Abstract
Gastrointestinal nociception is exacerbated by chronic stress through an unknown mechanism. The amygdala is a key nucleus involved in the autonomic and neuroendocrine responses to stress. The goal of this study was to test the hypothesis that prolonged exposure of the central amygdala (CeA) to stress or the stress hormone cortisol (or corticosterone in rats) induces nociceptive behaviors mediated by corticotropin-releasing factor (CRF) within the CeA. We selectively knocked down CRF in the CeA via antisense oligodeoxynucleotides (ASO) in animals with targeted, stereotaxically placed corticosterone (CORT) micropellets or following repeated water avoidance stress (WAS). CRF expression in the CeA was analyzed concurrently with the assessment of visceral hypersensitivity to colonic distension and mechanical somatic withdrawal threshold. The responses were characterized at 7 or 28 days post implantation of the CORT micropellet or following 7 days of WAS. Exposure of the CeA to elevated CORT or WAS increased CRF expression and heightened visceral and somatic sensitivity. Infusion of CRF ASO into the CeA decreased CRF expression and attenuated visceral and somatic hypersensitivity in both models. Our study provides important evidence for a CRF-mediated mechanism specifically within the CeA that regulates stress-induced visceral and somatic nociception.
Introduction
Irritable bowel syndrome (IBS) is a chronic functional gastrointestinal disorder, in which the patient experiences abdominal pain along with abnormal bowel habits1, 2 that is often comorbid with fibromyalgia.3 IBS symptoms can be worsened by elevated stress4 and multiple imaging studies have identified altered amygdala activation in IBS patients,5 providing support for a mechanism of abnormal descending pain modulation due to hyperactivation of the stress axis.3, 5 Chronic stress is known to have considerable detrimental effects on physical and mental health due to excessive corticosteroid levels.6 The neuroendocrine response to stress involves the activation of the hypothalamic–pituitary–adrenal (HPA) axis, which is initiated by the secretion of corticotropin-releasing factor (CRF) from the paraventricular nucleus of the hypothalamus leading to the release of cortisol from the adrenal cortex. In the healthy individuals, cortisol binds to mineralocorticoid receptors and glucocorticoid receptors within limbic brain regions to terminate the stress response through negative feedback mechanisms. In contrast, corticosterone (CORT), the rat equivalent of cortisol, binding in the amygdala increases expression of CRF within the central nucleus (CeA),7 which can then stimulate increased expression of CRF within the paraventricular nucleus of the hypothalamus7, 8 to facilitate stress axis activation. Clinical studies have demonstrated HPA dysregulation in both IBS and fibromyalgia patients,9, 10 suggesting that CRF may be an important mediator of the symptoms in these patients.
Our previous studies in two rodent models provide evidence for CeA modulation of both visceral and somatic nociceptive behaviors. The first model used targeted selective exposure of the CeA to CORT via stereotaxic micropellet placement. This model pharmacologically clamped the CeA at a level of CORT similar to a peak stress exposure for at least 7 days post implantation8 and reproduced many of the cardinal characteristics of IBS including increased anxiety-like behavior, as well as increased visceral and somatic nociceptive behaviors.11, 12, 13 Interestingly, at 28 days post implantation, the effect on the nociceptive behaviors persisted despite the depletion of the CORT-containing micropellet.14 Furthermore, we found that the nociceptive phenotypes induced by CORT were concomitant with an increase in CRF in the CeA both in the presence of (7 days) and after the depletion of (28 days) the CORT implant.15 The second model used repeated water avoidance stress (WAS), a psychological stressor, to activate the CeA with endogenous CORT released by the HPA axis. In this model, we demonstrated that increased colonic sensitivity was mediated by corticosteroid receptors within the CeA16 and was associated with increased CRF expression.17 To advance our previous studies, the objective of the present investigation was to determine if a causal relationship exists between increased CRF in the CeA and nociceptive behaviors. Using a loss-of-function approach, we tested the hypothesis that the increase in CRF is critical in the initiation and maintenance of nociceptive behaviors induced by elevated CORT in the CeA. Specifically, using both the targeted model of CORT micropellet implantation and the model of repeated WAS, we investigated whether increased CRF expression in the CeA is necessary to induce and maintain heightened visceral and somatic nociceptive behaviors.
Materials and methods
Animals
The experiments were performed on single-housed, adult male (250–350 g) Fischer-344 rats (Charles Rivers Laboratory, Wilmington, MA, USA). All the animals were maintained on a 12-h light/dark cycle (lights on at 0600 h) at 21 °C and 70% humidity with ad libitum access to food and water except during fasting 12–18-h before visceral sensitivity assessment. The animals were acclimated to the experimenter and the laboratory for a week before experimentation. The Institutional Animal Care and Use Committee of both the Oklahoma City Veterans Affairs Medical Center and the University of Oklahoma Health Sciences Center approved all the experiments.
Stereotaxic surgery
Animals were randomly assigned to the 7- or 28- day postimplant groups immediately before receiving bilateral stereotaxic implantation of either CORT or cholesterol (CHOL) micropellets (30 μg each) onto the dorsal margin of the CeA (bregma −2.5 mm, medial/lateral ±4.2 mm and anteroposterior −7.0 mm from dura) as well as bilateral 26-gauge guide cannula (Plastics One, Roanoke, VA, USA), as previously described.18 Another group of rats received only bilateral cannula targeting the CeA for administration of oligodeoxynucleotide (ODN) before being exposed to WAS or sham-WAS (SHAM).
ODN administration
Animals were treated with antisense ODN to knockdown CRF expression specifically within the CeA. Antisense ODN targeting of CRF is a validated method previously demonstrated to produce behaviorally relevant knockdown of CRF expression.19 Treatments consisted of infusions into the CeA via the cannulae with either CRF-specific antisense ODN (ASO) or non-targeting random sense ODN (RSO) dissolved in phosphate-buffered saline (pH 7.4). The previously validated ODNs19 were obtained from Life Technologies (Grand Island, NY, USA) and were polyacrylamide gel electrophoresis-purified phosphorothioate end-capped sequences of 18 bases with the following sequences: ASO: 5′-CAAGCGCAACATTTCATT-3′ RSO: 5′-GTAGTCTAGTGTAGTAGT-3′. An additional Alexa Fluor 594-conjugated RSO was used to determine the diffusion radius and cellular uptake in a subset of rats. On the basis of preliminary studies, 0.5 μl of ASO or RSO (6 μg μl−1) was bilaterally administered daily to each CeA starting 1 day before behavioral testing, so that each rat received at total of four doses (12 μg total per side). The ODN solution was drawn into a 33-gauge stainless steel injector (0.5 mm longer than the guide cannula) attached to a 10 μl Hamilton syringe with polyethylene tubing. The syringe was then attached to an infusion pump and ODN was delivered to the CeA of anesthetized rats (2% isoflurane inhalation) at a rate of 0.1 μl min−1. At the completion of the infusion, the injector was left in place for an additional 2 min to allow for complete diffusion.
WAS
Rats were randomly assigned to receive WAS or SHAM (exposure to an empty chamber) and either ASO or RSO. One day after stereotaxic surgery, rats were exposed to WAS or SHAM for 1 h per day for 7 days, as previously described.16, 17 Fecal pellet output was measured at the end of each session. ODN was administered 60 min before each session.
Somatic sensitivity assessment
Mechanical somatic threshold was determined as previously described20, 21 using an electronic von Frey anesthesiometer (series 2390, IITC Life Science, Woodland Hills, CA, USA) with a rigid tip probe. The procedure was repeated three times with 5-min intertrial intervals. The mean of the three trials was used to give the final withdrawal force for each animal.
Visceral sensitivity assessment
Using a similar protocol as in previous studies,14, 17, 21, 22 colorectal distension was performed in freely moving, conscious rats in their home cage at randomly presented graded pressures of 0, 20, 40 and 60 mm Hg for 10 min each with a 10-min recovery period between each distension period. The number of abdominal contractions was recorded visually each minute of the distension and summed for the entire 10-min distension period.
