Abstract
A 7-year-old, miniature dachshund was referred for examination and treatment of persistent anorexia, deep yellow-coloured urine and leucocytosis. The clinical sign of jaundice, results from a serum biochemistry profile and ultrasonographic images suggested a biliary tract obstruction. A cholecystectomy was performed to remove the obstruction. Histopathological assessment of the resected gallbladder and partial common bile duct indicated diffuse large B-cell lymphoma. Twelve days after the initial operation, a second procedure was performed due to bile leakage into the abdominal cavity. Chemotherapy was administered twice after the second operation but discontinued, because the dog showed adverse effects. The dog is still alive 24 months after the surgery. To the authors’ knowledge, this is the first description of canine gallbladder lymphoma.
Keywords: canine, cholecystectomy, gallbladder lymphoma, jaundice
Malignant lymphomas arise in lymphoid tissues, such as lymph nodes and spleen. Most extranodal lymphomas originate from the gastrointestinal tract [17]. Among them, gallbladder lymphoma is extremely rare. In human medicine, only 66 cases of lymphoma of the gallbladder have been reported in the literature [13]. Most reported cases of gallbladder lymphoma in humans have been of the diffuse large B-cell (DLBCL) or mucosa-associated lymphoid tissue (MALT) lymphoma types [5, 9, 14]. There have been no reports of canine gallbladder lymphoma, even though lymphoma is the most common hematopoietic tumor affecting dogs [17]. To the authors’ knowledge, this is the first report which details the characteristics of a canine gallbladder lymphoma.
A 7-year-old, spayed, female miniature dachshund, weighing 4.2 kg, was referred to the Animal Medical Center at Gifu University for examination and treatment for persistent anorexia of 16 days’ duration, discoloured urine of a deep yellow color and leucocytosis which did not respond to antibiotics. Prior to referral, the dog had received pharmacological treatment (cefazolin, enrofloxacin, glutathione, ursodeoxycholic acid and trepibutone) for 2 weeks at the practitioner. Following this treatment, the anorexia improved slightly, but the urine became a darker yellow color, and the leucocytosis remained unchanged. Prior to the initial presentation at our hospital, the dog did not have any past medical problems.
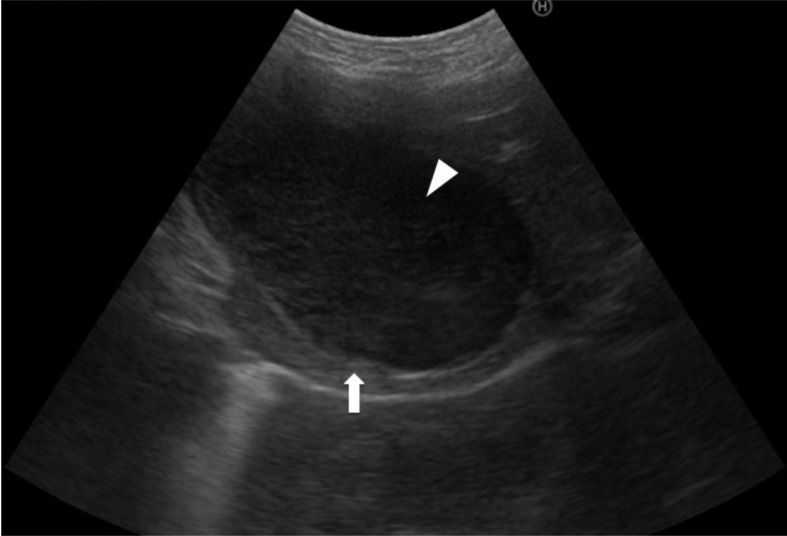
At the first visit to the Animal Medical Center at Gifu University, the dog was lethargic, and her conjunctiva and oral mucosa were markedly icteric. A complete blood cell count (CBC) showed apparent leucocytosis (31,500 cells/µl; reference range [RR], 6,000–17,000 cells/µl) with a mature neutrophilia (28,700 cells/µl; RR, 3,000–11,500 cells/µl). Serum biochemical analysis revealed an increased total bilirubin concentration (4.4 mg/dl; RR, 0.1–0.5 mg/dl) and markedly elevated activities of alanine aminotransferase (ALT; 1,846 U/l; RR, 17–78 U/l), aspartate aminotransferase (AST; 323 U/l; RR, 17–44 U/l), γ-glutamyltransferase (GGT; 86 U/l; RR, 5–14 U/l) and alkaline phosphatase (ALP; 10,500 U/l; RR, 47–254 U/l). Abdominal ultrasonographic evaluation confirmed the hyperechoic sludgy content and the hyperechoic gallbladder wall (Fig. 1) and identified a dilated common bile duct (CBD; diameter, 4.4 mm).
