Abstract
The ability to probe defined neural circuits in awake, freely-moving animals with cell-type specificity, spatial precision, and high temporal resolution has been a long sought tool for neuroscientists in the systems-level search for the neural circuitry governing complex behavioral states. Optogenetics is a cutting-edge tool that is revolutionizing the field of neuroscience and represents one of the first systematic approaches to enable causal testing regarding the relation between neural signaling events and behavior. By combining optical and genetic approaches, neural signaling can be bi-directionally controlled through expression of light-sensitive ion channels (opsins) in mammalian cells. The current protocol describes delivery of specific wavelengths of light to opsin-expressing cells in deep brain structures of awake, freely-moving rodents for neural circuit modulation. Theoretical principles of light transmission as an experimental consideration are discussed in the context of performing in vivo optogenetic stimulation. The protocol details the design and construction of both simple and complex laser configurations and describes tethering strategies to permit simultaneous stimulation of multiple animals for high-throughput behavioral testing.
Keywords: Neuroscience, Issue 95, optogenetics, rodent, behavior, opsin, channelrhodopsin, brain, fiber optics, laser, neural circuits
Introduction
Optogenetics has revolutionized systems-level neuroscience in its search for the neural circuit elements driving normal and disease-relevant behavioral states. The discovery that light-sensitive microbial opsins1 could be functionally expressed in mammalian cells provided the platform for using light to gain unprecedented control of neural activity with high spatial and temporal precision2. Unlike traditional electrophysiological or pharmacological approaches to the manipulation of neural activity, optogenetics allows for the control of specific cell types (based on genetic identify or spatial projection) within heterogeneous populations and at physiologically relevant timescales. The subsequent introduction of a neural-optical interface provided a practical tool for delivery of light to behaving animals3. This has allowed for real-time modulation of defined neural circuits in awake behaving rodents in order to causally test the role of these neural circuits in governance of behavioral states relevant to neurological and psychiatric disease4-6. Optogenetics, therefore, represents a powerful tool for introduction into any laboratory interested in investigating the functional relationship between brain activity and behavioral or physiological measures in animal models.
Successful design and completion of an optogenetic experiment involves various steps and considerations (see Figure 1). The goal of the current protocol is to provide individuals with the tools and components, along with the theoretical and practical knowledge, necessary to perform optogenetic stimulation in awake behaving rodents. Currently, there are two predominant wavelength ranges used to activate microbial opsin channels: in the blue spectra (commonly 473 nm) and green-yellow spectra (commonly 532 or 591 nm). Both lasers and light-emitting diodes (LEDs) can be used as light sources to deliver specific wavelengths of light to brain tissue. The non-coherent light emitted by LEDs, however, makes effective transmission of light difficult when coupling into the small core fibers required for in vivo rodent stimulation. Deciding on the appropriate laser assembly is a crucial initial step and will depend on the intended use of optogenetics in the lab. The current protocol describes two basic configurations that differ in their ease of assembly and use: single pre-coupled lasers and dual laser systems (see Figure 2). Single laser systems that are pre-coupled by the manufacturer are essentially ready-to-go upon arrival with little to no set-up required but have the disadvantage of minimal end-user customization. A dual laser system enables delivery of two different wavelengths down the same fiber. This will become increasingly important with the advent of combinatorial optogenetics whereby different wavelengths can be used to activate/inhibit distinct cell types that are spatially co-localized. This is also essential for use with bi-stable step function opsins where photocurrents are initiated and terminated by blue and yellow light, respectively7,8. Dual laser systems are also customizable as the user can add or remove components (e.g., external shutters, beam filters, inline power meters) from the beam path as required. Due to its versatility, the dual laser set-up is recommended if optogenetics is going to be a continued tool used in the lab. Coupling of the lasers, however, can present a challenge and so a quick, easy, and reliable coupling mechanism is provided in this protocol. Note, this protocol details the assembly of optical components and utilizes patch cords and components that are optimized for step-index multimode fibers with a 200 μm core and a numerical aperture (NA) of 0.22. Different core sizes and NA are available for purchase, however all components should ideally match in terms of core size and NA to avoid light loss at fiber connection points. Alternatively, at a fiber connection, light can pass from a smaller to a larger core size; and/or from a lower-NA to a higher-NA fiber without additional loss.
Tethering strategies are provided that allow for simultaneous stimulation of multiple mice for high-throughput behavioral testing. The protocols provided assume use of chronic implantable fibers for behavioral testing but can be modified for acute stimulation protocols. Acutely implanted fibers are advantageous for combining optogenetic stimulation with pharmacological manipulation, since the same cannula can used to deliver drugs and the tip of an optical fiber to the same location. The use of chronically-implanted fibers is, however, highly recommended for multiple-day behavioral testing as it reduces tissue damage associated with repeated insertion and removal of fibers and increases accuracy in terms of consistent placement of fiber for tissue illumination3. When combined with the tethering configurations described here, behavior can be recorded reliably across multiple days. Indeed, reliable light transmission has been reported months after fiber implantation9 such that chronic stimulation and behavioral testing paradigms can, theoretically, be carried out across multiple days and weeks. Additional notes on hardware components have been added to the protocol to allow the reader choice in the best product that will suit their individual needs, including cost-effective alternatives and products that can be made in-house. Important tips that are useful during setup and implementation are also provided.
Protocol
! CAUTION: This protocol involves the use of class 3b lasers and will require proper training and safety guidelines to be followed. Safety goggles must be worn at all times when operating lasers, with alignment procedures presenting an especially high risk. Contact the laser provider to determine the eyewear that will provide maximal attenuation for a given laser. If available, enroll in an institutional laser safety-training course. Never operate a laser without the appropriate safety eyewear and training.
1. Laser Apparatus Set-up
Where appropriate, steps in Section 1 are designated as (A) or (B) to differentiate between single or dual laser systems, respectively.
Attach and secure lasers to the breadboard. Breadboards are excellent heat conductors and act as a heat sink to prevent damage to internal laser components with prolonged use.
(A) Secure the pre-coupled laser to a 10” x 12” breadboard (or as required) using ¼-20” cap screws and washers (Figure 2A). If breadboard holes do not align with laser mounting holes, use small ‘table clamps’ to secure laser to breadboard.