Tissue collection
One hour following the final behavioral test, animals were euthanized by either transcardial perfusion of saline followed by 4% paraformaldehyde or decapitation under isoflurane anesthesia. Perfused brains were postfixed for 24 h and embedded in paraffin for postmortem analysis. Tissue sections (8 μm) containing the CeA (bregma −2.5 mm) were collected and used for implant localization and immunofluorescent detection of CRF expression. Implant localization was performed in conjunction with Nissl staining to examine the viability of CeA as previously described,21 and the location of the implants was determined on the basis of a stereotaxic atlas.23 Non-perfused brains were placed in a rat coronal precision brain slicer (Braintree Scientific, Braintree, MA, USA) and each side of the CeA was isolated using a1- mm punch (Braintree Scientific) with the tissue placed in RNAlater (Life Technologies) and stored at 4 °C until processed for CRF expression.
Quantification of CRF knockdown in the CeA
Quantitative reverse transcriptase PCR was performed as previously described,17 with minor modifications. Total RNA was isolated following the manufacturer's protocol with a PARIS Kit (Life Technologies). The concentration and quality of total RNA was determined using an Experion StdSens chip (Bio-Rad, Hercules, CA, USA) with an Experion automated electrophoresis system (Bio-Rad). cDNA was synthesized from the RNA samples with a QuantiTect Reverse Transcription Kit (Qiagen, Valencia, CA, USA). Samples and no template controls were processed for quantitative PCR in triplicate using QuantiFast SYBR Green Master Mix (Qiagen) in a final volume of 25 μl. Proprietary QuantiTect primers (Qiagen) for 28 S ribosomal RNA, GAPDH and CRF were used for all samples.
An Applied Biosystems StepOne Plus Real-Time PCR Thermal Cycler (Life Technologies) in ‘fast' mode was used following the protocol provided with the QuantiTect primers. Melting curves demonstrated single peaks at 84.3 °C for GAPDH, 87.2 °C for CRF, and 87.7 °C for 28 S ribosomal RNA. Fold change in CRF mRNA expression was calculated using the 2−ΔΔC(T) method24 with the geometric mean of the C(T) value for the two housekeeper genes25 used for the ΔC(T) calculation and the ΔΔC(T) based on the values in the CHOL+RSO or SHAM+RSO sample as appropriate.
Immunofluorescence was performed as previously described.15 Paraffin-embedded sections were processed for antigen retrieval and blocked with Rodent Block R (Biocare Medical, Concord, CA, USA). Slides were then incubated overnight at 4 °C in anti-CRF antibodies (Abcam, Cambridge, MA, USA) in 10% normal goat serum (Life Technologies), and negative controls were incubated in 10% normal goat serum (Life Technologies). Following primary incubation, the primary antibody solution was replaced with a secondary anti-rabbit antibody solution conjugated with Alexa Fluor 594 (Life Technologies). The nuclei were stained with 4',6-diamidino-2-phenylindole and the slides were coverslipped with ProLong Gold Anti-Fade solution (Life Technologies).
The samples from each animal were assigned to groups according to the last treatment that the animal received. All the samples were imaged using a fluorescent microscope (Axio Star Plus, Carl Zeiss MicroImaging, Thornwood, NY, USA). The images were captured from the medial subdivision of the CeA, which projects to key nuclei of the stress axis. Two randomly selected sections, from a total of six sections processed per animal, were imaged at × 40 magnification, and 480 000 pixels of the image were analyzed with ImageJ software (National Institutes of Health, Bethesda, MD, USA, http://imagej.nih.gov/ij) to quantify optical density. The densities were averaged into a single n-value used for statistical calculation.
Experimental design
The experimental design for the 7-day postimplant, 28-day postimplant or WAS studies are shown in Figures 1a, 2a and 3a, respectively. In all experimental series, filled circles indicate ODN treatment at least 60 min before a behavioral manipulation or tissue collection. Having previously demonstrated that the CORT implant model induces a long-term increase in nociceptive behaviors14 and CRF expression15 within the CeA and to take advantage of the transient nature of ASO knockdown of expression,19, 26 a crossover design was implemented for a subset of rats in the 7- and 28-day postimplant studies (Figures 1d and 2d) to examine whether the behaviors were reversible.
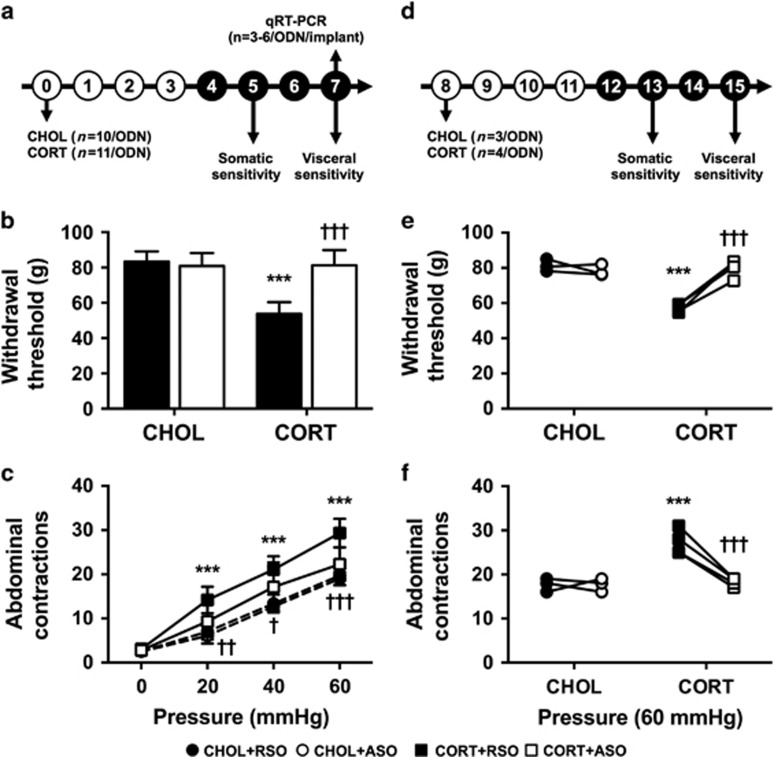
Figure 1.
Effect of ASO on CORT-induced behaviors 7 days post implant. (a–c) Primary administration of ODN; (d–f) crossover study with opposite ODN treatment. (a and d) Experimental design for the respective studies. Experimental day is listed in each circle. Filled circles represent ODN dosing. (b) Somatic withdrawal threshold was significantly reduced in CORT+RSO-treated rats, and was normalized in CORT+ASO-treated rats. (c) Visceral sensitivity was significantly increased in rats with CORT implants treated with RSO. Intra-CeA infusions of ASO in rats implanted with CORT significantly reduced visceral sensitivity to colonic distension. (e) Effect of ODN treatment on somatic withdrawal threshold in rats with CHOL or CORT implants. (f) Effect of ODN treatment on visceral sensitivity at 60 mm Hg in rats with CHOL or CORT implants. Data shown are mean±s.d. ***P<0.001 compared with CHOL+RSO, †P<0.05, ††P<0.01, †††P<0.001 compared with CORT+RSO, two- or three-factor analysis of variance with or without repeated measures, Tukey–Kramer post hoc analysis. ASO, antisense oligodeoxynucleotide; CeA, central amygdala; CHOL, cholesterol; CORT, corticosterone; ODN, oligodeoxynucleotide; RSO, random sense oligodeoxynucleotide.
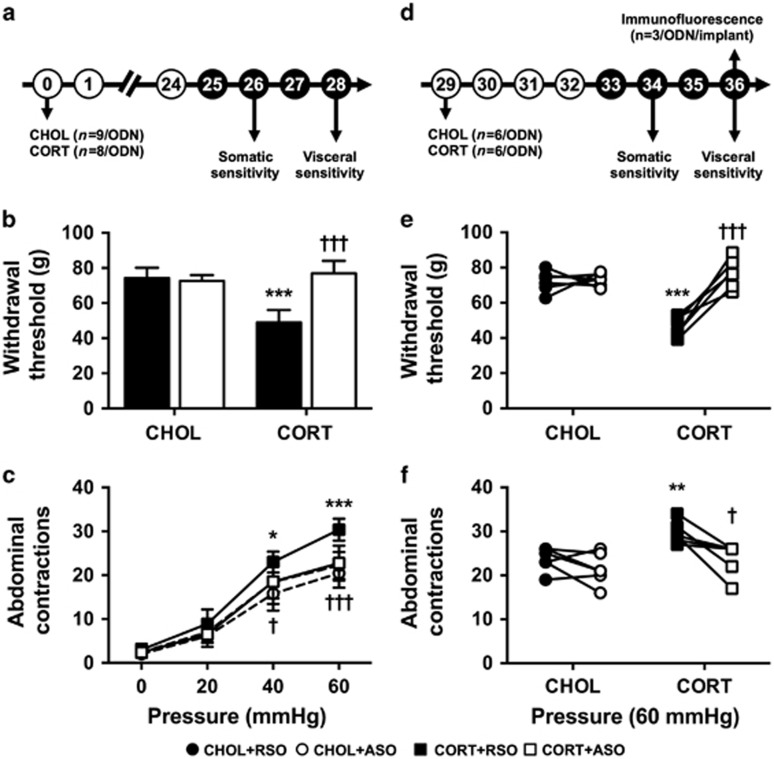
Figure 2.