Fig. 1.
Abdominal ultrasonography of the gallbladder. Abdominal ultrasonography of the gallbladder showing a hyperechoic wall (arrow) and hyperechoic sludgy content (arrowhead).
The jaundice, serum biochemical results and ultrasonographic images suggested a biliary tract obstruction. An emergency cholecystectomy was performed, because of the rapid deterioration in the patient’s general condition. The abdomen was surgically exposed with a ventral midline incision under general anesthesia. The gallbladder was prominently enlarged in the mid-abdominal cavity. The liver lobes appeared yellow in color and were mildly distended (Fig. 2A). Incision of the gallbladder wall revealed light green-colored muddy bile and mildly yellowed lumina of the gallbladder wall. Several gallstones were observed in the gallbladder (Fig. 2B). To confirm CBD patency, an 8Fr multipurpose tube (Atom Multipurpose Tube; Atom Medical Corporation, Tokyo, Japan) was inserted into the CBD from the resected end of the gallbladder. However, the tube became stuck in the middle of the CBD, because of an approximately 1-cm compression outside the CBD caused by proliferative tissue. The CBD was also partially dissected and subsequently sutured. To prevent postoperative stenosis, an indwelling feeding tube was placed in the CBD. The cholecystectomy was completed by removing the remaining gallbladder wall. A guillotine suture biopsy was performed on the quadrate lobe of the liver. A closed and continuous low-vacuum surgical drainage system (J-VAC®; Johnson & Johnson, NJ, U.S.A.) was placed at the gallbladder site, and peritoneal lavage using saline solution was performed. After the surgery, the enzyme activity levels of AST and ALT and the total bilirubin concentration gradually decreased. Initially, the postsurgical drainage fluid was deep yellow; however, ten days after surgery, the drainage fluid suddenly became dark green and remained unchanged for 2 days. Since the appearance of the drainage fluid suggested leakage of bile into the abdominal cavity, a second surgery was performed on day 12 after the first surgery. The abdomen was surgically exposed with a ventral midline incision under general anesthesia. The suture site of the CBD was widely obstructed and disconnected, and bile was leaking from a small opening in the CBD near the hepatic portal side. The original CBD was disconnected, because of a new compression caused by the proliferative tissue that was similar to the compression observed at the first surgery. A feeding tube (French size 8) was placed to bypass the obstructed part of the CBD to establish normal bile flow between the cranial part of the CBD and the duodenum. An indwelling closed and continuous low-vacuum surgical drainage system was placed after the surgery.
Fig. 2.
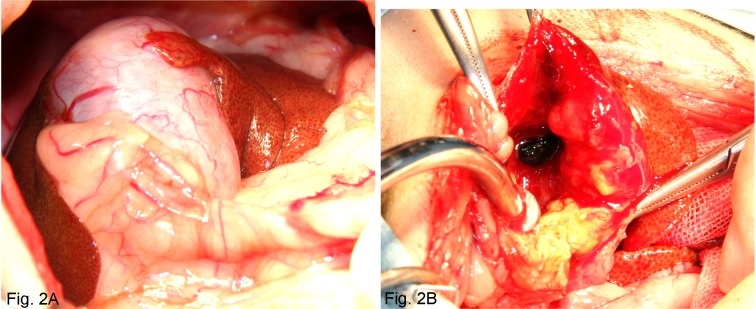
Intraoperative abdominal cavity findings (A) and Mucous membrane in the gallbladder (B).