(B) If the 2 lasers to be used have very different beam heights (>~1 cm), use small 4” x 6” breadboards to create a platform for one of the lasers. Attach these boards to the main large 12” x 18” breadboard using ¼-20” cap screws with washers, then attach the laser to the smaller boards using cap screws or table clamps as shown in Figure 2B. Attach the other laser directly to the breadboard using cap screws or a variable height table clamp. Critical Step: Breadboards, screws, and optical components can be purchased as imperial or metric so be consistent when purchasing items; the default for this protocol is imperial.
(A)Attach a thick-jacketed flat-cleave/physical contact (FC/PC) patch cord to the coupler (referred to as a coupler cord; see Figure 3) that is physically attached to the front of the laser (Figure 2A).
(B) Thread the coupler onto a ¾” optical post, then epoxy the joint between the coupler and the top of the post using J-B Kwik, or similar epoxy, to prevent loosening and misalignment during use. Attach the post to the breadboard (as the breadboard holes will not always line up with the required placement of optical components, a post holder, pedestal base adapter, and clamping fork are used to secure the coupler’s optical post in place). Attach a thick-jacketed patch cord (coupler cord) to the back of the coupler.
(B)Insert the first steering mirror for the blue laser into the kinematic holder, and attach it to the breadboard using a ¾” optical post. Attach this post to a base adapter and clamping fork. Position the clamping fork and post assembly directly in front of the blue laser with mirror angled at 45° to steer the laser towards the dichroic mirror. Use the grid pattern of holes on the breadboard as a rough alignment guide. Once roughly positioned, use a ¼”-20 cap screw to secure the clamping fork to the breadboard (see Figures 2B,C).
(B)Insert the dichroic mirror into a kinematic holder and attach it to a 1” optical post and secure directly to the breadboard. Position the dichroic mirror to the far left of, and in line with, the blue laser mirror. Angle the dichroic mirror at a 45° angle such that blue light reflected off the first mirror is reflected into the coupler, then tighten the screw attaching the kinematic holder to the post (see Figures 2B,C).
(B) Attach the first steering mirror for the yellow laser to a ¾” optical post. Attach the optical post to a base adapter and clamping fork. Position the clamping fork and post-ensemble directly in front of the yellow laser and angle the mirror at a 45° angle such that yellow light will be directed towards the second steering mirror. Secure the clamping fork in place with a ¼”-20 cap screw and washer.
(B) Attach the second steering mirror for the yellow laser to a 1” optical post. Angle the mirror such that the yellow light beam from the first mirror will be reflected through the dichroic and into the coupler (Figure 2C). Secure the post directly to the breadboard and tighten the mounting screw once the mirror is angled appropriately. Fine mirror adjustments will be made using the kinematic mirror mounts in a later step.
(B) Attach a neutral density filter wheel to a ¾” optical post and place the post into a post holder attached to a mounting base. Secure the ensemble to the breadboard between the first and second yellow mirrors using a single ¼”-20 cap screw. This wheel is used to adjust the power of yellow laser light reaching the coupler. Tip: Individually tighten all components (such as the screws holding kinematic mirror holders to the top of posts, and the threads holding base adapters to the bottom of posts) before attaching them to the base. Use a shaft of a small hex wrench in the provided through-holes in optical posts to get enough torque. This will prevent components coming loose during use, necessitating realignment.
Attach an FC/PC to FC/PC L-bracket adapter to the breadboard.
Optional: Secure a 1 x 2 50/50 mini cube fiber splitter directly to the breadboard for simultaneous in vivo stimulation of two or more animals. Additionally, handles can be added to assist in movement of breadboard assembly (as seen in Figure 2A).
2. Laser Coupling (Non-contact Style Coupling)
This section pertains to the dual-laser set-up (Figure 2B). Align the inner blue laser path before aligning the outer yellow laser path.
! CAUTION: Use a low light power coupling (~1 mW) to ensure eye safety. Wear safety goggles to power on the laser and until light intensity is measured and deemed to be safe.
Set the switches on the back of the laser to “Curr” (current) and transistor-transistor logic mode (TTL)+ for constant illumination (as opposed to Analog mode). Make sure that the power knob on the front of the driver is set at zero. Turn on the laser by turning on the driver first and then the laser key.
Slowly adjust the power knob located on the front of the laser driver so that ~1 mW laser light is being emitted. Wait 10 - 15 min (or as specified by manufacturer) for laser to warm up.
Connect the fiber optic cable tester directly to the free end of the coupler patch cord and turn on the cable tester (Figure 3A). Adjust the angle of the coupler so that the red beam travels straight back towards the center of the dichroic mirror. The beam path of the red light emitted from the cable tester is the exact path that the incoming laser light will need to follow in order to be coupled into the laser.
Perform a coarse alignment: Use the lateral and horizontal knobs on the kinematic mirrors to steer the beam of laser light into the coupler. The pedestal clamps may need to be loosened to slightly reposition the mirrors and the coupler. The kinematic mounts should still have some travel available for further fine adjustments. Do not be concerned if no blue light is being emitted out of the coupler-attached patch cord at this time.
Place a single piece of semi-translucent paper directly in front of the dichroic mirror, in between the dichroic and coupler. There will be both a blue and a red dot on this paper from the laser and the cable tester, respectively. Use paper that is translucent enough to see both the red and blue spots simultaneously from the same side of the paper.
Make fine adjustments to the first steering mirror (i.e., the one closer to the laser, not the dichroic) by carefully adjusting the lateral and horizontal knobs to align the center of the red dot with the blue dot.
Move the paper back towards the coupler so that it is directly in front of the coupler and adjust the knobs on the second (i.e., dichroic) mirror to align the laser beam with the red beam.
Iterate over steps 2.6 & 2.7 until the center of the blue/yellow and red beams are exactly aligned in both positions (i.e., until the red and blue beams are colinear).
Remove the cable tester from the coupler cord. Laser light should now be emitted from the end of the coupler patch cord.
Determine coupling efficiency by measuring the light power emitted from the fiber tip of the coupler patch cord using a power meter. Use the 500 mW setting on the power meter’s photodiode and change the wavelength setting (λ) to blue (473 nm) or yellow (635 nm) spectrum light depending on the laser being used.
Place the fiber tip perpendicular to the photodiode to obtain a power reading. Compare the light power entering the coupler to the light power emitted from the fiber end. A coupling efficiency of >80% is considered very good. Very small further adjustments of the second steering mirror can sometimes slightly improve coupling. In general, when the beam pattern from the end of the coupling fiber is a small, tight, central spot (with no rings surrounding it), coupling efficiency into the fiber core is optimal.