Effect of ASO on CORT-induced behaviors 28 days post implant. (a–c) Primary administration of ODN; (d–f) crossover study with opposite ODN treatment. (a and d) Experimental design for the respective studies. Experimental day is listed in each circle. Filled circles represent ODN dosing. (b) Following depletion of the CORT micropellet, the significantly decreased somatic withdrawal threshold in the CORT+RSO group was inhibited in CORT-implanted rats treated with ASO. (c) Visceral hypersensitivity to colonic distension that persisted in the CORT+RSO group was significantly decreased with ASO administration in the CORT-implanted group. (e) Somatic withdrawal threshold was dependent type of ODN administered in rats that had previously been implanted with CORT on the CeA. (f) Persistent visceral hyperalgesia in CORT+RSO-treated rats was significantly decreased with ASO treatment. Data shown are mean±s.d. *P<0.01, **P<0.01, ***P<0.001 compared with CHOL+RSO, †P<0.05, †††P<0.001 compared with CORT+RSO, two- or three-factor analysis of variance with or without repeated measures, Tukey–Kramer post hoc analysis. ASO, antisense oligodeoxynucleotide; CeA, central amygdala; CORT, corticosterone; ODN, oligodeoxynucleotide; RSO, random sense oligodeoxynucleotide.
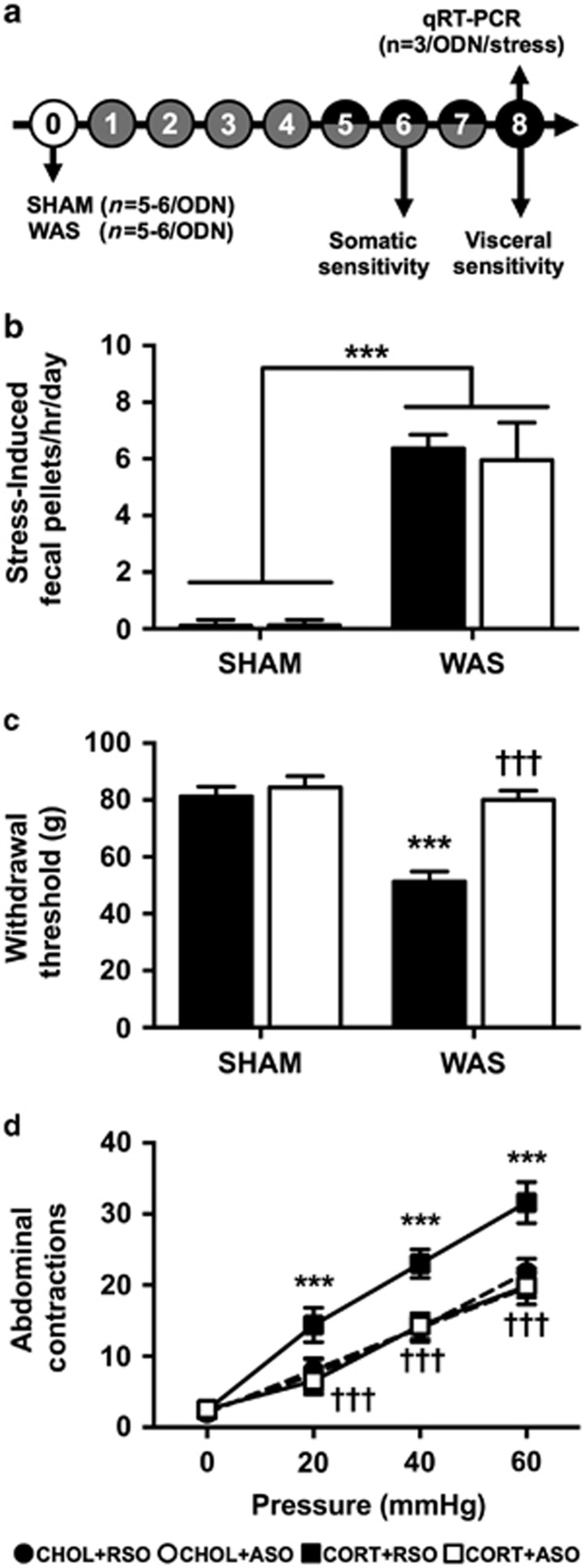
Figure 3.
Effect of ASO on WAS-induced behaviors. (a) Experimental design for repeated stress study. Experimental day is listed in each circle. Shaded circles indicate SHAM or WAS exposure. Filled circles represent ODN dosing. Both ODN dosing and stress exposure occurred on days 5–7. (b) WAS induced a significant increase in fecal pellet production on each day of exposure, which was not affected by ASO treatment. (c) WAS+RSO-treated rats demonstrated a significant decrease in withdrawal threshold that was not present in the WAS+ASO. (d) WAS+RSO induced a visceral hypersensitivity to colonic distension, which was normalized by ASO treatment. Data shown are mean±s.d. ***P<0.001 compared with SHAM+RSO, †††P<0.001 compared with WAS+RSO, two- or three-factor analysis of variance with or without repeated measures, Tukey–Kramer post hoc analysis. ASO, antisense oligodeoxynucleotide; ODN, oligodeoxynucleotide; RSO, random sense oligodeoxynucleotide; WAS, water avoidance stress.
Statistics
All data were presented as mean±s.d. and were collected by experimenters that were not masked due to the necessity of daily treatments. The n-values were based on previous studies using rats with CORT or CHOL implants onto the CeA14, 15 or following WAS.17 For somatic sensitivity, quantitative reverse transcriptase PCR and immunofluorescence, a two-way analysis of variance (ANOVA) was used followed by Tukey–Kramer post test. For visceral sensitivity, a three-way repeated measure ANOVA was used followed by Tukey–Kramer post test. P<0.05 was considered statistically significant. Statistical tests were performed with R-statistical package (v3.0.2, The R Foundation for Statistical Computing, www.R-project.org), graphs were generated with GraphPad Prism (v6.0e, GraphPad Software, La Jolla, CA, USA), and the final versions of the figures were completed in GIMP ver. 2.8 (GNU Project, www.gimp.org).
Results
Localization of micropellets, cannula and ODN infusion
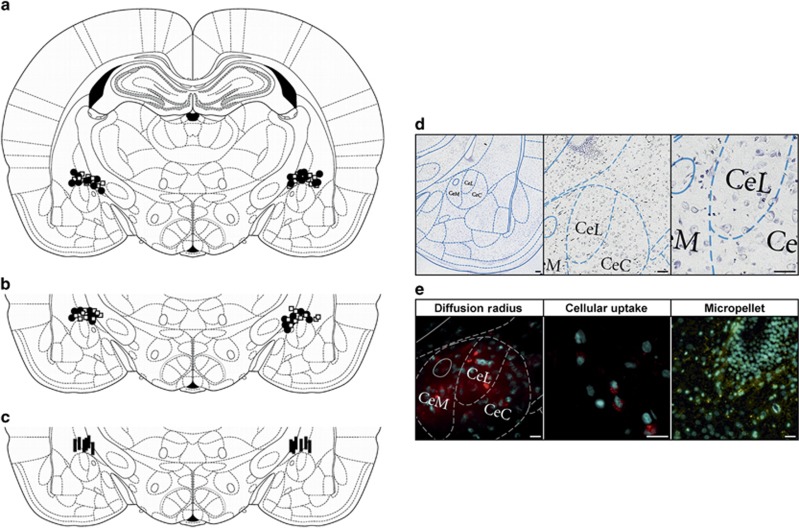
A summary of the localization, based on the rat brain atlas,23 is presented in Figure 4, with Figure 4a representing the 7-day postimplant, Figure 4b illustrating the 28-day postimplant and 1c showing the WAS studies. Nissl staining was performed in a subset of tissues to further verify the integrity of the CeA, with low-to-high magnification images presented in Figure 4d. Fluorescent visualization of tissue from animals infused with Alexa Fluor 594-conjugated ODNs demonstrated that the diffusion radius was restricted to the medial, the lateral and minor portions of the central CeA (Figure 4e). Increased magnification revealed cellular uptake of the ODNs within the neurons of the CeA (Figure 4e). These studies indicated that the surgeries successfully targeted the CeA as well as that the infusion of the ODN construct was limited to CeA.
