Abstract
Background and Aims Adventitious roots (ARs) are essential for vegetative propagation. The Arabidopsis thaliana transcription factors SHORT ROOT (SHR) and SCARECROW (SCR) affect primary/lateral root development, but their involvement in AR formation is uncertain. LAX3 and AUX1 auxin influx carriers contribute to primary/lateral root development. LAX3 expression is regulated by SHR, and LAX3 contributes to AR tip auxin maximum. In contrast, AUX1 involvement in AR development is unknown. Xylogenesis is induced by auxin plus cytokinin as is AR formation, but the genes involved are largely unknown. Stem thin cell layers (TCLs) form ARs and undergo xylogenesis under the same auxin plus cytokinin input. The aim of this research was to investigate SHR, SCR, AUX1 and LAX3 involvement in AR formation and xylogenesis in intact hypocotyls and stem TCLs in arabidopsis.
Methods Hypocotyls of scr-1, shr-1, lax3, aux1-21 and lax3/aux1-21 Arabidopsis thaliana null mutant seedlings grown with or without auxin plus cytokinin were examined histologically, as were stem TCLs cultured with auxin plus cytokinin. SCR and AUX1 expression was monitored using pSCR::GFP and AUX1::GUS lines, and LAX3 expression and auxin localization during xylogenesis were monitored by using LAX3::GUS and DR5::GUS lines.
Key Results AR formation was inhibited in all mutants, except lax3. SCR was expressed in pericycle anticlinally derived AR-forming cells of intact hypocotyls, and in cell clumps forming AR meristemoids of TCLs. The apex was anomalous in shr and scr ARs. In all mutant hypocotyls, the pericycle divided periclinally to produce xylogenesis. Xylary element maturation was favoured by auxin plus cytokinin in shr and aux1-21. Xylogenesis was enhanced in TCLs, and in aux1-21 and shr in particular. AUX1 was expressed before LAX3, i.e. in the early derivatives leading to either ARs or xylogenesis.
Conclusions AR formation and xylogenesis are developmental programmes that are inversely related, but they involve fine-tuning by the same proteins, namely SHR, SCR and AUX1. Pericycle activity is central for the equilibrium between xylary development and AR formation in the hypocotyl, with a role for AUX1 in switching between, and balancing of, the two developmental programmes.
Keywords: Adventitious rooting, Arabidopsis thaliana, auxin influx carriers, AUX1, LAX3, SCR, SHR, thin cell layers, xylogenesis
INTRODUCTION
Adventitious roots (ARs) may contribute together with the primary root (PR) and lateral roots (LRs) to form the root apparatus and are essential for vegetative propagation. The transcription factors SHORT ROOT (SHR) and SCARECROW (SCR), belonging to the GRAS family, are key regulators of PR stem cell definition/maintenance and radial patterning (Di Laurenzio et al., 1996; Helariutta et al., 2000; Sabatini et al., 2003). SHR is expressed in the stele, and SHR moves into the adjacent layer to control SCR transcription and endodermis specification (Koizumi et al., 2012). In contrast, SCR is expressed in the cortex/endodermis initial cell and the endodermis, its protein binds to its own promoter in the presence of SHR (Cui et al., 2007), and they jointly regulate quiescent centre (QC) markers (e.g. WOX5; Sarkar et al., 2007) and microRNAs (i.e. microRNA 165/6) involved in PR vascular differentiation (Carlsbecker et al., 2010).
Cytokinin is involved with auxin in controlling PR apical growth and vascular patterning, with SHR regulating cytokinin homeostasis in the xylem-associated pericycle (Cui et al., 2011; Zhang et al., 2013). Auxin and cytokinin are also involved in LR formation (Benková et al., 2003; Bielach et al., 2012), as are SHR (Lucas et al., 2011) and SCR (Malamy and Benfey, 1997), and SCR is auxin induced (Moubayidin et al., 2013).
AUXIN RESISTANT1 (AUX1) and LIKE AUXIN RESISTANT3 (LAX3) are auxin influx carriers of the same family. In arabidopsis they are required for QC organization in the embryonic radicle (Ugartechea-Chirino et al., 2010), are PR vascular markers also involved in different phases of the LR formation process (Swarup et al., 2008), and LAX3 is required for defining the auxin maximum and maintenance in the AR tip in planta (i.e. in ARs from intact hypocotyls) and in cultured thin cell layers (TCLs) (Della Rovere et al., 2013). LAX3 expression is regulated by SHR (Sozzani et al., 2010), but information about SHR involvement in AR formation is still contradictory (Scheres et al., 1995; Lucas et al., 2011). Stem TCLs are explants devoid of vasculature, i.e. the site of SHR expression in planta (Helariutta et al., 2000), but are capable of AR initiation by the endodermis (Falasca et al., 2004), i.e. the tissue lacking in the stems of shr and scr mutants (Fukaki et al., 1998; Wysocka-Diller et al., 2000). Moreover, AR formation in TCLs needs exogenous auxin [indole-3-butyric acid (IBA) (10 µm)] combined with cytokinin [kinetin (Kin) (0·1 µm)]. A role for AUX1 in controlling organ identity by mediating auxin–cytokinin interaction has been suggested in arabidopsis calli (Kakani et al., 2009), as a role for LAX3 in auxin/ cytokinin distribution during AR development (Della Rovere et al., 2013). However, whether these auxin influx carriers are involved with SHR and SCR in the control of AR formation from TCLs has never been investigated.
Both in planta and in cultured explants, xylogenesis consists of ectopic formation of tracheary-like cells, and in many species is an auxin plus cytokinin-mediated programme (Fukuda, 1997). Xylogenesis has led to important discoveries in the understanding of xylem formation in planta, and genes in common between the xylary processes in planta and in vitro have been found (Miyashima et al., 2013). Arabidopsis TCLs show xylogenesis in addition to AR formation under the same IBA + Kin input (Falasca et al., 2004), and the intact hypocotyl is induced to produce xylogenesis under specific auxin types/treatments (Falasca and Altamura, 2003). The possibility that AR formation and xylogenesis are under the control of common genes needs investigation.
The aim of the present research was to determine SHR and SCR involvement, in relation to the activities of AUX1 and LAX3, during AR formation and xylogenesis, in intact hypocotyls in the absence/presence of exogenous IBA and Kin, and in IBA + Kin-cultured TCLs, by investigating gene expression patterns and single/double mutant responses.
The results showed that AR formation and xylogenesis are inversely related, with SHR, SCR and AUX1 controlling the fine-tuning between the morphogenic programmes. The role of these transcription factors in the initiation of both processes, and of AUX1 in their switching, is discussed.
MATERIALS AND METHODS
Plant material and growth conditions
Arabidopsis thaliana seeds of the homozygous scr-1 and shr-1 [Wassilewskija (Ws) wild type (wt)] and lax3, aux1-21 and lax3/aux1-21 [Columbia (Col) wt] null mutants, and of pSCR::GFP (Ws background), AUX1::GUS, LAX3::GUS and DR5::GUS lines (Col background) were stratified and sterilized according to Della Rovere and co-workers (2013). These seeds (15–20 per plate, 20 plates per genotype, 12 × 12 cm each) were either sown on hormone-free (HF) medium and with IBA (10 µm) and Kin (0·1 µm) (IBA + Kin), or on a commercial soil, for seedling growth and plant production, respectively, according to Della Rovere et al. (2013). For seedling growth, the plates were placed in a vertical position at 22 ± 2 °C under continuous darkness for 14 d, after exposure to white light for 6 h (Takahashi et al., 2003).
TCL culture
Superficial TCLs, about 0·5 × 8 mm, 6–7 layers thick, were excised from the inflorescence–stem internodes of thirty 35-day-old plants of pSCR::GFP, AUX1::GUS, LAX3::GUS, DR5::GUS, shr-1, scr-1, lax3, aux1-21, lax3/aux1-21 and the corresponding wt. One hundred TCLs per genotype were cultured up to day 20 under IBA + Kin. Explants were examined under a Leica MZ8 stereomicroscope at the end of the culture period, and the response was evaluated as the mean number of ARs (± s.e.) per rooting explant.
Histological analysis under light and fluorescence/confocal microscopy
Thirty randomly selected seedlings per genotype and treatment were cleared, mounted and observed with Nomarski optics, and AUX1::GUS, LAX3::GUS and DR5::GUS seedlings were processed for β-glucuronidase (GUS) staining before clearing. In all mutants, and their wt, the hypocotyl length was measured under a Leica MZ8 stereomicroscope before fixation, and the number of ARs was expressed as mean density cm−1 (± s.e.).