Repeat steps 2.1 - 2.11 for yellow laser coupling, except use the two steering mirrors for the yellow laser (see Figure 2C). Do not adjust the position of the dichroic mirror or alignment of the blue laser will be lost.
3. In vivo Optogenetic Stimulation
Ensure that any procedures involving animal use are conducted in accordance with local and national guidelines and approved by the corresponding Institutional Animal Care and Use Committee.
Optic fiber set-up (see Figure 3B for identification of the different types of patch cords referred to below). To stimulate a single mouse,connect the coupler patch cord to a thick-jacketed patch cord using the FC/FC L-bracket adapter directly attached to the breadboard (see Figure 4A). To stimulate two animals from one laser, connect the coupler patch cord to two thick-jacketed patch cords using the 1 x 2 50/50 mini cube (see Figure 4B). To stimulate three or more animals, attach the coupler cord to a multimode fiber splitter using the 1 x 2 mini cube already attached to the breadboard (see Figure 4C).
Attach a commutator/rotary joint to the free ends of the thick-jacketed patch cord/fiber splitter. Commutators are essential as they allow rotation of the fiber with the rodent’s movement, which prevents accumulation of torque on the patch cord. Too much torque can twist cords, lead to breakage, and interfere with the animal’s natural movement during testing.
Attach the animal patch cord to the commutator.
Attach a connecting split sleeve to the free metal ferrule end of the animal patch cord (Figure 5). Do not force the sleeve all the way up the ferrule; leave ~0.5 cm of sleeve exposed as this is what connects to the implanted fiber optic affixed to the animal (Figure 6). Critical Step: Always purchase sleeves that contain a split to allow expansion of sleeve over implanted fiber ferrule during connection and removal. Too tight of a fit can cause severe trauma to the animal if the implant dislodges from the skull when attempting to disconnect the sleeve from the implant. If this does occur, the animal should be removed from the study and receive immediate veterinarian care. Similarly, before using a new sleeve for the first time, ‘break it in’ by plugging and unplugging a ferrule until it disconnects with the desired amount of force. Tip: It is easy to break a fiber while removing a sleeve that is tightly attached to a ferrule. To avoid this, push the ferrule out by inserting a small wooden rod into the open end of the sleeve (the handle of a standard cotton swab is the right size).
Connect the blue laser driver to a pulse generator using a BNC cable and turn the pulse generator on.
Put appropriate safety goggles on. Set the switches on the back of the laser to “Curr” and “TTL+” mode. Make sure that the power knob on the front of the driver is set at zero andturn the laser on (turn the driver on first and then the laser key).
Adjust the power knob on the front of the laser so that 5 - 10 mW is being emitted from the animal patch cord fiber tip as measured using a light power meter. 5 - 10 mW is a general guideline – the exact power intensity required to affect a given volume of tissue should be calculated prior to the start of the experiment, as in Aravanis et al.3
Switch the blue laser to “Analog” mode for in vivo stimulation. Note: Yellow DPSS lasers are operated in TTL+ mode for constant illumination. Wait 10-15 min for the laser to warm up.
Gently restrain the mouse and connect the split-sleeve on the animal patch cord to the chronic implantable fiber (see Figure 6). Make sure the ends of both fibers make physical contact with one another. Use the split on the connecting sleeve as a window to visualize direct contact between the two. Critical Step: Sometimes debris can collect on the metal ferrule of the animal’s implantable fiber and interfere with proper connection. In this case, use an ethanol wipe to gently clean the ferrule on the animal’s head prior to attachment. Never force a connecting sleeve over the ferrule as this can cause severe trauma to the animal. If a physical connection between fiber ends can not be made after cleaning, remove the animal from the study. Tip: Light leakage can occur at the connection point between the implanted fiber and animal patch cord. Visualization of this light by rodents may present an experimental confound10. Heat shrink tubing can be attached to patch cords and slid over the connecting point to minimize extraneous light.
Allow the mouse to recover for a few minutes prior to the start of behavioral testing. Tip: Depending on the behavioral test to be administered, it is best to habituate mice to the connection and tethering process 2 - 3 days prior, as the handling required to connect the animal may induce stress and confound behavioral testing.
Place the mouse in the behavioral testing apparatus ensuring that the connector cord is free of snags. Never leave an animal unattended during stimulation. Even with the use of commutators, patch cords do have a tendency to twist during extended periods of time and may interfere with behavioral testing.
Use a pulse generator to pulse the blue laser at a predetermined frequency that will activate opsin of choice. For yellow laser use: pulse the yellow laser with external shutters or by simply blocking the beam path with an opaque, non-reflective, non-flammable object.
4. Post In vivo Stimulation Considerations
This section is not intended to be a complete protocol but is offered as guidance for additional procedures that should be considered following in vivo optogenetic stimulation.
Upon completion of an experiment, confirm viral and fiber placement histologically for accurate interpretation of behavioral results. Euthanize animal according to institutional guidelines and perfuse the animal with ice-cold phosphate-buffered saline (PBS) and 4% (w/v) paraformaldehyde in PBS.
Remove implanted fiber optic by firmly grasping the exposed metal ferrule with pliers or hemostats. Pull up in one smooth, yet swift, motion. It is important to test the integrity of the implanted fiber by measuring the light output at the end of each experiment.
Post-fix the brains in paraformaldehyde for at least 24 - 48 hr before sectioning through the region of interest. (If using a freezing microtome, incubate brains in a 30% sucrose solution for several days before sectioning). Perform immunohistochemistry using standard protocols for detection of the appropriate opsin-tagged fluorophores, i.e., green florescent protein (GFP), enhanced yellow florescent protein (eYFP) or mCherry.
Check site of opsin expression and fiber implant under a microscope and visually confirm appropriate placement of virus injection and implant based on chosen coordinates.
