Abstract
BACKGROUND
Methods for direct measurement of glomerular filtration rate (GFR) are expensive and inconsistently applied across transplant centers. The Modified Diet in Renal Disease (MDRD) equation is commonly used for GFR estimation, but is inaccurate for GFRs > 60 ml/min per 1.73 m2. The Chronic Kidney Disease Epidemiology Collaboration (CKDEPI) and Wright equations have shown improved predictive capabilities in some patient populations. We compared these equations to determine which one correlates best with direct GFR measurement in lung transplant candidates.
METHODS
We conducted a retrospective cohort analysis of 274 lung transplant recipients. Pre-operative GFR was measured directly using a radionuclide GFR assay. Results from the MDRD, CKDEPI, Wright, and Cockroft–Gault equations were compared with direct measurement. Findings were validated using logistic regression models and receiver operating characteristic (ROC) analyses in looking at GFR as a predictor of mortality and renal function outcomes post-transplant.
RESULTS
Assessed against the radionuclide GFR measurement, CKDEPI provided the most consistent results, with low values for bias (0.78), relative standard error (0.03) and mean absolute percentage error (15.02). Greater deviation from radionuclide GFR was observed for all other equations. Pearson’s correlation between radionuclide and calculated GFR was significant for all equations. Regression and ROC analyses revealed equivalent utility of the radionuclide assay and GFR equations for predicting post-transplant acute kidney injury and chronic kidney disease (p < 0.05).
CONCLUSIONS
In patients being evaluated for lung transplantation, CKDEPI correlates closely with direct radionuclide GFR measurement and equivalently predicts post-operative renal outcomes. Transplant centers could consider replacing or supplementing direct GFR measurement with less expensive, more convenient estimation by using the CKDEPI equation.
Keywords: lung transplantation, renal function, GFR equations, CKDEPI, MDRD
Pre-operative renal function is associated with outcomes in surgical patients.1–3 This relationship is especially significant in lung transplant populations, given the increased susceptibility to kidney damage after transplantation.4 Several mechanisms contribute to renal deterioration in lung recipients, including iatrogenic factors related to diuresis and nephrotoxic treatment regimens.4 Glomerular filtration rate (GFR) is the primary indicator of renal function and its determination is an important part of the candidate evaluation process in lung transplantation.5 Direct GFR measurement using inulin clearance or radionuclide markers is the “gold standard” for GFR determination.6,7 However, direct measurement is more expensive and time-consuming. Therefore, estimated GFR based on serum creatinine is commonly employed in clinical practice.
The Modified Diet in Renal Disease (MDRD) equation is the most commonly used for GFR determination. The MDRD accounts for age, gender and race, and has been shown to accurately predict chronic kidney disease after lung transplant.8 However, the MDRD was developed in a population of patients with kidney disease.9 Studies have shown that the MDRD underestimates GFR for values >60 ml/min per 1.73 m2.10 In recent years, newer equations have been developed. The Chronic Kidney Disease Epidemiology Collaboration (CKDE-PI) equation was developed in a population that included healthy individuals and patients with various diseases.11 The CKDEPI has shown improved predictive capabilities in some patient populations.12,13 The Wright equation was developed in a cohort of oncology patients and has been shown to be useful for dosing chemotherapeutic agents.14,15 Similar to the MDRD, the Cockroft-Gault (CG) equation was developed in patients with kidney disease.14 The CG is consistently featured in the literature, amidst inconsistent conclusions about superiority compared with the MDRD.9,14,16,17
In this study we aimed to evaluate which of these four equations, the MDRD, CKDEPI, Wright or CG, most accurately estimates GFR in a population of end-stage lung disease patients being evaluated for lung transplantation, when compared with a direct radionuclide GFR (rGFR) measurement. In addition, we evaluated the utility of various GFR equations for predicting post-transplant outcomes, including acute kidney injury (AKI); Stage 3 chronic kidney disease at 6 months (CKD); dialysis requirement prior to discharge; and mortality at 1, 3 and 5 years.
