SUMMARY
Vertigo is generally due to a benign disorder, but it is the most common symptom associated with misdiagnosis of stroke. In this pilot study, we preliminarily assessed the diagnostic performance of a structured bedside algorithm to differentiate central from non-central acute vertigo (AV). Adult patients presenting to a single Emergency Department with vertigo were evaluated with STANDING (SponTAneous Nystagmus, Direction, head Impulse test, standiNG) by one of five trained emergency physicians or evaluated ordinarily by the rest of the medical staff (control group). The gold standard was a complete audiologic evaluation by a clinicians who are experts in assessing dizzy patients and neuroimaging. Reliability, sensibility and specificity of STANDING were calculated. Moreover, to evaluate the potential clinical impact of STANDING, neuroimaging and hospitalisation rates were compared with control group. A total of 292 patients were included, and 48 (16.4%) had a diagnosis of central AV. Ninety-eight (33.4%) patients were evaluated with STANDING. The test had good interobserver agreement (k = 0.76), with very high sensitivity (100%, 95%CI 72.3-100%) and specificity (94.3%, 95%CI 90.7-94.3%). Furthermore, hospitalisation and neuroimaging test rates were lower in the STANDING than in the control group (27.6% vs. 50.5% and 31.6% vs. 71.1%, respectively). In conclusion, STANDING seems to be a promising simple structured bedside algorithm that in this preliminary study identified central AV with a very high sensitivity, and was associated with significant reduction of neuroimaging and hospitalisation rates.
KEY WORDS: STANDING, Benign paroxysmal positional vertigo, Vestibular neuronitis, Bedside algorithm
RIASSUNTO
La vertigine è generalmente dovuta ad una patologia benigna, ma rappresenta il sintomo più comunemente associato ad una mancata diagnosi di stroke. In questo studio pilota, abbiamo valutato in modo preliminare la validità diagnostica di un algoritmo bedside strutturato per differenziare le vertigini acute (VA) di origine centrale da quelle di origine non centrale. I pazienti adulti che si presentavano presso il nostro Dipartimento di Emergenza con vertigini venivano valutati con lo STANDING (SponTAneous Nystagmus Direction, head Impulse test, standiNG) da uno dei cinque medici del Pronto Soccorso adeguatamente istruiti, o in maniera tradizionale dal resto dello staff medico (gruppo di controllo). Il gold standard era rappresentato da una valutazione audiologica completa effettuata da un audiologo esperto e associata agli esami per immagini. Sono state calcolate la ripetibilità, la sensibilità e la specificità dello STANDING. Inoltre, per valutare in modo preliminare il potenziale impatto clinico dello STANDING, sono state confrontate le percentuali di richiesta di esami per immagini e di ospedalizzazioni con quelle del gruppo di controllo. Sono stati reclutati 292 pazienti, per 48 dei quali (16,4%) era stata diagnosticata una vertigine di origine centrale. Novantotto pazienti (33,4%) sono stati valutati con lo STANDING. L'intero algoritmo ha mostrato una buona concordanza tra gli esaminatori (K = 0,76), con una sensibilità (100%, 95%IC 72,3-100%) e una specificità (94,3%, 95%IC 90,7- 94,3%) molto alte. Inoltre, le percentuali di ospedalizzazione e di richiesta di esami per immagini sono state più basse nel gruppo valutato con lo STANDING rispetto al gruppo di controllo (rispettivamente 27,6% vs. 50,5% e 31,6% vs. 71,1%). In conclusione, lo STANDING sembra un algoritmo semplice e promettente, identificando nella nostra popolazione non selezionata le VA di origine centrale con un'alta sensibilità e con una riduzione significativa del numero di esami per immagini e ospedalizzazioni.
Introduction
Vertigo is the illusion of the true rotational movement of self or surroundings and is a frequent complaint of patients presenting in the emergency department (ED) 1. It is often associated with the presence of nystagmus and is most likely due to vestibular system dysfunction. Imbalance or disequilibrium refers to a sense of unsteadiness often indistinguishable by patients, and often by physicians, from true vertigo 2 3. Many other symptoms of altered orientation in space are referred to as dizziness; the latter often represents several overlapping sensations and can be caused by many pathophysiological mechanisms and a variety of disorders, not necessarily vestibular in nature, such as presyncope (hyperventilation, orthostatic hypotension, vasovagal attacks, decreased cardiac output), anxiety disorders (panic syndrome, agoraphobia), hypoglycaemia and drug intoxication (alcohol, barbiturates, benzodiazepines): these conditions are defined as "pseudo-vertigo".
