Abstract
Hepatitis B virus (HBV) causes life-threatening liver disease. It is transmitted through a horizontal route or a mother-to-infant route, and the latter is the major route in endemic areas. Prevention of HBV infection by immunization is the best way to eliminate HBV-related diseases. The HBV vaccine is the first human vaccine using a viral antigen from infected persons, which is safe and effective. Either passive immunization by hepatitis B immunoglobulin (HBIG) or active immunization by HBV vaccine is effective, and a combination of both yields the best efficacy in preventing HBV infection. The impact of universal HBV immunization is huge, with 90%–95% effectiveness in preventing chronic HBV infection. It is the first cancer preventive vaccine with a protective efficacy against hepatocellular carcinoma (HCC) of ∼70%. Nevertheless, further effort is still needed to avoid vaccine failure and to increase the global coverage rate.
Immunization against hepatitis B virus (HBV) is remarkably effective in preventing HBV infection. Nevertheless, additional efforts to avoid vaccine failure and to increase the global coverage rate are needed.
Hepatitis B virus (HBV) infection can cause acute, fulminant, or chronic hepatitis, liver cirrhosis, and hepatocellular carcinoma (HCC). It is a potentially life-threatening infection, and is a major global health problem. Around 600,000 people die every year because of the acute or chronic consequences of the infection (www.who.int/mediacentre/factsheets/fs204/en). Once the infection becomes chronic, it is extremely refractory and is difficult to cure, despite recent developments of treatment measures (Chen 2011). Ultimately, around one-third of the chronically infected patients will eventually develop cirrhosis and HCC. HCC is one of the most common causes of cancer death worldwide because of a poor prognosis and a high recurrence rate after curative therapy. Besides liver disease, HBV infection also causes extrahepatic diseases, such as HBV-associated membranous nephropathy (HBVMN). Children with HBVMN are primarily infected via horizontal transmission. (Hsu et al. 1983; Kappus and Sterling 2013).
For hepatitis B, prevention is more effective than therapy. Despite the progress of antiviral therapy against HBV to suppress viral replication and to reduce complications in those with chronic hepatitis B, a cure for infection is still not possible. Hence, prevention of HBV infection by immunization is the best way to eliminate HBV-related diseases (Chen 2009). The development of an HBV vaccine using HBsAg protein from HBV carriers as the immunogen to induce anti-HBs, the protective antibody against HBV infection, is a successful pioneer in the history of vaccine development. During the past three decades, it is proved to be safe and successful in protecting people from HBV infection and the related diseases worldwide.
Nevertheless, there are problems that hinder the success of HBV prevention globally. Looking back at the history of the development of the HBV vaccine and immunization strategies and exploring the existing problems might help to eliminate HBV-related diseases.
TRANSMISSION ROUTE OF HBV AND OUTCOMES OF HBV INFECTION AT DIFFERENT AGES
To prevent HBV infection effectively, it is crucial to understand its route of transmission. HBV infection is transmitted through either a horizontal route or through a mother-to-infant route. Mother-to-infant transmission was called “vertical transmission” in the past, but this term is less frequently used now because it caused confusion with genetic transmission, which does not occur with HBV. In endemic areas, perinatal mother-to-infant transmission is the most important route of transmission; HBV infection is encountered mainly during infancy and early childhood.
Age at infection and source of infection affect the outcome of HBV infection (Beasley 2009). Without immunoprophylaxis, perinatal transmission from highly infectious (HBeAg-positive) hepatitis B carrier mothers results in chronic infection in 90% of their infants (Stevens et al. 1975). In contrast, only 5% of infants of HBeAg-negative HBsAg carrier mothers become chronic carriers and a small fraction may develop acute or fulminant hepatitis B (Shiraki et al. 1980; Chang et al. 1987). Among 2- to 4-yr-old toddlers, ∼25% will become chronic carriers (Beasley et al. 1982). In contrast, only 2.7% of the newly HBV-infected 18- to 19-yr-old university students became chronic carriers (Beasley et al. 1983a). Thus, the younger the age at infection, the higher the HBV carriage rate (Table 1).
Table 1.
Outcome of HBV infection in subjects of different age at transmission with no immunoprophylaxis
| Age at infection | Annual infection rate, (% anti-HBc seroconversion/yr) | Chronicity rate among infected (% HBsAg+ > 6 mo) | References |
|---|---|---|---|
| Infant | Stevens et al. 1975 | ||
| Mother HBsAg+, HBeAg+ | >95% | >90% | |
| Mother HBsAg+, HBeAg− | 5–10% | <5% | |
| Toddler | 5.0% | 23% | Beasley et al. 1982 |
| Young adult | 1.5% | 2.7% | Beasley et al. 1983a |
PREVENTION AGAINST HBV INFECTION: IMMUNIZATION AND OTHER STRATEGIES
HBV immunization can be classified into passive and active immunization. Passive immunization using hepatitis B immunoglobulin (HBIG) provides temporary immunity, whereas active immunization by the vaccine yields long-term immunity. Because in endemic areas the major infection route comes from maternal transmission, and the outcome of perinatal transmission results in a very high rate (90%) of chronic infection, the best timing of initial HBV immunization, therefore, should be within 24 h after birth, followed by subsequent doses of HBV vaccine during infancy.
Other prevention strategies, such as screening the blood products, proper sterilization of injection needles and syringes, and avoidance of risky behaviors, such as parenteral drug abuse, tattoo, or skin piercing, may preclude HBV horizontal transmission. Education to avoid high-risk behaviors should be advocated in addition to vaccination. In addition to the program for infants, many countries with low prevalence of HBV infection also have HBV vaccination programs for adolescents to prevent exposure to HBV by sexual contacts or other risk behaviors.