Representative Results
Behavioral results obtained with in vivo optogenetic stimulation are entirely dependent on the neural circuit being targeted, the animal model used, and the modulation parameters. For current demonstrative purposes, dopamine neurons in the ventral tegmental area, or VTA, of tyrosine hydroxylase::Cre mice were transduced with a stable step-function opsin (SSFO)8, or control virus (eYFP), and a fiber implant was chronically implanted. The use of TH::Cre transgenic mice ensures that opsin expression is restricted to TH+ cells (dopamine) in the VTA. Figure 7 depicts representative behavioral results obtained using the current described laser set-up for simultaneous stimulation of multiple mice. Here, mice were tethered and stimulated at the same time using separate lasers (3 mice/laser as in Figure 4C) and locomotor behavior was recorded for 1 hr. Repeated stimulation of dopamine neurons in the VTA resulted in a hyperactive phenotype that persisted throughout the duration of stimulation. No change in locomotor behavior was seen in eYFP mice (see Video 1). Following behavioral testing, immunohistochemistry was performed to verify accurate viral targeting to VTA dopamine neurons and fiber placement was visually confirmed (see Figure 7).
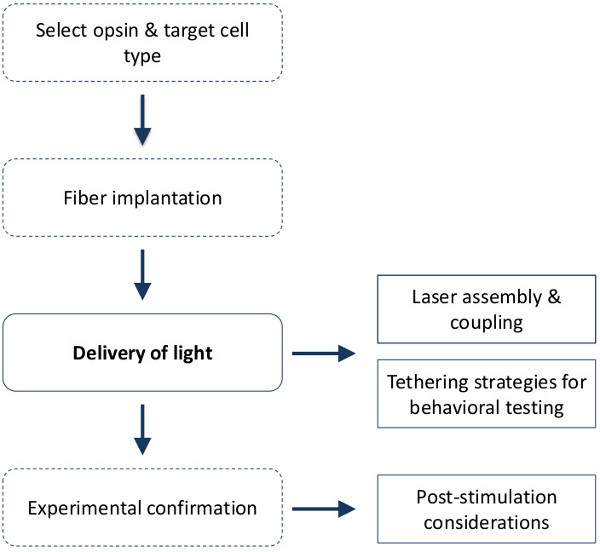 Figure 1. Experimental steps for in vivo optogenetic stimulation. There are four general steps involved when designing and performing in vivo optogenetic stimulation. This protocol specifically details the steps involved in the delivery of light from a laser light source to deep brain structures in the behaving rodent and includes 1) laser system assembly and light coupling; 2) tethering strategies for connecting multiple animals to a light source for high-throughput behavioral testing and 3) provides guidelines to confirm the targeting strategy for light delivery – a step that is essential for data interpretation. Note: although this protocol is not exclusive to chronic implantable fibers for tethering purposes, it is recommended and assumed when combining optogenetic stimulation with behavioral testing. See both Ung & Arenkiel, 201218 and Sparta et al., 20129 for in-house production and implantation of chronic optical fibers. Solid lines = steps covered in this protocol.
Figure 1. Experimental steps for in vivo optogenetic stimulation. There are four general steps involved when designing and performing in vivo optogenetic stimulation. This protocol specifically details the steps involved in the delivery of light from a laser light source to deep brain structures in the behaving rodent and includes 1) laser system assembly and light coupling; 2) tethering strategies for connecting multiple animals to a light source for high-throughput behavioral testing and 3) provides guidelines to confirm the targeting strategy for light delivery – a step that is essential for data interpretation. Note: although this protocol is not exclusive to chronic implantable fibers for tethering purposes, it is recommended and assumed when combining optogenetic stimulation with behavioral testing. See both Ung & Arenkiel, 201218 and Sparta et al., 20129 for in-house production and implantation of chronic optical fibers. Solid lines = steps covered in this protocol.
 Figure 2. Laser systems used for in vivo optogenetic stimulation. (A) Single laser system for in vivo stimulation. This laser is physically pre-coupled by manufacturer and requires little end user set-up. (B) Dual laser system. Two lasers are coupled into a single fiber through the use of mirrors that act to steer each beam path into a non-contact style coupler. This is the most versatile set-up as optical components can be removed or added as needed but presents more of a challenge in terms of efficient laser coupling. (C) Schematic of dual laser system shown in (B) indicating placement of lasers and mirrors with the corresponding laser light beam path (arrows) depicted. Here, the dichroic mirror “D” is used to deflect blue wavelengths of light while transmitting yellow wavelengths through to the coupler “C” and into the attached coupler patch cord. B = blue laser; C = non-contact style coupler; D = dichroic mirror; FW = filter wheel; M = Mirror; Y = yellow laser. Please click here to view a larger version of this figure.
Figure 2. Laser systems used for in vivo optogenetic stimulation. (A) Single laser system for in vivo stimulation. This laser is physically pre-coupled by manufacturer and requires little end user set-up. (B) Dual laser system. Two lasers are coupled into a single fiber through the use of mirrors that act to steer each beam path into a non-contact style coupler. This is the most versatile set-up as optical components can be removed or added as needed but presents more of a challenge in terms of efficient laser coupling. (C) Schematic of dual laser system shown in (B) indicating placement of lasers and mirrors with the corresponding laser light beam path (arrows) depicted. Here, the dichroic mirror “D” is used to deflect blue wavelengths of light while transmitting yellow wavelengths through to the coupler “C” and into the attached coupler patch cord. B = blue laser; C = non-contact style coupler; D = dichroic mirror; FW = filter wheel; M = Mirror; Y = yellow laser. Please click here to view a larger version of this figure.
 Figure 3. (A) Cable tester used in the non-physical coupling protocol. Bottom: cable tester directly connected to a patch cord. Insert depicts connection point of tester to cable. (B) Patch cords referred to throughout protocol. From outer to inner: Multimode fiber splitter, black-jacketed animal patch cord with white zirconia split sleeve attached to the flat-cleave (FC) end, thick-jacketed patch cord (also referred to as a “coupler cord”). Thick-jacketed patch cords are coated with polyvinyl chloride (PVC) tubing for extra protection. For these cables, industry standard color codes are used to distinguish between different fiber types, where orange = multimode fiber. Animal patch cords are thinner jacketed to allow flexibility for animal movement during behavioral testing. Note that dust caps are placed on FC/PC ends when cables are not in use. Please click here to view a larger version of this figure.
Figure 3. (A) Cable tester used in the non-physical coupling protocol. Bottom: cable tester directly connected to a patch cord. Insert depicts connection point of tester to cable. (B) Patch cords referred to throughout protocol. From outer to inner: Multimode fiber splitter, black-jacketed animal patch cord with white zirconia split sleeve attached to the flat-cleave (FC) end, thick-jacketed patch cord (also referred to as a “coupler cord”). Thick-jacketed patch cords are coated with polyvinyl chloride (PVC) tubing for extra protection. For these cables, industry standard color codes are used to distinguish between different fiber types, where orange = multimode fiber. Animal patch cords are thinner jacketed to allow flexibility for animal movement during behavioral testing. Note that dust caps are placed on FC/PC ends when cables are not in use. Please click here to view a larger version of this figure.