Figure 4.
Localization of stereotaxic implants, tissue viability and diffusion of ODN. Summary images of micropellet placement on the dorsal margin of the CeA at 7 days (a) or 28 days (b) post implantation surgery and point of cannula termination for stress study (c). Open squares are CHOL implants, filled circles are CORT implants and filled bars indicate the end of the cannula. All pellets or cannula were on the dorsal surface of the CeA between −1.80 and −2.92 mm from bregma. However, the localizations are shown on the bregma −2.5 mm schematic.23 (d) Representative Nissl staining of coronal sections at × 10, × 20 and × 40 (left to right) shows minimal damage to the CeA as a result of stereotaxic implants. (e) Fluorescent micrograph of coronal sections from animals infused with RSO conjugated to Alexa Fluor 549 show diffusion radius ( × 20) and cellular uptake ( × 40) of the ODN. As an additional verification of tissue integrity, the third panel shows a DAPI stain of the tissue immediately surrounding a micropellet ( × 20). Scale bar, 150 μm in all the images. CeA, central amygdala; CHOL, cholesterol; CORT, corticosterone; DAPI, 4',6-diamidino-2-phenylindole; ODN, oligodeoxynucleotide; RSO, random sense oligodeoxynucleotide.
Effect of ODN on CRF expression in the CeA
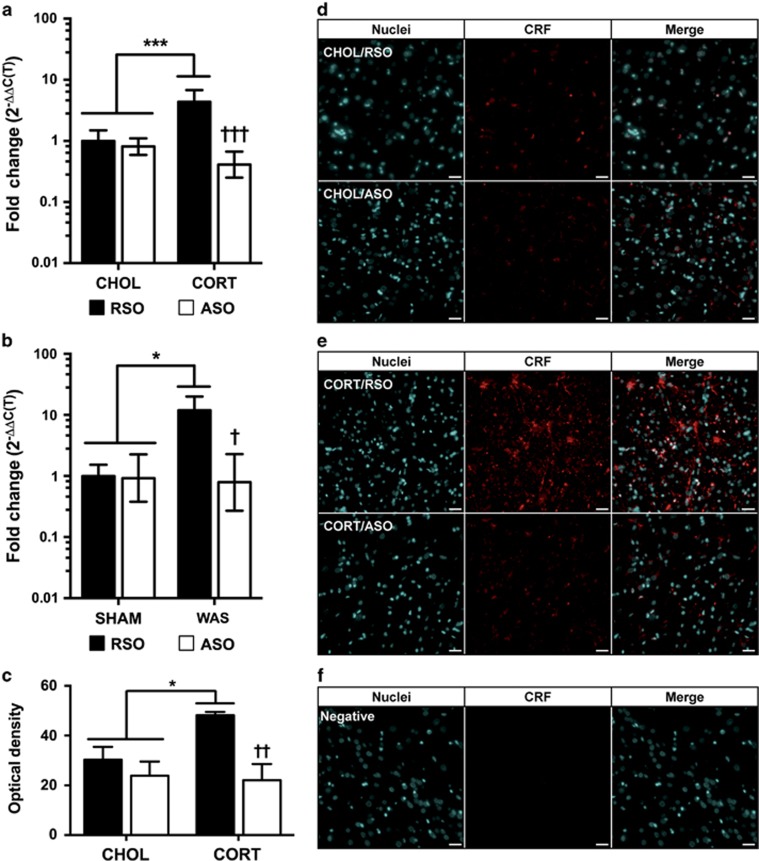
The amount of knockdown of CRF by the ASO used in this study has been previously reported.19 As we have previously quantified the increase in CRF mRNA in both models,15, 17, 22 we initially used quantitative reverse transcriptase PCR to validate CRF knockdown in this study. As shown in Figure 5a, there were significant main effects for CeA implant (F(1,17)=4.53, P=0.048) and ODN treatment (F(1,17)=49.58, P<0.001) on CRF mRNA expression, as well as a significant interaction between the terms (F(1,17)=34.34, P<0.001) at 7 days post implantation. Post hoc analysis confirmed that CRF mRNA was increased in the CORT+RSO group (4.4-fold increase compared with CHOL+RSO) and that ASO significantly knocked down CRF in the rats with CORT implants (59% decrease from CHOL+RSO, 90% less than CORT+RSO), whereas not significantly changing expression in CHOL-implanted rats. As shown in Figure 5b, a similar effect was found with WAS-induced changes in CRF mRNA expression. There were significant main effects for stress exposure (F(1,8)=6.77, P=0.032), ODN treatment (F(1,8)=9.81, P=0.014), and the stress exposure:ODN treatment interaction (F(1,8)=8.73, P=0.002). Post tests demonstrated that CRF mRNA expression was significantly elevated in the WAS+RSO group (11.9-fold greater than SHAM+RSO) and that ASO significantly knocked down CRF mRNA in the rats exposed to WAS (21% less than SHAM+RSO, 93% less than WAS+RSO), without changing the expression in the SHAM+RSO group. To determine whether the change in mRNA produced a functional change in protein expression and to compare the knockdown in our study with the published literature,19 immunohistochemistry was used to verify the specificity of the CRF expression to the CeA in the 28-day postimplantation series. As illustrated in Figure 5c, quantification of the optical density demonstrated a marginal effect of CeA implant (F(1,8)=5.08, P=0.054), and significant effects of ODN treatment (F(1,8)=23.02, P=0.014) and the interaction term (F(1,8)=8.52, P=0.019), after the depletion of the CORT micropellet. There was a significant increase in CRF immunofluorescence in rats that previously had a CORT implant and were subsequently treated with RSO (1.6-fold increase compared with CHOL+RSO) that was comparable to our previous study quantifying mRNA.15 The CRF signal was significantly reduced following the ASO treatment (56% lower than CORT+RSO) to levels that were identical to the CHOL+ASO treatment. Representative images of the quantified CRF immunofluorescence for each treatment group are shown in Figures 5d and e, and the specificity of the CRF antibody is shown in Figure 5f. Overall, the efficiency of knockdown was similar to the levels previously reported using the same ODN infused into the CeA.19, 27
Figure 5.
Knockdown of CRF expression in the CeA with ASO treatment. (a) CRF mRNA from CeA micropunches following treatment with RSO or ASO at 7 days post implantation (n=3–6 per group). (b) CRF mRNA from CeA micropunches following treatment with RSO or ASO after SHAM or WAS exposure (n=3 per group). (c) Quantification of CRF immunofluorescence in the CeA following RSO or ASO treatment 28 days post implantation (n=3 per group). (d and e) Representative fluorescent micrographs at × 20 following immunofluorescent detection of CRF in animals 28 days post CHOL or CORT implantation treated with RSO or ASO, respectively. (f) The × 20 image of the negative control for immunofluorescence. Scale bar, 150 μm in all the images. Data shown are mean±s.d. *P<0.05, ***P<0.001 compared with CHOL/SHAM+RSO and †P<0.05, ††P<0.01 compared with CORT/WAS+RSO by two-factor analysis of variance with Tukey–Kramer post hoc analysis. ASO, antisense oligodeoxynucleotide; CeA, central amygdala; CHOL, cholesterol; CORT, corticosterone; CRF, corticotropin-releasing factor; mRNA, messenger RNA; ODN, oligodeoxynucleotide; RSO, random sense oligodeoxynucleotide; WAS, water avoidance stress.
