map onto the conventional partitioning of differentiation subsets defined by CD27 and CD45RO expression (30). The panels display antigen-reactive cells as pixels, colour-coded by function, overlaid on monochrome cloud plots showing the phenotypic distribution of all CD4 (for EBNA1) or CD8 (for LMP2 or EBNA3A) T-cells. Effectors secreting MIP-1β were predominantly CD27loCD45ROlo both before and after vaccination, suggesting a state of terminal differentiation. In contrast, EBNA1-specific CD4 T-cells secreting TNFα and/or IL-2 were predominantly CD45RO+, particularly after vaccination. Similarly, LMP2-specific CD8 T-cells secreting TNFα and/or IFNγ were strictly confined within the CD27+CD45RO+ compartment, most apparently after vaccination. Thus, in general, MVA-EL increased the frequency and functional diversity of effector T-cells without driving terminal differentiation. Nonetheless, highly differentiated CD107a+ LMP2-specific CD8 T-cells, likely representing potent cytotoxic effectors (31), were clearly present after vaccination.
Data combination with the phase IA trial in Hong Kong
The phase I trial described here and the parallel trial in Hong Kong (14) used the same vaccine batch, dose escalation scheme and ELISpot screening protocol, allowing us to explore the combined results. Table 2 shows the proportions of patients at each dose level with a >2-fold post-vaccination increase in T-cells specific for each of the four antigens. Of the 27 patients for whom data were available, 18 and 12 responded to EBNA1 and LMP2, respectively. In contrast, only one and three patients showed increased responses to the non-vaccine target antigens, EBNA3A and FLU, respectively. Overall, 20/27 patients (74 %) showed increased T-cell responses to EBNA1 and/or LMP2; indeed, all patients vaccinated at dose level 3 or above responded to one or both of these vaccine-encoded antigens.
Table 2. Combined immunological response data from two Phase 1 clinical trials of MVA-EBNA1/LMP2.
| Vaccine target antigens | Viral non-vaccine antigens | |||
|---|---|---|---|---|
| EBNA1 | LMP2 | EBNA3A | FLU | |
| Dose level 1 | 3/8 | 3/8 | 0/8 | 1/8 |
| Dose level 2 | 2/5 | 1/5 | 0/5 | 0/5 |
| Dose level 3 | 4/4 | 1/4 | 0/4 | 0/4 |
| Dose level 4 | 3/4 | 2/4 | 0/4 | 1/4 |
| Dose level 5 | 6/6 | 5/6 | 1/6 | 1/6 |
| All patients | 18/27 | 12/27 | 1/27 | 3/27 |
PBMCs were tested with the indicated antigen pepmixes in IFNγ ELISpot assays at one or two timepoints in a blood sample taken 28-77 days after receiving the first vaccine dose (cycle 2 day 8 and cycle 3 day 29). A T-cell response was defined as a >2-fold amplification of specific T cell frequency post-vaccination, using the mean of two readings if available and >10 sfc/well. The proportion of patients who had a T cell response at each dose level is shown, as well as the total number of patients responding overall.
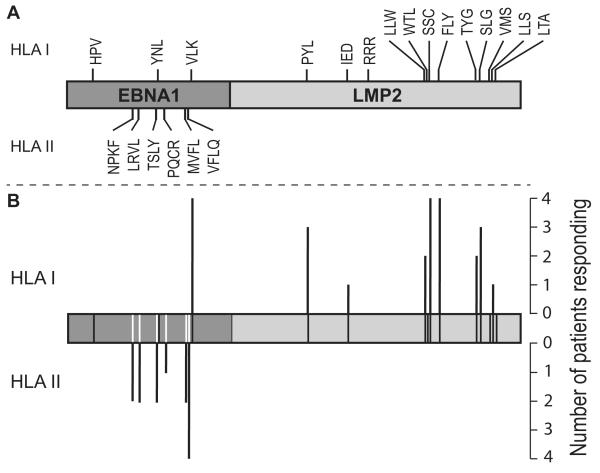
Both trials also included response screening based on individual HLA-matched EBNA1 and LMP2 epitope peptides; overall, 30 patients were analysed in this way. Figure 5A presents a linear map of the vaccine-encoded EBNA1/LMP2 antigen sequence depicting the position of all CD4 and CD8 T-cell epitopes tested during the course of the two trials. Figure 5B identifies the epitopes against which a specific response was observed post-vaccination and the number of patients responding in each case. The vaccine was clearly immunogenic in the context of multiple HLA alleles. Thus, responses were induced against 9/15 HLA class I-restricted epitopes and 6/6 HLA class II-restricted epitopes tested, with up to four patients responding per epitope.