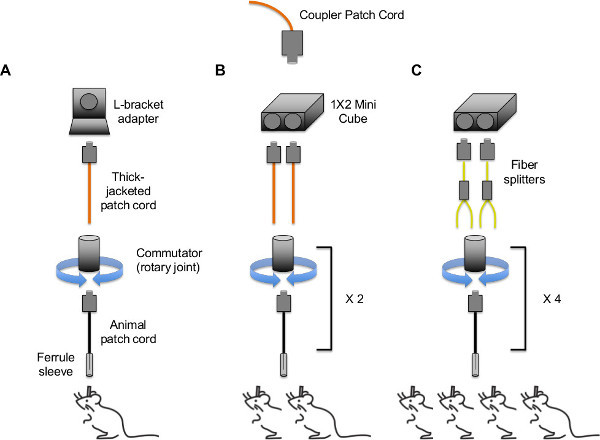 Figure 4. Tethering strategies for in vivo optogenetic stimulation of (A) a single animal, (B) two animals; (C) three or four animals. Possible configurations are not limited to those shown above – multiple configurations are possible through the unique combination of adapters, fiber splitters, and branching patch cords that are available commercially or by custom order. Note: patch cords and fiber splitters contain FC/PC connectors on both ends (only one end is depicted). Please click here to view a larger version of this figure.
Figure 4. Tethering strategies for in vivo optogenetic stimulation of (A) a single animal, (B) two animals; (C) three or four animals. Possible configurations are not limited to those shown above – multiple configurations are possible through the unique combination of adapters, fiber splitters, and branching patch cords that are available commercially or by custom order. Note: patch cords and fiber splitters contain FC/PC connectors on both ends (only one end is depicted). Please click here to view a larger version of this figure.
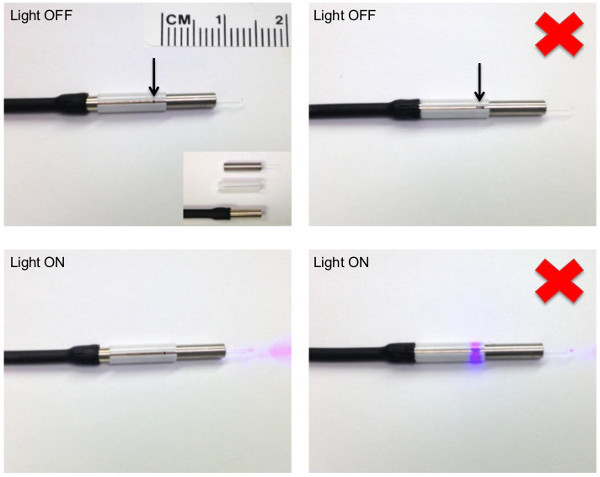 Figure 5. Correct and incorrect (red x) connection of a patch cord to an implantable fiber optic using a split-sleeve.(Left panel) A zirconia split-sleeve is used to connect a patch cord to the ferrule of an implantable fiber optic (shown here not affixed to an animal). Arrow is pointing to the connection point between the patch cord and implantable fiber optic. Compare to (Right panel) where a gap exists between the patch cord and implantable fiber optic, as visualized through the split of the connecting sleeve. Note the light leakage that can occur with an improper connection (bottom right). Bottom insert on upper left panel depicts individual components used. From top to bottom of insert: Doric implantable fiber optic cannula, white zirconia split-sleeve, flat-cleeve (FC) end of a black-jacketed animal patch cord (full patch cord depicted in Figure 3B). In all panels, note that the connecting sleeve is not flush with the FC end of the patch cord. Leave ~0.5 cm of an over-hang for connection to the implantable fiber optic affixed to the animal. Please click here to view a larger version of this figure.
Figure 5. Correct and incorrect (red x) connection of a patch cord to an implantable fiber optic using a split-sleeve.(Left panel) A zirconia split-sleeve is used to connect a patch cord to the ferrule of an implantable fiber optic (shown here not affixed to an animal). Arrow is pointing to the connection point between the patch cord and implantable fiber optic. Compare to (Right panel) where a gap exists between the patch cord and implantable fiber optic, as visualized through the split of the connecting sleeve. Note the light leakage that can occur with an improper connection (bottom right). Bottom insert on upper left panel depicts individual components used. From top to bottom of insert: Doric implantable fiber optic cannula, white zirconia split-sleeve, flat-cleeve (FC) end of a black-jacketed animal patch cord (full patch cord depicted in Figure 3B). In all panels, note that the connecting sleeve is not flush with the FC end of the patch cord. Leave ~0.5 cm of an over-hang for connection to the implantable fiber optic affixed to the animal. Please click here to view a larger version of this figure.
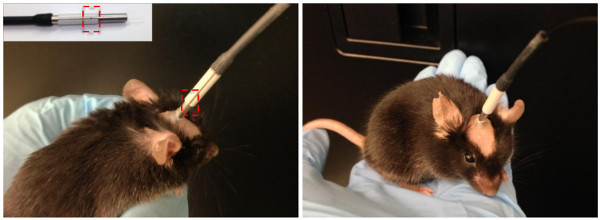 Figure 6. Side (left) and frontal (right) view of a mouse with an implanted fiber optic connected to a patch cord. Use the split on the connecting sleeve to help visualize proper connection of the patch cord to the ferrule of the implanted fiber optic. The connection point is highlighted by a red dashed box and also depicted in the upper insert. Please click here to view a larger version of this figure.
Figure 6. Side (left) and frontal (right) view of a mouse with an implanted fiber optic connected to a patch cord. Use the split on the connecting sleeve to help visualize proper connection of the patch cord to the ferrule of the implanted fiber optic. The connection point is highlighted by a red dashed box and also depicted in the upper insert. Please click here to view a larger version of this figure.