The enlarged gallbladder and the yellow and mildly distended liver lobes are apparent (A). Mildly yellowed gallbladder wall lumina and several gallstones are observed in the gallbladder during surgery (B).
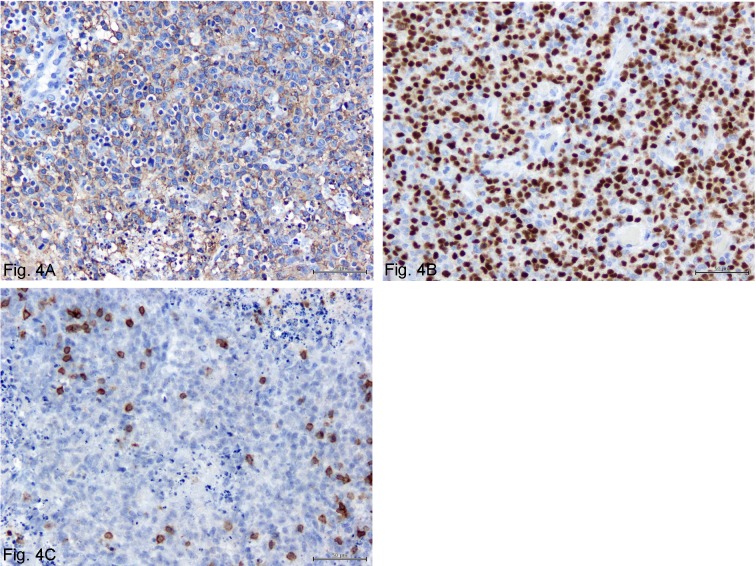
The dissected gallbladder, proliferating tissue from the CBD, and liver biopsy collected during the first surgery were histopathologically examined. Tissue from the gallbladder and the CBD comprised diffuse large lymphoid neoplastic cells with large vesicular nuclei which had coarse chromatin and one or several prominent nucleoli (Fig. 3). Several mitotic figures were observed throughout the neoplastic tissues (Fig. 3B). The proliferative neoplastic cells were predominant in the perivascular area, because severe necrosis obscured the remaining tissues. In a marginal area assumed to be the gallbladder wall, moderate to severe fibrosis was observed, and the mucosal epithelial cells were absent. Immunohistochemical evaluation of the neoplastic lymphoid cells showed they stained with anti-CD20 and anti-Pax5 antibodies, but did not stain with anti-CD3 antibodies; only infiltrative small mature lymphocytes were positive for CD3 (Fig. 4). Based on the histopathological and immunohistochemical results, the present neoplasm was diagnosed as diffuse large B-cell lymphoma. In hepatic tissue, moderate to severe neutrophilic infiltration, hepatocyte vacuolation consistent with the accumulation of lipid and fibrosis with moderate proliferation of cholangiole cells in Glisson’s capsules were observed. Hepatic infiltration with neoplastic cells was not observed.
Fig. 3.
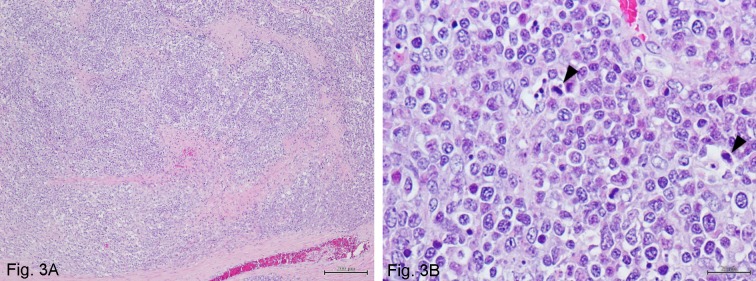
Overview (A) and detail (B) of the histopathological aspects of the gallbladder.