Vertigo is caused in 24-43% of cases by a benign peripheral disorder 4 such as benign paroxysmal positional vertigo (BPPV) or vestibular neuronitis (VN). However, although the most common causes of dizziness and vertigo are benign, differential diagnosis must include potentially life-threatening central disease 5; indeed, vertigo can be the manifestation of central neurological disease such as cerebellar or brainstem stroke 6. BPPV is characterised by recurrent short lasting vertigo triggered by head movements and can be revealed by diagnostic manoeuvres such as the Dix-Hallpike and Pagnini-McClure positionings 7-9. The clinical features of VN are the subacute onset of vertigo associated with spontaneous nystagmus lasting days to weeks. Vestibular neuronitis is generally self-limiting and commonly attributed to viral aetiology. Similar clinical symptoms commonly occur in cerebellar infarction, sometimes without any accompanying neurological symptoms or signs except for acute vertigo (AV) and gait ataxia 3 10 11. Several clinical tests to differentiate central from non-central AV have been investigated, but none reaches adequate sensitivity and specificity to be used as stand-alone test 11. For this reason, clinical evaluation of patients with vertigo is often difficult and rarely conclusive, usually leading to an overuse of consultants and neuroimaging tests. Moreover, computed tomography (CT) brain scan, the test most commonly performed in the ED on a patient with dizziness 12, can easily miss central disease because of its low sensitivity, particularly in the posterior fossa 13 14. Although magnetic resonance imaging (MRI) of the brain is more sensitive, it is not always readily available and is not a practical screening test in the emergency setting. All these pitfalls and technical obstacles contribute to the fact that in practice dizziness (a term used to encompass vertigo, pseudovertigo, imbalance or disequilibrium) is the symptom most commonly associated with a missed diagnosis of stroke 14 15. We believe that the development of simple, reliable and accurate predictors is a crucial step to optimise the use of neuroimaging studies, improve diagnostic accuracy, enhance patient flow through the ED and reduce unnecessary hospitalisation.
The aim of this pilot study was to preliminarily assess the reliability and diagnostic accuracy of a simple structured clinical algorithm (STANDING: SponTAneous Nystagmus, Direction, head Impulse test, standiNG) that we developed to differentiate central from non-central AV in the emergency setting, and to evaluate in an explorative fashion if its use might be associated with a reduction of the neuroimaging burden and hospitalisation.
Materials and methods
Clinical setting and selection of participants
Consecutive adult patients presenting to the ED with AV with no associated focal neurological deficit (isolated vertigo) were prospectively evaluated in a single level III ED (mean attendance 60,000 people/year), between May 2011 and January 2012. All patients underwent clinical anamnesis and complete neurological examination. Exclusion criteria were the presence of pseudo-vertigo, severe cognitive impairment, or severe symptoms of dizziness that prevented the patient's cooperation, as well as refusal to participate the study.
Management strategies
Patients with isolated vertigo underwent ordinary clinical examination (control group) or clinical examination together with simple structured clinical algorithm (STANDING) by one of 5 emergency physicians, who had already completed a workshop managed by an expert clinician in assessing dizzy patients, consisting of 5 hours didactic and practical sessions, comprehensive of 15 STANDING proctored examinations.
After initial clinical assessment, with or without STANDING, the referring ED physician determined if the vertigo was of central or non-central origin and as necessary ordered further tests (in suspicious of a central AV, usually a head CT scan). Afterwards, within 24 hours, all patients underwent complete examination by a clinician expert in assessing dizzy patients. If central origin was suspected or uncertain (disagreement between expert physician and the attending emergency physician), the diagnosis was corroborated by brain MRI (see below) and in-hospital observation. Central vestibulopathy was diagnosed by the presence of a lesion in the posterior fossa in brain imaging or by the presence of a possible transient insult in the same region that required active treatment (vertebro-basilar transient ischaemic attack, TIA) 16. When central vestibulopathy was diagnosed, patients were admitted and treated accordingly. Otherwise, when both the attending emergency physician and the expert physician agreed on the non-central origin of vertigo, neuroimaging tests and in-hospital observation were not mandatory. The hospital's Institutional Review Board approved the study.