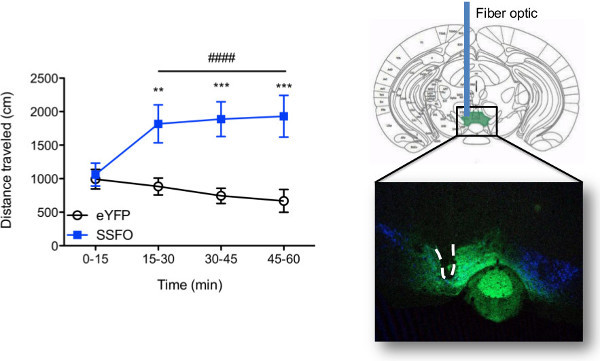 Figure 7. Representative results. (Left) Behavioral readout of in vivo optogenetic stimulation. Example of behavior that can be obtained using the described laser set-up and tethering protocol. Locomotor activity was recorded during optogenetic stimulation of the ventral tegmental area (VTA) in tyrosine hydroxylase (TH)::Cre mice (n = 7 - 8/group) transduced with either a step-function opsin (AAV5-DIO-SSFO-eYFP) or control virus (AAV5-DIO-eYFP) in the VTA. Groups of three mice were simultaneously tethered to a single laser as depicted in Figure 4C and stimulated with a 5 sec pulse of 447 or 473 nm light delivered once every 15 min. Two-way repeated measures ANOVA revealed a significant group x time interaction (F3,39 = 15.27, p < 0.0001) and a significant main effect of time (F3,39 = 4.67, p = 0.007) whereby optogenetic stimulation increased locomotor activity only in SSFO mice (bonferroni post-hoc p < 0.0001, relative to t = 0 - 15 time bin) resulting in an overall increase in locomotor activity compared with eYFP mice (main effect of group: F1,39 = 10.69, p = 0.0061; bonferroni post-hoc p < 0.01 at t = 15 - 30 and p < 0.001 at t = 30 - 45 and t = 45 - 60). Fiber specs: 200 μm core, 0.22 NA. Light irradiance = 6 - 66 mW/mm2, corresponding to fiber tip distance of 0.1 - 0.6 mm from viral injection site with 5 mW light power emitted at fiber tip prior to tethering. Error bars represent standard error of the mean. eYFP vs. SSFO: **p < 0.01; ***p < 0.001; time effect: #### p < 0.0001. (Right) Histological confirmation of viral and fiber optic placement. Confocal fluorescence image acquired on a Leica TCS SP5 scanning laser microscope was used to visualize fiber placement (dotted-line) and viral-mediated expression (green) in the mouse ventral tegmental area following in vivo optogenetic stimulation. Dopamine neurons (TH+) are seen in blue. Please click here to view a larger version of this figure.
Figure 7. Representative results. (Left) Behavioral readout of in vivo optogenetic stimulation. Example of behavior that can be obtained using the described laser set-up and tethering protocol. Locomotor activity was recorded during optogenetic stimulation of the ventral tegmental area (VTA) in tyrosine hydroxylase (TH)::Cre mice (n = 7 - 8/group) transduced with either a step-function opsin (AAV5-DIO-SSFO-eYFP) or control virus (AAV5-DIO-eYFP) in the VTA. Groups of three mice were simultaneously tethered to a single laser as depicted in Figure 4C and stimulated with a 5 sec pulse of 447 or 473 nm light delivered once every 15 min. Two-way repeated measures ANOVA revealed a significant group x time interaction (F3,39 = 15.27, p < 0.0001) and a significant main effect of time (F3,39 = 4.67, p = 0.007) whereby optogenetic stimulation increased locomotor activity only in SSFO mice (bonferroni post-hoc p < 0.0001, relative to t = 0 - 15 time bin) resulting in an overall increase in locomotor activity compared with eYFP mice (main effect of group: F1,39 = 10.69, p = 0.0061; bonferroni post-hoc p < 0.01 at t = 15 - 30 and p < 0.001 at t = 30 - 45 and t = 45 - 60). Fiber specs: 200 μm core, 0.22 NA. Light irradiance = 6 - 66 mW/mm2, corresponding to fiber tip distance of 0.1 - 0.6 mm from viral injection site with 5 mW light power emitted at fiber tip prior to tethering. Error bars represent standard error of the mean. eYFP vs. SSFO: **p < 0.01; ***p < 0.001; time effect: #### p < 0.0001. (Right) Histological confirmation of viral and fiber optic placement. Confocal fluorescence image acquired on a Leica TCS SP5 scanning laser microscope was used to visualize fiber placement (dotted-line) and viral-mediated expression (green) in the mouse ventral tegmental area following in vivo optogenetic stimulation. Dopamine neurons (TH+) are seen in blue. Please click here to view a larger version of this figure.
Video 1.In vivo optogenetic stimulation: Hyperactivity during VTA stimulation using SSFO in TH::Cre mice. Please click here to view this video.
| Table 1. Light irradiances required to activate commonly used opsins. | ||||
| Opsin Variant | λ | Power Density (/mm2) | Properties | |
| On/Off Kinetics | ||||
| Optical excitation: Fast-acting channelrhodopsins | ||||
| ChR22 | 470 | 1 - 5mW | 1.21/12 msec | Fires up to 40 Hz |
| ChETA19 | 490 | 5 mW | 0.86/8.5 msec | Fires up to 200 Hz |
| ChIEF20 | 450 | 1.65 mW | 1.62/12 msec | Non-desensitizing form of ChR2 |
| C1V18 | 540 - 630 | 8 mW (540nm) | 5/34 msec at 540nm | Red-shifted |
| 3.2 mW (630nm) | 67 msec (on) at 630nm | |||
| Optical excitation: Slow-acting channel rhodopsins | ||||
| Stable step function opsin (SFO)8 | 470/590 | 8 μW (470nm) | 20 msec/29 min | New SFO variant; longer open state. Opened by 470 nm, closed by 590 nm |
| Optical Inhibition | ||||
| eNpHR3.021 | 560 - 630 | 3 - 5 mW | 2.5 msec/<10 msec | Sustained inhibition for 30 min22 with constant light* |
| ArchT3.011,23 | 520 - 560 | 1 - 5 mW | 2/<10 msec | More sensitive with larger photocurrents than eNpHR3.0 |
| This table is provided as a guide only; specific light irradiances required for neural modulation should be independently confirmed. | ||||
| Experimental validation is important to verify that the opsin, targeting strategy, and light stimulation parameters modulate neural firing in the intended manner5. | ||||
| Power density (mW/mm2) refers to the power of light illuminated on a given area of brain tissue and does not refer to light power emitted from the fiber tip. | ||||
| *always use the lowest light intensity possible, especially with prolonged light stimulation. |
Table 1.Light irradiances required to activate commonly used opsins.Abbreviations AAV = adeno-associated virus DPSS = diode-pumped solid state eYFP = enhanced yellow florescent protein FC/PC = flat cleave/physical contact GFP = green florescent protein PBS = phosphate buffered saline PVC = polyvinyl chloride mW = milliwatt NA = numerical aperture SSFO = stable step-function opsin TH = tyrosine hydroxylase TTL = transistor-transistor logic V = voltage VTA = ventral tegmental area
Discussion
The current described laser set-ups and tethering strategies are compatible with a wide range of rodent behavioral tests. Indeed, a variety of behavioral tests have been used following, or accompanying, in vivo optogenetic stimulation that include emotive behavioral tasks, behavioral conditioning, learning and memory paradigms, sleep, arousal, and appetitive tasks to name a few (see Nieh et al.6 for a comprehensive review). Optogenetics has changed the way traditional behavioral tests are conducted in that multiple-day studies can now be condensed into a single session in which behavior is compared, within-subjects, during distinct epochs of light ‘on’ versus ‘off’5. Of note, behavioral apparatuses that contain doorways, closed compartments or other obstructions may have to be modified to accommodate passage of tethered fibers.