Figure 5. MVA-EL vaccination of NPC patients stimulates broad T-cell immunity specific for multiple epitopes in EBNA1 and LMP2.
A. Diagram showing the design of the EBNA1/LMP2 fusion protein encoded by the MVA-EL vaccine. The location of defined epitopes used as peptide targets in IFNγ ELISpot assays are also indicated. HLA class I-restricted CD8 T cell epitopes are shown above the fusion protein; HLA class II-restricted CD4 T-cell epitopes are shown below. B. Diagram showing the breadth of T-cell responses stimulated by MVA-EL. Epitope location is represented by black (HLA class I) and white (HLA class II) lines on the fusion protein. Bar height above (HLA class I) and below (HLA class II) the fusion protein indicates the number of donors from the Hong Kong and UK phase IA trials that responded to the indicated epitope.
Discussion
EBV is found in the malignant cells of several different cancers and represents a rational target for therapeutic vaccination (2). NPC is a particularly common tumour among South-East Asian, especially Southern Chinese people, but occurs as a rare cancer in other populations (1,2). Although MVA-EL was designed to treat NPC (10), the vaccine’s clinical utility could potentially extend to other EBV+ malignancies such as ENKTL, HL and GC that similarly express EBNA1 and LMP2. These malignancies occur worldwide and variation in human populations and viral strains could therefore potentially affect their immune control. To identify if the vaccine had broad applicability it was important to conduct a Phase I trial of similar design to that conducted in Hong Kong (14) but in a different population group. Here we present the results of this trial, performed in parallel with the Hong Kong study and of similar design but performed in NPC patients in the UK. Of the 16 patients vaccinated in the UK, 11 were of unspecified European origin, termed White, and 5 were of South East Asian descent.
Sequence variation is a feature of prevalent EBV strains across different populations and some polymorphisms can affect viral epitope presentation (18-23). In the present cohort of 16 patients, we were able to genotype the resident EBV strain in eight cases. Natural strain variation was detected consistent with previously described patterns in White and Chinese individuals (19). There were five genotypes in addition to the prototypic European strain, B95.8. This is relevant because the synthetic peptides used in our screening assays were based on B95.8 sequences. It therefore seems likely that strain-specific variation in individual EBNA1/LMP2 epitope sequences was present as described previously (17, 24). Importantly, such variation did not prevent the Chinese-origin LMP2 sequence encoded by MVA-EL from expanding T-cell populations originally generated by diverse viral strains.
Substantial HLA diversity also exists between different populations, both in terms of the prevalent alleles (32) and allelic subtypes that can impact epitope selection (24). For example, although HLA-A2 was the single most common allele in both UK (50%) and Hong Kong (78%) trial patients, 7/8 UK patients carried the HLA-A*02.01 subtype compared to only 1/14 patients in Hong Kong (14). Importantly, we found that vaccination increased the frequencies of several HLA-A*02.01-restricted LMP2-specific T-cell responses. Given the high frequency of HLA-A*02.01 in Western populations, this result is encouraging for worldwide deployment of the MVA-EL vaccine.
Combining ELISpot data from the Hong Kong and UK trials of MVA-EL showed that vaccination increased T-cell responses to EBNA1 and/or LMP2 in 20/27 patients. Although the response rate in UK patients (8/14) was lower compared to the Hong Kong cohort (15/18), the UK data does not include any patients treated at the highest vaccine dose and, due to the expansion of the first dose cohort, includes six patients treated at the lowest dose. As MVA-EL immunogenicity increases with dosage (14), the UK response rate is likely to be an underestimate of what will be achieved in future trials using the highest, most immunogenic, dose.
Many vaccine trials involving tumour-associated antigens of cellular origin are dealing with very low abundance reactivities and their analysis requires in vitro expansion, which may lead to phenotypic perturbations. In contrast, even though EBNA1 and LMP2 are generally subdominant EBV antigens, the size of these T-cell responses after vaccination may be sufficient to allow direct ex vivo analysis. This was the case with one of our vaccine recipients, UK0102, from whom additional blood samples were available to permit a more detailed characterization of MVA-EL-induced T-cell populations.