Methods
Study design and patient selection
We conducted a retrospective cohort analysis of lung recipients at a single center from January 1, 2006 to March 1, 2012. The study protocol was approved by the institutional review board at Duke University Medical Center, Durham, NC. Patients included in the study underwent lung transplantation before March 2012 and had available pre-operative serum creatinine data. Patients were excluded if they underwent repeat or multiple-organ transplantation.
Since 2006, our center protocol has included the rGFR assay in lung transplant evaluation for patients believed to be at increased risk for developing post-operative kidney disease. Risk determination is based on several factors, including age, history of kidney dysfunction and functional status. At our center, patients with rGFR <50 ml/min per 1.73 m2 are excluded from transplant with rare exceptions. Therefore, the GFR is a critical component in the evaluation of potential lung recipients.
In this study we assessed the correlation between GFR equations and the rGFR assay in patients who had available radionuclide study results and sufficient data to calculate GFR using the various equations. These patients underwent rGFR testing because they were considered at risk for developing post-transplant kidney disease. Validation was performed by considering the utility of GFR equations for predicting post-operative outcomes, including: mortality at 1, 3 and 5 years; AKI; dialysis requirement before discharge; and Stage 3 CKD at 6 months. Similar to National Surgical Quality Improvement Program (NSQIP) definitions, AKI was defined as serum creatinine >3 mg/dl within the first week after transplant. CKD was defined as GFR <60 ml/min per 1.73 m2 within the first 6 months after transplant. Further validation of results was performed in a separate cohort of patients who had sufficient data to calculate GFR but no rGFR study results—the validation cohort.
Radionuclide GFR measurement
Technetium-99m diethylene triamine pentaacetic acid (Tc-99m DTPA) is filtered but neither reabsorbed nor excreted in kidney tubules. The Tc-99m DTPA approach is well known as an accurate measure of renal function, but it is not widely used due to concerns about cost and convenience.6 Studies have demonstrated an excellent correlation between this method and the inulin clearance method for measuring GFR.6,18 To determine GFR, 0.93 mCi of Tc-99m DTPA was administered to patients intravenously. Sequential blood draws were performed 1 and 3 hours after radiotracer injection. Measured GFR values were normalized to a body surface area of 1.73 m2.
Data collection
Our institution maintains a data warehouse containing administrative, financial and clinical information generated during patient care.19 This system was used to acquire demographic information, pre-existing comorbidities, operative characteristics, post-operative complications and survival information. Data were supplemented and validated with manual chart review and with information from our institutional database for cardiovascular disease. Survival data were cross-referenced with the Social Security Death Index, providing for a more complete survival follow-up.
Statistical analysis
Statistical analyses were carried out with SAS version 9.2 (SAS Institute, Inc., Cary, NC). Results from the four equations were compared with data from the nuclear assay based on the following parameters:
Linear regression models were generated for all relationships after calculation of Pearson’s coefficients. Additional comparisons were made using Student’s t-test for paired samples. Bland–Altman plots were used to provide graphical representation of the agreement between equations and the nuclear measurement.20
Logistic regression models were created to assess the value of the rGFR study and the two closest correlating GFR equations for predicting post-transplant outcomes, including mortality at 1, 3 and 5 years; AKI; CKD; and dialysis requirement before discharge. Receiver operating characteristic (ROC) analyses were performed to assess the relative efficiencies of GFR measures for predicting these same outcomes.
Results
Patients’ characteristics
Two hundred seventy-four patients underwent the radionuclide GFR assay and were included in the study. The mean age for the group was 64.6 ± 7.9 years and the mean lung allocation score at the time of transplant was 48.8 ± 16.1 years. Female patients made up 26.4% (n = 73) of the study population (Table 1).
Table 1.