The gallbladder and proliferating tissue of the common bile duct comprise diffuse large lymphoid neoplastic cells with large vesicular nuclei with coarse chromatin and one or several prominent nucleoli. Several mitotic figures are observed throughout the neoplastic tissues (arrowhead). Hematoxylin and Eosin stain. Bar=200 µm (A) and 20 µm (B).
Fig. 4.
Immunohistochemical staining of the dissected gallbladder.
(A) Neoplastic lymphoid cells are positive for CD20 in the cytoplasmic membrane. Immunolabeling with anti-CD20, Hematoxylin counterstain. (B) The nuclei of neoplastic lymphoid cells are positive for Pax5. Immunolabeling with anti-Pax5, Hematoxylin counterstain. (C) The majority of neoplastic lymphoid cells are negative for CD3. Small mature lymphocytes infiltrating into the neoplastic tissue are positive for CD3. Immunolabeling with anti-CD3, Hematoxylin counterstain. All bars=50 µm.
Before the second surgery, computed tomography (CT) was performed to detect lymphoma cell infiltration in additional tissues. There was no apparent lymph node enlargement or neoplastic lesions in the thoracic or abdominal cavity.
The dog was hospitalized for 5 days after the second surgery. After discharge, the patient received daily total parenteral nutrition, antibiotics and drainage fluid clearing. The drainage tube was removed 18 days after the second surgery. Routine clinical, hematological and biochemistry examinations were periodically performed. The dog’s condition improved, and her appetite stabilized approximately 1 month after the second surgery. Only antibiotic therapy was continued for an additional month. Chemotherapy for lymphoma included low-dose chlorambucil (Leukeran®; GlaxoSmithKline LLC, Philadelphia, PA, U.S.A., 2 mg/m2) administered orally once a day beginning at 3 months after the second surgery. However, chemotherapy was discontinued due to an elevation in the patient’s liver enzyme activities and nausea. Because the liver enzyme activities decreased after one month, chemotherapy was restarted at the same dose, but the schedule was changed to twice a day. However, chemotherapy was stopped after the liver enzyme activities increased and the nausea reappeared. Since then, the dog has not received any chemotherapy, but has been placed on a commercial therapeutic diet for gastrointestinal care (i/d® Canine Gastrointestinal Health; Hill’s Pet Nutrition, Topeka, KS, U.S.A.) and nutritional supplements (vegetable support; W/I System Co., Tokyo, Japan). Routine follow-up examinations, including hematological and biochemical examinations, are performed monthly. The dog no longer appears icteric, and most laboratory values are within the reference range, except for GGT (372 U/l) and ALP (1,064 U/l). The dog’s weight increased slightly from 4.2 to 4.5 kg. The dog’s overall condition at 24 months from the first visit is good.
In veterinary medicine, only two cases of feline gallbladder lymphoma have been reported to date [2, 7]. One cat was diagnosed with lymphoma based on cytologic evaluation of fine-needle aspirates of the gallbladder, and a urine sediment evaluation identified concurrent B-cell lymphoma in the urinary bladder [7]. Therefore, the immunophenotype of the lymphoma in the gallbladder in the cat was speculated to be of B-cell lineage; however, because immunohistochemistry was not performed for the cells from the gallbladder, it remains unclear whether this cat had two independent tumors or whether the same clonal expansion of lymphoma cells occurred in both the urinary bladder and the gallbladder. Histopathological examination with immunohistochemistry in another cat was consistent with a diagnosis of small-cell lymphoma of T cells within the gallbladder, liver and small intestine. In humans, the majority of immunophenotypes were of B-cell lineage and included extranodal marginal zone lymphoma (24%), diffuse large B-cell lymphoma (24%) and follicular lymphoma (15%) [13]. In the dog from our report, the lymphoma was demonstrated to be of B-cell origin, similar to the former case described in a cat and to humans. In general, lymphoma of B-cell origin shows a positive response to chemotherapy, compared with those of T-cell origin [4]. In the current case, we chose low dose chlorambucil chemotherapy that is considered a safe and effective treatment for dogs [12], because the dog’s general condition was poor. However, chemotherapy was not continued, because the patient experienced adverse side effects associated with the treatment. Therefore, we were unable to assess the response to the chemotherapy for gallbladder lymphoma in this dog; however, the gallbladder lymphoma in the present case was not clinically malignant. Without potent and long-term chemotherapy, the dog has maintained her quality of life 24 months from the first visit.