Green fluorescent protein (GFP) fluorescence in pSCR::GFP was observed either under the Leica DMRB microscope equipped with a double wavelength filter set (BP 490/20 and BP 575/30) with dichroic filters RKPs 505 and 600 and emission filters BPs 525/20 and 635/40, or under confocal microscopy using a ×63 oil-immersion lens on a Leica TCS-SP5 confocal microscope supplied with the Leica application suite advanced fluorescence (LAS AF Lite) software (Leica Microsystems). Propidium iodide staining and detection were according to Della Rovere et al. (2013).
Basal stem internodes of scr-1, shr-1 and wt plants were dehydrated, embedded, cross-sectioned (4 µm thickness) and stained with toluidine blue, according to Della Rovere and co-workers (2013).
Ten TCLs per genotype were harvested periodically for histology in bright field, as were 30 seedlings per genotype and treatment. Explants and seedlings were fixed, dehydrated and embedded, as above, longitudinally sectioned (4 µm thickness) and stained according to Della Rovere et al. (2013). Explants of the GUS marker lines were sectioned at 12 µm.
The histological image acquisition, the computerized analysis and numbering of the AR stages were according to Della Rovere and co-workers (2013).
Xylogenesis counting
Twenty TCLs per mutant, and the corresponding wt, were fixed, dehydrated, sectioned and stained as above, and vascular strands, nodules and single lignified cells were counted under light microscopy at 70 µm intervals [interval established on the basis of the mean dimensions of the xylary cells, i.e. 42·2 (±3·6) × 11·6 (±1·7) × 10 (±1·3) µm]. The presence of lignin thickenings in the cell walls was verified by detecting lignin autofluorescence under a Zeiss Axiolab epifluorescence microscope, equipped with a 50 W HBO mercury lamp, using the BP 365, FT 395 and LP 397 filter set.
Statistical analysis
Data were expressed as means (± s.e.). A normality test was applied before analysis of variance (ANOVA; GraphPad Instat 3). One-way/two-way ANOVA (P < 0·05) was used to compare the effects of genotypes, and genotypes and treatments, respectively, followed by Tukey’s post-test (GraphPad Prism 6.0). Experiments were repeated three times in two consecutive years, with similar results (data of the second year are shown).
RESULTS
The hypocotyl pericycle shows periclinal divisions leading to ectopic metaxylem formation in all the null mutants
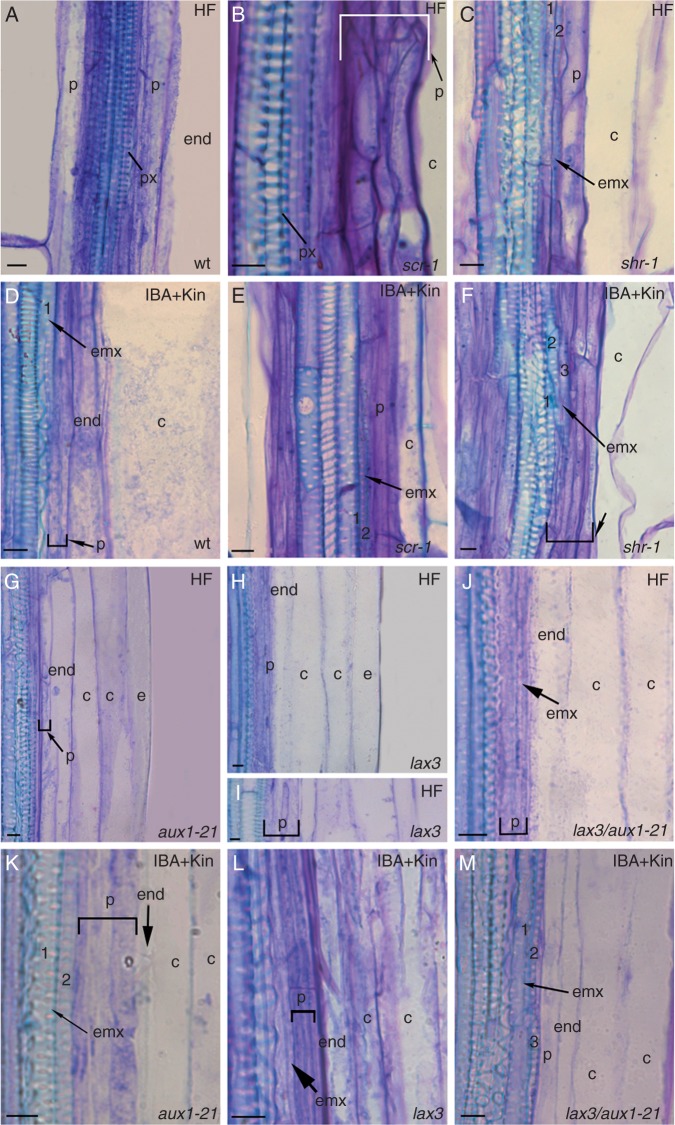
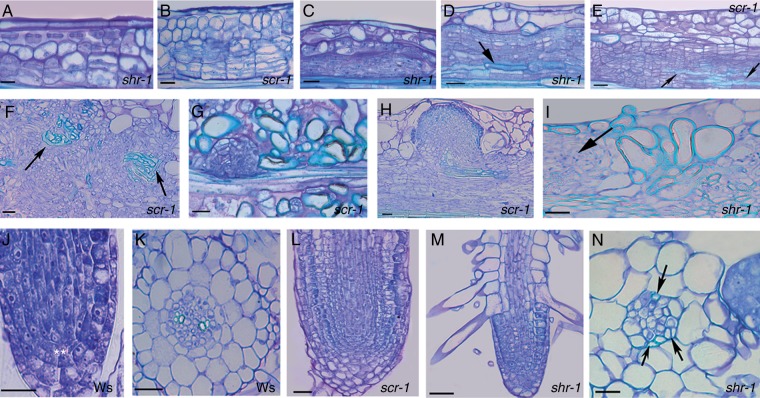
The hypocotyl of shr-1 and scr-1 HF-grown seedlings exhibited two irregular cortical layers, no endodermis, exarch metaxylem (i.e. metaxylem in a peripheral position in the stele); and endarch protoxylem (i.e. protoxylem located internally to the metaxylem), in contrast to the regular structure of the wt (Fig. 1A–C). However, as in the wt, protoxylem was formed by tracheary elements with annular or helical thickenings, and metaxylem by pitted elements (Fig. 1A–C). At the shr-1 and scr-1 hypocotyl–PR junction, the pericycle divided periclinally, generating a multilayered bundle of meristematic cells. However, in shr-1, immature ectopic metaxylem (i.e. xylem elements not yet lignified but with pitted secondary thickenings forming) appeared in the bundle under HF conditions, and mature elements (i.e. dead pitted elements with lignified cell walls) appeared under IBA + Kin treatment (Fig. 1C, F). Immature ectopic metaxylem was observed in scr-1, and occasionally in the wt, under IBA + Kin treatment only (Fig. 1D, E).
Fig. 1.
Ectopic metaxylem formation in pericycle proliferation at the hypocotyl–PR junction of 14-day-old wt (Ws, A, D), scr-1 (B, E), shr-1 (C, F), aux1-21 (G, K), lax3 (H, I, L) and lax3/aux1-21 (J, M) seedlings grown under continuous darkness without hormones (HF) and with IBA + Kin. Longitudinal sections stained with toluidine blue. Numbers mark immature/mature ectopic metaxylem elements, and square brackets indicate the pericycle periclinal proliferation. Exarch metaxylem within the hypocotyl stele of scr-1 (B) and shr-1 (C) in comparison with the exarch protoxylem of the wt (A), aux1-21 (G), lax3 (H, I) and lax3/aux1-21 (J) is shown. c, cortical parenchyma, e, epidermis, end, endodermis, emx, ectopic-metaxylem, p, pericycle, px, protoxylem. Scale bars = 10 µm.
The hypocotyl of aux1-21, lax3 and lax3/aux1-21 HF seedlings showed the same radial pattern as the wt, with the addition of a multilayered bundle of periclinally divided pericycle cells at its junction with the PR (Fig. 1G–J), where ectopic immature metaxylem occurred in the double mutant (Fig. 1J). Under IBA + Kin treatment, ectopic immature metaxylem appeared in all mutants (Fig. 1K–M), and occasionally in the wt, but mature elements appeared only in aux1-21 and lax3/aux1-21 (Fig. 1K, M).