HEPATITIS B VACCINE DEVELOPMENT
In 1965, Baruch S. Blumberg reported a “new” antigen, the Australia antigen, in sera of patients with leukemia. He later discovered that the antigen is closely associated with a causal agent of viral hepatitis (Blumberg et al. 1967, 1969). Australia antigen was later found to be the hepatitis B surface antigen (HBsAg). Antibody to HBsAg (anti-HBs) was later shown to be a neutralizing antibody against HBV infection.
Maurice Hilleman was the pioneer of several important human vaccines, and the HBV vaccine is one of the most important. He hypothesized that, by injecting HBsAg, an antibody to HBsAg could be produced. HBsAg thus became the backbone of the hepatitis B vaccine. A highly purified and inactivated vaccine was made of HBsAg from HBV carrier plasma. Through stringent treatments with pepsin, urea, and formaldehyde, together with rigorous filtration, a safe vaccine was produced that was devoid of infectious agents, including human immunodeficiency virus (HIV). To ensure safety, the vaccine was tested exhaustively by ordinary procedures as well as in chimpanzees and marmosets. It was highly potent and it induced antibodies in guinea pigs, grivet monkeys, and chimpanzees after subcutaneous inoculation. Chimpanzees given three doses of vaccine were protected against challenges with 1000 chimpanzee-infectious doses of HBV given intravenously (Buynak et al. 1976) The first large-scale trials for the plasma vaccine were performed on homosexual men belonging to a high-risk group (Szmuness et al. 1980) The plasma-derived vaccine was approved by the Food and Drug Administration (FDA) in 1981. A similar vaccine was developed in France at almost the same time (Maupas et al. 1981).
Currently, there are two kinds of HBV vaccines, a plasma-derived vaccine, the first-generation vaccine, and the recombinant vaccine, the second-generation vaccine. The recombinant vaccine was produced by expressing the HBsAg gene in the yeast Saccharomyces cerevisiae (Valenzuela et al. 1982; Hilleman 1987). Both are safe and effective.
ESTABLISHMENT OF THE STRATEGIES OF HBV IMMUNIZATION IN INFANCY BY PILOT CLINICAL TRIALS
History of the development of the HBV vaccine and immunization program is an excellent model for the development of other preventive and therapeutic vaccines against infectious agent-related diseases and cancer.
Passive Immunization by HBIG
Before an HBV vaccine was available, a randomized double-blind, placebo-controlled efficacy trial of passive immunization using HBIG injection for the prevention of mother-to-infant transmission of HBV infection was conducted in Taiwan. HBIG was given within 1 h after birth to infants of HBeAg-positive HBsAg carrier mothers. The HBsAg carrier rate of the placebo-treated infants was 92%. The HBsAg carrier rate was 26% among infants who received three doses of HBIG at birth, 3, and 6 mo old, and was 54% in those who received a single dose at birth. The prevention efficacy was 42% by one dose of HBIG and 71% by three doses of HBIG, respectively (Table 2) (Beasley et al. 1983b).
Table 2.
Comparisons of the prevention of HBV infection in infants or immunogenicity of different strategies among different risk groupsa
| Infants of HBsAg+, HBeAg+ mothers | Infants of HBsAg+, HBeAg− mothers | Immunogenicity in low-risk infants (infants or children of HBsAg− mothers) | |
|---|---|---|---|
| HBIG only (Beasley et al. 1983b) | |||
| One dose, 1.0 mL | HBsAg+, 54%, efficacy 45% | – | – |
| Three doses, 0.5 mL | HBsAg+, 26%, efficacy 75% | ||
| Placebo | HBsAg+, 92% | ||
| HBV plasma vaccine | Vaccine, three doses (RP Beasley and L-Y Hwang, unpubl.) 1 wk, 1, 6 mo, HBsAg+, 23% 1, 2, 6 mo, HBsAg+, 38% Placebo HBsAg+, 88% |
Vaccine, four doses (Tsai et al. 1984), HBsAg+, 0% Placebo, HBsAg+, 5.3% |
Anti-HBs (Lee et al. 1983) After one dose, 48% After two doses, 96% After three doses, 100% |
| Combination of HBV plasma vaccine and HBIG, 0.5 mL/dose | HBIG birth, 3 mo + vaccine, 3, 4, 9 mo, HBsAg 2.0% (Beasley et al. 1983c) | HBIG + vaccine, 0, 1, 6 mo, HBsAg+, <1% (Yang et al. 2003) | – |
| HBIG birth + vaccine 0, 1, 6 mo, HBsAg+, 6.0% (Beasley et al. 1983c) | Vaccine, 0, 1, 6 mo, HBsAg+ <1% | ||
| HBIG birth + vaccine, 1, 2, 7 mo, HBsAg+, 8.6% (Beasley et al. 1983c) | |||
| HBIG birth + vaccine, 0, 1, 2, 12 mo, HBsAg+, 11% (Hsu et al. 1988) | |||
| Placebo, HBsAg+, 88.1% (Beasley et al. 1983c) | |||
| Combination of HBV recombinant vaccine + one dose HBIG, 0.5 mL | HBIG + vaccine 0, 1, 6 mo, HBsAg+, 3.3% (Lee et al. 1991) HBIG + vaccine, 0, 1, 6 mo, HBsAg+, 9.26% (Chen et al. 2012) |
HBIG + vaccine 0, 1, 6 mo, HBsAg+, 0.14% (Chen et al. 2012) Vaccine, 0, 1, 6 mo, HBsAg+, 0.29% |
– |
aHBsAg+ rate in high-risk infants (in comparison to controls with no vaccine or HBIG).
