Myeloid and lymphoid neoplasms with PDGFRA gene rearrangements are a category of rare diseases that typically manifest with peripheral blood eosinophilia accompanied by eosinophilic tissue infiltrates, and may be associated with typical or atypical mast cell proliferations. These tumors can present as myeloproliferative neoplasms, acute myeloid leukemias or lymphoblastic leukemias/lymphomas and are, consequently, classified within the larger category of ‘Myeloid and lymphoid neoplasms with eosinophilia and abnormalities of PDGFRA, PDGFRB or FGFR1' in the 2008 World Health Organization (WHO) classification. For neoplasms harboring PDGFRA rearrangements, FIP1L1–PDGFRA is by far the most common fusion detected, reflecting an 800-kb intrachromosomal deletion [del(4)(q12q12)].1
Identification of PDGFRA rearrangement is both an important diagnostic and predictive marker, as these neoplasms are typically responsive to treatment with imatinib.2, 3 The atypical mast cell proliferations that accompany these tumors may result in a mistaken diagnosis of systemic mastocytosis (SM),4, 5 which is associated with an activating D816V alteration in KIT.6 The discrimination of these entities is important, as PDGFRA-rearranged tumors with eosinophilia, unlike SM harboring KIT D816V, are typically imatinib responsive.7
We describe a novel TNKS2–PDGFRA fusion in a myeloid neoplasm with eosinophilia, detected via comprehensive genomic profiling with a next-generation sequencing-based assay (FoundationOne), the presence of which correlated with the results from multiprobe fluorescent in situ hybridization (FISH) testing. Initially this patient was diagnosed with an aggressive SM (aSM); however, detection of this novel fusion, a translocation between chromosomes 4(q12) and 10(q23.3), resulted in diagnostic reclassification and led to targeted drug therapy with a resulting dramatic clinical response.
A 58-year-old Caucasian woman presented with left upper quadrant pain and hepatosplenomegaly with the splenic tip palpable to 15 cm below the left costal margin and liver palpable 5 cm below the right costal margin, with 6 years of chronic, untreated hepatitis C and hepatic cirrhosis diagnosed 2 years prior. An abdominal magnetic resonance imaging scan confirmed splenomegaly with the evidence of splenic infarct and a nodular liver, consistent with cirrhosis. A small amount of ascites was present. Her medical history was significant for mild thrombocytopenia of several years duration with no evidence of bleeding.
A bone marrow core biopsy was performed and was hypercellular (100% cellularity) with dense infiltrates and aggregates of >15 spindled mast cells as well as increased eosinophils. Mast cells were positive for CD2 and CD25 expression by immunohistochemistry. Targeted genomic analysis for mutations in KIT was ordered, but ultimately failed due to insufficient sample material. Cytogenetic analysis was ordered but the results were not immediately available, given the inherent time needed for processing. On the basis of these findings, the patient was initially diagnosed with aSM. A peripheral blood count performed a month after initial diagnosis revealed leukoerythroblastic features with a marked absolute eosinophilia (white blood cells of 20.6 × 109/l, absolute eosinophil count of 3708/μl) and no circulating blasts. An expanded blast population was not seen either in the marrow aspirate smear or using CD34 immunohistochemistry on the core biopsy. The patient's clinical status deteriorated rapidly with worsening thrombocytopenia and progressive anasarca, necessitating hospitalization. Diuretics were of little benefit.
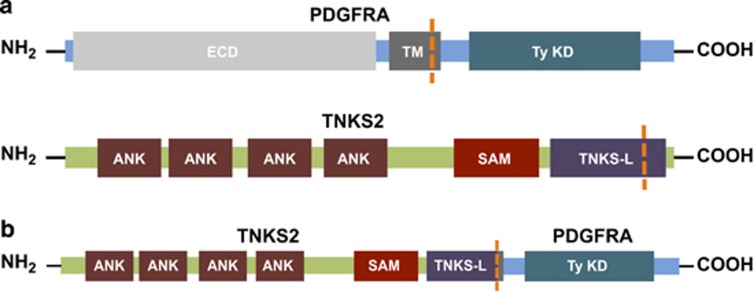
Subsequent FISH testing was reported positive for a PDGFRA gene rearrangement and was attributed to presence of the canonical FIP1L1–PDGFRA fusion. However, karyotyping identified a translocation involving chromosomes 4 and 10. Comprehensive genomic profiling by FoundationOne performed on a bone marrow aspirate fully characterized the presence of a t(4:10) abnormality, identified as a novel fusion of TNKS2 and PDGFRA (Figure 1). Further, a KRAS G12D point mutation was detected. These findings resulted in reclassification of this disease from aSM to a myeloid neoplasm with eosinophilia harboring a PDGFRA rearrangement.
Figure 1.
Intrachromosomal rearrangement of TNKS2 and PDGFRA results in a likely fusion gene. ANK, ankyrin domains; ECD, extracellular domain; SAM, sterile alpha motif; TM, transmembrane; TNKS-L, tankyrase-like; TyKD, kinase domain.