SCR and AUX1 are expressed from the AR-forming divisions in the hypocotyl pericycle
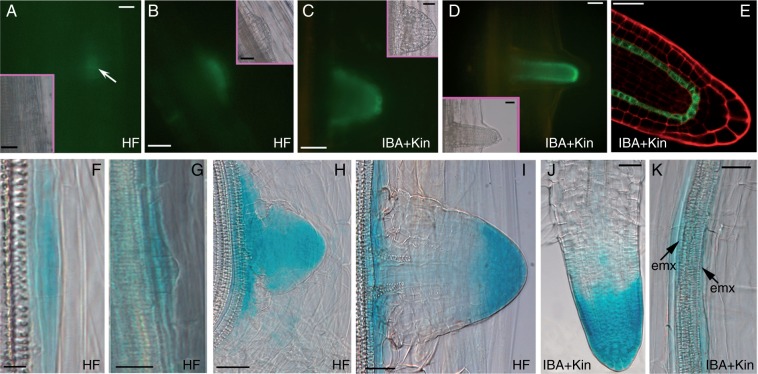
SCR signal, monitored in SCR::GFP seedlings grown with or without exogenous hormones, appeared in the first anticlinally formed derivatives of the hypocotyl pericycle (AR stage I, Fig. 2A, arrow), increased at AR stage II/III (Fig. 2B) and was increased further at stage VII, localizing in the QC, endodermis/cortical initials and derived cells (Fig. 2C). SCR expression extended from the latter cells to the differentiating endodermis in AR primordia (ARPs) and mature ARs (Fig. 2D, E).
Fig. 2.
SCR (A–E), AUX1 (F, H–K) and LAX3 (G) expression in the AR-forming hypocotyl of 14-day-old pSCR::GFP, AUX1::GUS and LAX3::GUS seedlings grown under continuous darkness, either under HF conditions (A, B, F–I) or with IBA + Kin (C–E, J, K). (A) Appearance of SCR signal (arrow) at AR stage I and (B) signal presence at AR stage III. (C, D) SCR expression in the QC, endodermis/cortical initials and derivatives at AR stage VII (C) and in the ARP tip (D). (E) SCR expression in the QC, endodermis/cortical initials/derivatives and endodermis in the AR tip. (A–D, microscopic fluorescence pictures, with corresponding bright-field images in the insets; E confocal microscopy). (F, G) AUX1 expression at stage I (F) and LAX3 expression at stage II (G). (H, I) Uniform AUX1 expression at stage VII (H) and expression limited to the tip and base in the ARP (I). (J) Detail of a mature AR expressing AUX1 in all tip tissues. (K) AUX1 expression in IBA + Kin periclinally divided pericycle and in the ectopic metaxylem (emx, arrows). Scale bars = 10 µm (A, F and inset in A) and 30 µm (B–E, G–K and insets in B–D).
Endogenous auxin accumulated in the pericycle of the DR5::GUS seedlings at the hypocotyl–PR junction before any morphogenic event, increased at AR stage I and continued to be present at further AR stages, progressively localizing at the AR tip and the AR vascular connection with the hypocotyl. Auxin signal increased under IBA + Kin treatment, confirming previous results (Della Rovere et al., 2013).
In AUX1::GUS seedlings, under both HF and IBA + Kin conditions, AUX1 was expressed in the hypocotyl pericycle AR stage I onwards(Fig. 2F), preceding the expression of LAX3, which initiated at AR stage II (Fig. 2G). AUX1 continued to be uniformly expressed up to stage VII (Fig. 2H). In protruding ARPs and mature ARs, AUX1continued to be expressed over the whole tip, and in the procambium and differentiating protoxylem at the organ base (Fig. 2I, J). In contrast, LAX3 expression was excluded from the AR tip, with the exception of the inner cap layer, but was present in all the AR differentiating/mature stele tissues (Della Rovere et al., 2013; and data not shown). In contrast to LAX3, AUX1 expression did not increase under IBA + Kin treatment in comparison with HF conditions. Interestingly, occasional pericycle periclinally divided cells engaged in ectopic metaxylem formation showed AUX1 expression (Fig. 2K, arrows).
AR formation in intact hypocotyls is highly reduced in all mutants, except lax3
At day 14, independently of the treatment, the density of the emerged and non-emerged ARs on the hypocotyls was significantly lower than in the wt (Fig. 3A). The IBA + Kin treatment increased the AR density, significantly more in scr-1 than in shr-1 (Fig. 3A).
Fig. 3.

AR mean density (± s.e.) in the hypocotyl of 14-day-old wt (Ws), scr-1 and shr-1 (A), and wt (Col), lax3, aux1-21 and lax3/aux1-21 (B) seedlings grown under continuous darkness on HF and IBA + Kin. Emerged ARs and ARs only histologically detectable (ANERs) were counted together (A, B). The asterisk shows no AR response. ANER density alone is shown in the inset. aP < 0·0001 with other genotypes within the IBA + Kin treatment, bP < 0·0001 with other genotypes within the HF treatment. cP < 0·05 with scr-1 within the IBA + Kin treatment. dP < 0·0001 within the same genotype. eP < 0·05 within the same genotype. Columns with no letter/the same letters are not significantly different (two-way ANOVA followed by Tukey’s post-test). n = 30 (second replicate of the second year).
Macroscopic ARs were rare, and non-emerged ARs (ANERs) were prevalent on both HF-treated wt and lax3 seedlings, without differences between the two (Fig. 3B and inset). In contrast, the AR density of HF-treated aux1-21 seedlings was very low, and almost totally constituted by ANERs, whereas no AR stage occurred in the HF-treated lax3/aux1-21 seedlings (Fig. 3B and inset). Under IBA + Kin treatment, in contrast to lax3 and the wt, the aux1-21 AR response remained almost totally constituted by ANERs, but the density increased in comparison with the HF treatment, as in lax3 and the wt, remaining, however, less than the half that of the latter genotypes (Fig. 3B and inset). Moreover, the IBA + Kin treatment caused AR formation in the double mutant, with a response similar to aux1-21 (Fig. 3B and inset).
AR formation in intact hypocotyls is altered in all mutants, although at different levels
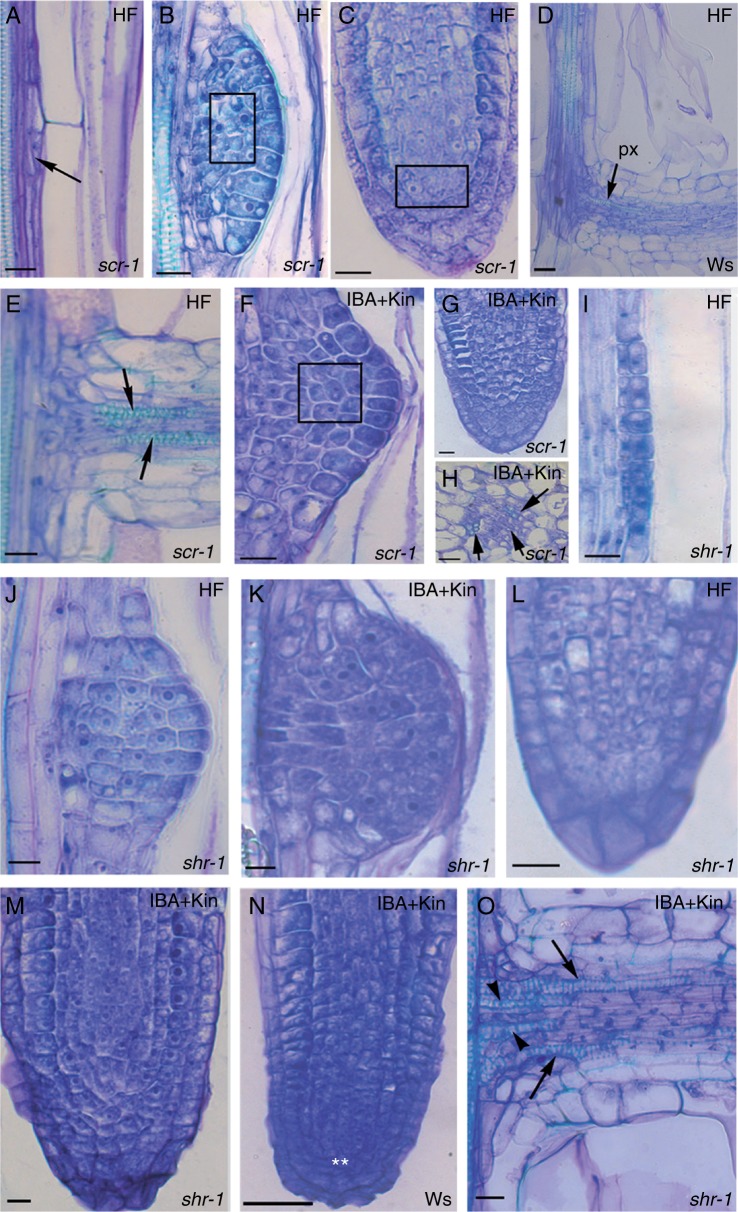
At the hypocotyl–PR junction of HF-treated scr-1 seedlings, the external derivatives of the periclinally divided pericycle bundle occasionally showed oblique/anticlinal divisions (Fig. 4A, arrow), followed by AR formation. In comparison with the wt (Della Rovere et al., 2013), in the scr-1 mutant, the QC was not defined either at stage VII or later, resulting in absence of the QC in the apex of the rare mature ARs (Fig. 4B, C). However, as in the wt, vascular elements with protoxylem-like thickenings were formed in the scr-1 mutant at the point of connection of the AR with the hypocotyl vasculature, and differentiated into the AR exarch protoxylem (Fig. 4D, E, arrows). The HF-treated scr-1 ARs were diarch, with the same wt xylem patterning, but lacking the endodermis. In the presence of IBA + Kin, the AR-forming divisions in the pericycle also occurred sporadically along the hypocotyl, but resulted in anomalous stage VII domes (Fig. 4F), as under HF treatment. However, the apex of the mature ARs was enriched in meristematic cells in comparison with HF treatment, even if there continued to be a lack of cells with QC morphology (Fig. 4G). The endodermis was absent in the IBA + Kin-treated scr-1 ARs as was the case under HF treatment, but xylem formation was enhanced in the stele, frequently leading to triarch ARs (Fig. 4H). At the hypocotyl–PR junction of HF- and IBA + Kin-grown shr-1 seedlings, AR-forming divisions occurred sporadically, but always in the outer layer of the periclinally divided pericycle bundle (Fig. 4I). In contrast to the wt, in shr-1, as for scr-1, QC definition did not occur either at stage VII or later (Fig. 4J, K), resulting in the QC being absent in mature ARs in both treatments (Fig. 4L, M; compare with C, G, N). However the apex of the ARs formed under IBA + Kin treatment was enriched in meristematic and root cap cells in comparison with the HF treatment (Fig. 4L, M), and the AR connection with the hypocotyl was enriched in vascular elements (Fig. 4O, arrows). In the AR primary structure, endodermis was absent, and protoxylem was exarch. Triarch ARs appeared under IBA + Kin treatment, as in scr-1 under the same treatment.