Effect of knockdown of CRF on day 7 post implant
The first series of experiments evaluated the effect of knockdown of CRF in the CeA in a model of somatic and visceral sensitivity induced by constant exposure of the CeA to elevated CORT via stereotaxically placed micropellets.8 The initial study evaluated the effect of ODN on both somatic withdrawal threshold and visceral sensitivity to colonic balloon distension, as illustrated in the design presented in Figure 1a. For somatic withdrawal threshold (Figure 1b), there were significant main effects for CeA implant (F(1,31)=43.26, P<0.001), ODN treatment (F(1,31)=20.42, P<0.001) and the interaction (F(1,31)=32.63, P<0.001). Post hoc testing revealed that CORT-implanted rats treated with RSO had a significantly reduced withdrawal threshold compared with CHOL+RSO, which was normalized in the CORT+ASO group. The visceral sensitivity data shown in Figure 1c demonstrated significant main effects of CeA implant (F(1,31)=51.49, P<0.001) and ODN treatment (F(1,31)=5.73, P=0.023), without a significant interaction (F(1,31)=1.87, P=0.182). The main effect of the repeated measure, pressure, was also significant (F(3,93)=690.1, P<0.001), as were the interactions of CeA implant:pressure (F(3,93)=18.83, P<0.001) and ODN treatment:pressure (F(3,93)=3.13, P=0.029). Post hoc comparisons determined that the number of abdominal contractions in response to colonic distension was significantly reduced with ASO treatment in the CORT-implant group compared with the CORT+RSO group at all distension pressures.
To determine whether the effects of ODN treatment were reversible, a subset of rats were used in a crossover study as shown in the experimental design in Figure 1d. The responses to somatic withdrawal threshold of RSO or ASO treatment in the same animals are as demonstrated in Figure 1e. The ANOVA modeling revealed significant main effects for CeA implant (F(1,5)=28.45, P=0.003), ODN treatment (F(1,5)=38.49, P=0.002) and the interaction term (F(1,5)=45.60, P=0.001). As with the initial study, the CORT-implanted rats treated with RSO exhibited a significantly decreased withdrawal threshold that was reversed with ASO treatment. The effects of ASO treatment on visceral sensitivity were also reversible as demonstrated by significant main effects for CeA implant (F(1,5)=13.84, P=0.014), ODN treatment (F(1,20)=51.01, P<0.001) and pressure (F(3,15)=590.8, P<0.001). There were also significant interaction terms for the crossover study: CeA implant:ODN treatment (F(1,20)=42.38, P<0.001), CeA implant:pressure (F(3,20)=5.00, P=0.009), ODN treatment:pressure (F(3,20)=5.00, P=0.009), and CeA implant:ODN treatment:pressure (F(3,20)=4.14, P=0.019). Although Figure 1e illustrates only the post hoc comparison of the response to 60 mm Hg distension for each ODN treatment in the same animal, the responses to 20 mm Hg and 40 mm Hg were also significantly decreased in the CORT-implanted rats treated with ASO (P<0.001 for each pressure).
Effect of knockdown of CRF on day 28 post implant
The next series of experiments aimed to evaluate the effect of CRF knockdown in the CeA in a model of persistent somatic and visceral sensitivity that occurs despite the dissolution of the CORT implant.14 A similar experimental design was used (Figure 2a). There was a persistent change in somatic sensitivity as demonstrated by significant main effects for CeA implant (F(1,18)=36.55, P<0.001) and CeA implant:ODN treatment (F(1,18)=17.44, P<0.001), without a significant effect of ODN treatment (F(1,18)=1.91, P=0.184) due to the mixed treatment effects in the ANOVA model. As shown in Figure 2b, post hoc comparisons confirmed that a significant decrease in somatic withdrawal threshold was maintained in the CORT+RSO group, compared with the CHOL+RSO. ASO infusion in the rats that had previously received CORT implants normalized the withdrawal response. Evaluating the response to colonic distension indicated significant main effects for CeA implant (F(1,18)=15.37, P=0.001), ODN treatment (F(1,18)=6.16, P=0.023) and the CeA implant:ODN treatment interaction (F(1,18)=7.34, P=0.014). The repeated measure of pressure was also significant (F(3,54)=482.0, P<0.001), along with the interaction term of CeA implant:pressure (F(3,54)=5.95, P=0.001). Figure 2c illustrates the post hoc analysis of the responses to each distension pressure. Colonic sensitivity was not affected by ODN treatment in CHOL-implanted rats, whereas a significant colonic hyperalgesia to distension was measured in the CORT+RSO group that was completely inhibited with ASO treatment.
In a subset of rats, we used a crossover design (Figure 2d) for our next series of experiments in which we investigated whether the effect of ODN was reversible. ANOVA modeling indicated main effects for CeA implant (F(1,10)=26.64, P<0.001), ODN treatment (F(1,10)=29.98, P<0.001), and the interaction (F(1,10)=26.45, P<0.001). Figure 2e illustrates the post hoc analysis of somatic sensitivity, which confirms that the decreased somatic withdrawal threshold in the CORT+RSO group was inhibited in the same rats when treated with ASO. For visceral sensitivity, there were significant effects for CeA implant (F(1,10)=5.14, P=0.047), ODN treatment (F(1,40)=20.31, P<0.001), pressure (F(3,30)=416.7, P<0.001), CeA implant:pressure (F(3,30)=3.29, P=0.034), and ODN treatment:pressure (F(3,40)=3.10, P=0.038). Post hoc analysis of the intra-animal effects of RSO or ASO in the CORT-implanted rats revealed significant decreases in sensitivity at 40 and 60 mm Hg distension, with the 60 mm Hg response shown in Figure 2e.
Effect of knockdown of CRF following WAS
The final series of experiments used a model of repeated psychological stress, WAS, to investigate whether CRF knockdown in the CeA could inhibit stress-induced somatic and visceral nociceptive behaviors. The experimental design is shown in Figure 3a. To ensure that the stress exposure consistently activated the HPA axis, fecal pellet output in response to WAS or SHAM was measured daily as shown in Figure 3b. There was the expected main effect of stress exposure (F(1,19)=446.5, P<0.001), but neither ODN treatment (F(1,19)=0.49, P=0.493) nor the day of stress exposure (F(6,114)=1.01, P=0.43) affected the fecal pellet output. Thus, WAS produced the predicted increase in fecal pellet output, without habituation and was not affected by ODN treatment. Stress-induced somatic sensitivity was demonstrated by significant main effects for stress exposure (F(1,19)=115.5, P<0.001), ODN treatment (F(1,19)=107.6, P<0.001) and the interaction term (F(1,19)=73.93, P<0.001). As shown in Figure 3c, a decreased somatic withdrawal threshold was observed in the WAS+RSO group compared with the SHAM+RSO. In the WAS+ASO group, the stress-induced somatic hypersensitivity was inhibited (Figure 3c). The analysis of the response to colonic distension indicated significant main effects for stress exposure (F(1,17)=46.07, P<0.001), ODN treatment (F(1,17)=88.70, P<0.001) and pressure (F(3,51)=565.4, P<0.001). There were significant interaction terms for stress exposure:ODN treatment (F(1,17)=56.99, P<0.001), stress exposure:pressure (F(3,51)=6.48, P<0.001), ODN treatment:pressure (F(3,51)=16.18, P<0.001) and stress exposure:ODN treatment:pressure (F(3,51)=8.08, P<0.001). Figure 3d illustrates that the response to colonic distension in the SHAM+RSO and SHAM+ASO groups were identical, whereas a significant colonic hypersensitivity to distension was measured in the WAS+RSO, which was inhibited in the WAS+ASO group.