Patients’ Characteristics
| Characteristic | N (%) or mean (SD) |
|---|---|
| Number of patients | 274 (100)a |
| Age of recipient (years) | 64.6 (7.9)b |
| Female gender | 73 (26.4)a |
| Caucasian ethnicity | 251 (91.6)a |
| Body mass index | 24.9 (2.8)b |
| Radionuclide GFR (ml/min per 1.73 m2) | 84.6 (18.1)b |
| Diagnosis | |
| Obstructive disease | 80 (29.2)a |
| Restrictive disease | 176 (64.2)a |
| Cystic fibrosis/bronchiectasis | 8 (2.9)a |
| Primary pulmonary hypertension | 5 (1.8)a |
| Other | 5 (1.8)a |
| Bilateral transplant | 181 (66.1)a |
| Diabetes mellitus | 43 (15.7)a |
| Congestive heart failure | 27 (9.9)a |
| Age of donor (years) | 36.0 (15.1)b |
| Ischemic time (hours) (n = 417) | 6.9 (2.0)b |
| Lung allocation score (n = 506) | 48.8 (16.1)b |
N(5%).
Mean (SD).
Correlation between GFR equations and radionuclide GFR assay
For patients with available rGFR data, the mean radionuclide GFR was 84.6 (18.1) ml/min per 1.73 m2. Mean calculated GFR was 88.2 (26.2) ml/min per 1.73 m2 for the MDRD, 85.4 (16.7) ml/min per 1.73 m2 for the CKDEPI, 99.2 (26.9) ml/min per 1.73 m2 for the Wright equation and 89.4 (26.2) ml/min per 1.73 m2 for the CG equation.
Overall, the CKDEPI provided the most consistent results, with low values for bias (mean standard error = 0.78), relative standard error (mean RSE = 0.03), mean percentage error (MPE = 3.12%), mean absolute percentage error (MAPE = 15.02%) and precision (interquartile range [IQR] = 19.55) (Table 2). Greater deviation from rGFR was observed for all other equations.
Table 2.
Statistical Analysis of Differences Between Direct Measured (Radionuclide) GFR and GFR Equations
| Equation | Mean (ml/min) | SD (ml/min) | MPE (%) | MAPE (%) | MSE (ml/min) | MRSE (ml/min) |
|---|---|---|---|---|---|---|
| Radionuclide | 84.6 | 18.1 | NA | NA | NA | NA |
| MDRD | 88.2 | 26.2 | 5.3 | 18.3 | 3.6 | 0.05 |
| CKDEPI | 85.4 | 16.7 | 3.1 | 15.0 | 0.8 | 0.03 |
| Wright | 99.2 | 26.9 | 19.1 | 23.9 | 14.6 | 0.19 |
| CG | 89.4 | 26.2 | 7.3 | 20.5 | 4.8 | 0.07 |
CKDEPI, Chronic Kidney Disease Epidemiology Collaboration; CG, Cockroft–Gault; MDRD, Modified Diet in Renal Disease; MAPE, mean absolute percentage error; MPE, mean percentage error; MRSE, mean relative standard error; MSE, mean standard error; SD, standard deviation.
Pearson’s correlation between rGFR and calculated GFR was significant for all equations (p < 0.001). When comparing data using Student’s t-test for paired samples, rGFR values were significantly different from those obtained by all equations except CKDEPI (p = 0.45). This is further demonstrated when mean values and 95% confidence intervals are plotted (Figure 1).
Figure 1.
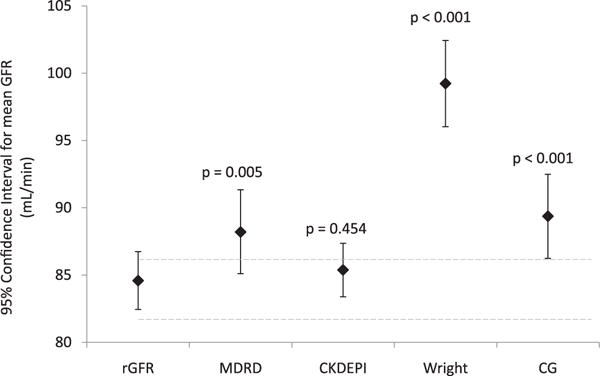
Mean values and 95% confidence intervals for directly measured (radionuclide) GFR and for GFR equations.
Graphical representation of results using the Bland–Altman method demonstrates a trend of worsening correlation with increasing GFR values for the MDRD, Wright and CG equations. Conversely, the CKDEPI plot shows a consistently close correlation through the entire range. Bland–Altman plots are presented in the Supplementary material (available online at www.jhltonline.org).