Active Immunization by HBV Vaccine
The immunogenicity of the hepatitis B virus vaccine was studied in 38 neonates of noncarrier mothers, 23 of whom were healthy and susceptible to HBV infection, and 15 who had anti-HBs passively acquired from their mothers. Initially, each neonate received two 20-μg doses of the HBV plasma vaccine by the intramuscular route, with a 1-mo interval between doses. The first dose of vaccine stimulated the production of anti-HBs in 48% of the neonates within 1 mo. Anti-HBs was detectable in the serum of 91% of vaccine recipients at the age of 3 mo, 2 mo after the second dose. By the age of 6 mo, 96% of vaccinees had detectable anti-HBs. At the age of 7 mo, all had developed anti-HBs after a third dose. High levels of anti-HBs were sustained after vaccination in infants who initially had passive antibodies, indicating that the preexisting anti-HBs did not interfere with the immunogenicity of the vaccine (Lee et al. 1983) (Table 2).
Since the HBV vaccine became available in 1982, more than one billion doses of the vaccine have been used worldwide. Active immunization with three or four doses of HBV vaccine without HBIG was proved to be immunogenic in more than 90% of infants of noncarrier mothers or HBeAg-negative carrier mothers. In infants of highly infectious (HBeAg seropositive HBsAg carrier) mothers, the prevention efficacy of using HBV vaccines was only ∼75% (Table 2) (Beasley et al. 1983c).
Combination of Passive and Active Immunization
A pilot clinical trial combining active and passive immunization with injection of HBIG within 24 h after birth followed by three doses of HBV vaccine further reduced the HBsAg carrier rate to 3% in infants. The results of early clinical trials for HBV immunization conducted in Taiwan established the ground for the universal HBV immunization strategies and programs used currently.
In the first randomized blind controlled trial combining HBIG plus the HBV vaccine, infants of HBeAg-positive, HBsAg carrier mothers were given HBIG immediately after birth, followed by one of the three schedules of HBV vaccination. No difference in efficacy between the three schedules was noted; the combined prevention efficacy was 94% versus HBIG alone (71%) or vaccine alone (75%). Persistent HBs antigenemia developed in only 6% of infants receiving prophylaxis, but in 88% of the controls (without prophylaxis). Anti-HBs developed in all those who did not become antigenemic (Beasley et al. 1983c). The best result of HBV prevention against perinatal transmission of HBV infection by highly infectious mothers was thus established to be the combination of HBIG with the hepatitis B vaccine. This study recommended that HBIG should be given as soon as possible after birth and need not be given again if the infant is subsequently vaccinated. A similar study was performed also in Taiwan with another plasma-derived vaccine with a slightly different schedule (Lo et al. 1985). The result also showed that combining HBIG with a hepatitis B vaccine had the highest efficacy reaching 90% versus 74% in those who received only the vaccine. These studies provided the basis for the subsequent HBV universal immunization programs.
Prevention Efficacy of Recombinant HBV Vaccine with HBIG
Among infants who received HBIG at birth and three 5-μg doses of yeast-recombinant hepatitis B vaccine, only 4.8% became chronic carriers, a greater than 90% level of protection and a rate comparable with that seen with HBIG and plasma-derived hepatitis B vaccine. These results suggested that, in this high-risk setting, the yeast-recombinant vaccine was at least as effective as the plasma-derived vaccine in preventing HBV infection and the chronic carrier state (Stevens et al. 1987).
THE EARLIEST UNIVERSAL HBV MASS IMMUNIZATION PROGRAM
The first universal hepatitis B mass vaccination program in the world was launched in Taiwan in July 1984. Pregnant women were screened for HBsAg and then HBeAg. Infants of mothers negative for HBeAg or HBsAg received the plasma-derived hepatitis B vaccine only at 0, 1, 2, and 12 mo. The vaccine was shifted to a recombinant hepatitis B vaccine at 0, 1, and 6 mo after October 1992. The immunization program in the initial 2 years (July 1984 to June 1986) covered only infants of HBsAg carrier mothers. From the third year on, all of the infants were covered. Infants of highly infectious mothers with positive HBeAg received HBIG within 24 h after birth in addition to the hepatitis B vaccine. The coverage rate of hepatitis B vaccine for neonates was ∼94% in initial years, and was ∼99% recently. Catch-up vaccination was also given to preschool and then school children (Chen et al. 1987).
The efficacy of the Taiwanese mass hepatitis B immunization program was evaluated in 3464 18-mo-old vaccinated infants of HBsAg carrier mothers. In 786 infants born to highly infectious mothers who had received HBIG and vaccine on schedule, the protective efficacy was ∼85%. Overall, 11% of infants still carried HBsAg, and 81% had anti-HBs (>10 mIU/mL in more than 90%) (Table 2). The geometric mean titers of anti-HBs were >200 mIU/mL in every group of infants. According to a random sampling study of children in Taiwan 3 years after implementing the universal HBV vaccination program, the protective efficacy was 86% in the HBIG plus HBV vaccine group and was 78% in those with three doses of HBV vaccine alone (Hsu et al. 1988).