On the basis of this diagnosis, the patient was started on a course of imatinib (400 mg, daily) and the clinical benefit was dramatic with immediate onset of diuresis, resolution of the anasarca, reduction in splenomegaly and steady improvement in the platelet count.
At the time of publication, the patient remains on a maintenance dose of imatinib (100 mg, daily) and a subsequent bone marrow biopsy 4 months after initiation of therapy revealed a dramatic normalization in marrow cellularity without eosinophilia or mast cell proliferation. Follow-up multiprobe FISH for PDGFRA abnormality at this time was negative.
A bone marrow aspirate was collected via needle biopsy and comprehensive genomic profiling (FoundationOne) was performed in a Clinical Laboratory Improvement Amendments-certified laboratory, College of American Pathologists and New York State accredited (Foundation Medicine, Cambridge, MA, USA). Methods of the clinical cancer gene assay used to analyze this patient have been previously published and the assay performance has been validated rigorously.9 Here we provide a brief summary. DNA is extracted from formalin-fixed, paraffin-embedded tissue (⩾1 mm3) containing no less than 20% tumor nuclei by enzymatic digestion and subsequent purification. DNA is fragmented by sonication to 200 bp segments. Indexed sequencing adapters are ligated to the DNA fragments and PCR amplified to yield 500 ng of sequencing library. Hybridization selection is performed using individually synthesized baits targeting 3679 exons of 236 cancer-related genes and 47 introns of 19 genes frequently rearranged in cancer. The Illumina HiSeq 2500 (Illumina Inc, San Diego, CA, USA) platform is used in 49 × 49 paired-end sequencing. Sequence data are mapped to the human genome (hg19) using BWA aligner v0.5.9.10 Sequence data are analyzed through a computational analysis pipeline to call variants present in the sample, including substitutions, short insertions and deletions, rearrangements and copy-number variants.
FISH studies were performed with a standard commercially available multiprobe assay. Red and green probes hybridize to the 3' and 5' end of FIP1L1, respectively. An aqua probe hybridizes to PDGFRA. Loss of the 3' end of FIP1L1, as evidenced by an absent red signal, is considered a positive result for PDGFRA–FIP1L1 fusion.
We present a patient initially diagnosed as harboring aSM and ultimately reclassified most appropriately as having a myeloid neoplasm with rearrangement of PDGFRA with a novel fusion partner, TNKS2, identified by comprehensive genomic profiling. Since the first detection of FIP1L1–PDGFRA fusions was in 2003,1 many other fusion partners of PDGFRA have been discovered. These include translocations with chromosomes 4, 12 and 22, and partial insertions of chromosome 9.11 We have observed a translocation of TNKS2 and PDGFRA with breakpoint in intron 25 and exon 12, respectively. The discovery of a novel fusion of TNKS2 with PDGFRA further demonstrates the diversity of alterations possible in these myeloid neoplasms with eosinophilia.
From our clinical observations, it is unknown whether TNKS2 has a role identical to that of FIP1L1 in the context of a fusion with PDGFRA. Further in vitro studies are needed to elucidate whether the identity of the fusion partner in a PDGFRA rearrangement has functional and clinical implications.
Unlike the near pathognomonic KIT D816V alteration in SM, FIP1L1–PDGFRA fusions are associated with a favorable response to imatinib therapy2, 3 and mandate the diagnosis of a myeloid neoplasm with PDGFRA rearrangement. Other known fusion partners of PDGFRA described by Gotlib and Cools11 have not yet been known to respond to imatinib.12 The TNKS2–PDGFRA fusion described further extends the set of known targets for imatinib therapy. Interestingly, a concurrent KRAS G12D mutation was also detected in this patient. In some settings, this mutation has been associated with a poor response to tyrosine–kinase inhibition;8 yet the patient's response to imatinib was remarkable and has been durable to date. Others have described similar findings with crizotinib, demonstrating clinical efficacy despite the presence of concurrent KRAS and MET alterations in the same tumor.13
Unlike the well-characterized intrachromosomal FIP1L1–PDGFRA fusion of 4q12, the TNKS2–PDGFRA fusion observed in this patient is an interchromosomal translocation. In performing diagnostic assays, direct interrogation for the presence of FIP1L1–PDGFRA may not always be performed and patients are, instead, tested for deletion of CHIC2, a gene that lies in the region between FIP1L1 and PDGFRA. This surrogate assay may be most effective for detecting recurrence of a typical FIP1L1–PDGFRA,5 but would not have resolved an interchromosomal event such as this one.
In summary, this report describes a novel TNKS2–PDGFRA fusion that diagnostically defines a distinct category of myeloid neoplasms with eosinophilia as defined within the WHO 2008 classification schema. Using FISH analyses, these neoplasms may be incorrectly interpreted as harboring a FIP1L1–PDGFRA rearrangement or may even be interpreted as fusion negative if the fusion is complex and FISH probes do not fully border the regions of interest. Identification of a PDGFRA rearrangement is important for both diagnosis and therapy as these neoplasms are responsive to tyrosine kinase inhibitors. However, the implications for durability of therapeutic response and prognosis, particularly with respect to the specific PDGFRA fusion partner, are not yet known. Comprehensive genomic profiling offers an accurate and detailed analysis platform that reliably detects the complex interchromosomal events at gene-level resolution.