The STANDING test
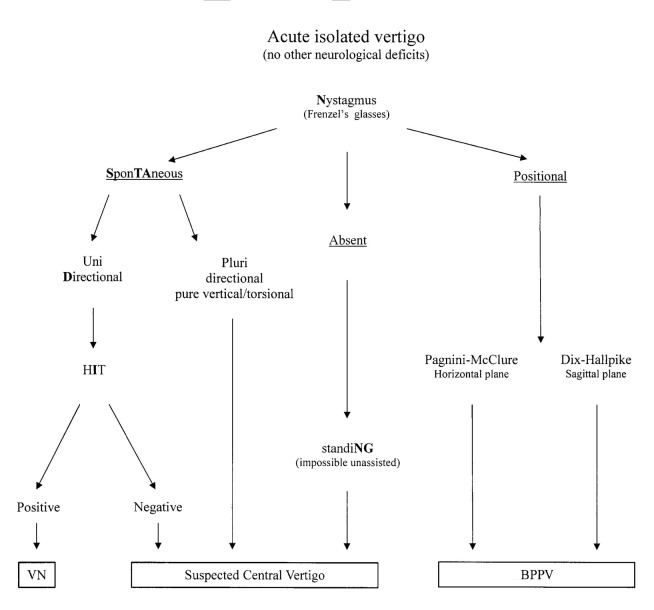
The STANDING test is a structured diagnostic algorithm based on previously described diagnostic signs and bedside manoeuvres that we logically assembled in four sequential steps (Fig. 1).
Fig. 1.
Diagram of the STANDING approach. VN = Vestibular neuronitis; HIT = head impluse test; BPPV = benign paroxysmal positional vertigo.
First, the presence of nystagmus was assessed with Frenzel's glasses in supine position after at least 5 minutes of rest. When no spontaneous nystagmus was present in the main gaze positions, the presence of a positional nystagmus was assessed by the Pagnini-McClure manoeuvres first and then by the Dix-Hallpike positionings 7. The presence of a positional nystagmus of the paroxysmal type was considered typical of BPPV.
Instead, when spontaneous nystagmus was already present in supine position and was persistent, the direction was examined: multidirectional nystagmus, such as bidirectional gaze-evoked nystagmus (i.e. right beating nystagmus present with gaze toward the right and left beating nystagmus present with gaze toward the left side), and a pure vertical (up or down beating) or torsional nystagmus were considered signs of central vertigo.
When the nystagmus was unidirectional (i.e. nystagmus beating on the same side independent of gaze direction and head position), we performed the head impulse test (HIT) 17. When an acute lesion occurs in one labyrinth, the input from the opposite side is unopposed and as a result, when the head is rapidly moved toward the affected side, the eyes will be initially pushed toward that side and, immediately after, a corrective eye movement (corrective "saccade") back to the point of reference is seen. When the corrective "saccade" is present the HIT is considered positive and indicates non-central AV, whereas a negative HIT indicates central vertigo 18.
Patients showing neither spontaneous nor positional nystagmus were invited to stand and gait was evaluated. When there was an inability to maintain an upright stance without assistance, they were suspected to have central disease (Fig. 1).
Neuroimaging
We performed a CT brain scan using a Somatom Definition AS128 instrument (Siemens, Erlangen, Germany) on every patient suspected to harbour central vertigo. When the CT was negative but central vertigo was still suspected, patients underwent MRI within 24-72 hours after initial evaluation. Patients underwent brain MRI stroke-protocol with a 1.5 Tesla Magnetom Vision/Plus (Siemens, Enlargen, Germany) instrument including; 1) multi-planar T1; 2) axial T2 or fluid attenuated inversion recovery (FLAIR); and 3) axial DWI sequences.
Statistical analysis
We express continuous variables as means ± standard deviation (SD), and dichotomous variables as percentages. The inter-observer reliability of two emergency physician was calculated by Cohen's k for each step of STANDING in a subgroup of patients (n = 30). We also tested the inter-observer agreement between the STANDING test and audiological evaluation. We assessed the diagnostic accuracy for central vestibulopathy of the STANDING test, calculating sensitivity, specificity, positive and negative predictive values with 95% confidence intervals (CI). To assess the potential clinical impact of the STANDING test, we compared the baseline characteristics and the neuroimaging test and hospitalisation rates of patients examined with the STANDING test with those of a control group, using a student's t-test for continuous variables and Fisher's exact test for dichotomous variables.
Calculations were performed using the SPSS statistical package (version 17.0, SPSS, Chicago, Illinois, USA).