GLOBAL VIEW OF HBV IMMUNIZATION
In 2009, the World Health Organization (WHO) recommended that all infants receive the hepatitis B vaccine as soon as possible after birth, preferably within 24 h. The birth dose should be followed by two or three doses to complete the primary series. In 1992, the World Health Assembly passed a resolution to recommend global vaccination against hepatitis B. Furthermore, as of July 2011, 93 member states have introduced the hepatitis B birth dose (www.who.int/mediacentre/factsheets/fs204/en). WHO recommended that high-risk groups should be vaccinated. They include (1) people who frequently require blood and/or blood products, dialysis patients, recipients of solid organ transplantations; (2) people interned in prisons; (3) IV drug users; (4) household and sexual contacts of people with chronic HBV infection; (5) people with multiple sexual partners, health-care workers, and others who are exposed to blood or blood products through work; and (6) travelers who have not completed their hepatitis B vaccination series before leaving for endemic areas.
The global hepatitis B vaccine coverage rate was estimated at 75%, and was as high as 91% in the Western Pacific and 89% in the Americas. Coverage in Southeast Asia reached 52% in 2010. In 2011, 180 countries had introduced infant HBV vaccination, and the global HBV vaccination coverage rate for the third dose was estimated to be ∼78%.
Universal HBV vaccination in infancy is more effective than selective immunization for high-risk groups. Slightly different immunization strategies or schedules are used in different countries, depending on epidemiologic features of HBV infection and available resources (Chen 2009). In many endemic countries with limited resources, three doses of hepatitis B vaccine are given to all infants, regardless of the maternal HBsAg status. This strategy offers an efficacy of ∼75% to 80% for infants of highly infectious HBeAg-positive mothers. Nevertheless, the cost of maternal screening and subsequent use of HBIG in the newborns can be avoided. In countries with low prevalence of HBV infection and better resources, HBIG is given to newborns of all HBsAg-positive mothers regardless of their HBeAg status, and all infants received three doses of the HBV vaccine. Although the cost of maternal screening for HBeAg can be saved, the wider use of HBIG increases the cost substantially.
PREVENTION OF DISEASES BY HBV VACCINATION
Hepatitis B vaccine provides protection against HBV infection and its complications, including fulminant hepatitis, chronic hepatitis, liver cirrhosis, and HCC, as well as membranous nephropathy. It is the first vaccine against a cancer, the first vaccine protecting from a sexually transmitted infection, and the first vaccine against a chronic disease (Lavanchy 2012).
Prevention of HBV Infection, Hepatitis B, and Nephropathy
After universal HBV immunization, the incidence of acute hepatitis B declined (Wasley et al. 2007; McMahon et al. 2011; Su et al. 2012). Owing to breakthrough HBV infection from mother-to-infant transmission, vaccinated infants had higher rates of infection (0.78/100,000) than those aged 1–14 yr (0.04/100,000), who had the lowest rates. After 25 years of universal HBV immunization in Taiwan, acute hepatitis B among adolescents and young adults ≤25 yr old was markedly reduced, rendering the previously unvaccinated 25- to 39-yr-old cohort additional targets for the prevention of acute hepatitis B (Kao and Chen 2008).
The mortality associated with fulminant hepatitis in infants has also declined significantly (Kao et al. 2001; Chiang et al. 2013). In addition, HBVMN in the vaccinated children also decreased drastically, very likely because of the reduction in horizontal transmission of HBV infection after universal HBV vaccination (Liao et al. 2011).
An ∼90% reduction of chronic HBV infection rate has been achieved after the HBV immunization program. We conducted serial seroepidemiologic surveys of HBV markers before and 5, 10, 15, and 20 years after the implementation of the vaccination program in Taipei, Taiwan (Hsu et al 1986; Chen et al. 1996; Ni et al. 2001, 2007, 2012). The HBsAg carrier rate decreased steadily from ∼10% before the vaccination program to 0.6%–0.7% afterward in vaccinated children younger than 20 yr of age. The infection rate (anti-HBc seropositive rate) declined from 38% to 16% and further to 4.6% in children 15–20 years after the program (Ni et al. 2007).
Similar effects have also been observed in many other countries where universal vaccination programs have been successfully conducted. The HBsAg seropositive rates declined to below 1% in most countries after hepatitis B immunization (Chen 2009). In Gambia, all nonimmune children under the age of 5 yr in the villages of Keneba and Manduar were vaccinated against HBV in 1984. Overall, vaccine efficacies in 1993 against HBV infection and chronic HBsAg carriage were 94.7% and 95.3%, respectively (Whittle et al. 1995). In 2007–2008, a seroepidemiologic study in Gambia showed the effectiveness of the hepatitis B vaccination that was implemented in 1986. Comparing fully vaccinated versus unvaccinated Gambians, HBV infection was 0.8% (2/255) versus 12.4% (59/475), indicating an efficacy of 94%. For HBV infection, the anti-HBc positivity rate was 27.4% (70/255) and 56.0% (267/475), respectively. The protection yielded by HBV vaccination persisted beyond the onset of sexual activities, suggesting that there was no need for a booster dose (Peto et al. 2014).
Prevention of HCC
Taiwan has a high prevalence of HBV infection and HCC both in children and adults. Children with HCC in Taiwan are nearly 100% HBsAg seropositive, 86% of them are HBeAg negative, and their mothers are mostly (94%) HBsAg seropositive, suggesting maternal transmission as the source of HBV infection (Chang et al. 1989).
Prevention of chronic HBV infection by immunization can reduce the incidence of liver cancer. As shown in our previous studies, the incidence of HCC among children 6–19 yr old born after the universal HBV vaccination program was reduced to ∼30%–32% of that among those born before the program was in effect in Taiwan (Chang et al. 1997, 2009).