The described tethering strategies permit simultaneous stimulation of multiple mice from a single laser. High throughput optogenetic behavioral testing can therefore be achieved through the use of multiple lasers and testing equipment. The number of animals that can be simultaneously stimulated, however, will be limited by the maximum light power that can be achieved at each fiber tip. Maximum power output at the fiber tip is dependent upon the 1) starting power of the laser; 2) coupling efficiency and 3) number of beam splits. For a 100 mW blue laser with ~80% coupling efficiency and up to 4 beam splits (as depicted in Figure 4C), average power at the fiber tip can range between 5-10 mW when using 200 μm core, 0.22 NA fiber patch cords (n.b. expect transmission loss from rotary joints to be <15%). Measuring light output at the fiber tip is essential for determining adequate light power for opsin activation as opsins differ in their sensitivity to light and therefore the light power density (mW/mm2) required for activation11. For instance, the stable step-function opsin (SSFO) acts as a photon accumulator and therefore requires very little light power density for activation (<8 μW/mm2)8. Compare this to the traditional channel rhodopsin (ChR2) that requires a minimum of 1 mW/mm2 of light to elicit action potentials2. Table 1 is provided as a quick reference for known minimum light irradiances required to activate the most common opsins currently in use. Lastly, one must consider that light scatters and absorbs as it travels through brain tissue such that more light power is required for deeper brain structures3. A useful online resource is available at http://www.stanford.edu/group/dlab/cgi-bin/graph/chart.php that will calculate the light intensity at various depths through brain tissue by taking into account the fiber core size, numerical aperture, wavelength of light used, and the starting light power at the fiber tip. For an excellent overview of the theoretical principles underlying these calculations, see Foutz et al. (2012)12. Examples of how to apply these principles and calculations to experimental design are demonstrated in Aravanis et al. (2007)3 and Tye et al. (2012)13. Performing these calculations prior to the start of an experiment is crucial to ensure adequate light irradiance for opsin activation. Given these considerations, it is advantageous to purchase higher-powered lasers to ensure adequate power output. Lasers with a power output between 100-200 mW are generally sufficient to compensate for small core fibers, multiple fiber splitting, coupling inefficiency and transmission loses7. If using high power lasers, however, care must be taken to avoid neural damage or heat and light-associated artifacts that can occur with prolonged and/or high powered light illumination7. A safe range for in vivo experiments is up to 75 mW/mm2.14
Deciding on the type of laser to purchase can be a complicated matter as there are many factors to consider. For instance, direct diode lasers provide more stable and repeatable pulsed output than do diode-pumped solid-state (DPSS) lasers, and are more reliable over time in a lab environment. In some cases, however, direct diode lasers may emit a lower light power, ~0.1 mW, even when the command voltage is 0 V due to a constant bias current being sent to the diode by the laser’s control electronics. This ‘spontaneous’ emission has a broader spectrum than does laser emission from the same laser, so can be specifically reduced by installing a narrow band-pass (or ‘cleanup’) filter between the laser and coupler (see parts list). This filter will also reduce power output by ~50% when lasing, so purchase a higher-powered laser accordingly. It should be noted that yellow DPSS lasers are extremely sensitive and may behave erratically and have reduced lifespan if rapidly modulated by a pulse generator. Adjustment of yellow laser power should be done through external density filter wheels placed in the beam path (Section 1.7) while operating the laser in TTL+ mode. Alternatively, purchasing a green 532 nm DPSS laser is a cost-effective alternative that can activate both halorhodopsins and archaerhodopsins.
The numerical aperture (NA) of a fiber is important to consider when designing and purchasing fiber components for laser assembly set-up. The NA of an optical fiber determines the angles of light rays that can be accepted and emitted at the tip of a fiber. If a higher-NA fiber is mated to a lower-NA fiber, significant loss will occur at that interface, so it is important to be consistent with fiber NA within a single setup (or to ensure that NA increases along the light path). The effect of fiber NA on the volume of brain tissue illuminated is less important, since brain tissue is highly scattering, and since the light coupled from a laser source will tend to ‘underfill’ high-NA fibers; however optical fibers with an NA of 0.22 and 0.37 are commonly used. Similarly, coupling from a larger-core to a smaller-core fiber will also result in significant losses, so always be sure to use increasing or equal core diameters when progressing from the laser source to the animal implant. On a general note, fiber ends should always be capped when not in use to prevent dust and particulate build-up. It is a good idea to regularly clean fiber ends and connectors (70% isopropyl alcohol works well) to ensure maximal light power output, and to test light power output through a ‘dummy implant’ before beginning each day’s experiments.