Results
Patient characteristics
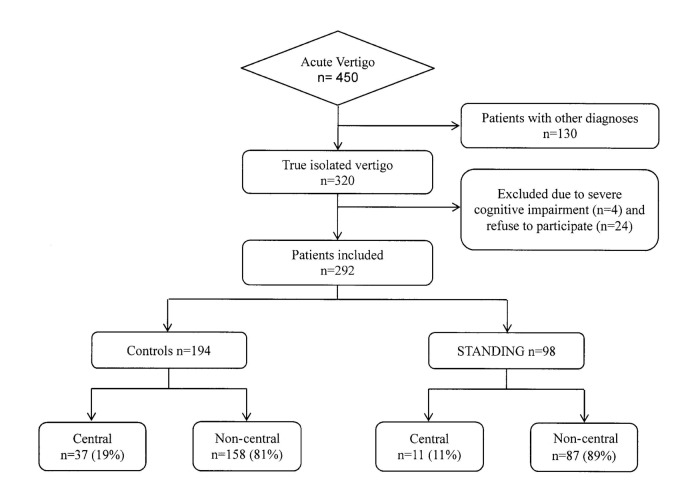
A total of 450 patients complaining of vertigo (Fig. 2) were evaluated in our ED during 8 months (0.8% of the overall presentations): among these, 130 (28.8%) were actually pseudo-vertigo, 4 (0.8%) patients presented a severe cognitive impairment and 24 (5.3%) refused to participate in the study and a definite diagnosis could not be made. The remaining 292 patients were included. The study population had a mean age of 58.2 years and 61% were females (Table I); at least one cardiovascular risk factor was present in 44.8% of patients.
Fig. 2.
Study flow diagram.
Table I.
Baseline characteristics, neuroimaging tests and hospitalisation rates of ED patients presenting with acute vertigo.
| All patients n = 292 |
STANDING n = 98 |
Controls n = 194 |
Difference* % (95% CI) |
|
|---|---|---|---|---|
| Females (%) | 178 (61%) | 56 (57.1%) | 121 (62.4%) | -5.2 (-17.9, +7.2) |
| Age (Mean ± Ds) | 58.2 ± 16.3 | 60 ± 16.3 | 57.3 ± 11.3 | +2.7 ± 22.1 |
| CV risk factors (%) | 131 (44.8%) | 45 (45.9%) | 86 (44.3%) | +1.6 (-11.1, +14.4) |
| Central Vertigo (%) | 48 (16.4.%) | 11 (11.2%) | 37 (19.1%) | -8 (-15.3, +1.9) |
| CT brain scan (%) | 169 (57.9%) | 31 (31.6%) | 138 (71.1) | -39.5 (-50.7, -27) |
| Brain MRI (%) | 19 (6.5%) | 10 (10.2%) | 9 (4.6%) | 5.6 (-1, +11.9) |
| Hospitalisation (%) | 125 (42.8%) | 27 (27.6%) | 98 (50.5%) | -23 (-34.1, -10.4) |
CT: computed tomography; MRI: magnetic resonance imaging; CV risk factor: at least one of the following cardiovascular risk factors: diabetes, blood hypertension, smoke, dyslipidaemia; hospitalisation included both admission to general or neurological wards and in the observation unit; * absolute differences between STANDING and control groups.
Forty-eight patients (16.4%) of 292 had a final diagnosis of central disease: among these, 21 (43.8%) had a vertebro- basilar TIA and 14 had a stroke (29.2%), (Table II). A total of 244 patients (83.6%) had non-central AV, most often BPPV or VN (Table II).
Table II.
Specific diagnosis in patients evaluated with STANDING or routine tests (controls).
| STANDING n = 98 (%) |
Controls n = 194 (%) |
|
|---|---|---|
| Central vertigo | 11(11.2) | 37 (19) |
| Ischaemic stroke | 3 (3.1) | 8 (4.1) |
| Haemorragic stroke | 1 (1.0) | 2 (1.0) |
| Cerebral tumour | 2 (2.0) | 3 (1.5) |
| Vertebrobasilar TIA | 4 (4.1) | 17 (8.7) |
| Other central diseases | 1 (1.0) | 6 (3.1) |
| Non-central vertigo | 87 (88.7) | 157 (80.9) |
| BPPV | 60 (61.2) | 104 (53.6) |
| VN | 18 (18.3) | 25 (12.9) |
| Other causes | 9 (9.1) | 28 (14.4) |
TIA: transient ischaemic attack; other central disease: hydrocephalus, multiple sclerosis, epilepsy; BPPV: benign paroxysmal positional vertigo; VN: vestibular neuronitis; other causes: Menière's disease, migraineous vertigo.
CT brain scan was performed on 169 (57.9%) patients and revealed central disease in 26 (15.4%); 19 patients (6.5%) underwent head MRI that showed central disease in 7 (36.8%). A total of 125 patients (42.8%) were hospitalised: 91 (31.1%) of these patients were observed inhospital for at least 24 hours; the other 34 (11.6%) were admitted to an internal medicine or neurology ward.