Fig. 4.
scr-1 (A–C, E–H) and shr-1 (I–M, O) AR formation at the hypocotyl–PR junction of 14-day-old seedlings grown under continuous darkness under either HF or IBA + Kin treatment, in comparison with the wt (D, N). (A–C) Anomalous divisions at stage I (A, arrow), and no QC definition in the region marked by a rectangle either at stage VII (B) or in the AR apex (C) of HF scr-1-seedlings. (D, E) Exarch protoxylem differentiation (px, arrows) at the base of scr-1 ARs (E) and wt-ARs (D) under HF treatment. (F, G) Anomalous AR development in IBA + Kin scr-1 seedlings from stage VII (F) to mature tip formation (G). The square in F shows the region where the QC is not defined. (H) Xylem overproduction (arrows) in the stele of scr-1 ARs produced under IBA + Kin treatment. (I) Outer pericycle derivatives leading to AR stage II in HF shr-1 seedlings. (J, K) shr-1 stage VII lacking QC definition under HF (J) and IBA + Kin treatment (K). (L, M) shr-1 mature AR tips with no QC under HF (L) and IBA + Kin (M) treatment, and with a cell enrichment in the latter. (N) Regular apical structure of an AR formed by a IBA + Kin-treated wt seedling. The QC cells are marked by the asterisks. (O) Vascular connection with the hypocotyl of an IBA + Kin-treated shr-1 AR showing exarch protoxylem (arrows) and endarch metaxylem (arrowheads). Longitudinal (A–G and I–O) and transverse (H) sections stained with toluidine blue. Scale bars = 10 µm (G, I–M, O), 20 µm (A–C, E, F, H) and 30 µm (D, N).
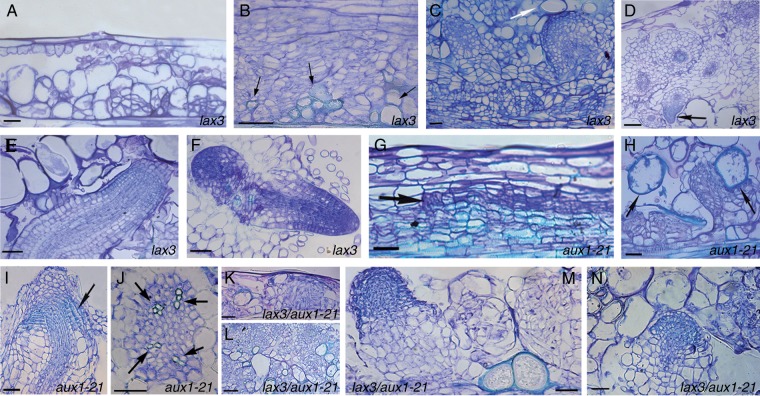
In both HF- and IBA + Kin-treated aux1-21 seedlings initial AR stages (Fig. 5A) were more frequent than advanced AR stages. The rare aux1-21 stage VII domes showed regular QC definition (Fig. 5B, asterisks), and developed into ARPs with regular apical structure. The rare protruding aux1-21 ARs were located at the hypocotyl–PR junction, and showed a regular primary radial pattern, but with overproduction of xylem (Fig. 5C, arrows).
Fig. 5.
AR formation in aux1-21 (A–C), lax3 (D–G) and lax3/aux1-21 (H, I) hypocotyl of 14-day-old seedlings grown under continuous darkness with either IBA + Kin or HF treatment. (A, B) First AR stages along an aux1-21 hypocotyl (A) and more advanced stages, e.g. stage VII (B), at its junction with the PR under IBA + Kin treatment. The regular presence of the QC at stageVII is marked by asterisks. (C) Regular radial pattern but xylem overproduction within the stele (arrows) in mature ARs (IBA + Kin). (D, E) AR stage VII lacking QC definition, in IBA + Kin-treated lax3 seedlings (D), and QC presence, marked by the asterisks, in the tip of a not yet protruded ARP of a lax3 HF seedling (E). (F, G) Details of a mature AR of a IBA + Kin-treated lax3 seedling showing an apex, regular in structure, but enriched in root cap cells (F), and primary structure definition with a regular radial pattern (G). (H, I) AR stages in IBA + Kin-treated lax3/aux1-21 seedlings. Anomalous divisions at stage V (H) and in the root cap initials at stage VII (I) are shown by the arrows. Longitudinal (A, B, D–F, H, I) and transverse (C, G) sections stained with toluidine blue. Scale bars = 20 µm.
The first pericycle divisions in lax3 seedlings were regularly anticlinal, under both HF and IBA + Kin treatment, and led to dome-shaped ARPs, with QC definition not occurring at stage VII (Fig. 5D), but instead occurring later (Fig. 5E, asterisks). There were numerous ARPs advanced in development, and they showed periclinal divisions in root cap initials leading to ARs with multilayered root caps (Fig. 5F). Under both treatments, in lax3 ARs the definition of the primary radial pattern was regular (Fig. 5G), as was the acropetal differentiation and the position of the protoxylem.
At the hypocotyl–PR junction of lax3/aux1-21 seedlings, only anomalous divisions in the outermost layers of the proliferated pericycle bundle occurred, whereas all AR stages were present under IBA + Kin treatment, even though they were characterized by anomalous cell divisions (Fig. 5H, I, arrows). As in IBA + Kin-treated aux1-21 seedlings, early AR stages were prevalent, and, as in lax3, the rare protruding ARs showed a multilayered root cap. Moreover, as in the single mutants, the AR radial pattern was regular, as was the acropetal differentiation of protoxylem and its exarch position.
SCR and AUX1 are expressed in the AR meristemoid founder cells in TCLs, and AUX1 is also expressed in cells leading to xylogenesis
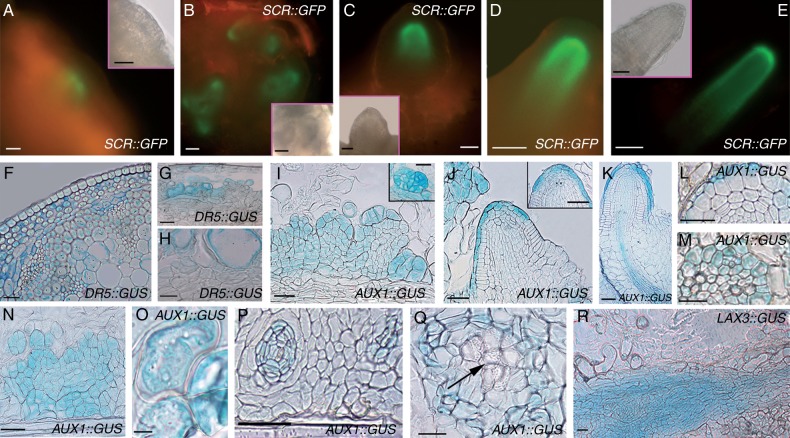
SCR expression was regularly present in the stem endodermis of the basal inflorescence of SCR::GFP plants, but disappeared in the endodermal derivatives in SCR::GFP TCLs. Expression reappeared in the meristematic clumps leading to AR meristemoids, and continued to be present in the latter (Fig. 6A, B). In stage VII domes, protruding ARPs and elongating ARs, SCR expression was localized in the same AR stages from intact hypocotyls (Fig. 6C–E; compare with Fig. 2C–E).