Logistic regression and ROC models assessing GFR as a predictor of outcomes
Bivariate and multivariable logistic regression models were created to assess the utility of the rGFR assay as well as the CKDEPI and MDRD equations for predicting outcomes. On bivariate analysis, none of the GFR measures, including the rGFR assay, was a significant predictor of dialysis requirement before discharge or of mortality at 1, 3 or 5 years (all p > 0.05; Table 3). On the other hand, all GFR measures were significant predictors of AKI and CKD (p < 0.05). These relationships remained significant on multivariable analysis, controlling for donor and recipient factors (Table 3).
Table 3.
Logistic Regression Models Evaluating GFR Measures as Predictors of Outcomes
| Assay/equation | Bivariate analysis p-value (OR; 95% CI) | Multivariable analysisa p-value (OR; 95% CI) |
|---|---|---|
| Acute kidney injury | ||
| Radionuclide GFR Assay | 0.018 (0.974; 0.954–0.996)b | 0.096 (NA) |
| MDRD | 0.002 (0.973; 0.956–0.990)b | 0.031 (0.980; 0.963–0.998)b |
| CKDEPI | 0.001 (0.968; 0.949–0.987)b | 0.018 (0.974; 0.954–0.995)b |
| Stage 3 chronic kidney disease at 6 months | ||
| Radionuclide GFR Assay | <0.001 (0.969; 0.951–0.987)b | 0.002 (0.968; 0.949–0.988)b |
| MDRD | 0.007 (0.985; 0.974–0.996)b | 0.033 (0.988; 0.976–0.999)b |
| CKDEPI | <0.001 (0.960; 0.940–0.981)b | <0.001 (0.961; 0.939–0.983)b |
| Dialysis requirement prior to discharge | ||
| Radionuclide GFR assay | 0.935 (0.999; 0.968–1.030) | |
| MDRD | 0.354 (0.989; 0.965–1.013) | NAc |
| CKDEPI | 0.510 (0.990; 0.961–1.020) | |
| 1-year mortality | ||
| Radionuclide GFR assay | 0.317 (0.989; 0.968–1.011) | |
| MDRD | 0.155 (0.988; 0.973–1.004) | NAc |
| CKDEPI | 0.200 (0.987; 0.967–1.007) | |
| 3-year mortality | ||
| Radionuclide GFR assay | 0.457 (0.992; 0.971–1.014) | |
| MDRD | 0.462 (0.994; 0.978–1.010) | NAc |
| CKDEPI | 0.391 (0.991; 0.970–1.012) | |
| 5-year mortality | ||
| Radionuclide GFR assay | 0.705 (1.011; 0.956–1.068) | |
| MDRD | 0.398 (0.983; 0.946–1.022) | NAc |
| CKDEPI | 0.295 (0.972; 0.920–1.025) | |
CI, confidence interval; CKDEPI, chronic kidney disease epidemiology collaboration; GFR, glomerular filtration rate; OR, odds ratio; MDRD, modified diet in renal disease.
Multivariate analysis controlling for donor and recipient factors including age, gender, diagnosis, year of transplant, ischemic time and lung allocation score. The final models were selected using a backwards elimination process.
Statistically significant at the 95% level.
Regression models not created given non-significance on bivariate analysis.
ROC curves were created to assess the value of GFR measures for identifying patients at greatest risk for AKI, CKD and 1-year mortality (Figure 2). None of the GFR measures were useful for predicting 1-year mortality with 95% confidence intervals for areas under the curve (AUCs) that included 0.5 (AUC = 0.5 is observed for tests that give each individual a 50% chance of having the outcome). AUCs for predicting 1-year mortality were 0.550 (0.428 to 0.671) for rGFR, 0.584 (0.469 to 0.700) for the CKDEPI and 0.589 (0.478 to 0.700) for the MDRD.
Figure 2.