Polychromatic flow cytometry revealed the true size and breadth of the immune response in patient UK0102, which was much larger than that measured by IFNγ ELISpot alone. Summing all five effector functions (IFNγ, IL-2, TNFα, MIP-1β and CD107a), the CD4 T-cell response to EBNA1 reached over 0.2% of all CD4 T-cells in the blood. Similarly, the CD8 T-cell response to LMP2 reached over 0.6% of all peripheral CD8 T-cells, a response of similar magnitude to that seen against EBNA3A, an immunodominant EBV antigen. Furthermore, different functional profiles were detected within these responses, such that vaccination increased not only the total numbers of EBNA1/LMP2-reactive T-cells, but also the proportion of those expanded populations with polyfunctional capabilities. To some extent, the different functional subsets were linked with cell phenotype. In particular, MIP-1β-producing cells were typically separate from cells with other functional outputs and segregated within a highly differentiated (CD27loCD45ROlo) subpopulation. Importantly, MVA-EL vaccination did not appear to drive other functionally responsive cells into this terminally differentiated compartment, potentially enabling more durable immune memory. However, these results were obtained from a single patient and further work is needed to determine their broader significance. Multiplexed measurements enabled by polychromatic flow cytometry will therefore play an important role in the evaluation of vaccine-induced T-cell responses in our ongoing phase IB and II trials of MVA-EL.
Other important factors determining widespread use of a therapy are its safety and tolerability. In the present trial, MVA-EL was generally well tolerated and 14 patients completed all three vaccination cycles at dose levels up to 5×108 pfu with reports of only grade 1 and 2 ISRs and systemic toxicities. The very first patient exhibited a florid ISR on cycle 1 (albeit grade 2) and a cautious approach led to an expansion of that dose level. On dose level 2, a patient withdrew following grade 2 ‘flu-like symptoms. Taken together with the Hong Kong data, the maximum tolerated dose has not been identified in the dose range tested, and the highest dose tested (5×108 pfu) appears to give the most consistent immune response across both antigens. For these reasons, the recommended dose for further investigation is 5×108 pfu.
The UK trial included 16 patients, all of whom underwent radiotherapy or chemoradiotherapy for NPC and, unlike the Hong Kong trial, six patients with residual disease at trial entry. Using plasma EBV DNA levels to demarcate tumour load and the risk of subsequent progression (33, 34), we noted two patients with apparent disease stabilization after vaccination (UK0103 and UK0104). In UK0103, we speculate that fluctuations in circulating EBV genome levels during vaccination might reflect T-cell-mediated cytotoxic activity against the tumour, as reported previously in the context of radiotherapy (35) and vaccination with MVA-EL (14). Patient UK0103 exhibited a response against a pool of HLA class II epitopes although cells were not available for testing with whole antigen pepmixes. Patient UK0104 recognized both EBNA1 and LMP2 prior to vaccination, but no amplifications were detected after MVA-EL administration. In two other patients, vaccination had no apparent clinical impact. Indeed, EBV levels rose relentlessly in one of these patients despite detectable immune responses to vaccination. Testing for a relationship between detectable immune reactivity and clinical benefit will require an expanded cohort of patients with residual disease consistently treated at the recommended dose. This trial is currently ongoing [ClinicalTrials.gov NCT01094405].
As for extending MVA-EL to a wider range of malignancies, we note that several studies using adoptive T-cell transfer to boost EBNA1- or LMP2-specific effectors have reported clinical responses in patients with various EBV+ lymphomas (36-39). Using MVA-EL vaccination to boost the same T-cell responses may well lead to similar outcomes. The future of immunotherapy almost certainly lies in the development of rationally designed combination treatments. In this regard, the development of MVA-EL, with well characterised antigens and T-cell reactivities will allow us to investigate potential synergism between therapeutic vaccination and other modalities such as additive T-cell therapy, which has been applied to a range of EBV-associated malignancies including NPC (36-43), or immune checkpoint inhibitors such as anti-CTLA4 or anti-PD1 (44).