Discussion
Our previous studies demonstrated that targeted exposure of the CeA for 7 days to elevated levels of CORT contained in bilaterally placed micropellets induced visceral and somatic nociceptive behaviors.11, 13, 14 Importantly, the visceral and somatic hypersensitivity persisted concomitantly with a sustained increase in CRF expression in the CeA15 despite dissipation of the CORT-containing micropellets.11, 13, 14 In addition, we demonstrated a similar increase in visceral sensitivity to colonic distension along with an increase in CRF expression in the CeA following repeated exposure to WAS.16, 17 However, the causal relationship between enhanced CRF expression in the CeA and the nociceptive behaviors remained to be established. In the current study, we directly investigated the relationship between the sustained increase in CRF expression in the CeA and the persistent nociceptive behaviors. Specifically, we knocked down CRF expression induced by elevated CeA CORT with targeted antisense ODN and then examined visceral and somatic nociceptive behaviors at 7 and 28 days post CeA manipulation or following WAS. Our experiments provide strong evidence for the effectiveness of the ASO for reducing expression of CRF in each experimental manipulation. At 7 days post implantation, blocking CRF with ASO prevented the decrease in somatic withdrawal threshold and significantly inhibited visceral hypersensitivity, verifying that CRF within the CeA is a pivotal mediator of colonic and somatic hypersensitivity. At 28 days post implantation, after the complete diffusion of the CORT micropellet, knocking down the persistent increase in CRF with ASO normalized both somatic and visceral sensitivity, demonstrating that CRF in the CeA is necessary for the maintenance of nociceptive behaviors. In addition, the effects of ODN treatment on somatic and visceral nociceptive behaviors were completely reversible at both 7 and 28 days post implant. Similarly, in a model of repeated stress, knocking down CRF in the CeA was also able to produce a significant decrease in somatic and visceral nociceptive behaviors. In total, these studies provide compelling evidence to support a role of CRF in the CeA in both the initiation and maintenance of stress-associated nociceptive behaviors.
To our knowledge, this is the first study to directly examine the effect of CRF expression on nociceptive behaviors in models of targeted and generalized CeA exposure to CORT. We explored the relationship between CORT and CRF in the CeA by showing that targeted knockdown of CRF expression in the CeA attenuates the CORT- and stress-induced nociceptive behaviors. In addition, our experiments advanced these observations by showing in long-term studies that the involvement of CRF is pivotal to maintaining the exaggerated nociceptive phenotypes induced by a transient exposure of the CeA to elevated levels of CORT. Although this is the first study to directly investigate the role of CRF expression in the CeA on nociceptive behaviors, other studies have demonstrated that CRF expression in the amygdala increases in response to elevated systemic CORT,7, 8, 15, 28, 29 tissue injury30, 31 or psychological stress.32, 33 In addition, exogenous CRF can sensitize somatic and visceral nociceptive responses when directly infused into the amygdala.34, 35
Our manipulation of the CeA with a CORT micropellet produced an increase in CRF expression that was maintained throughout the duration of study. On the basis of the evidence provided by the ASO treatment, we can conclude that the increased CRF expression induced a decrease in somatic withdrawal threshold, suggestive of a mechanical allodynia, and an increase in visceral sensitivity to colonic distension, supporting the role of CRF expression in the CeA in nociceptive behaviors. Infusion of ASO not only decreased expression of CRF, but also restored somatic and colonic sensitivity to values that were identical to control treatments. Following washout of the ASO treatment, somatic and visceral nociceptive behaviors returned, illustrating the reversibility of the ASO treatment. Although not directly tested in this study, we can speculate on the specificity of the CRF knockdown for the CeA. We have previously demonstrated that CORT implants onto the dorsal margins of the caudate, the ventral hippocampus or the anterolateral bed nucleus of the stria terminals (BNST) failed to induce visceral hypersensitivity to colonic distension.21, 36 However, CORT implants on the CeA increased expression of CRF in not only the CeA, but also the BNST and the paraventricular nucleus of the hypothalamus at 7 days post implantation.8, 15, 21, 29, 37 The goal of future studies will be to evaluate the effect of CRF ASO in other nuclei that respond with increased CRF expression due to targeting the CeA with CORT micropellets.
Although few studies have examined visceral pain, our current results are supported by recent studies of the role of CRF in amygdala-mediated mechanical allodynia. First, direct administration of CRF into the CeA produced an acute decrease in hindpaw withdrawal threshold to mechanical stimulation in healthy rats,38 an effect dependent on the type-1 CRF receptor (CRF1). A second study used a CRF binding protein antagonist to increase CRF within the CeA in a model of neuropathic pain, which produced significant decreases in mechanical threshold in both the injured and uninjured limbs.39 A third study, using a model of nicotine withdrawal, demonstrated both an increase in CRF and CRF1 mRNA expression in the CeA and a decrease in withdrawal threshold that was CRF1 mediated.40 Thus, there is convergent evidence that CRF in the CeA modulates acute pain through CRF1, which is expanded upon by our study in which we have demonstrated a role for CRF in both CORT- and stress-induced nociceptive behaviors.
Conversely, a role for CRF-induced antinociception has been suggested on the basis of anatomical studies that found co-localization of CRF and dynorphin terminals within the CeA, BNST,41 dorsal raphe42 and locus coeruleus,43 thus demonstrating a limbic circuit that may be used in response to an acute threat. As predicted by this circuit, multiple studies have shown increased thresholds to nociceptive behaviors in nonstressed animals after exogenous administration of CRF, such as: increased withdrawal threshold to tail shock,44, 45 and increased mechanical and thermal withdrawal thresholds.46 However, the key difference in those studies compared with our current findings was site specificity. We recently demonstrated in the extended amygdala that the net effect of CRF is region specific due to the differential distribution of CRF1 and the type-2 (CRF2) receptors.22 Our present findings also support a mechanism by which prolonged exposure of the CeA to elevated CORT induced stable reprogramming of CRF gene expression through histone deacetylation of the CRF promoter.47 Thus, with regard to nociceptive behaviors induced by CORT in the CeA, interventions targeting mechanisms that regulate CRF may potentially be more efficacious than CRF receptor antagonists.
Our finding that repeated WAS-induced nociceptive behaviors were inhibited by knocking down CRF in the CeA provides additional evidence implicating CRF in the CeA in the development of stress-induced nociceptive behaviors. The lack of effect of ASO on fecal pellet output is in agreement with our previous studies that found only a modest reduction16 or no change in stress-induced fecal pellet output with administration of corticoid receptor antagonists on the CeA, but rather a change in colonic motility immediately after the stress exposure.48 Although there is evidence for a persistent colonic hypersensitivity with 10 days of repeated WAS,49 there is no evidence for a sustained increase in CRF within the CeA with WAS. Thus, a limitation of the current study was an inability to investigate, using a crossover design, the reversibility of the ASO effect because a second, 7-day stress exposure would have been required. Although peripheral administration of CRF1 receptor antagonists have been shown to inhibit repeated WAS-induced visceral hypersensitivity,50 the site of action of the CRF1 antagonist effect remains unclear. Moreover, although this current study does not exclude a role for peripheral CRF mechanisms in WAS-induced nociceptive behaviors, the targeted administration of CRF ASO to the CeA indicates that central expression of CRF is sufficient to induce somatic and visceral nociceptive behaviors. In addition, similar to the CORT-implant model, we have recently shown that WAS also induces an increase in CRF expression in the anterolateral BNST.22 In that study, both CRF1 and CRF2 antagonists infused directly into the anterolateral BNST inhibited both somatic and visceral hypersensitivity,22 providing a rationale for investigating the effect of CRF ASO in the BNST in future studies. Although the mechanism by which CRF in the CeA modulates WAS-induced nociceptive behaviors was not determined in the current study, we have previously demonstrated that the WAS-induced increase in CRF expression in the CeA was associated with de-methylation of the CRF promoter and that intracerebroventricular administration of a histone deacetylase inhibitor blocked visceral hypersensitivity.17 Recent literature also provides evidence for epigenetic changes in the dorsal root ganglia that innervate the colon following WAS.51 Thus, future studies will further investigate the role of epigenetic mechanisms responsible for WAS-induced nociceptive behaviors.
In conclusion, our studies have demonstrated that both targeted and stress-induced elevation of CORT in the CeA induces persistent visceral and somatic nociceptive behaviors through an increase in CRF expression. Inhibiting the increase in CRF expression using targeted antisense oligodeoxynucleotides attenuated the visceral and somatic nociceptive phenotypes induced by elevated amygdala CORT. The results of our studies suggest that the augmented CRF expression in the CeA is required to induce nociceptive behaviors following a stressor, which likely represents an important factor to consider when developing therapies and interventions for chronic stress-related conditions such as IBS.