ZRC, SMA, GMF, JSR, RY, NAP, JAE, JC, PJS, VAM, RE and PVC are employed by and have equity interest in Foundation Medicine, Inc. The remaining authors declared no conflict of interest.
References
- Cools J, DeAngelo DJ, Gotlib J, Stover EH, Legare RD, Cortes J, et al. A tyrosine kinase created by fusion of the PDGFRA and FIP1L1 genes as a therapeutic target of imatinib in idiopathic hypereosinophilic syndrome. N Engl J Med. 2003;348:1201–1214. doi: 10.1056/NEJMoa025217. [DOI] [PubMed] [Google Scholar]
- Pardanani A, D'Souza A, Knudson RA, Hanson CA, Ketterling RP, Tefferi A. Long-term follow-up of FIP1L1-PDGFRA-mutated patients with eosinophilia: survival and clinical outcome. Leukemia. 2012;26:2439–2441. doi: 10.1038/leu.2012.162. [DOI] [PubMed] [Google Scholar]
- Jain N, Cortes J, Quintás-Cardama A, Manshouri T, Luthra R, Garcia-Manero G, et al. Imatinib has limited therapeutic activity for hypereosinophilic syndrome patients with unknown or negative PDGFRα mutation status. Leuk Res. 2009;33:837–839. doi: 10.1016/j.leukres.2008.10.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tefferi A, Verstovsek S, Pardanani A. How we diagnose and treat WHO-defined systemic mastocytosis in adults. Haematologica. 2008;93:6–9. doi: 10.3324/haematol.12324. [DOI] [PubMed] [Google Scholar]
- Pardanani A, Ketterling RP, Brockman SR, Flynn HC, Paternoster SF, Shearer BM, et al. CHIC2 deletion, a surrogate for FIP1L1-PDGFRA fusion, occurs in systemic mastocytosis associated with eosinophilia and predicts response to imatinib mesylate therapy. Blood. 2003;102:3093–3096. doi: 10.1182/blood-2003-05-1627. [DOI] [PubMed] [Google Scholar]
- Garcia-Montero AC, Jara-Acevedo M, Teodosio C, Sanchez ML, Nunez R, Prados A, et al. KIT mutation in mast cells and other bone marrow hematopoietic cell lineages in systemic mast cell disorders: a prospective study of the Spanish Network on Mastocytosis (REMA) in a series of 113 patients. Blood. 2006;108:2366–2372. doi: 10.1182/blood-2006-04-015545. [DOI] [PubMed] [Google Scholar]
- Ma Y, Zeng S, Metcalfe DD, Akin C, Dimitrijevic S, Butterfield JH, et al. The c-KIT mutation causing human mastocytosis is resistant to STI571 and other KIT kinase inhibitors; kinases with enzymatic site mutations show different inhibitor sensitivity profiles than wild-type kinases and those with regulatory-type mutations. Blood. 2002;99:1741–1744. doi: 10.1182/blood.v99.5.1741. [DOI] [PubMed] [Google Scholar]
- Miranda C, Nucifora M, Molinari F, Conca E, Anania MC, Bordoni A, et al. KRAS and BRAF mutations predict primary resistance to imatinib in gastrointestinal stromal tumors. Clin Cancer Res Off J Am Assoc Cancer Res. 2012;18:1769–1776. doi: 10.1158/1078-0432.CCR-11-2230. [DOI] [PubMed] [Google Scholar]
- Frampton GM, Fichtenholtz A, Otto GA, Wang K, Downing SR, He J, et al. Development and validation of a clinical cancer genomic profiling test based on massively parallel DNA sequencing. Nat Biotechnol. 2013;31:1023–1031. doi: 10.1038/nbt.2696. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li H, Durbin R. Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics. 2009;25:1754–1760. doi: 10.1093/bioinformatics/btp324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gotlib J, Cools J. Five years since the discovery of FIP1L1-PDGFRA: what we have learned about the fusion and other molecularly defined eosinophilias. Leukemia. 2008;22:1999–2010. doi: 10.1038/leu.2008.287. [DOI] [PubMed] [Google Scholar]
- Stover EH, Chen J, Folens C, Lee BH, Mentens N, Marynen P, et al. Activation of FIP1L1–PDGFRalpha requires disruption of the juxtamembrane domain of PDGFRalpha and is FIP1L1-independent. Proc Natl Acad Sci USA. 2006;103:8078–8083. doi: 10.1073/pnas.0601192103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Palma NA, Ali SM, O'Connor J, Dutta D, Wang K, Soman S, et al. Durable response to crizotinib in a MET-amplified, KRAS-mutated carcinoma of unknown primary. Case Rep Oncol. 2014;7:503–508. doi: 10.1159/000365326. [DOI] [PMC free article] [PubMed] [Google Scholar]