During behavioral testing, it is imperative that steps be taken to control for the effects of viral infection, exogenous protein expression, visible light, and possible tissue heating effects and artifacts on animal behavior. Therefore, the proper control group should consist of animals transduced with a control virus (e.g., GFP, eYFP, mCherry) that receive identical light stimulation parameters. Experimental verification is a crucial final step as the behavioral data used for analysis is entirely dependent on proper opsin and fiber optic placement in the region of interest. Specifically, in animals where no immunohistochemical signal is detected, or where placement of signal or fiber is not in the region of interest, then behavioral data for that animal should be removed from the experiment. Additionally, it is essential to test light output at the fiber tip both before surgical implantation and again post-mortem to ensure adequate light power for opsin activation. In animals where severe light loss has occurred through the fiber after experimentation (>30%)9, data for that animal should be considered for removal. Criteria for removal should be established a priori. Finally, one must consider the pulse frequency required to modulate neural firing, which will depend on the brain structure and neuronal sub-types being targeted. Published optogenetic light stimulation parameters exist for multiple neuronal sub-types, however, the ability to modulate neural firing should be independently confirmed through in vivo or brain slice electrophysiological recordings.
As one becomes adept with laser use and modification of laser set-ups, combinations of different wavelengths can be tethered to multiple fibers on a single animal or delivered down the same fiber for combinatorial optogenetics8. Multi-wavelength stimulation will become increasingly important given the rapid development of red-shifted channelrhodopsins8, the engineering of blue-shifted hyperpolarizing opsins15, the use of bistable step-function opsins8,16,17, and the general expanding list of opsins with distinct activation spectra11. This expansion of the optogenetic toolbox will permit the unprecedented control of multiple neural sub-types both within and across brain regions to determine their role in governing complex behavioral states.
Disclosures
The authors have no conflicts of interest to report.
Acknowledgments
These studies were funded by grants received from the NIH (MH082876, DA023988).
References
- Zhang F, et al. The microbial opsin family of optogenetic tools. Cell. 2011;147:1446–1457. doi: 10.1016/j.cell.2011.12.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boyden ES, Zhang F, Bamberg E, Nagel G, Deisseroth K. Millisecond-timescale genetically targeted optical control of neural activity. Nat Neurosci. 2005;8:1263–1268. doi: 10.1038/nn1525. [DOI] [PubMed] [Google Scholar]
- Aravanis AM, et al. An optical neural interface: in vivo control of rodent motor cortex with integrated fiberoptic and optogenetic technology. J Neural Eng. 2007;4:S143–156. doi: 10.1088/1741-2560/4/3/S02. [DOI] [PubMed] [Google Scholar]
- Sidor MM. Psychiatry's age of enlightenment: optogenetics and the discovery of novel targets for the treatment of psychiatric disorders. J Psychiatry Neurosci. 2012;37:4–6. doi: 10.1503/jpn.110175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tye KM, Deisseroth K. Optogenetic investigation of neural circuits underlying brain disease in animal models. Nat Rev Neurosci. 2012;13:251–266. doi: 10.1038/nrn3171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nieh EH, Kim SY, Namburi P, Tye KM. Optogenetic dissection of neural circuits underlying emotional valence and motivated behaviors. Brain Res. 2013;1511:73–92. doi: 10.1016/j.brainres.2012.11.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yizhar O, Fenno LE, Davidson TJ, Mogri M, Deisseroth K. Optogenetics in neural systems. Neuron. 2011;71:9–34. doi: 10.1016/j.neuron.2011.06.004. [DOI] [PubMed] [Google Scholar]
- Yizhar O, et al. Neocortical excitation/inhibition balance in information processing and social dysfunction. Nature. 2011;477:171–178. doi: 10.1038/nature10360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sparta DR, et al. Construction of implantable optical fibers for long-term optogenetic manipulation of neural circuits. Nat Protoc. 2012;7:12–23. doi: 10.1038/nprot.2011.413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kravitz AV, Owen SF, Kreitzer AC. Optogenetic identification of striatal projection neuron subtypes during in vivo recordings. Brain Res. 2013;1511:21–32. doi: 10.1016/j.brainres.2012.11.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mattis J, et al. Principles for applying optogenetic tools derived from direct comparative analysis of microbial opsins. Nat Methods. 2012;9:159–172. doi: 10.1038/nmeth.1808. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Foutz TJ, Arlow RL, McIntyre CC. Theoretical principles underlying optical stimulation of a channelrhodopsin-2 positive pyramidal neuron. J Neurophysiol. 2012;107:3235–3245. doi: 10.1152/jn.00501.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tye KM, et al. Amygdala circuitry mediating reversible and bidirectional control of anxiety. Nature. 2011;471:358–362. doi: 10.1038/nature09820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cardin JA, et al. Targeted optogenetic stimulation and recording of neurons in vivo using cell-type-specific expression of Channelrhodopsin-2. Nat Protoc. 2010;5:247–254. doi: 10.1038/nprot.2009.228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chow BY, et al. High-performance genetically targetable optical neural silencing by light-driven proton pumps. Nature. 2010;463:98–102. doi: 10.1038/nature08652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Diester I, et al. An optogenetic toolbox designed for primates. Nat Neurosci. 2011;14:387–397. doi: 10.1038/nn.2749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berndt A, Yizhar O, Gunaydin LA, Hegemann P, Deisseroth K. Bi-stable neural state switches. Nat Neurosci. 2009;12:229–234. doi: 10.1038/nn.2247. [DOI] [PubMed] [Google Scholar]
- Ung K, Arenkiel BR. Fiber-optic implantation for chronic optogenetic stimulation of brain tissue. J Vis Exp. 2012. p. e50004. [DOI] [PMC free article] [PubMed]
- Gunaydin LA, et al. Ultrafast optogenetic control. Nat Neurosci. 2010;13:387–392. doi: 10.1038/nn.2495. [DOI] [PubMed] [Google Scholar]
- Lin JY, Lin MZ, Steinbach P, Tsien RY. Characterization of engineered channelrhodopsin variants with improved properties and kinetics. Biophys J. 2009;96:1803–1814. doi: 10.1016/j.bpj.2008.11.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gradinaru V, et al. Molecular and cellular approaches for diversifying and extending optogenetics. Cell. 2010;141:154–165. doi: 10.1016/j.cell.2010.02.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goshen I, et al. Dynamics of retrieval strategies for remote memories. Cell. 2011;147:678–689. doi: 10.1016/j.cell.2011.09.033. [DOI] [PubMed] [Google Scholar]
- Han X, et al. A high-light sensitivity optical neural silencer: development and application to optogenetic control of non-human primate cortex. Front Syst Neurosci. 2011;5(18) doi: 10.3389/fnsys.2011.00018. [DOI] [PMC free article] [PubMed] [Google Scholar]