Fig. 6.
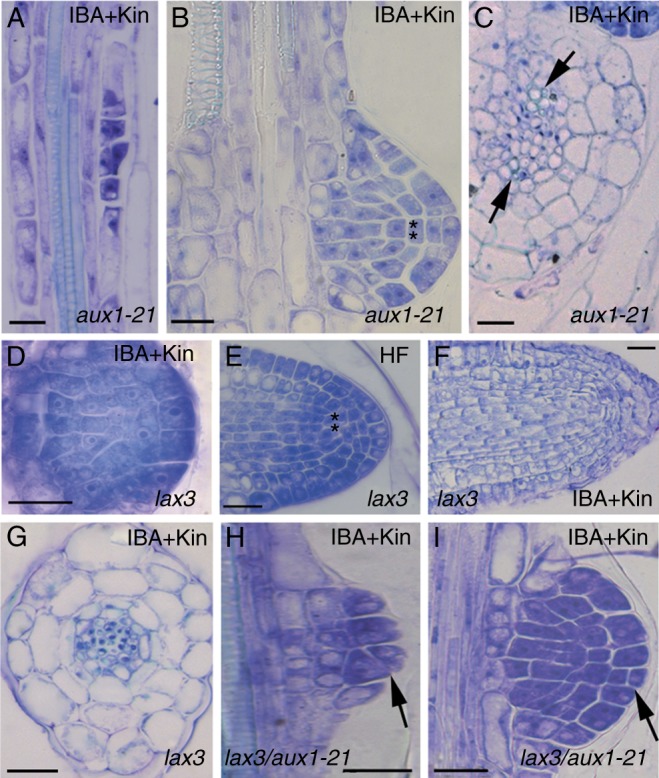
Expression of SCR (A–E), AUX1 (I–Q) and LAX3 (R) during AR formation and xylogenesis in TCLs, and endogenous auxin presence before and during TCL culture (F–H), by the use of SCR::GFP, AUX1::GUS, LAX3::GUS and DR5::GUS lines. (A) Weak SCR expression in a superficial meristematic clump. (B) High SCR expression in the root meristemoids. (C–E) SCR signal localization in a stage VII AR (C), ARP (D) and mature AR apex (E). (A–E, microscopic fluorescence pictures. Insets in A–C and E are corresponding bright-field images). (F) Pattern of DR5::GUS in the basal inflorescence stem before culture. (G, H) Auxin accumulation in forming (G) and maturing (H) pitted cells. (I) AUX1 expression in meristematic clumps and root meristemoids (inset). (J) Protruding ARP with high AUX1 expression in the root cap, and weak expression in the QC (asterisks in the inset). (K) Protruded AR with strong AUX1 expression in the lateral cap, and forming epidermis and vasculature. (L, M) AUX1 expression in a few epidermal cells (L), and pericycle, protoxylem and protophloem poles (M) in the AR primary structure. (N–Q) AUX1 expression in callus cells (N), in scattered cells with degenerating protoplast and lignifying cell wall (O), and in forming xylary nodules (P), and disappearance of expression in mature xylary elements (arrow) (Q). (R) LAX3 expression in a vascular strand. Longitudinal sections (A–E, G–K, N–R) and cross-sections (F, L, M). Scale bars = 10 µm (M, O), 30 µm (F, H, R), 50 µm (A–E, I–L, N, P, Q and insets in A, C, E, I, J) and 100 µm (G and inset in B).
In the basal stem inflorescence internodes of DR5::GUS plants, auxin was detected in randomly located cortical and endodermal cells (Fig. 6F). In the TCLs excised from these internodes and cultured with IBA + Kin, auxin accumulated in root meristemoids and ARP tips, but also in the QC, inner columella and differentiating protoxylem of the mature ARs, confirming previous results (Della Rovere et al., 2013). In addition, the cells involved in xylogenesis showed auxin accumulation (Fig. 6G, H).
AUX1 was precociously expressed in cultured AUX1::GUS TCLs, marking the meristematic clumps and the AR meristemoids (Fig. 6I and inset). Expression continued in ARPs, i.e. forming cap, epidermis and, faintly, QC and surrounding initials (Fig. 6J and inset), and in protruding ARs, i.e. the lateral cap, and forming epidermis and vasculature (Fig. 6K). In the primary structure of the elongated ARs, AUX1 was expressed in some epidermal cells, pericycle and protophloem and protoxylem poles (Fig. 6L, M). AUX1 expression was also present in groups of callus cells arising from proliferated inner endodermis (Fig. 6N), and in differentiating vascular strands and nodules originating from them, as in single/small groups of cells directly developing into pitted elements (Fig. 6O, P), with expression ceasing after the completion of cell wall lignification and developmental cell death (Fig. 6Q). Also LAX3 expression occurred in all the xylogenic cell types, e.g. the vascular strands (Fig. 6R). Moreover, it occurred in the AR meristemoids, and ARPs/ARs as in intact hypocotyls under the same hormonal condition (Della Rovere et al., 2013, and data not shown).
SCR-1, SHR-1 and AUX1 are necessary for AR formation in TCLs
The basal stem internodes of scr-1 and shr-1 showed three irregular cortical layers between the epidermis and fibres, and no endodermis, as in the apical stem internodes (Fukaki et al., 1998). At the end of the culture period, macroscopic ARs only sporadically appeared on the basal internode TCLs of both mutants. Moreover, the mean number of ARs per rooting TCL was about 3-fold lower than in the wt, without significant differences between the two (Fig. 7A).
Fig. 7.
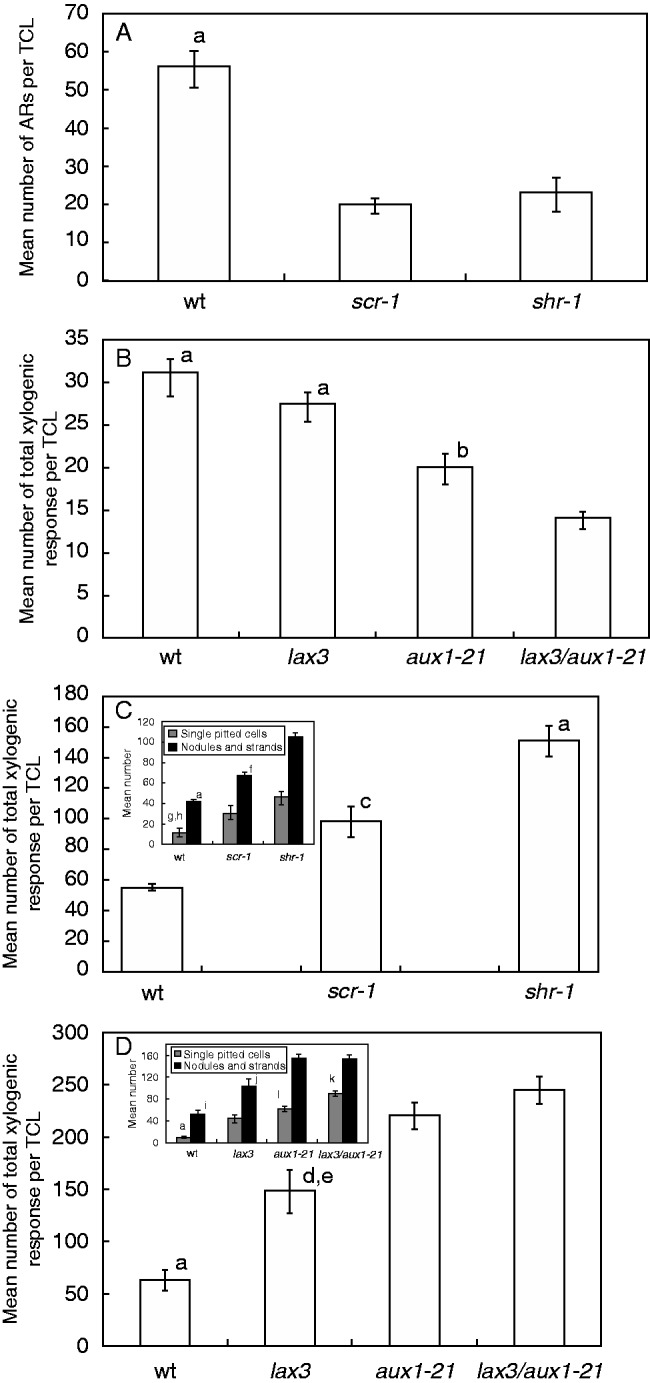
Macroscopic AR response (A, B) and xylogenic response (C, D) in TCLs from Ws (wt), scr-1 and shr-1 (A, C), and Col (wt), lax3, aux1-21 and lax3/aux1-21 (B, D) after 20 d of culture with IBA + Kin under darkness. (A, B) Mean number/TCL (±s.e.) of protruded ARs plus ARPs dissected from the callus under the stereomicroscope. (C, D) Mean number/TCL (± s.e.) of total xylogenic response, i.e. single cells with pitted cell walls, and xylogenic nodules and strands, counted together, in Ws (wt), scr-1 and shr-1 (C), and Col (wt), lax3, aux1-21 and lax3/aux1-21 (D). Insets in C and D show the mean number/TCL (± s.e.) of single pitted cells, and of nodules and strands. aP < 0·001 with the other genotypes. bP < 0·05 with lax3/aux1-21. cP < 0·01 with the wt. dP < 0·01 with aux1-21. eP < 0·001 with lax3/aux1-21. f,gP < 0·001 with shr-1 within the same category. hP < 0·05 with scr-1 within the same category. iP < 0·001 with aux1-21 and lax3/aux1-21 within the same category. jP < 0·01 with the wt, aux1-21 and lax3/aux1-21 within the same category. kP < 0·001 with aux1-21 and lax3 within the same category. lP < 0·05 with lax3 within the same category. Columns with no letter/the same letter are not significantly different (one-way ANOVA followed by Tukey’s post-test). n = 40 (A, B), n = 20 (C, D). Second replicate of the second year.