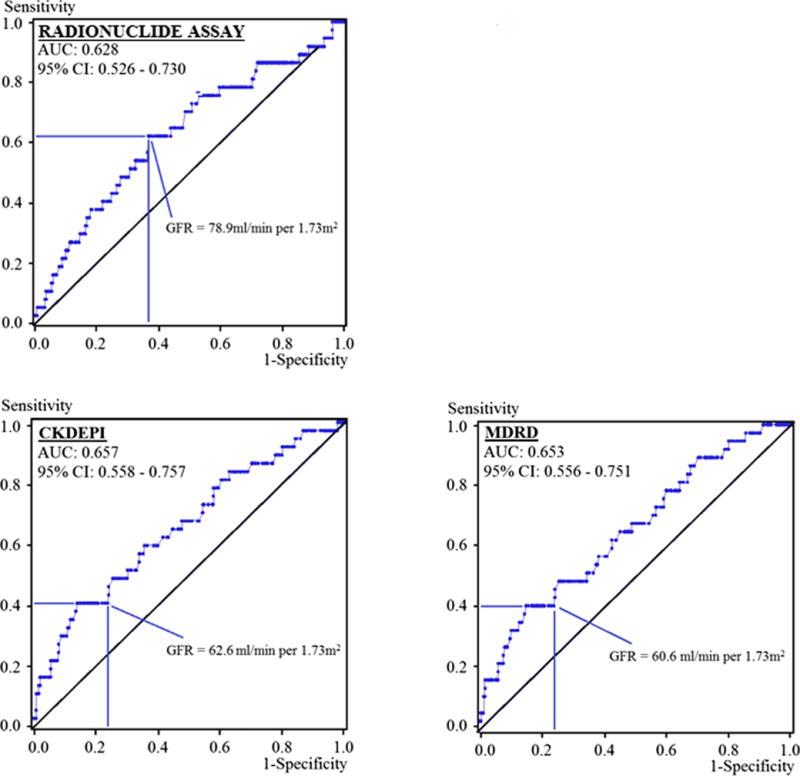
Receiver operating characteristic (ROC) curves displaying the usefulness of GFR measures for predicting post-operative acute kidney injury.
GFR measures performed more reliably for predicting AKI (Figure 2). AUCs were 0.628 (0.526 to 0.730) for rGFR, 0.657 (0.558 to 0.757) for the CKDEPI and 0.653 (0.556 to 0.751) for the MDRD (Figure 2). Chi-square analysis comparing these AUCs demonstrated no significant advantage for any particular GFR measure (p > 0.05). As with the AKI analysis, ROC investigations into the value of GFR for predicting CKD generated AUCs that were statistically indistinguishable when comparing the rGFR assay to both MDRD and CKDEPI equations (p > 0.05).
Logistic regression models to verify results in the validation cohort
Comparative patient characteristics and results from statistical modeling in the validation cohort (N =243) are presented in the Supplementary material available online at www.jhltonline.org. Findings on logistic regression analysis in the validation cohort were similar to those observed with models created using the primary study cohort.
CKDEPI GFR as a screening test before direct GFR measurement
Using CKDEPI GFR ≤60 ml/min per 1.73 m2 as a screening test before performing the rGFR study, 235 patients (86%) would not have needed to undergo evaluation with the rGFR assay. Of these patients, 224 (82% of the study population) ultimately had rGFR >60 ml/min per 1.73 m2 and would thus have been appropriately spared from undergoing the more costly radionuclide test. For the remaining 11 patients (4% of the study population), the rGFR assay would have been omitted inappropriately.
Increasing the cut-off for screening to CKDEPI GFR ≤70 ml/min per 1.73 m2, 201 patients (73%) would not have needed to undergo evaluation with the rGFR assay. Of these patients, 195 (71% of the study population) ultimately had rGFR >60 ml/min per 1.73 m2 and would thus have been appropriately spared from undergoing the radionuclide test. For the remaining 6 patients (2% of the study population), the rGFR assay would have been omitted inappropriately.
A consideration of the results of using various CKDEPI cut-offs (ranging from 50 ml/min per 1.73 m2 to 100 ml/min per 1.73 m2) is presented in the Supplementary Material available online (see Table S4).