The most common clinical symptom in human patients with gallbladder lymphoma is upper abdominal pain [1, 3, 5,6,7,8, 10, 11, 14,15,16, 18]. One of 2 cats with gallbladder lymphoma also showed a pain response with palpation of the cranial abdomen [7]. In the dog of this report, we could not elicit upper abdominal pain, suggesting it may be difficult to identify upper abdominal pain in animals with gallbladder lymphoma. Most human patients had cholecystectomies, and the diagnosis of gallbladder lymphoma was histologically confirmed [6,7,8, 10, 11, 13,14,15,16, 18, 19]. Canine gallbladder lymphoma is extremely rare and difficult to diagnose on the basis of symptoms, ultrasonography or hematology and biochemical results, but based on the findings in human medicine, gallbladder lymphoma should be a differential diagnosis for biliary tract obstruction in dogs.
There were several surgically related findings in the present case. Substituting a feeding tube temporarily for the CBD was extremely challenging, but worked well. Using a closed-type vacuum drain may be valuable for patients with gallbladder rupture or biliary tract damage, because its use prevents peritonitis spread by the continuous suction of leaked bile, inflammatory mediators and exudates. Thus, the surgical site around the CBD may heal faster without systemic inflammation.
In conclusion, gallbladder lymphoma is exceedingly rare, but should be a differential diagnosis for a biliary tract obstruction in dogs, and postoperative histopathological evaluation is essential for accurate diagnosis. This is the first report of canine gallbladder lymphoma.
REFERENCES
- 1.Abe Y., Takatsuki H., Okada Y., Saito A., Kimura T., Nishimura J.1999. Mucosa-associated lymphoid tissue type lymphoma of the gallbladder associated with acute myeloid leukemia. Intern. Med. 38: 442–444. doi: 10.2169/internalmedicine.38.442 [DOI] [PubMed] [Google Scholar]
- 2.Baxter K. J., Hill R. C., Parfitt S. L., Berry C. R., Heskett T. W., Sheppard B. J.2012. Gastrointestinal small-cell lymphoma with gall bladder involvement in a cat. J. Feline Med. Surg. 14: 267–271. doi: 10.1177/1098612X11434820 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bisig B., Copie-Bergman C., Baia M., Gaulard P., Delbecque K., Fassotte M. F., Boniver J., de Leval L.2009. Primary mucosa-associated lymphoid tissue lymphoma of the gallbladder: report of a case harboring API2/MALT1 gene fusion. Hum. Pathol. 40: 1504–1509. doi: 10.1016/j.humpath.2009.02.018 [DOI] [PubMed] [Google Scholar]
- 4.Carter R. F., Valli V. E., Lumsden J. H.1986. The cytology, histology and prevalence of cell types in canine lymphoma classified according to the National Cancer Institute Working Formulation. Can. J. Vet. Res. 50: 154–164. [PMC free article] [PubMed] [Google Scholar]
- 5.Ferluga D., Luzar B., Gadzijev E. M.2003. Follicular lymphoma of the gallbladder and extrahepatic bile ducts. Virchows Arch. 442: 136–140. [DOI] [PubMed] [Google Scholar]