The basal stem internodes of lax3, aux1-21 and lax3/aux1-21 mutants showed the same regular structure as the wt internodes. At the end of the culture period, the mean number of ARs per rooting TCL was significantly reduced on aux1-21 and lax3/aux1-21 TCLs in comparison with the wt and lax3, whereas there was no significant difference between the latter two (Fig. 7B).
AR formation is competitively inhibited by xylogenesis in shr-1 and scr-1 TCLs
In both shr-1- and scr-1 TCLs, cell groups in the innermost mutant cortical layer (i.e. that positioned as the endodermis in the wt) divided periclinally (Fig. 8A, B) at the same time (day 3) as the endodermis in wt TCLs. In contrast to the scattered groups generated by the wt endodermis, shr-1 and scr-1 periclinal derivatives formed a homogeneous meristematic bundle of superimposed cell files all along the explant (Fig. 8C). Strands of xylary elements with spiral/annular thickenings, but mainly pitted thickenings, appeared conspicuously within the meristematic bundle (Fig. 8D, E, arrows), whereas meristematic clumps, leading to AR meristemoids, appeared sporadically at its surface. In contrast, xylogenesis was reduced, but root meristemoid formation was enhanced, in the endodermis derivatives of wt TCLs. At day 7, the first xylogenic nodules and AR domes, surrounded by cells with pitted cell walls, appeared in shr-1 and scr-1 TCLs (Fig. 8F, G). In contrast, xylogenesis remained confined to the innermost part of the proliferating endodermis in wt TCLs, and the AR domes were not surrounded by lignified cells. During the second week, ARPs grew toward the epidermis in wt TCLs, and occasionally also in shr-1/scr-1 TCLs (Fig. 8H); however, in the mutants, in contrast to the wt, the late-forming root meristemoids remained entrapped by lignified cells (Fig. 8I, arrow). At the end of the culture period, as in intact hypocotyls under the same treatment, and in contrast to the regular apical structure and the diarch xylem pattern of the ARs in wt TCLs (Fig. 8J, K), shr-1 and scr-1 ARs showed an anomalous stem cell niche and root cap, and no QC and endodermis. However, they showed precocious epidermis differentiation (Fig. 8L, M) and xylem overproduction leading to triarch/polyarch ARs (Fig. 8N). Moreover, xylogenesis was quantified as single pitted elements, nodules and strands. The total xylogenic response in the TCLs of both mutants was significantly higher than in wt TCLs, and significantly higher in shr-1 TCLs than in scr-1 TCLs (Fig. 7C); however, in both mutants, the main typology of the xylogenic response was made up of cells with pitted thickenings, both singly (Fig. 7C, inset) and located within nodules and strands (Fig. 8D, E, I).
Fig. 8.
AR formation and xylogenesis in shr-1 and scr-1 TCLs during culture with IBA + Kin under darkness. (A, B) Early periclinal divisions in the innermost irregular cortical layer in shr-1 (A) and scr-1 (B) (day 3). (C) Bundle of meristematic cell files formed by the innermost irregular cortical layer in shr-1. (D, E) Xylary elements in the inner part of the meristematic bundle in shr-1 (D) and scr-1 (E) (day 7). The arrows show elements with pitted cell wall thickenings. (F) Details of xylogenic nodules (arrows) in a scr-1 TCL. (G) Domed root meristemoid in scr-1 TCLs, surrounded by lignified callus cells (day 7). (H) ARP not yet protruded from the explant in scr-1. (I) Callus cells highly expanded and with pitted cell walls around a late-formed meristemoid (arrow) in shr-1 (day 14). (J, K) Regular AR tip, with the QC marked by the asterisks (J) and regular diarch primary structure (K) of ARs from wt (Ws) TCLs (day 20). (L, M) AR tips with anomalous stem cell niche and root cap cell enrichment in scr-1 (L) and shr-1 (M), and precocious differentiation of the epidermis with rhizoblasts in the latter (day 20). (N) Triarch AR from shr-1 (xylem poles shown by the arrows). Longitudinal (A–J) and transverse (K, N) sections stained with toluidine blue. Scale bars = 20 µm (A, N), 30 µm (J, K) and 40 µm (B–I, L, M).
Xylogenesis is also enhanced in aux1-21 and lax3/aux1-21 TCLs concomitantly with AR reduction
The first divisions in the stem endodermis of lax3 TCLs occurred as early as in wt TCLs, and similarly originated groups of derivative cells (Fig. 9A), forming scattered cell groups where vascular strands, nodules and single/small groups of pitted cells (Fig. 9B, arrows) differentiated. Moreover, as in wt TCLs, a lot of AR meristemoids were formed; however, lax3 AR meristemoids developed into ARPs with a disorganized niche, numerous root cap initials and no QC. These ARPs were impeded in terms of protrusion by cells with pitted thickenings (Fig. 9C, arrow); nonetheless they became ARs, with most remaining entrapped within the explant (Fig. 9D). These ARs exhibited a flattened, cell-enriched, apex with a multilayered root cap (Fig. 9E), as in the intact hypocotyls under the same hormonal treatment, and formed a lot of lateral root primordia (LRPs) (Fig. 9D, arrow, and F). The few elongated ARs on lax3 TCLs were frequently triarch/polyarch.
Fig. 9.
AR formation and xylogenesis in lax3 (A–F), aux1-21 (G–J) and lax3/aux1-21 (K–N) TCLs during culture with IBA + Kin under darkness. (A) Early formation of scattered endodermis derivatives in lax3 TCLs (day 3). (B) Cells with pitted thickenings (arrows) in the callus of a lax3 TCL. (C) Anomalous lax3 ARPs surmounted by pitted elements (arrow) (day 14). (D, E) Mature ARs within a day 20 lax3 explant showing LRPs (arrow in D), a flattened apex and a multilayered root cap (E). (F) Primary region of a lax3 AR showing two LRPs. (G) Bundle of endodermis periclinal derivatives in an aux1-21 TCL with a xylogenic strand enriched in pitted elements (arrowheads) and a root clump in the outermost bundle layer (arrow) (day 7). (H) Meristemoids and domed ARPs confined with xylogenic cells (arrows) in aux1-21 (day 14). (I) AR apex with early differentiation/expansion in the cortex, and meristematization in the epidermis and lateral cap (arrow) (aux1-21 TCL, day 20). (J) Tetrarch xylem (arrows) within the stele of an aux1-21 AR. (K) Formation of the bundle of endodermis derivatives in a lax3/aux1-21 TCL (day 3). (L) Xylogenesis, mainly formed by pitted cells, in the callus of an aux1-21 TCL (day 14). (M) A lax3/aux1-21 ARP with anomalous structure near xylogenesis (day 14). (N) LRP on a lax3/aux1-21 AR, with an excess of meristematization (day 20). Longitudinal (A–E, G–I, K–M) and transverse (F, J, N) sections stained with toluidine blue. Scale bars = 50 µm (A–C, E–K, M, N) and 100 µm (D, L).