Discussion
This study represents the first systematic comparison of several renal function equations used in the pre-transplant period, based on correlation with direct GFR testing and prediction of post-transplant outcomes. Although the CKDEPI best correlated with the radionuclide GFR assay, neither predicted post-transplant survival at 1, 3 or 5 years. However, both tests are equivalent predictors of renal function after lung transplantation, including AKI and CKD at 6 months. The observed relevance for predicting post-transplant renal function provides strong support for the CKDEPI equation as an adequate measure of GFR before lung transplantation.
The study findings are in accordance with much of the literature produced in other patient populations since the CKDEPI equation was created in 2009. In the original report, Levey et al demonstrated improved precision and accuracy, as well as decreased bias of the CKDEPI equation when compared with the MDRD. Equations were compared with a direct GFR assay using iothoalamate, and results were validated in a cohort of 3,900 patients.11 Subsequent studies provided further evidence in favor of the CKDEPI equation. Lujan et al compared the CKDEPI with the MDRD in 85 adults undergoing evaluation prior to kidney donation.10 They showed lower bias and higher accuracy with the CKDEPI equation. A similar study by Kilbride et al compared the CKDEPI and MDRD in 398 elderly patients.21 They demonstrated better CKDEPI performance. However, none of these studies included lung recipients or patients with end-stage lung disease, and none correlated GFR with outcomes after lung transplantation.
In aggregate, the literature suggests that the CKDEPI is the best serum creatinine–based equation for estimating GFR across a broad range of populations, but with possibly less value in certain unique groups. For example, in their study of 105 diabetic patients, Silveiro et al found that both the CKDEPI and MDRD underestimated GFR in diabetic populations, especially in those patients with high GFR values.22 Tent et al studied 65 non-diabetic patients with a history of CKD.23 Although the CKDEPI performed better than the MDRD for long-term GFR monitoring, neither equation was useful for identifying progressive renal deterioration in patients who already had CKD.
Our study has demonstrated that, in a cohort of patients with end-stage lung disease, the CKDEPI equation correlates best with the radionuclide GFR test. In addition, we found that creatinine-based equations can assist in stratifying patients at risk of developing AKI or CKD after lung transplantation. Based on these findings, transplant centers that use the radionuclide assay can consider replacing direct measurement with GFR estimation using the CKDEPI equation. It is worth noting that study results may not be directly applicable to centers that use alternative assays for direct GFR measurement, such as creatinine or inulin clearance. Similar studies in patients undergoing each of these direct GFR measurements will be necessary before results can be extended to these centers. In addition to the previously described benefits related to using creatinine-based equations, performing similar studies at these centers will improve nationwide standardization of candidate evaluation practices, ultimately allowing for better intercenter comparisons and increased collaboration.
An alternative to completely replacing radionuclide and other direct GFR measures involves the use of the CKDEPI as a screening assay to determine which patients need to undergo direct GFR testing. As noted in the Results, with a CKDEPI screening threshold of 60 ml/min per 1.73 m2, only 15% of patients would need to undergo direct testing with only 4% of the entire sample missing a direct test that would have been appropriate. If this threshold was increased to 70 ml/min per 1.73 m2, 29% of patients would need to undergo direct testing with only 2% of the entire sample missing a direct test that would have been appropriate. The benefit of CKDEPI as a screening test is clear in both situations, but the latter (with the cut-off at 70 ml/min per 1.73 m2) may be more appropriate given the relative importance of identifying candidates with poor renal function.
The typical U.S. hospital charge for the radionuclide GFR assay is about $1,000 and the test takes >5 hours to complete. In contrast, GFR by the CKDEPI equation can be calculated based on serum creatinine data obtainable from a single blood draw. The benefits in terms of convenience are obvious. On the financial side, the average charge associated with obtaining serum creatinine data is significantly lower at about $60. In addition, this result is available as part of serum chemistry panels, routinely obtained in transplant candidate evaluation. Finally, serial CKDEPI calculations can be performed readily and inexpensively during the evaluation and waitlist period. This is important, as it is well described that GFR values can vary with hydration status, medication use and other changing recipient conditions. The outlined potential for economic gain is worthy of serious consideration, particularly given the present-day emphasis on high-quality, cost-conscious care within the U.S. healthcare system.