STANDING reliability and accuracy
Ninety-eight (33.6%) of the 292 patients were initially evaluated by ED physicians using the STANDING test. Of these, 60 patients (61.2%) had paroxysmal positional nystagmus, while 24 (24.5%) had spontaneous nystagmus that was pluridirectional in 2 (8.3%) and unidirectional in 22 (91.7%) cases. Among these, the prevalence of right and left beating nystagmus was similar (52.4% left and 47.6% right). HIT was performed in 23 patients and was negative in 4 (17.4%) and positive in 19 (82.6%) patients. In one (4.1%) of 24 cases, HIT was not applicable due to patient intolerance.
Fourteen patients (14.3%) did not show nystagmus; 10 of these patients, when invited to stand, revealed an inability to maintain an upright stance, and were diagnosed with potential central disease.
The reliability of the STANDING test between two ED physicians was tested in 30 patients. The Cohen's kappa of the first (spontaneous vs. positional nystagmus), second (unidirectional vs. pluridirectional or pure vertical/torsional nystagmus), third (HIT) and fourth (standing) step was 0.86, 0.93, 0.73 and 0.78, respectively. The Cohen's kappa of the final result of the test (central vs. non-central AV) was 0.76.
After performing the STANDING test, central vertigo was suspected by ED physicians in 16 (16.3%) of 98 patients and was confirmed by a clinician who was expert in assessing dizzy patients in 13 patients (13.2%). The STANDING test showed high agreement (95.9%) with audiological examination corresponding to a Cohen's kappa of 0.86.
Eleven (68.7%) of the 16 patients with suspected central vertigo, according to STANDING, had a final diagnosis of central vestibulopathy, whereas no patient with negative STANDING had a final diagnosis of central disease. Test characteristics are reported in Table III.
Table III.
STANDING test characteristics.
| Central vertigo Final diagnosis | Non-central vertigo Final diagnosis | Total | |
|---|---|---|---|
| Central vertigo STANDING | 11 | 5 | 16 |
| Non-central vertigo STANDING | 0 | 82 | 82 |
| Total | 11 | 87 | 98 |
Sensitivity: 100% (95% CI: 72.3-100%); specificity: 94.3% (95% CI: 90.7-94.3%); positive predictive value: 68.8% (95% CI: 49.7-68.8%); negative predictive value: 100% (95% CI: 96.3-100%).
STANDING test vs. ordinary evaluation
When we compared the STANDING group to the control group, there were no statistically significant differences in gender, age, or prevalence of cardiovascular risk factors (Table I). Central vertigo was slightly more common in the control group than in the STANDING group, but the difference was not significant.
In the STANDING group, 31 patients (31.6%) underwent CT brain scans that were positive in 3 patients (9.6%); 10 patients (10.2%) also underwent a brain MRI that was positive in 5 (50%). Hospitalisation was requested in 27 patients (27.6%), in most cases 24-48 hours observation (18 patients, 66.7%). CT and hospitalisation rates were significantly lower in the STANDING group than in the control group (Table I).
Discussion
In this study, a structured bedside algorithm (STANDING) performed by emergency physicians showed good reliability and high accuracy for detecting central vestibulopathy in an unselected population presenting with acute vertigo. The application of STANDING was associated with lower neuroimaging and hospitalisation rates than in controls.
Vertigo is a relatively common complaint that is often diagnosed and treated in the ED. In our study, conducted in an unselected population presenting to a level III ED, we found that about 1% of overall attendances presented with vertigo and that about 70% of these patients had true vertigo. In previous studies, similar results were found 1 18, but the prevalence was higher (1-10%) when all forms of dizziness were included 19.