In summary, the present trial provides three key advances. First, it provides further evidence of the safety and tolerability of MVA-EL, and supports the selection of dose level 5 for ongoing trials in NPC patients. Second, it demonstrates the immunogenicity of MVA-EL across diverse ethnic backgrounds, thereby supporting proposals to trial this vaccine against other EBV+ cancers, including ENKTL, HL and GC. These malignancies occur worldwide in all ethnic groups and together cause a greater disease burden than NPC (45). Third, it demonstrates the potential importance of multiplexed functional and phenotypic measurements in the full characterization of vaccine-induced T-cell responses. Collectively, these new data justify the ongoing development of MVA-EL through the next phase of trials with a view to eventual clinical use in the adjuvant setting to maintain remission following primary treatment or as treatment of relapsed disease, either alone or in combination with other modalities.
Supplementary Material
Statement of translational relevance.
Activated tumour-specific T-cells represent a powerful selective mechanism to control malignancy. Therapeutic vaccination provides a way to selectively amplify and arm tumour-specific T-cells that is scalable and thus suitable for treating large numbers of patients. This phase I trial establishes the safety and immunogenicity of therapeutic vaccination against Epstein-Barr virus (EBV), which is resident in several human cancers totalling 200,000 cases each year worldwide. It pilots detailed functional dissection of the immune response to vaccination as a pharmacodynamic readout. As well as being of direct clinical relevance, the ability to readily characterise vaccine-induced T-cell responses against non-self tumour antigens makes EBV an ideal model system in which to explore novel immunotherapeutic strategies. Our trial therefore lays the groundwork for future large-scale efficacy trials of the vaccine and for trials testing a combinatorial approach to immunotherapy, for example using non antigen-specific immune checkpoint inhibitors with immunogenic vaccination against tumour antigens.
Acknowledgments
Financial support: This work was funded by grants from Cancer Research UK, the UK Experimental Cancer Medicine Centres, the Hong Kong Cancer Fund and the Wellcome Trust; DAP is a Wellcome Trust Senior Investigator.
Footnotes
Conflict of interest statement: The authors declare no conflicts of interest.
References
- 1.Parkin DM. The global health burden of infection-associated cancers in the year 2002. Int. J. Cancer. 2006;118:3030–44. doi: 10.1002/ijc.21731. [DOI] [PubMed] [Google Scholar]
- 2.Young LS, Rickinson AB. Epstein–barr virus: 40 years on. Nat. Rev. Cancer. 2004;4:757–768. doi: 10.1038/nrc1452. [DOI] [PubMed] [Google Scholar]
- 3.Baujat B, Audry H, Bourhis J, Chan A, Onat H, Chua D, et al. Chemotherapy as an adjunct to radiotherapy in locally advanced nasopharyngeal carcinoma ( Review ) Cochrane Database Syst Rev. 2006:CD004329. doi: 10.1002/14651858.CD004329.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Lu H, Peng L, Yuan X, Hao Y, Lu Z, Chen J, et al. Concurrent chemoradiotherapy in locally advanced nasopharyngeal carcinoma: a treatment paradigm also applicable to patients in Southeast Asia. Cancer Treat. Rev. 2009;35:345–53. doi: 10.1016/j.ctrv.2009.01.002. [DOI] [PubMed] [Google Scholar]
- 5.Bensouda Y, Kaikani W, Ahbeddou N, Rahhali R, Jabri M, Mrabti H, et al. Treatment for metastatic nasopharyngeal carcinoma. Eur. Ann. Otorhinolaryngol. Head Neck Dis. 2011;128:79–85. doi: 10.1016/j.anorl.2010.10.003. [DOI] [PubMed] [Google Scholar]
- 6.Razak ARA, Siu LL, Liu FF, Ito E, O’Sullivan B, Chan K. Nasopharyngeal carcinoma: the next challenges. Eur. J. Cancer. 2010;46:1967–78. doi: 10.1016/j.ejca.2010.04.004. [DOI] [PubMed] [Google Scholar]