Our study has a few limitations. Most important is the fact that we considered only patients who received transplants, excluding transplant candidates who were denied organs. Renal dysfunction was a contributory factor in the exclusion of some of these patients from transplantation. Thus, study findings must be applied cautiously to the transplant evaluation process. An additional limitation is the fact that the radio-nuclide study used at our center is one of several “direct” tests available for GFR measurement. Results from other assays, such as inulin clearance and 24-hour creatinine clearance, were not available for patients in our study. Variation between the radionuclide assay and those used at other centers could diminish the value of comparisons made across studies. Finally, this study was a single-center, retrospective analysis, with all the limitations in generalizability and ascertainment of causality associated with such an approach.
In conclusion, results from our study support the conclusion that CKDEPI correlates most closely with direct pre-operative GFR measurement in lung recipients. Our study also identified a correlation between pre-operative CKDEPI values and outcomes such as post-transplant AKI or the development of CKD. Thus, transplant centers could consider replacing direct radionuclide GFR measurements with more convenient GFR estimation using the CKDEPI equation. Alternatively, CKDEPI GFR may be used as a screening assay to determine which patients need further testing with direct GFR measurements. Ultimately, widespread use of a uniform GFR estimate will foster standardization of candidate evaluation across centers, allowing for better inter-center comparisons of transplantation practices.
Supplementary Material
Acknowledgments
This project was funded by the Bollinger Research Grant Committee within the Department of Surgery at Duke University Medical Center. M.G.H. is supported by the Cardiothoracic Surgery Trials Network, funded by the National Institutes of Health.
Footnotes
Disclosure statement
The authors have no conflicts of interest to disclose.
Supplementary materials
Supplementary materials associated with this article can be found in the online version at www.jhltonline.org.
References
- 1.Sanchez EQ, Melton LB, Chinnakotla S, et al. Predicting renal failure after liver transplantation from measured glomerular filtration rate: review of up to 15 years of follow-up. Transplantation. 2010;89:232–5. doi: 10.1097/TP.0b013e3181c42ff9. [DOI] [PubMed] [Google Scholar]
- 2.Brown JR, Cochran RP, Leavitt BJ, et al. Multivariable prediction of renal insufficiency developing after cardiac surgery. Circulation. 2007;116:139–43. doi: 10.1161/CIRCULATIONAHA.106.677070. [DOI] [PubMed] [Google Scholar]
- 3.Cooper WA, O’Brien SM, Thourani VH, et al. Impact of renal dysfunction on outcomes of coronary artery bypass surgery: results from the Society of Thoracic Surgeons National Adult Cardiac Database. Circulation. 2006;113:1063–70. doi: 10.1161/CIRCULATIONAHA.105.580084. [DOI] [PubMed] [Google Scholar]
- 4.George TJ, Arnaoutakis GJ, Beaty CA, et al. Acute kidney injury increases mortality after lung transplantation. Ann Thorac Surg. 2012;94:185–92. doi: 10.1016/j.athoracsur.2011.11.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Davis SQ, Garrity ER. Organ allocation in lung transplant. Chest. 2007;132:1646–51. doi: 10.1378/chest.07-0011. [DOI] [PubMed] [Google Scholar]
- 6.Assadi M, Eftekhari M, Hozhabrosadati M, et al. Comparison of methods for determination of glomerular filtration rate: low and high-dose Tc-99m-DTPA renography, predicted creatinine clearance method, and plasma sample method. Int Urol Nephrol. 2008;40:1059–65. doi: 10.1007/s11255-008-9446-4. [DOI] [PubMed] [Google Scholar]
- 7.Sundaraiya S, Mendichovszky I, Biassoni L, et al. Tc-99m DTPA renography in children following renal transplantation: its value in the evaluation of rejection. Pediatr Transplant. 2007;11:771–6. doi: 10.1111/j.1399-3046.2007.00747.x. [DOI] [PubMed] [Google Scholar]