Studies elsewhere also showed declines in the HCC incidence in children as a result of infant HBV immunization in Khon Kaen, Thailand, and Alaska, United States. The incidence of HCC is significantly lower in Thai children who receive the hepatitis B vaccine at birth. The age-standardized incidence rates for liver cancer in Thai children over 10 yr of age of nonvaccinated and vaccinated children were 0.88 and 0.07 per million, respectively (Wichajarn et al. 2008). After 25 years of universal newborn vaccination coupled with mass screening and immunization of susceptible Alaska Natives, HCC was eliminated among Alaska Native children. The incidence of HCC in adolescence below the age of 20 decreased from three per 100,000 in 1984–1988 to zero in 1995–1999, and no cases have occurred since 1999 (McMahon et al. 2011).
PROBLEMS TO BE SOLVED FOR A BETTER CONTROL OF HEPATITIS B AND ITS SEQUELAE
Inadequate Resources
Universal HBV immunization programs still have not been established all over the world, mainly because of limited resources. In some areas, universal HBV vaccination has been implemented, yet the cost of vaccination is not covered by the government. Further increase of the global coverage rates of infant HBV vaccination is an important issue toward a better control of hepatitis B and HCC. Coverage of the expenses of HBV vaccination in developing countries by the government or establishing a vaccine fund is important to expand the HBV vaccine coverage. It is particularly urgent in areas where HBV infection and HCC are endemic (Lerkhachonsuk et al. 2013).
Poor Compliance
In areas with adequate resources and an established program of universal HBV immunization, poor compliance may still be a problem. The number of people with ignorance or opposition to the HBV vaccines may be reduced after a better understanding of the extremely low incidence of vaccine-related side effects. For instance, although there is little supporting evidence, an association between CNS demyelinating diseases and hepatitis B vaccine has been implied repeatedly (Chen 2009). On the basis of critical systematic review of current data, no significant increased risk in onset or relapse of any diseases was associated with hepatitis B vaccination (Martínez-Sernández and Figueiras 2013). Clarification of some other poorly documented side effects of the vaccines may help to reduce anxiety about the risks of the vaccine and, as a consequence, enhance HBV vaccine coverage rate. Furthermore, in some developed countries, because of the competition from other new vaccines, HBV has not captured sufficient attention from the government (Van Herck et al. 2008).
Vaccine Failure
The causes of breakthrough HBV infection in the vaccinated subjects include high maternal viral load, intrauterine infection (Lin et al. 1987; Tang et al. 1998), surface-gene mutants (Hsu et al. 2010), genetic hyporesponsiveness to the vaccine (Kruskall et al. 1992), and immune compromised hosts. In the era of post-HBV vaccination, even after complete immunization, vaccine failure still occurs. The leading cause of vaccine failure is the mother-to-infant transmission of HBV from highly infectious mothers (Chen et al. 2012). In children born to HBeAg-seropositive HBsAg carrier mothers, despite HBIG and three doses of the HBV vaccine, 9.26% still became HBsAg positive. In contrast, in children born to HBeAg-negative, HBsAg-seropositive mothers, only 0.29% became HBsAg positive if no HBIG was given at birth, and 0.14% became HBsAg positive if HBIG was given at birth (P = 0.65). The predictive infection rates of vaccinated infants at maternal viral load levels of 7, 8, and 9 log10 copies/mL were 6.6%, 14.6%, and 27.7%, respectively (Wen et al. 2013).
Vaccine failure has been attributed to hepatitis B surface–gene mutants. Compared with the unvaccinated subjects (7.8%), the prevalence rate of the hepatitis B surface gene a mutant increased to 22%–28% among vaccinated HBsAg carriers. However, the prevalence rate of the mutants among the total population has remained stationary for 20 years because HBV vaccination reduced the HBsAg seropositive rates in the vaccinated population. Less infectivity of G145R, recombinant vaccine use, and mutant loss with older age seem to decrease the a mutant prevalence in an immunized population over time (Hsu et al. 2010, 2013).
Infants of HBsAg carrier mothers with positive HBeAg and high viral load are the high-risk group of breakthrough HBV infection, despite immune prophylaxis with a combination of passive (HBIG) and active (vaccine) immunization. Continuing efforts are needed to prevent breakthrough infection of HBV by highly infectious mothers. Preliminary results using anti-HBV nucleoside analog during the last trimester of pregnancy to prevent mother-to-infant transmission have been reported (van Zonneveld et al. 2003; Xu et al. 2009; Han et al. 2011). However, further studies are needed to determine the efficacy versus adverse responses and cost effectiveness in reducing mother-to-infant transmission from highly viremic mothers.
Duration of Protection after HBV Immunization in Infancy
Anti-HBs titers gradually decline with age in children immunized in infancy with hepatitis B vaccine, which is also seen in those who received a booster dose during childhood (Wang et al. 2005). In a prospective long-term follow-up study of 1200 HBV vaccinees in the general population (from 7 to 16 yr old), the annual new anti-HBc seropositive rate was low (0.13%), and no new chronic HBV infection was detected. The decay rate of anti-HBs titer during age 7–16 yr was ∼20% of the titer of the previous year. Among the uninfected children who had anti-HBs <10 mIU/mL, the boosted and nonboosted children developed new anti-HBc positivity at a similar rate, indicating that a vaccine booster at this age is not necessary (Lin et al. 2003).