- 7.Fox CP, Haigh TA, Taylor GS, Long HM, Lee SP, Shannon-Lowe C, et al. A novel latent membrane 2 transcript expressed in Epstein-Barr virus-positive NK- and T-cell lymphoproliferative disease encodes a target for cellular immunotherapy. Blood. 2010;116:3695–704. doi: 10.1182/blood-2010-06-292268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Pallesen G, Hamilton-Dutoit SJ, Rowe M, Young LS. Expression of Epstein-Barr virus latent gene products in tumour cells of Hodgkin’s disease. Lancet. 1991;337:320–322. doi: 10.1016/0140-6736(91)90943-j. [DOI] [PubMed] [Google Scholar]
- 9.Van Beek J, zur Hausen A, Klein Kranenbarg E, van de Velde CJH, Middeldorp JM, van den Brule AJC, et al. EBV-positive gastric adenocarcinomas: a distinct clinicopathologic entity with a low frequency of lymph node involvement. J. Clin. Oncol. 2004;22:664–70. doi: 10.1200/JCO.2004.08.061. [DOI] [PubMed] [Google Scholar]
- 10.Taylor GS, Haigh TA, Gudgeon NH, Phelps RJ, Lee SP, Steven NM, et al. Dual Stimulation of Epstein-Barr Virus (EBV)-specific CD4+ and CD8+ T-cell responses by a chimeric antigen construct : potential therapeutic vaccine for EBV-Positive nasopharyngeal carcinoma. J. Virol. 2004;78:768–778. doi: 10.1128/JVI.78.2.768-778.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Voo KS, Fu T, Wang HY, Tellam J, Heslop HE, Brenner MK, et al. Evidence for the presentation of major histocompatibility complex class I-restricted Epstein-Barr virus nuclear antigen 1 peptides to CD8+ T lymphocytes. J. Exp. Med. 2004;199:459–70. doi: 10.1084/jem.20031219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Tellam J, Connolly G, Green KJ, Miles JJ, Moss DJ, Burrows SR, et al. Endogenous presentation of CD8+ T cell epitopes from Epstein-Barr virus-encoded nuclear antigen 1. J. Exp. Med. 2004;199:1421–31. doi: 10.1084/jem.20040191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Lee SP, Brooks JM, Al-Jarrah H, Thomas WA, Haigh TA, Taylor GS, et al. CD8 T cell recognition of endogenously expressed Epstein-Barr virus nuclear antigen 1. J. Exp. Med. 2004;199:1409–20. doi: 10.1084/jem.20040121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Hui EP, Taylor GS, Jia H, Ma BBY, Chan SL, Ho R, et al. Phase I trial of recombinant modified vaccinia ankara encoding Epstein-Barr viral tumor antigens in nasopharyngeal carcinoma patients. Cancer Res. 2013;73:1676–88. doi: 10.1158/0008-5472.CAN-12-2448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Lee SP, Chan AT, Cheung ST, Thomas WA, Croom-Carter D, Dawson CW, et al. CTL control of EBV in nasopharyngeal carcinoma (NPC): EBV-specific CTL responses in the blood and tumors of NPC patients and the antigen-processing function of the tumor cells. J. Immunol. 2000;165:573–82. doi: 10.4049/jimmunol.165.1.573. [DOI] [PubMed] [Google Scholar]
- 16.Lin X, Gudgeon NH, Hui EP, Jia H, Qun X, Taylor GS, et al. CD4 and CD8 T cell responses to tumour-associated Epstein-Barr virus antigens in nasopharyngeal carcinoma patients. Cancer Immunol. Immunother. 2008;57:963–75. doi: 10.1007/s00262-007-0427-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Tsang CW, Lin X, Gudgeon NH, Taylor GS, Jia H, Hui EP, et al. CD4+ T-cell responses to Epstein-Barr virus nuclear antigen EBNA1 in Chinese populations are highly focused on novel C-terminal domain-derived epitopes. J. Virol. 2006;80:8263–6. doi: 10.1128/JVI.00400-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Midgley RS, Bell AI, Mcgeoch DJ, Rickinson AB. Latent gene sequencing reveals familial relationships among Chinese Epstein-Barr virus strains and evidence for positive selection of A11 epitope changes. J. Virol. 2003;77:11517–11530. doi: 10.1128/JVI.77.21.11517-11530.