Although vertigo is usually ascribable to benign aetiologies such as peripheral vertigo, in previous studies up to 25% of patients had central nervous system disease 1 20 and up to 5% of acute vertigo may be due to cerebrovascular disease 1. In our cohort, a significant fraction (16.4%) had a central disease. Because of this concern, ED evaluations for vertigo are often lengthy, involve substantial use of diagnostic resources, and require many consultants. Although the use of neuroimaging and admission in patients with vertigo are disproportionately high, this does not correspond with improvements in the overall diagnostic yield for stroke 1 12. According
Accordingly, in our cohort, CT brain scan was performed in more than half of the population with a low diagnostic yield (15.4%). In order to optimise both patient care and use of healthcare resources, some bedside techniques have recently been developed to assess stroke in patients with acute vertigo. Early studies investigated the association between individual symptoms, signs, or risk factors with the presence of central nervous system disease. Among these, multiple prodromal episodes of dizziness, neurologic symptoms including diplopia 21 and age over 50 years 13 were strongly associated with stroke. However, these studies provided a low level of evidence 11 due to their retrospective nature. More recently 22, in a series of 120 patients with vertigo/dizziness, Ozono et al. reported that the risk factors for cerebrovascular disease such as hypertension, heart disease and diabetes were also risk factors for central vertigo/dizziness; moreover, to predict a central origin for vertigo/dizziness, only gaze nystagmus was a significant factor. Cnyrim et al. considered the usefulness of finding skew deviation, gaze-evoked nystagmus, negative HIT, impaired vertical smooth pursuit and deviation of subjective visual vertical in a population of 83 patients with rotatory vertigo, postural imbalance and horizontal-rotational nystagmus, without additional inner ear, brainstem or cerebellar symptoms; the authors found that when all 5 signs were combined, the sensitivity and specificity in diagnosing central vertigo increased to 92% 23. Similarly, based on the presence of negative HIT, central-type nystagmus, skew deviation and abnormal vertical smooth pursuit, classification of acute vestibular syndromes (distinguishing between vestibular neuritis and cerebellar or brainstem infarction) appeared to be reliable even in a stroke unit, as pointed out by Chen et al. 24: despite the relative inexperience in neuro-otology of the stroke team, the sensitivity and specificity of bedside ocular motor testing were comparable to those reported by expert neuro-otologist. In another study, a structured bedside clinical examination was proposed 10. Kattah et al. described a 3-step bedside oculomotor examination called HINTS (Head Impulse- Nystagmus-Test of Skew) for differentiating stroke from acute peripheral vestibulopathy. The results of their study confirmed that a normal HIT is the single best bedside predictor of stroke, and showed that the HINTS appears to be more sensitive for stroke than early MRI. Furthermore, a fourth step (HINTS "plus") has been recently added to the HINTS protocol that includes assessing the presence of new hearing loss, generally unilateral and on the side of the abnormal head impulse test 25: recent evidence suggests that the presence of such hearing loss more often indicates a vascular rather than viral cause of the acute vestibular syndrome presentation. Thus, in cases of inner ear strokes, in which HINTS eye movements are indistinguishable from vestibular neuritis, comorbid sudden hearing loss may be the only clue to stroke.
There are at least three important differences between our study and that of Kattah et al. First, we included all patients with acute vertigo without overt neurological signs, thus an unselected population which included not only patients with acute vestibular syndrome but also patients with other vestibulopathies and even those who did not have vestibular disease. We believe that in practice, to rule out a life-threatening disorder such as posterior cerebrovascular disease, the most effective way is to "rule in" one of the non-central specific disorders. Therefore, the STANDING algorithm provides the essential tools to recognise the most frequent peripheral vestibular diseases (BPPV and VN) and can help emergency physicians to identify the population of patients with central disease. However, by including all kind of vertigo we failed to submit all patients to a strong gold standard (brain MRI) as Kattah et al. have done.
Second, we propose a diagnostic algorithm, which includes nystagmus examination performed by emergency physicians, while in the HINTS study oculomotor examination was performed by expert neuro-opthalmologists. Nystagmus assessment is a key diagnostic feature in patients presenting with dizziness because the presence of specific types of nystagmus may be the only indicator of a potentially serious pathology, even if CT or MRI imaging are negative 26. One prior study showed that ED physicians report in charts the presence or absence of nystagmus in most patients presenting with acute dizziness, but that they do not utilize this sign for diagnostic purposes 27. In our study, STANDING showed good reliability and high accuracy in emergency physician hands. Finally, we point out that patients with presumed vertigo at the end of clinical examination not infrequently (14.3%) showed any signs of nystagmus. Our results indicate that these patients, due to the high prevalence of central disease (36%), should be carefully assessed.
In a recent study, Navi et al. 28 reported that the ABCD2 score is a useful tool to differentiate cerebrovascular from non-cerebrovascular causes of dizziness. However, the authors noted several limitations to their approach. In particular, the retrospective nature of the study may have overestimated the performance of the score. Moreover, the ABCD2 score does not include nystagmus examination, precluding comparison with the STANDING and the HINTS that remain the two diagnostic algorithms specifically developed for vertigo examination.
In an era in which efficiency and cost containments are warranted, STANDING may be a quick and inexpensive method that reduces healthcare costs. Indeed, STANDING was associated with a significant reduction of neuroimaging and hospitalisation rates. To our knowledge, this is the first study showing the potential clinical impact of using a structured bedside diagnostic algorithm in vertiginous patients presenting to the ED.