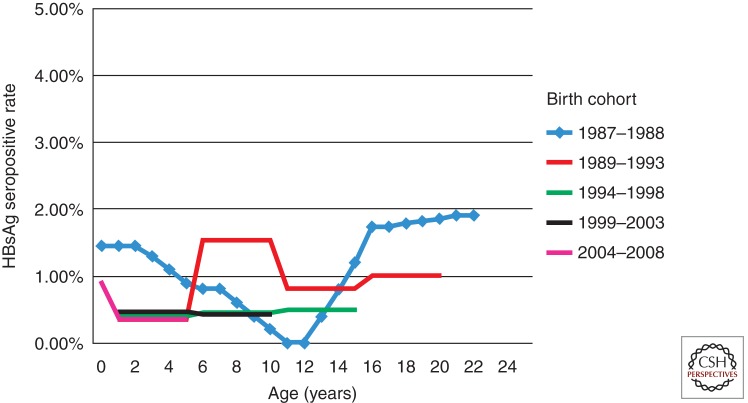
How long can the protection by HBV vaccination last? The proportions of postbooster anti-HBs titer <10 mIU/mL among 15–23 yr olds was 24%–28% (Jan et al. 2010). In another study, a booster dose of HBV vaccination was administered to 1974 HBsAg- and anti-HBs-negative subjects. The proportions of postbooster anti-HBs titer <10 mIU/mL was 27.9% (Wu et al. 2013). Even if studies show a loss of immune memory by this method, this does not necessarily mean that booster doses are required (Hall 2010). Loss of antibody among those who received HBV vaccination during infancy may not indicate loss of immunity or protection. Evidence on several critical end points, such as protection against infection and rates of anti-HBc conversion in vaccinated populations by age, and protection against diseases are more important than the anti-HBs titers (Chen 2005). There had been no increase of HBsAg seropositive rates in cohorts born after the HBV vaccination program was set into place as found in the five consecutive surveys in Taiwan (Fig. 1) (Ni et al. 2012). For those who had received vaccinations in infancy, routine booster vaccination may not be required to protect against chronic HBV infection at least before 25 yr of age. The WHO also suggested that routine universal boosters are not recommended for children or adolescents. Nevertheless, it should be considered for those at high risk of HBV infection with anti-HBs <10 mIU/mL after primary HBV vaccination (www.who.int/mediacentre/factsheets/fs204/en).
Figure 1.
Long-term serial seropidemiologic surveys showing no increase of HBsAg-positive rates for at least 20 years in Taiwan (Ni et al. 2012).
CONCLUDING REMARKS
Prevention is critical in the control of HBV infection. This is particularly important in places where HBV infection, chronic liver disease, and HCC are prevalent. The hepatitis B vaccine is the first vaccine against a major human cancer. To achieve better results in HBV prevention worldwide, higher coverage rates of HBV vaccination, better strategies against breakthrough infection and for nonresponders, and good long-term protection are needed. It is of vital importance to convince the governments of countries without a universal HBV vaccination program to establish and implement such a program, and to help the countries with insufficient coverage rates to improve their vaccination programs. With the universal hepatitis B vaccination program starting from neonates in most countries, HBV infection and its sequelae will be further reduced, or even eradicated (Chen 2009). The concept of a cancer preventive vaccine, using HBV as an example, can be extended further to other infectious agents and their related cancers.
ACKNOWLEDGMENTS
We appreciate Professor R. Palmer Beasley, Dr. Lu-Yu Hwang, and Mrs. R. P Beasley for allowing us to use the unpublished data from their study.
Footnotes
Editors: Christoph Seeger and Stephen Locarnini
Additional Perspectives on Hepatitis B and Delta Viruses available at www.perspectivesinmedicine.org
REFERENCES
- Beasley RP 2009. Rocks along the road to the control of HBV and HCC. Ann Epidemiol 19: 231–234. [DOI] [PubMed] [Google Scholar]
- Beasley RP, Hwang LY, Lin CC, Leu ML, Stevens CE, Szmuness W, Chen KP 1982. Incidence of hepatitis B virus infections in preschool children in Taiwan. J Infect Dis 146: 198–204. [DOI] [PubMed] [Google Scholar]
- Beasley RP, Hwang LY, Lin CC, Ko YC, Twu SJ 1983a. Incidence of hepatitis among students at a university in Taiwan. Am J Epidemiol 117: 213–222. [DOI] [PubMed] [Google Scholar]
- Beasley RP, Hwang LY, Stevens CE, Lin CC, Hsieh FJ, Wang KY, Sun TS, Szmuness W 1983b. Efficacy of hepatitis B immune globulin for prevention of perinatal transmission of the hepatitis B virus carrier state: Final report of a randomized double-blind, placebo-controlled trial. Hepatology 3: 135–141. [DOI] [PubMed] [Google Scholar]
- Beasley RP, Hwang LY, Lee GC, Lan CC, Roan CH, Huang FY, Chen CL 1983c. Prevention of perinatally transmitted hepatitis B virus infections with hepatitis B immune globulin and hepatitis B vaccine. Lancet 2: 1099–1102. [DOI] [PubMed] [Google Scholar]
- Blumberg BS, Gerstley BJ, Hungerford DA, London WT, Sutnick AI 1967. A serum antigen (Australia antigen) in Down’s syndrome, leukemia, and hepatitis. Ann Intern Med 66: 924–931. [DOI] [PubMed] [Google Scholar]
- Blumberg BS, Sutnick AI, London WT 1969. Australia antigen and hepatitis. JAMA 207: 1895–1896. [PubMed] [Google Scholar]