Acknowledgments
BG-VM acknowledges the generous funding support for her Research Career Scientist and Merit Review Awards from the Department of Veterans Affairs [I01BX002188]. ACJ acknowledges the funding support from a predoctoral fellowship from the National Institutes of Health National Institute of Diabetes and Digestive and Kidney Diseases [F31DK089871]. Imaging support provided by the Oklahoma Medical Research Foundation Imaging Core Facility.
The authors declare no conflict of interest.
References
- Longstreth GF, Thompson WG, Chey WD, Houghton LA, Mearin F, Spiller RC. Functional bowel disorders. Gastroenterology. 2006;130:1480–1491. doi: 10.1053/j.gastro.2005.11.061. [DOI] [PubMed] [Google Scholar]
- Drossman DA. The functional gastrointestinal disorders and the Rome III process. Gastroenterology. 2006;130:1377–1390. doi: 10.1053/j.gastro.2006.03.008. [DOI] [PubMed] [Google Scholar]
- Kim SE, Chang L. Overlap between functional GI disorders and other functional syndromes: what are the underlying mechanisms. Neurogastroenterol Motil. 2012;24:895–913. doi: 10.1111/j.1365-2982.2012.01993.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mayer EA, Naliboff BD, Chang L, Coutinho SV. V. Stress and irritable bowel syndrome. Am J Physiol Gastrointest Liver Physiol. 2001;280:G519–G524. doi: 10.1152/ajpgi.2001.280.4.G519. [DOI] [PubMed] [Google Scholar]
- Wilder-Smith CH. The balancing act: endogenous modulation of pain in functional gastrointestinal disorders. Gut. 2011;60:1589–1599. doi: 10.1136/gutjnl-2011-300253. [DOI] [PubMed] [Google Scholar]
- McEwen BS. Protective and damaging effects of stress mediators: central role of the brain. Dialogues Clin Neurosci. 2006;8:367–381. doi: 10.31887/DCNS.2006.8.4/bmcewen. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schulkin J, Gold PW, McEwen BS. Induction of corticotropin-releasing hormone gene expression by glucocorticoids: implication for understanding the states of fear and anxiety and allostatic load. Psychoneuroendocrinology. 1998;23:219–243. doi: 10.1016/s0306-4530(97)00099-1. [DOI] [PubMed] [Google Scholar]
- Shepard JD, Barron KW, Myers DA. Stereotaxic localization of corticosterone to the amygdala enhances hypothalamo-pituitary-adrenal responses to behavioral stress. Brain Res. 2003;963:203–213. doi: 10.1016/s0006-8993(02)03978-1. [DOI] [PubMed] [Google Scholar]
- Dinan TG, Quigley EM, Ahmed SM, Scully P, O'Brien S, O'Mahony L, et al. Hypothalamic-pituitary-gut axis dysregulation in irritable bowel syndrome: plasma cytokines as a potential biomarker. Gastroenterology. 2006;130:304–311. doi: 10.1053/j.gastro.2005.11.033. [DOI] [PubMed] [Google Scholar]
- Riedel W, Schlapp U, Leck S, Netter P, Neeck G. Blunted ACTH and cortisol responses to systemic injection of corticotropin-releasing hormone (CRH) in fibromyalgia: role of somatostatin and CRH-binding protein. Ann N Y Acad Sci. 2002;966:483–490. doi: 10.1111/j.1749-6632.2002.tb04251.x. [DOI] [PubMed] [Google Scholar]
- Greenwood-Van Meerveld B, Gibson M, Gunter W, Shepard J, Foreman R, Myers D. Stereotaxic delivery of corticosterone to the amygdala modulates colonic sensitivity in rats. Brain Res. 2001;893:135–142. doi: 10.1016/s0006-8993(00)03305-9. [DOI] [PubMed] [Google Scholar]
- Myers DA, Gibson M, Schulkin J, Greenwood Van-Meerveld B. Corticosterone implants to the amygdala and type 1 CRH receptor regulation: effects on behavior and colonic sensitivity. Behav Brain Res. 2005;161:39–44. doi: 10.1016/j.bbr.2005.03.001. [DOI] [PubMed] [Google Scholar]
- Myers B, Dittmeyer K, Greenwood-Van Meerveld B. Involvement of amygdaloid corticosterone in altered visceral and somatic sensation. Behav Brain Res. 2007;181:163–167. doi: 10.1016/j.bbr.2007.03.031. [DOI] [PubMed] [Google Scholar]
- Myers B, Greenwood-Van Meerveld B. Elevated corticosterone in the amygdala leads to persistent increases in anxiety-like behavior and pain sensitivity. Behav Brain Res. 2010;214:465–469. doi: 10.1016/j.bbr.2010.05.049. [DOI] [PubMed] [Google Scholar]
- Tran L, Greenwood-Van Meerveld B. Altered expression of glucocorticoid receptor and corticotropin-releasing factor in the central amygdala in response to elevated corticosterone. Behav Brain Res. 2012;234:380–385. doi: 10.1016/j.bbr.2012.07.010. [DOI] [PubMed] [Google Scholar]
- Myers B, Greenwood-Van Meerveld B. Differential involvement of amygdala corticosteroid receptors in visceral hyperalgesia following acute or repeated stress. Am J Physiol Gastrointest Liver Physiol. 2012;302:G260–G266. doi: 10.1152/ajpgi.00353.2011. [DOI] [PubMed] [Google Scholar]
- Tran L, Chaloner A, Sawalha AH, Greenwood Van-Meerveld B. Importance of epigenetic mechanisms in visceral pain induced by chronic water avoidance stress. Psychoneuroendocrinology. 2013;38:898–906. doi: 10.1016/j.psyneuen.2012.09.016. [DOI] [PubMed] [Google Scholar]
- Johnson AC, Tran L, Schulkin J, Greenwood-Van Meerveld B.Importance of stress receptor-mediated mechanisms in the amygdala on visceral pain perception in an intrinsically anxious rat Neurogastroenterol Motil 201224479–486.e219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pitts MW, Todorovic C, Blank T, Takahashi LK. The central nucleus of the amygdala and corticotropin-releasing factor: insights into contextual fear memory. J Neurosci. 2009;29:7379–7388. doi: 10.1523/JNEUROSCI.0740-09.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Myers B, Greenwood-Van Meerveld B. Divergent effects of amygdala glucocorticoid and mineralocorticoid receptors in the regulation of visceral and somatic pain. Am J Physiol Gastrointest Liver Physiol. 2010;298:G295–G303. doi: 10.1152/ajpgi.00298.2009. [DOI] [PubMed] [Google Scholar]
- Tran L, Wiskur B, Greenwood-Van Meerveld B. The role of the anteriolateral bed nucleus of the stria terminalis in stress-induced nociception. Am J Physiol Gastrointest Liver Physiol. 2012;302:G1301–G1309. doi: 10.1152/ajpgi.00501.2011. [DOI] [PubMed] [Google Scholar]
- Tran L, Schulkin J, Greenwood-Van Meerveld B. Importance of CRF receptor-mediated mechanisms of the bed nucleus of the stria terminalis in the processing of anxiety and pain. Neuropsychopharmacology. 2014;39:2633–2645. doi: 10.1038/npp.2014.117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paxinos G, Watson C.The Rat Brain in Stereotaxic Coordinates6th edn. Academic Press/Elsevier: Amsterdam, The Netherlands/Boston, MA, USA; 2007 [Google Scholar]
- Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods. 2001;25:402–408. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
- Vandesompele J, De Preter K, Pattyn F, Poppe B, Van Roy N, De Paepe A, et al. Accurate normalization of real-time quantitative RT-PCR data by geometric averaging of multiple internal control genes. Genome Biol. 2002;3:RESEARCH0034. doi: 10.1186/gb-2002-3-7-research0034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Landgraf R, Naruo T, Vecsernyes M, Neumann I. Neuroendocrine and behavioral effects of antisense oligonucleotides. Eur J Endocrinol. 1997;137:326–335. doi: 10.1530/eje.0.1370326. [DOI] [PubMed] [Google Scholar]