The quantification of xylogenesis at the end of the culture period revealed that the total response of lax3 TCLs was significantly higher than in the wt explants (Fig. 7D), with pitted cells prevalent, both as single elements and within the strands/nodules (Figs 7D inset, and 9B).
In contrast to lax3 TCLs, aux1 TCLs exhibited a uniform bundle of endodermis periclinal derivatives along the explant, and an enrichment in meristematic cells and vascular strands, resembling shr-1 and scr-1 TCL responses (Fig. 9G; compare with Fig. 8D, E). AR clumps/meristemoids were randomly formed by the outermost layers of the bundle (Fig. 9G, arrow), and most remained entrapped within the pitted cells (Fig. 9H, arrows). The few meristemoids continuing development produced ARPs with early expanding cortex, but with a meristematic epidermis and lateral cap (Fig. 9I, arrow). The elongated aux1-21 ARs were usually tetrarch (Fig. 9J). The quantification of the total xylogenic response showed that aux1-21 TCLs produced significantly more xylary cells than lax3 TCLs, and pitted cells in particular (Fig. 7D inset; Fig. 9G, arrowheads).
The bundle of endodermis derivatives formed in lax3/aux1-21 TCLs was scattered as in lax3 TCLs, but enriched in meristematic cells as in aux1-21 TCLs (Fig. 9K; compare with A). Xylogenesis was enhanced as in aux1-21 TCLs (Figs 7D and 9L), and similarly irregular domed ARPs appeared sporadically (Fig. 9M), and developed into ARs with the same anomalies as aux1-21 ARs, but able to form LRPs as lax3 ARs (Fig. 9N).
DISCUSSION
The results show that SHR, SCR and AUX1 are needed for AR formation in intact hypocotyls and cultured TCLs, and the reduction in AR formation in shr-1, scr-1, aux1-21 and lax3/aux1-21 mutants is coupled to a promotion of xylogenesis.
SHR and SCR mark AR progenitor cells, with AUX1 exhibiting a pivotal role
We demonstrated that SCR expression started in the AR founder cells in both intact hypocotyls and cultured TCLs, suggesting that SCR affected the initiation of ARs in the initiating tissues. SCR is activated by SHR in the PR (Helariutta et al., 2000), and the formation of a complex between the two proteins follows, and activates numerous downstream genes, including cell cycle components (Sozzani et al., 2010). It is possible that SCR is also activated by SHR during AR initiation, and that the transcription factors together activate downstream genes necessary for AR construction. Thus, the two genes might be necessary for the priming of the cell identity progenitors of the AR formation process, independently of the starting tissue (hypocotyl pericycle vs. stem endodermis), as is supported by the finding of similar, and strongly reduced, AR response in the hypocotyls and TCLs of their mutants. The same transcription factors were also necessary later in AR development, because QC definition and primary radial patterning depended on their action. In fact, SCR expression occurred at stage VII, when the QC is defined in the ARP (Della Rovere et al., 2013). In addition, the QC was not defined, and the endodermis was lacking, in the ARs of scr-1 and shr-1 TCLs and seedlings, as in their PR (Di Laurenzio et al., 1996; Helariutta et al., 2000).
Endogenous indole acetic acid (IAA) accumulation is an early event in LR formation (Benková et al., 2003). The present results show that it is also important in AR formation, because IAA accumulation in the pericycle at the hypocotyl–PR junction and in the basal inflorescence stem occurred before the first AR-forming divisions, and increased at their appearance in both intact hypocotyls and TCLs. In the PR tip, the cortex/endodermis initial cells need the presence of SHR and SCR, and of a local auxin increase, for the induction of downstream genes (Cruz-Ramírez et al., 2012), but a high level of AUX1 transcription also occurs (Sozzani et al., 2010). We observed that the increase in endogenous IAA accumulation at AR stage I in the hypocotyl, and in the AR meristematic clumps in TCLs, was concomitant with the appearance of AUX1 expression. This suggests that AUX1, mediating the auxin influx specifically in the AR-initiating cells, might be related to the priming activity of SHR and SCR. In contrast, LAX3 expression occurred only from AR stage II onwards, suggesting that LAX3 was involved later than AUX1 in the AR formation process. In agreement with this, AR formation of aux1-21 and lax3/aux1-21 seedlings and TCLs was strongly inhibited, as in shr-1 and scr-1, whereas that of lax3 was comparable with that of the wt. It has been suggested that AUX1 recognizes endogenous IAA and not IBA, whereas IBA may be a substrate of LAX3 (Liu et al., 2012). In agreement with this, AUX1 expression was not enhanced by IBA + Kin, in contrast to LAX3 (Della Rovere et al., 2013; this study). It is thus possible that AUX1, and not LAX3, is involved in creating the initial endogenous IAA distribution essential for AR initiation by SHR and SCR. In contrast, the role of LAX3 is restricted to AR development, being necessary only from stage II onwards.
There is an inverse relationship between AR formation and xylogenesis, governed by the same genes
Arabidopsis TCLs underwent xylogenesis in addition to AR formation under the exogenous IBA + Kin treatment (Falasca et al., 2004; this study), and the same treatment was necessary to cause ectopic metaxylem formation in the wt hypocotyls, even if this xylogenic response was limited in comparison with AR formation. Interestingly, xylogenesis increased in the seedlings and TCLs of all mutants, and this increase was always associated with increased xylem formation in the stele of the ARs, demonstrating that the two xylary processes were strictly related, sharing SHR, SCR, AUX1 and LAX3 activities.
Moreover, in seedlings and TCLs, xylogenesis began, and continued, where endogenous IAA accumulated, showing that high levels of IAA were essential to the process. We observed that ectopic metaxylem was the only xylogenic component in the intact hypocotyls, and ectopic metaxylem-like cells were the predominant component in TCLs. Metaxylem specification is known to be promoted by high auxin and high expression of the ZD-ZIP III genes (Ursache et al., 2014), and the HD-ZIP III transcription factor ATHB-8 is induced by auxin and involved in xylem specification in planta, and in xylogenesis in TCLs (Baima et al., 2000, 2014). ATHB-8, like other members of the family, is regulated by microRNA 165/6 produced by the SHR–SCR complex (Carlsbecker et al., 2010). Moreover, shr-1 and scr-1 seedlings are known to exhibit a high endogenous auxin content (Lucas et al., 2011; Moubayidin et al., 2013), and no formation of microRNA 165/6 (Muraro et al., 2014). Thus, a high endogenous auxin level and the absence of degradation of HD-ZIP III transcription factors might explain the observed enhancement of xylogenesis in the seedlings and TCLs of these mutants, and support the importance of SHR and SCR also in the xylogenic process. The exclusive ectopic metaxylem which occurs in the hypocotyl of lax3, aux1-21 and their double mutant, and the prevalent ectopic metaxylem-like production of their TCLs, might be indicative of an endogenous IAA accumulation also in the latter mutants. Xylogenesis was enhanced in hypocotyls and TCLs of aux1-21 and lax3/aux1-21 in comparison with lax3, and this event was concomitant with a highly reduced rooting in the aux1 mutants only. The non-utilization of the endogenous auxin in the AR process might have caused an excess of endogenous auxin in the aux1 mutants, enhancing xylogenesis as an alternative to rooting. Consistent with this, high xylogenesis is caused by IAA in walnut microcuttings recalcitrant to rooting (Reverberi et al., 2001).
Thus, there is an inverse relationship between AR formation and xylogenesis, but with an important difference between the auxin influx carriers, highlighted by the response of their mutant TCLs. In fact, in lax3 TCLs, ARs were formed, but xylogenesis trapped them within the explant, blocking only their macroscopic protrusion. In contrast, in the TCLs of aux1-21 single/double mutants, the trapping effect by the xylogenic cells was limited, because the bulk of the ARs were arrested at early stages, whereas xylogenesis was overproduced. This means that xylogenesis occurred as an alternative programme to AR formation in aux1-21 single/double mutant TCLs, from the first cell divisions onwards. Thus, in contrast to LAX3, AUX1 might exert a pivotal role in rhizogenesis/xylogenesis switching, in accordance with its role in other developmental processes, including in vitro organogenesis (Kakani et al., 2009; Péret et al., 2012).
In conclusion, AR formation and xylogenesis are developmental programmes which are inversely related, but involve a fine-tuning by the same proteins, i.e. SHR, SCR and AUX1. The pericycle activity is central for the equilibrium between xylary development and AR formation in the hypocotyl, with a role for AUX1 in the switch between the programmes, necessary for their balance. The results also open the way to understanding the genetic basis of AR recalcitrance in in vitro cultured cuttings in which block of AR formation is associated with enhanced xylogenesis resulting in unsuccessful micropropagation.