- Buynak EB, Roehm RR, Tytell AA, Bertland AU II, Lampson GP, Hilleman MR 1976. Vaccine against human hepatitis B. JAMA 235: 2832–2834. [PubMed] [Google Scholar]
- Chang MH, Lee CY, Chen DS, Hsu HC, Lai MY 1987. Fulminant hepatitis in children in Taiwan: The important role of hepatitis B virus. J Pediatr 111: 34–39. [DOI] [PubMed] [Google Scholar]
- Chang MH, Chen DS, Hsu HC, Hsu HY, Lee CY 1989. Maternal transmission of hepatitis B virus in childhood HCC. Cancer 64: 2377–2380. [DOI] [PubMed] [Google Scholar]
- Chang MH, Chen CJ, Lai MS, Hsu HM, Wu TC, Kong MS, Liang DC, Shau WY, Chen DS, Taiwan Childhood Hepatoma Study Group. 1997. Universal hepatitis B vaccination in Taiwan and the incidence of hepatocellular carcinoma in children. N Engl J Med 336: 1855–1859. [DOI] [PubMed] [Google Scholar]
- Chang MH, You SL, Chen CJ, Liu CJ, Lee CM, Lin SM, Chu HC, Wu TC, Yang SS, Kuo HS, et al. 2009. Decreased incidence of hepatocellular carcinoma in hepatitis B vaccinees: A 20-year follow-up study. J Natl Cancer Inst 101: 1348–1355. [DOI] [PubMed] [Google Scholar]
- Chen DS 2005. Ling-term protection of hepatitis B vaccine: Lessons from Alaskan experience after 15 years. Ann Intern Med 142: 384–385. [DOI] [PubMed] [Google Scholar]
- Chen DS 2009. Hepatitis B vaccination: The key towards elimination and eradication of hepatitis B. J Hepatol 50: 805–816. [DOI] [PubMed] [Google Scholar]
- Chen DS 2011. Fighting against hepatitis B: Lessons from Taiwan. Hepatology 54: 381–392. [DOI] [PubMed] [Google Scholar]
- Chen DS, Hsu NHM, Sung JL, Hsu TC, Hsu ST, Kuo YT, Lo KJ, Shih YT 1987. The Hepatitis Steering Committee, The Hepatitis Control Committee: A mass vaccination program in Taiwan against hepatitis B virus infection in infants of hepatitis B surface antigen carrier-mothers. JAMA 257: 2597–2603. [PubMed] [Google Scholar]
- Chen HL, Chang MH, Ni YH, Hsu HY, Lee PI, Lee CY, Chen DS 1996. Seroepidemiology of hepatitis B virus infection in children: Ten years of mass vaccination in Taiwan. JAMA 276: 906–908. [PubMed] [Google Scholar]
- Chen HL, Lin LH, Hu FC, Lee JT, Lin WT, Yang YJ, Huang FC, Wu SF, Chen SC, Wen WH, et al. 2012. Effects of maternal screening and universal immunization to prevent mother-to-infant transmission of HBV. Gastroenterology 142: 773–781. [DOI] [PubMed] [Google Scholar]
- Chiang CJ, Yang YW, You SL, Lai MS, Chen CJ 2013. Thirty-year outcomes of the National Hepatitis B Immunization Program in Taiwan. JAMA 310: 974–976. [DOI] [PubMed] [Google Scholar]
- Hall AJ 2010. Boosters for hepatitis B vaccination? Need for an evidence-based policy. Hepatology 51: 1485–1486. [DOI] [PubMed] [Google Scholar]
- Han GR, Cao MK, Zhao W, Jiang HX, Wang CM, Bai SF, Yue X, Wang GJ, Tang X, Fang ZX 2011. A prospective and open-label study for the efficacy and safety of telbivudine in pregnancy for the prevention of perinatal transmission of hepatitis B virus infection. J Hepatol 55: 1215–1221. [DOI] [PubMed] [Google Scholar]
- Hilleman MR 1987. Yeast recombinant hepatitis B vaccine. Infection 15: 3–7. [DOI] [PubMed] [Google Scholar]
- Hsu HC, Lin GH, Chang MH, Chen HC 1983. Association of hepatitis B surface antigenemia and membranous nephropathy in children in Taiwan. Clin Nephrol 20: 121–129. [PubMed] [Google Scholar]
- Hsu HY, Chang MH, Chen DS, Lee CY, Sung JL 1986. Baseline seroepidemiology of hepatitis B virus infection in children in Taipei, 1984: A study just before mass hepatitis B vaccination program in Taiwan. J Med Virol 18: 301–307. [DOI] [PubMed] [Google Scholar]
- Hsu HM, Chen DS, Chuang CH, Lu JC, Jwo DM, Lee CC, Lu HC, Cheng SH, Wang YF, Wang CY 1988. Efficacy of a mass hepatitis B vaccination program in Taiwan: Studies on 3464 infants of hepatitis B surface antigen-carrier mothers. JAMA 260: 2231–2235. [PubMed] [Google Scholar]
- Hsu HY, Chang MH, Ni YH, Chiang CL, Chen HL, Wu JF, Chen PJ 2010. No increase in prevalence of hepatitis B surface antigen mutant in a population of children and adolescents who were fully covered by universal infant immunization. J Infect Dis 201: 1192–1200. [DOI] [PubMed] [Google Scholar]
- Hsu HY, Chang MH, Ni YH, Jeng YM, Chiang CL, Chen HL, Wu JF, Chen PJ 2013. Long-term follow-up of children with postnatal immunoprophylaxis failure who were infected with hepatitis B virus surface antigen gene mutant. J Infect Dis 207: 1047–1057. [DOI] [PubMed] [Google Scholar]