- 6.Friedman E. P., Lazda E., Grant D., Davis J.1993. Primary lymphoma of the gallbladder. Postgrad. Med. J. 69: 585–587. doi: 10.1136/pgmj.69.813.585 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Geigy C. A., Dandrieux J., Miclard J., Kircher P., Howard J.2010. Extranodal B-cell lymphoma in the urinary bladder with cytological evidence of concurrent involvement of the gall bladder in a cat. J. Small Anim. Pract. 51: 280–287. doi: 10.1111/j.1748-5827.2010.00911.x [DOI] [PubMed] [Google Scholar]
- 8.Huang W. T., Chuang S. S., Huang C. C., Lu C. L., Eng H. L.2007. Primary diffuse large B-cell lymphoma of the gallbladder with cholelithiasis masquerading as acute cholecystitis: case report and literature review. N. Z. Med. J. 120: U2470. [PubMed] [Google Scholar]
- 9.Jelic T. M., Barreta T. M., Yu M., Frame J. N., Estallila O. C., Mellen P. F., Newman S. S., Chang H. H.2004. Primary, extranodal, follicular non-Hodgkin lymphoma of the gallbladder: case report and a review of the literature. Leuk. Lymphoma 45: 381–387. doi: 10.1080/10428190310001597919 [DOI] [PubMed] [Google Scholar]
- 10.Jho D. H., Jho D. J., Chejfec G., Ahn M., Ong E. S., Espat N. J.2007. Primary biliary B-cell lymphoma of the cystic duct causing obstructive jaundice. Am. Surg. 73: 508–510. [PubMed] [Google Scholar]
- 11.Koshy M., Zhao F., Garofalo M. C.2008. Primary MALT lymphoma of the gallbladder. Case report. J. Gastrointestin. Liver Dis. 17: 207–210. [PubMed] [Google Scholar]
- 12.Leach T. N., Childress M. O., Greene S. N., Mohamed A. S., Moore G. E., Schrempp D. R., Lahrman S. R., Knapp D. W.2012. Prospective trial of metronomic chlorambucil chemotherapy in dogs with naturally occurring cancer. Vet. Comp. Oncol. 10: 102–112. doi: 10.1111/j.1476-5829.2011.00280.x [DOI] [PubMed] [Google Scholar]
- 13.Mani H., Climent F., Colomo L., Pittaluga S., Raffeld M., Jaffe E. S.2010. Gall bladder and extrahepatic bile duct lymphomas: clinicopathological observations and biological implications. Am. J. Surg. Pathol. 34: 1277–1286. doi: 10.1097/PAS.0b013e3181e9bb8b [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Ono A., Tanoue S., Yamada Y., Takaji Y., Okada F., Matsumoto S., Mori H.2009. Primary malignant lymphoma of the gallbladder: a case report and literature review. Br. J. Radiol. 82: e15–e19. doi: 10.1259/bjr/30768802 [DOI] [PubMed] [Google Scholar]
- 15.Rajesh L. S., Nada R., Yadav T. D., Joshi K.2003. Primary low-grade B-cell lymphoma of the mucosa-associated lymphoid tissue of the gallbladder. Histopathology 43: 300–301. doi: 10.1046/j.1365-2559.2003.01655.x [DOI] [PubMed] [Google Scholar]
- 16.Sezgin O., Altintas E., Polat A., Tombak A.2009. Ultrasonographic features of a large B-cell lymphoma of the gallbladder. Turk. J. Gastroenterol. 20: 239–240. [DOI] [PubMed] [Google Scholar]
- 17.Vail M. D.2012. Hematopoietic Tumors, Saunders Elsevier, St. Louis. [Google Scholar]
- 18.Willingham D. L., Menke D. M., Satyanarayana R.2009. Gallbladder lymphoma in primary sclerosing cholangitis. Clin. Gastroenterol. Hepatol. 7: A26. doi: 10.1016/j.cgh.2008.08.007 [DOI] [PubMed] [Google Scholar]
- 19.Yamamoto T., Kawanishi M., Yoshiba H., Kanehira E., Itoh H.2005. Primary non-Hodgkin’s lymphoma of the gallbladder. AJR Am. J. Roentgenol. 184: S86–S87. doi: 10.2214/ajr.184.3_supplement.01840s86 [DOI] [PubMed] [Google Scholar]