Limitations
Our data should be interpreted in the context of several limitations. First, it was limited to a single tertiary care referral centre with daily audiologist consultations and thus it is uncertain that STANDING will yield similar results in other settings. Second, our study lacks a strong gold standard (e.g. MR in all patients), and thus the derived sensitivity, specificity and accuracy of the test may have been overestimated. Third, the study was not randomised. Thus, the different CT and hospitalisation rates may be not accurate. Fourth, among central causes of vertigo we identified 40% of patients diagnosed with central diseases with vertebro-basilar TIA. Although National Institute of Neurological Disorders and Stroke criteria state that isolated vertigo should not be defined as TIAs, a recent study reports that in patients with definite vertebrobasilar stroke, isolated vertigo is the most common symptom preceding vertebrobasilar stroke 29. Moreover, since patients with vertebrobasilar TIA were reported to have the same risk of subsequent stroke as those with carotid TIA 17, it may be useful to have a practical clinical prediction algorithm to identify subgroups of patients at high-risk of vertebrobasilar stroke.
Conclusions
Although our results should be interpreted with caution, in our unselected cohort the STANDING test appears to show high sensitivity and specificity to detect central vestibulopathy, with good reliability in the emergency setting. STANDING seems to be associated with a reduction of neuroimaging burden and hospital admission rates, and may thus be promising tool for evaluation of acute vertigo. This data, largely exploratory, should be confirmed in a properly designed clinical trial.
References
- 1.Newman-Toker DE, Hsieh YH, Camargo CA, Jr, et al. Spectrum of dizziness visits to US emergency departments: crosssectional analysis from a nationally representative sample. Mayo Clin Proc. 2008;83:765–775. doi: 10.4065/83.7.765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Newman-Toker DE, Cannon LM, Stofferahn ME, et al. Imprecision in patient reports of dizziness symptom quality: a cross-sectional study conducted in an acute care setting. Mayo Clin Proc. 2007;82:1329–1340. doi: 10.4065/82.11.1329. [DOI] [PubMed] [Google Scholar]
- 3.Kerber KA, Brown DL, Lisabeth LD, et al. Stroke among patients with dizziness, vertigo, and imbalance in the emergency department: a population-based study. Stroke. 2006;37:2484–2487. doi: 10.1161/01.STR.0000240329.48263.0d. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Herr RD, Zun L, Mathews JJ. A directed approach to the dizzy patient. Ann Emerg Med. 1989;18:664–672. doi: 10.1016/s0196-0644(89)80524-4. [DOI] [PubMed] [Google Scholar]
- 5.Armato E, Ferri E, Pinzani A, et al. Cerebellar haemorrhage mimicking acute peripheral vestibulopathy: the role of the video head impulse test in differential diagnosis. Acta Otorhinolaryngol Ital. 2014;34:288–291. [PMC free article] [PubMed] [Google Scholar]
- 6.Casani AP, Dallan I, Cerchiai N, et al. Cerebellar infarctions mimicking acute peripheral vertigo: how to avoid misdiagnosis? Otolaryngol Head Neck Surg. 2013;148:475–481. doi: 10.1177/0194599812472614. [DOI] [PubMed] [Google Scholar]
- 7.Bhattacharyya N, Baugh RF, Orvidas L, et al. Clinical practice guideline: benign paroxysmal positional vertigo. Otolaryngol Head Neck Surg. 2008;139:47–81. doi: 10.1016/j.otohns.2008.08.022. [DOI] [PubMed] [Google Scholar]
- 8.Califano L, Vassallo A, Melillo MG, et al. Direction-fixed paroxysmal nystagmus lateral canal benign paroxysmal positioning vertigo (BPPV): another form of lateral canalolithiasis. Acta Otorhinolaryngol Ital. 2013;33:254–260. [PMC free article] [PubMed] [Google Scholar]
- 9.Califano L, Salafia F, Mazzone S, et al. Anterior canal BPPV and apogeotropic posterior canal BPPV: two rare forms of vertical canalolithiasis. Acta Otorhinolaryngol Ital. 2014;34:189–197. [PMC free article] [PubMed] [Google Scholar]