- Pitts MW, Takahashi LK. The central amygdala nucleus via corticotropin-releasing factor is necessary for time-limited consolidation processing but not storage of contextual fear memory. Neurobiol Learn Mem. 2011;95:86–91. doi: 10.1016/j.nlm.2010.11.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Makino S, Gold PW, Schulkin J. Corticosterone effects on corticotropin-releasing hormone mRNA in the central nucleus of the amygdala and the parvocellular region of the paraventricular nucleus of the hypothalamus. Brain Res. 1994;640:105–112. doi: 10.1016/0006-8993(94)91862-7. [DOI] [PubMed] [Google Scholar]
- Shepard JD, Barron KW, Myers DA. Corticosterone delivery to the amygdala increases corticotropin-releasing factor mRNA in the central amygdaloid nucleus and anxiety-like behavior. Brain Res. 2000;861:288–295. doi: 10.1016/s0006-8993(00)02019-9. [DOI] [PubMed] [Google Scholar]
- Nishii H, Nomura M, Aono H, Fujimoto N, Matsumoto T. Up-regulation of galanin and corticotropin-releasing hormone mRNAs in the key hypothalamic and amygdaloid nuclei in a mouse model of visceral pain. Regul Pept. 2007;141:105–112. doi: 10.1016/j.regpep.2006.12.022. [DOI] [PubMed] [Google Scholar]
- Ulrich-Lai YM, Xie W, Meij JT, Dolgas CM, Yu L, Herman JP. Limbic and HPA axis function in an animal model of chronic neuropathic pain. Physiol Behav. 2006;88:67–76. doi: 10.1016/j.physbeh.2006.03.012. [DOI] [PubMed] [Google Scholar]
- McNally GP, Akil H. Role of corticotropin-releasing hormone in the amygdala and bed nucleus of the stria terminalis in the behavioral, pain modulatory, and endocrine consequences of opiate withdrawal. Neuroscience. 2002;112:605–617. doi: 10.1016/s0306-4522(02)00105-7. [DOI] [PubMed] [Google Scholar]
- Iwasaki-Sekino A, Mano-Otagiri A, Ohata H, Yamauchi N, Shibasaki T. Gender differences in corticotropin and corticosterone secretion and corticotropin-releasing factor mRNA expression in the paraventricular nucleus of the hypothalamus and the central nucleus of the amygdala in response to footshock stress or psychological stress in rats. Psychoneuroendocrinology. 2009;34:226–237. doi: 10.1016/j.psyneuen.2008.09.003. [DOI] [PubMed] [Google Scholar]
- Ji G, Neugebauer V. Pro- and anti-nociceptive effects of corticotropin-releasing factor (CRF) in central amygdala neurons are mediated through different receptors. J Neurophysiol. 2008;99:1201–1212. doi: 10.1152/jn.01148.2007. [DOI] [PubMed] [Google Scholar]
- Gue M, Del Rio-Lacheze C, Eutamene H, Theodorou V, Fioramonti J, Bueno L. Stress-induced visceral hypersensitivity to rectal distension in rats: role of CRF and mast cells. Neurogastroenterol Motil. 1997;9:271–279. doi: 10.1046/j.1365-2982.1997.d01-63.x. [DOI] [PubMed] [Google Scholar]
- Myers B, Greenwood-Van Meerveld B. Corticosteroid receptor-mediated mechanisms in the amygdala regulate anxiety and colonic sensitivity. Am J Physiol Gastrointest Liver Physiol. 2007;292:G1622–G1629. doi: 10.1152/ajpgi.00080.2007. [DOI] [PubMed] [Google Scholar]
- Shepard JD, Schulkin J, Myers DA. Chronically elevated corticosterone in the amygdala increases corticotropin releasing factor mRNA in the dorsolateral bed nucleus of stria terminalis following duress. Behav Brain Res. 2006;174:193–196. doi: 10.1016/j.bbr.2006.07.019. [DOI] [PubMed] [Google Scholar]
- Ji G, Fu Y, Adwanikar H, Neugebauer V. Non-pain-related CRF1 activation in the amygdala facilitates synaptic transmission and pain responses. Mol Pain. 2013;9:2. doi: 10.1186/1744-8069-9-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bourbia N, Ansah OB, Pertovaara A. Corticotropin-releasing factor in the rat amygdala differentially influences sensory-discriminative and emotional-like pain response in peripheral neuropathy. J Pain. 2010;11:1461–1471. doi: 10.1016/j.jpain.2010.05.004. [DOI] [PubMed] [Google Scholar]
- Baiamonte BA, Valenza M, Roltsch EA, Whitaker AM, Baynes BB, Sabino V, et al. Nicotine dependence produces hyperalgesia: role of corticotropin-releasing factor-1 receptors (CRF1Rs) in the central amygdala (CeA) Neuropharmacology. 2014;77:217–223. doi: 10.1016/j.neuropharm.2013.09.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marchant NJ, Densmore VS, Osborne PB. Coexpression of prodynorphin and corticotrophin-releasing hormone in the rat central amygdala: evidence of two distinct endogenous opioid systems in the lateral division. J Comp Neurol. 2007;504:702–715. doi: 10.1002/cne.21464. [DOI] [PubMed] [Google Scholar]
- Retson TA, Van Bockstaele EJ. Coordinate regulation of noradrenergic and serotonergic brain regions by amygdalar neurons. J Chem Neuroanat. 2013;52:9–19. doi: 10.1016/j.jchemneu.2013.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kravets JL, Reyes BA, Unterwald EM, Van Bockstaele EJ. Direct targeting of peptidergic amygdalar neurons by noradrenergic afferents: linking stress-integrative circuitry. Brain Struct Funct. 2013;220:541–558. doi: 10.1007/s00429-013-0674-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yarushkina NI, Bagaeva TR, Filaretova LP. Analgesic actions of corticotropin-releasing factor (CRF) on somatic pain sensitivity: involvement of glucocorticoid and CRF-2 receptors. Neurosci Behav Physiol. 2009;39:819–823. doi: 10.1007/s11055-009-9212-9. [DOI] [PubMed] [Google Scholar]
- Yarushkina NI, Bagaeva TR, Filaretova LP. Central corticotropin-releasing factor (CRF) may attenuate somatic pain sensitivity through involvement of glucocorticoids. J Physiol Pharmacol. 2011;62:541–548. [PubMed] [Google Scholar]
- Vit JP, Clauw DJ, Moallem T, Boudah A, Ohara PT, Jasmin L. Analgesia and hyperalgesia from CRF receptor modulation in the central nervous system of Fischer and Lewis rats. Pain. 2006;121:241–260. doi: 10.1016/j.pain.2005.12.024. [DOI] [PubMed] [Google Scholar]
- Tran L, Schulkin J, Ligon CO, Greenwood Van-Meerveld B.Epigenetic modulation of chronic anxiety and pain by histone deacetylation Mol Psychiatry 2015. doi: 10.1038/mp.2014.122(in press). [DOI] [PubMed]
- Venkova K, Johnson AC, Myers B, Greenwood-Van Meerveld B. Exposure of the amygdala to elevated levels of corticosterone alters colonic motility in response to acute psychological stress. Neuropharmacology. 2010;58:1161–1167. doi: 10.1016/j.neuropharm.2010.02.012. [DOI] [PubMed] [Google Scholar]
- Bradesi S, Schwetz I, Ennes HS, Lamy CM, Ohning G, Fanselow M, et al. Repeated exposure to water avoidance stress in rats: a new model for sustained visceral hyperalgesia. Am J Physiol Gastrointest Liver Physiol. 2005;289:G42–G53. doi: 10.1152/ajpgi.00500.2004. [DOI] [PubMed] [Google Scholar]
- Larauche M, Bradesi S, Million M, McLean P, Tache Y, Mayer EA, et al. Corticotropin-releasing factor type 1 receptors mediate the visceral hyperalgesia induced by repeated psychological stress in rats. Am J Physiol Gastrointest Liver Physiol. 2008;294:G1033–G1040. doi: 10.1152/ajpgi.00507.2007. [DOI] [PubMed] [Google Scholar]
- Hong S, Zheng G, Wiley JW. Epigenetic regulation of genes that modulate chronic stress-induced visceral pain in the peripheral nervous system. Gastroenterology. 2014;148:148–157. doi: 10.1053/j.gastro.2014.09.032. [DOI] [PMC free article] [PubMed] [Google Scholar]