- 8.Al-Naamani N, Maarouf OH, Wilt JS, et al. The modification of diet in renal disease (MDRD) and the prediction of kidney outcomes after lung transplantation. J Heart Lung Transplant. 2008;27:1191–7. doi: 10.1016/j.healun.2008.07.023. [DOI] [PubMed] [Google Scholar]
- 9.Lin J. A comparison of prediction equations for estimating glomerular filtration rate in adults without kidney disease. J Am Soc Nephrol. 2003;14:2573–80. doi: 10.1097/01.asn.0000088721.98173.4b. [DOI] [PubMed] [Google Scholar]
- 10.Lujan PR, Chiurchiu C, Douthat W, et al. CKD-EPI instead of MDRD for candidates to kidney donation. Transplantation. 2012;94:637–41. doi: 10.1097/TP.0b013e3182603260. [DOI] [PubMed] [Google Scholar]
- 11.Levey AS, Stevens LA, Schmid CH, et al. A new equation to estimate glomerular filtration rate. Ann Intern Med. 2009;150:604–12. doi: 10.7326/0003-4819-150-9-200905050-00006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Hsu C. CKD-EPI eGFR categories were better than MDRD categories for predicting mortality in a range of populations. Ann Intern Med. 2012;157:JC5–12. doi: 10.7326/0003-4819-157-10-201211200-02012. [DOI] [PubMed] [Google Scholar]
- 13.Matsushita K, Mahmoodi BK, Woodward M, et al. Comparison of risk prediction using the CKD-EPI equation and the MDRD study equation for estimated glomerular filtration rate. JAMA. 2012;307:1941–51. doi: 10.1001/jama.2012.3954. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Trobec K, Knez L, Meško Brguljan P, et al. Estimation of renal function in lung cancer patients. Lung Cancer. 2012;76:397–402. doi: 10.1016/j.lungcan.2011.11.016. [DOI] [PubMed] [Google Scholar]
- 15.Wright JG, Boddy AV, Highley M, et al. Estimation of glomerular filtration rate in cancer patients. Br J Cancer. 2001;84:452–9. doi: 10.1054/bjoc.2000.1643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Al-Naamani N, Maarouf OH, Ahya VN, et al. Assessment of kidney function in lung transplant candidates. J Heart Lung Transplant. 2008;27:635–41. doi: 10.1016/j.healun.2008.02.008. [DOI] [PubMed] [Google Scholar]
- 17.Cantarovich M, Yoshida EM, Peltekian KM, et al. Poor prediction of the glomerular filtration rate using current formulas in de novo liver transplant patients. Transplantation. 2006;82:433–6. doi: 10.1097/01.tp.0000228888.31242.2b. [DOI] [PubMed] [Google Scholar]
- 18.Chen L-I, Kuo M-C, Hwang S-J, et al. Comparisons of technetium-99m diethylenetriaminepentaacetic acid plasma clearance and renal dynamic imaging with inulin clearance. Am J Kidney Dis. 2011;58:1043–5. doi: 10.1053/j.ajkd.2011.08.026. [DOI] [PubMed] [Google Scholar]
- 19.Horvath MM, Winfield S, Evans S, et al. The DEDUCE Guided Query tool: providing simplified access to clinical data for research and quality improvement. J Biomed Inform. 2011;44:266–76. doi: 10.1016/j.jbi.2010.11.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Bland JM, Altman DG. Statistical methods for assessing agreement between two methods of clinical measurement. Lancet. 1986;1:307–10. [PubMed] [Google Scholar]
- 21.Kilbride HS, Stevens PE, Eaglestone G, et al. Accuracy of the MDRD (Modification of Diet in Renal Disease) study and CKD-EPI (CKD Epidemiology Collaboration) equations for estimation of GFR in the elderly. Am J Kidney Dis. 2013;61:57–66. doi: 10.1053/j.ajkd.2012.06.016. [DOI] [PubMed] [Google Scholar]
- 22.Silveiro SP, Araújo GN, Ferreira MN, et al. Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) equation pronouncedly underestimates glomerular filtration rate in type 2 diabetes. Diabetes Care. 2011;34:2353–5. doi: 10.2337/dc11-1282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Tent H, Waanders F, Krikken JA, et al. Performance of MDRD study and CKD-EPI equations for long-term follow-up of nondiabetic patients with chronic kidney disease. Nephrol Dial Transplant. 2012;27(suppl 3):89–95. doi: 10.1093/ndt/gfr235. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