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Midgley R, Bell A, Yao Q, Croom-Carter D, Hislop A, Whitney B, et al. HLA-A11-restricted epitope polymorphism among Epstein-Barr virus strains in the highly HLA-A11-positive Chinese population: incidence and immunogenicity of variant epitope sequences. J. Virol. 2003;77:11507–11516. doi: 10.1128/JVI.77.21.11507-11516.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.De Campos-Lima PO, Levitsky V, Brooks J, Lee SP, Hu LF, Rickinson AB, et al. T cell responses and virus evolution: loss of HLA A11-restricted CTL epitopes in Epstein-Barr virus isolates from highly A11-positive populations by selective mutation of anchor residues. J. Exp. Med. 1994;179:1297–305. doi: 10.1084/jem.179.4.1297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Edwards RH, Sitki-green D, Moore DT, Raab-Traub N. Potential Selection of LMP1 Variants in Nasopharyngeal Carcinoma. J. Virol. 2004;78:868–881. doi: 10.1128/JVI.78.2.868-881.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Levitsky V, Zhang QJ, Levitskaya J, Kurilla MG, Masucci MG. Natural variants of the immunodominant HLA A11-restricted CTL epitope of the EBV nuclear antigen-4 are nonimmunogenic due to intracellular dissociation from MHC class I:peptide complexes. J. Immunol. 1997;159:5383–90. [PubMed] [Google Scholar]
- 23.Burrows JM, Burrows SR, Poulsen LM, Sculley TB, Moss DJ, Khanna R. Unusually high frequency of Epstein-Barr virus genetic variants in Papua New Guinea that can escape cytotoxic T-cell recognition: implications for virus evolution. J. Virol. 1996;70:2490–6. doi: 10.1128/jvi.70.4.2490-2496.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Lee SP, Tierney RJ, Thomas WA, Brooks JM, Rickinson AB. Conserved CTL epitopes within EBV latent membrane protein 2. J. Immunol. 1997;158:3325–3334. [PubMed] [Google Scholar]
- 25.Roederer M, Nozzi JL, Nason MC. SPICE: exploration and analysis of post-cytometric complex multivariate datasets. Cytometry A. 2011;79:167–74. doi: 10.1002/cyto.a.21015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Liu W, Putnam AL, Xu-Yu Z, Szot GL, Lee MR, Zhu S, et al. CD127 expression inversely correlates with FoxP3 and suppressive function of human CD4+ T reg cells. J. Exp. Med. 2006;203:1701–1711. doi: 10.1084/jem.20060772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Betts MR, Brenchley JM, Price DA, De Rosa SC, Douek DC, Roederer M, et al. Sensitive and viable identification of antigen-specific CD8+ T cells by a flow cytometric assay for degranulation. J. Immunol. Methods. 2003;281:65–78. doi: 10.1016/s0022-1759(03)00265-5. [DOI] [PubMed] [Google Scholar]
- 28.Smith C, Beagley L, Khanna R. Acquisition of polyfunctionality by Epstein-Barr virus-specific CD8+ T cells correlates with increased resistance to galectin-1-mediated suppression. J. Virol. 2009;83:6192–8. doi: 10.1128/JVI.00239-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Ning RJ, Xu XQ, Chan KH, Chiang AKS. Long-term carriers generate Epstein-Barr virus (EBV)-specific CD4(+) and CD8(+) polyfunctional T-cell responses which show immunodominance hierarchies of EBV proteins. Immunology. 2011;134:161–71. doi: 10.1111/j.1365-2567.2011.03476.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Liddy N, Bossi G, Adams KJ, Lissina A, Mahon TM, Hassan NJ, et al. Monoclonal TCR-redirected tumor cell killing. Nat. Med. 2012;18:980–7. doi: 10.1038/nm.2764. [DOI] [PubMed] [Google Scholar]
- 31.Chattopadhyay PK, Betts MR, Price DA, Gostick E, Horton H, Roederer M, et al. The cytolytic enzymes granyzme A, granzyme B, and perforin: expression patterns, cell distribution, and their relationship to cell maturity and bright CD57 expression. J Leukoc Biol. 2009;85:88–97. doi: 10.1189/jlb.0208107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Tsuji K, Aizawa M, Sasazuki T, editors. HLA 1991. Proceedings of the 11th International Histocompatibility Workshop and Conference; Oxford: Oxford University Press; 1991. [Google Scholar]