ACKNOWLEDGEMENTS
The authors acknowledge D. Serafini Fracassini for critical reading of the manuscript, F. Pantanella for technical support, P. Benfey for pSCR::GFP and shr-1 null mutant seeds, S. Sabatini for DR5::GUS and scr-1 null mutant seeds, and M. Bennett for AUX1::GUS, LAX3::GUS, and aux1-21, lax3 and lax3/aux1-21 null mutant seeds. Funding was provided by Università degli Studi di ROMA “La Sapienza” Progetti di Ateneo to G.F.
LITERATURE CITED
- Baima S, Tomassi M, Matteucci A, Altamura MM, Ruberti I, Morelli G. 2000. Role of the ATHB-8 gene in xylem formation. In: Savidge RA, Barnett JR, Napier R, eds. Cell and molecular biology of wood formation . Oxford: BIOS Scientific Publishers, 445–455. [Google Scholar]
- Baima S, Forte V, Possenti M, et al. 2014. Negative feedback regulation of auxin signaling by ATHB8/ACL5–BUD2 transcription module. Molecular Plant 7: 1006–1025. [DOI] [PubMed] [Google Scholar]
- Benková E, Michniewicz M, Sauer M, et al. 2003. Local, efflux-dependent auxin gradients as a common module for plant organ formation. Cell 115: 591–602. [DOI] [PubMed] [Google Scholar]
- Bielach A, Podlešáková K, Marhavý P, et al. 2012. Spatiotemporal regulation of lateral root organogenesis in Arabidopsis by cytokinin. The Plant Cell 24: 3967–3981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carlsbecker A, Lee JY, Roberts CJ, et al. 2010. Cell signalling by microRNA165/6 directs gene dose-dependent root cell fate. Nature 465: 316–320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cruz-Ramírez A, Díaz-Triviño S, Blilou I, et al. 2012. A bistable circuit involving SCARECROW–RETINOBLASTOMA integrates cues to inform asymmetric stem cell division. Cell 150: 1002–1015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cui H, Levesque MP, Vernoux T, et al. 2007. An evolutionarily conserved mechanism delimiting SHR movement defines a single layer of endodermis in plants. Science 316: 421–425. [DOI] [PubMed] [Google Scholar]
- Cui H, Hao Y, Kovtun M, et al. 2011. Genome-wide direct target analysis reveals a role for SHORT-ROOT in root vascular patterning through cytokinin homeostasis. Plant Physiology 157: 1221–1231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Della Rovere F, Fattorini L, D’Angeli S, Veloccia A, Falasca G, Altamura MM. 2013. Auxin and cytokinin control formation of the quiescent centre in the adventitious root apex of arabidopsis. Annals of Botany 112: 1395–1407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Di Laurenzio L, Wysocka-Diller J, Malamy JE, et al. 1996. The SCARECROW gene regulates an asymmetric cell division that is essential for generating the radial organization of the Arabidopsis root. Cell 86: 423–433. [DOI] [PubMed] [Google Scholar]
- Falasca G, Altamura MM. 2003. Histological analysis of adventitious rooting in Arabidopsis thaliana (L.) Heynh seedlings. Plant Biosystems 137: 265–274. [Google Scholar]
- Falasca G, Zaghi D, Possenti M, Altamura MM. 2004. Adventitious root formation in Arabidopsis thaliana thin cell layers. Plant Cell Reports 23: 17–25. [DOI] [PubMed] [Google Scholar]
- Fukaki H, Wysocka-Diller J, Kato T, Fujisawa H, Benfey PN, Tasaka M. 1998. Genetic evidence that the endodermis is essential for shoot gravitropism in Arabidopsis thaliana . The Plant Journal 14: 425–430. [DOI] [PubMed] [Google Scholar]
- Fukuda H. 1997. Tracheary element differentiation. The Plant Cell 9: 1147–1156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Helariutta Y, Fukaki H, Wysocka-Diller J, et al. 2000. The SHORT-ROOT gene controls radial patterning of the Arabidopsis root through radial signaling. Cell 101: 555–567. [DOI] [PubMed] [Google Scholar]
- Kakani A, Li G, Peng Z. 2009. Role of AUX1 in the control of organ identity during in vitro organogenesis and in mediating tissue specific auxin and cytokinin interaction in Arabidopsis. Planta 229: 645–657. [DOI] [PubMed] [Google Scholar]
- Koizumi K, Hayashi T, Gallagher KL. 2012. SCARECROW reinforces SHORT-ROOT signaling and inhibits periclinal cell divisions in the ground tissue by maintaining SHR at high levels in the endodermis. Plant Signaling and Behavior 7: 1573–1577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu X, Barkawi L, Gardner G, Cohen JD. 2012. Transport of indole-3-butyric acid and indole-3-acetic acid in Arabidopsis hypocotyls using stable isotope labelling. Plant Physiology 158: 1988–2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lucas M, Swarup R, Paponov IA, et al. 2011. SHORT-ROOT regulates primary, lateral, and adventitious root development in Arabidopsis. Plant Physiology 155: 384–398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Malamy JE, Benfey PN. 1997. Organization and cell differentiation in lateral roots of Arabidopsis thaliana . Development 124: 33–44. [DOI] [PubMed] [Google Scholar]
- Miyashima S, Sebastian J, Lee JY, Helariutta Y. 2013. Stem cell function during plant vascular development. EMBO Journal 32: 178–193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moubayidin L, Di Mambro R, Sozzani R, et al. 2013. Spatial coordination between stem cell activity and cell differentiation in the root meristem. Developmental Cell 26: 405–415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Muraro D, Mellor N, Pound MP, et al. 2014. Integration of hormonal signaling networks and mobile microRNAs is required for vascular patterning in Arabidopsis roots. Proceedings of the National Academy of Sciences, USA 111: 857–862. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Péret B, Swarup K, Ferguson A, et al. 2012. AUX/LAX genes encode a family of auxin influx transporters that perform distinct functions during Arabidopsis development. The Plant Cell 24: 2874–2885. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reverberi M, Falasca G, Lauri P, Caboni E, Altamura MM. 2001. Indoleacetic acid induces xylem formation instead of rooting in walnut (Juglans regia L.) microcuttings. Plant Biosystems 135: 71–77. [Google Scholar]
- Sabatini S, Heidstra R, Wildwater M, Scheres B. 2003. SCARECROW is involved in positioning the stem cell niche in the Arabidopsis root meristem. Genes and Development 17: 354–358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sarkar AK, Luijten M, Miyashima S, et al. 2007. Conserved factors regulate signalling in Arabidopsis thaliana shoot and root stem cell organizers. Nature 446: 811–814. [DOI] [PubMed] [Google Scholar]
- Scheres B, Di Laurenzio L, Willemsen V, et al. 1995. Mutations affecting the radial organisation of the Arabidopsis root display specific defects throughout the embryonic axis. Development 121: 53–62. [Google Scholar]
- Sozzani R, Cui H, Moreno-Risueno MA, et al. 2010. Spatiotemporal regulation of cell-cycle genes by SHORTROOT links patterning and growth. Nature 466: 128–132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Swarup K, Benková E, Swarup R, et al. 2008. The auxin influx carrier LAX3 promotes lateral root emergence. Nature Cell Biology 10: 946–954. [DOI] [PubMed] [Google Scholar]
- Takahashi F, Sato-Nara K, Kobayashi K, Suzuki M, Suzuki H. 2003. Sugar-induced adventitious roots in Arabidopsis seedlings. Journal of Plant Research 116: 83–91. [DOI] [PubMed] [Google Scholar]
- Ugartechea-Chirino Y, Swarup R, Swarup K, et al. 2010. The AUX1 LAX family of auxin influx carriers is required for establishment of embryonic root cell organization in Arabidopsis thaliana . Annals of Botany 105: 277–289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ursache R, Miyashima S, Chen Q, et al. 2014. Tryptophan-dependent auxin biosynthesis is required for HD-ZIP III-mediated xylem patterning. Development 141: 1250–1259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wysocka-Diller JW, Helariutta Y, Fukaki H, Malamy JE, Benfey PN. 2000. Molecular analysis of SCARECROW function reveals a radial patterning mechanism common to root and shoot. Development 127: 595–603. [DOI] [PubMed] [Google Scholar]
- Zhang W, Swarup R, Bennett M, Schaller GE, Kieber JJ. 2013. Cytokinin induces cell division in the quiescent center of the Arabidopsis root apical meristem. Current Biology 23: 1979–1989. [DOI] [PubMed] [Google Scholar]