- 10.Kattah JC, Talkad AV, Wang DZ, et al. HINTS to diagnose stroke in the acute vestibular syndrome: three-step bedside oculomotor examination more sensitive than early MRI diffusion- weighted imaging. Stroke. 2009;40:3504–3510. doi: 10.1161/STROKEAHA.109.551234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Tarnutzer AA, Berkowitz AL, Robinson KA, et al. Does my dizzy patient have a stroke? A systematic review of bedside diagnosis in acute vestibular syndrome. CMAJ. 2011;183:571–592. doi: 10.1503/cmaj.100174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kerber KA, Meurer WJ, West BT, et al. Dizziness presentations in U.S. emergency departments, 1995-2004. Acad Emerg Med. 2008;15:744–750. doi: 10.1111/j.1553-2712.2008.00189.x. [DOI] [PubMed] [Google Scholar]
- 13.Simmons Z, Biller J, Adams HP, Jr, et al. Cerebellar infarction: comparison of computed tomography and magnetic resonance imaging. Annals of Neurology. 1986;19:291–293. doi: 10.1002/ana.410190312. [DOI] [PubMed] [Google Scholar]
- 14.Edlow JA, Newman-Toker DE, Savitz SI. Diagnosis and initial management of cerebellar infarction. Lancet Neurol. 2008;7:951–964. doi: 10.1016/S1474-4422(08)70216-3. [DOI] [PubMed] [Google Scholar]
- 15.Moulin T, Sablot D, Vidry E, et al. Impact of emergency room neurologists on patient management and outcome. Eur.Neurol. 2003;50:207–214. doi: 10.1159/000073861. [DOI] [PubMed] [Google Scholar]
- 16.Flossmann E, Rothwell PM. Prognosis of vertebrobasilar transient ischaemic attack and minor stroke. Brain. 2003;126:1940–1954. doi: 10.1093/brain/awg197. [DOI] [PubMed] [Google Scholar]
- 17.Kerber KA. Vertigo and dizziness in the emergency department. Emerg Med Clin North Am. 2009;27:39–50. doi: 10.1016/j.emc.2008.09.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Cheung CS, Mak PS, Manley KV, et al. Predictors of important neurological causes of dizziness among patients presenting to the emergency department. Emerg Med J. 2010;27:517–521. doi: 10.1136/emj.2009.078014. [DOI] [PubMed] [Google Scholar]
- 19.Schappert SM, Burt CW. Ambulatory care visits to physician offices, hospital outpatient departments, and emergency departments: United States, 2001-02. Vital Health Stat 13. 2006;(159):1–66. [PubMed] [Google Scholar]
- 20.Neuhauser HK. Epidemiology of vertigo. Curr Opin Neurol. 2007;20:40–46. doi: 10.1097/WCO.0b013e328013f432. [DOI] [PubMed] [Google Scholar]
- 21.Gomez CR, Cruz-Flores S, Malkoff MD, et al. Isolated vertigo as a manifestation of vertebrobasilar ischemia. Neurology. 1996;47:94–97. doi: 10.1212/wnl.47.1.94. [DOI] [PubMed] [Google Scholar]
- 22.Ozono Y, Kitahara T, Fukushima M, et al. Differential diagnosis of vertigo and dizziness in the emergency department. Acta Otolaryngol. 2014;134:140–145. doi: 10.3109/00016489.2013.832377. [DOI] [PubMed] [Google Scholar]
- 23.Cnyrim DD, Newman-Toker D, Karch C, et al. Bedside differentiation of vestibular neuritis from central "vestibular pseudoneuritis". J Neurol Neurosurg Psychiatry. 2008;79:458–460. doi: 10.1136/jnnp.2007.123596. [DOI] [PubMed] [Google Scholar]
- 24.Chen L, Lee W, Chambers BR, et al. Diagnostic accuracy of acute vestibukar syndrome at the bedside in a stroke unit. J Neurol. 2011;258:855–861. doi: 10.1007/s00415-010-5853-4. [DOI] [PubMed] [Google Scholar]
- 25.Newman-Toker DE, Kerber KA, Hsieh Y-H, et al. HINTS outperforms ABCD2 to screen for stroke in acute continuous vertigo and dizziness. Acad Emerg Med. 2013;20:987–996. doi: 10.1111/acem.12223. [DOI] [PubMed] [Google Scholar]
- 26.Grad A, Baloh RW. Vertigo of vascular origin. Clinical and electronystagmographic features in 84 cases. Arch Neurol. 1989;46:281–284. doi: 10.1001/archneur.1989.00520390047014. [DOI] [PubMed] [Google Scholar]
- 27.Kerber KA, Morgenstern LB, Meurer WJ, et al. Nystagmus assessments documented by emergency physicians in acute dizziness presentations: a target for decision support? Acad Emerg Med. 2011;18:619–626. doi: 10.1111/j.1553-2712.2011.01093.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Navi BB, Kamel H, Shah MP, et al. Application of the ABCD2 score to identify cerebrovascular causes of dizziness in the emergency department. Stroke. 2012;43:1484–1489. doi: 10.1161/STROKEAHA.111.646414. [DOI] [PubMed] [Google Scholar]
- 29.Paul NL, Simoni M, Rothwell PM. Transient isolated brainstem symptoms preceding posterior circulation stroke: a population-based study. Lancet Neurol. 2013;12:65–71. doi: 10.1016/S1474-4422(12)70299-5. [DOI] [PMC free article] [PubMed] [Google Scholar]