- 33.Han BL, Xu XY, Zhang CZ, Wu JJ, Han CF, Wang H, et al. Systematic review on Epstein-Barr virus (EBV) DNA in diagnosis of nasopharyngeal carcinoma in Asian populations. Asian Pac. J. Cancer Prev. 2012;13:2577–81. doi: 10.7314/apjcp.2012.13.6.2577. [DOI] [PubMed] [Google Scholar]
- 34.Chan KCA, Lo YMD. Circulating EBV DNA as a tumor marker for nasopharyngeal carcinoma. Semin. Cancer Biol. 2002;12:489–496. doi: 10.1016/s1044579x02000913. [DOI] [PubMed] [Google Scholar]
- 35.Lo YMD, Leung SF, Chan LY, Chan ATC, Lo KW, Johnson PJ, et al. Kinetics of Plasma Epstein-Barr Virus DNA during Radiation Therapy for Nasopharyngeal Carcinoma Advances in Brief Kinetics of Plasma Epstein-Barr Virus DNA during Radiation Therapy for Nasopharyngeal Carcinoma. Cancer Res. 2000;60:2351–2355. [PubMed] [Google Scholar]
- 36.Icheva V, Kayser S, Wolff D, Tuve S, Kyzirakos C, Bethge W, et al. Adoptive transfer of Epstein-Barr virus (EBV) nuclear antigen 1-specific t cells as treatment for EBV reactivation and lymphoproliferative disorders after allogeneic stem-cell transplantation. J. Clin. Oncol. 2013;31:39–48. doi: 10.1200/JCO.2011.39.8495. [DOI] [PubMed] [Google Scholar]
- 37.Bollard CM, Aguilar L, Straathof KC, Gahn B, Huls MH, Rousseau A, et al. Cytotoxic T lymphocyte therapy for Epstein-Barr virus+ Hodgkin’ s disease. J. Exp. Med. 2004;200:1623–1633. doi: 10.1084/jem.20040890. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Bollard CM, Gottschalk S, Leen AM, Weiss H, Straathof KC, Carrum G, et al. Complete responses of relapsed lymphoma following genetic modification of tumor-antigen presenting cells and T-lymphocyte transfer. Blood. 2007;110:2838–45. doi: 10.1182/blood-2007-05-091280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Bollard CM, Gottschalk S, Torrano V, Diouf O, Ku S, Hazrat Y, et al. Sustained Complete Responses in Patients With Lymphoma Receiving Autologous Cytotoxic T Lymphocytes Targeting Epstein-Barr Virus Latent Membrane Proteins. J. Clin. Oncol. 2013;31:1–14. doi: 10.1200/JCO.2013.51.5304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Comoli P, Pedrazzoli P, Maccario R, Basso S, Carminati O, Labirio M, et al. Cell therapy of stage IV nasopharyngeal carcinoma with autologous Epstein-Barr virus-targeted cytotoxic T lymphocytes. J Clin Oncol. 2005;23:8942–9. doi: 10.1200/JCO.2005.02.6195. [DOI] [PubMed] [Google Scholar]
- 41.Straathof KC, Bollard CM, Popat U, Huls MH, Lopez T, Morriss MC, et al. Treatment of nasopharyngeal carcinoma with Epstein-Barr virus--specific T lymphocytes. Blood. 2005;105:1898–904. doi: 10.1182/blood-2004-07-2975. [DOI] [PubMed] [Google Scholar]
- 42.Louis CU, Straathof K, Bollard CM, Ennamuri S, Gerken C, Lopez TT, et al. Adoptive transfer of EBV-specific T cells results in sustained clinical responses in patients with locoregional nasopharyngeal carcinoma. J Immunother. 2010;33:983–90. doi: 10.1097/CJI.0b013e3181f3cbf4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Chia WK, Teo M, Wang WW, Lee B, Ang SF, Tai WM, et al. Adoptive T-cell transfer and chemotherapy in the first-line treatment of metastatic and/or locally recurrent nasopharyngeal carcinoma. Mol Ther. 2014;22:132–9. doi: 10.1038/mt.2013.242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Pardoll DM. The blockade of immune checkpoints in cancer immunotherapy. Nat Rev Cancer. 2012;12:252–64. doi: 10.1038/nrc3239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Cohen JI, Fauci AS, Varmus H, Nabel GJ. Epstein-Barr virus: an important vaccine target for cancer prevention. Sci. Transl. Med. 2011;3:1–3. doi: 10.1126/scitranslmed.3002878. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.