- Jan CF, Huang KC, Chien YC, Greydanus DE, Davies HD, Chiu TY, Huang LM, Chen CJ, Chen DS 2010. Determination of immune memory to hepatitis B vaccination through early booster response in college students. Hepatology 51: 1547–1554. [DOI] [PubMed] [Google Scholar]
- Kao JH, Chen DS 2008. Universal hepatitis B vaccination: Killing 2 birds with 1 stone. Am J Med 121: 1029–1031. [DOI] [PubMed] [Google Scholar]
- Kao JH, Hsu HM, Shau WY, Chang MH, Chen DS 2001. Universal hepatitis B vaccination and the decreased mortality from fulminant hepatitis in infants in Taiwan. J Pediatr 139: 349–352. [DOI] [PubMed] [Google Scholar]
- Kappus MR, Sterling RK 2013. Extrahepatic manifestations of acute hepatitis B virus infection. Gastroenterol Hepatol 9: 123–126. [PMC free article] [PubMed] [Google Scholar]
- Kruskall MS, Alper CA, Awdeh Z, Yunis EJ, Marcus-Bagley D 1992. The immune response to hepatitis B vaccine in humans: Inheritance patterns in families. J Exp Med 175: 495–502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lavanchy D 2012. Viral hepatitis: Global goals for vaccination. J Clin Virol 55: 296–302. [DOI] [PubMed] [Google Scholar]
- Lee GC, Hwang LY, Beasley RP, Chen SH, Lee TY 1983. Immunogenicity of hepatitis B virus vaccine in healthy Chinese neonates. J Infect Dis 148: 526–529. [DOI] [PubMed] [Google Scholar]
- Lee CY, Huang LM, Chang MH, Hsu CY, Wu SJ, Sung JL, Safary A 1991. The protective efficacy of recombinant hepatitis B vaccine in newborn infants of hepatitis B e antigen-positive-hepatitis B surface antigen carrier mothers. Pediatr Infect Dis J 10: 299–303. [DOI] [PubMed] [Google Scholar]
- Lerkhachonsuk AA, Yip CH, Khuhaprema T, Chen DS, Piummer M, Jee SH, Toi M, Willailak S, Asian Oncology Summit. 2013. Cancer prevention in Asia: Resource-stratified guidelines from the Asian Oncology Summit 2013. Lancet Oncol 14: 497–507. [DOI] [PubMed] [Google Scholar]
- Liao MT, Chang MH, Lin FG, Tsai IJ, Chang YW, Tsau YK 2011. Universal hepatitis B vaccination reduces childhood hepatitis B virus-associated membranous nephropathy. Pediatrics 128: e600–e604. [DOI] [PubMed] [Google Scholar]
- Lin HH, Lee TY, Chen DS, Sung JL, Ohto H, Etoh T, Kawana T, Mizuno M 1987. Transplacental leakage of HBeAg-positive maternal blood as the most likely route in causing intrauterine infection with hepatitis B virus. J Pediatr 111: 877–881. [DOI] [PubMed] [Google Scholar]
- Lin YC, Chang MH, Ni YH, Hsu HY, Chen DS 2003. Long-term immunogenicity and efficacy of universal hepatitis B virus vaccination in Taiwan. J Infect Dis 187: 134–138. [DOI] [PubMed] [Google Scholar]
- Lo KJ, Tsai YT, Lee SD, Yeh CL, Wang JY, Chiang BN, Wu TC, Yeh PS, Goudeau A, Coursaget P 1985. Combined passive and active immunization for interruption of perinatal transmission of hepatitis B virus in Taiwan. Hepatogastroenterology 32: 65–68. [PubMed] [Google Scholar]
- Martínez-Sernández V, Figueiras A 2013. Central nervous system demyelinating diseases and recombinant hepatitis B vaccination: A critical systematic review of scientific production. J Neurol 260: 1951–1959. [DOI] [PubMed] [Google Scholar]
- Maupas P, Chiron JP, Barin F, Coursaget P, Goudeau A, Perrin J, Denis F, Mair ID 1981. Efficacy of hepatitis B vaccine in prevention of early HBsAg carrier state in children. Controlled trial in an endemic area (Senegal). Lancet i: 289–292. [DOI] [PubMed] [Google Scholar]
- McMahon BJ, Bulkow LR, Singleton RJ, Williams J, Snowball M, Homan C, Parkinson AJ 2011. Elimination of hepatocellular carcinoma and acute hepatitis B in children 25 years after a hepatitis B newborn and catch-up immunization program. Hepatology 54: 801–807. [DOI] [PubMed] [Google Scholar]
- Ni YH, Chang MH, Huang LM, Chen HL, Hsu HY, Chiu TY, Tsai KS, Chen DS 2001. Hepatitis B virus infection in children and adolescents in a hyperendemic area: 15 years after mass hepatitis B vaccination. Ann Intern Med 135: 796–800. [DOI] [PubMed] [Google Scholar]
- Ni YH, Huang LM, Chang MH, Yen CJ, Lu CY, You SL, Kao JH, Lin YC, Chen HL, Hsu HY, et al. 2007. Two decades of universal hepatitis B vaccination in Taiwan: Impact and implication for future strategies. Gastroenterology 132: 1287–1293. [DOI] [PubMed] [Google Scholar]
- Ni YH, Chang MH, Wu JF, Hsu HY, Chen HL, Chen DS 2012. Minimization of hepatitis B infection by a 25-year universal vaccination program. J Hepatol 57: 730–735. [DOI] [PubMed] [Google Scholar]
- Peto TJ, Mendy ME, Lowe Y, Webb EL, Whittle HC, Hall AJ 2014. Efficacy and effectiveness of infant vaccination against chronic hepatitis B in the Gambia Hepatitis Intervention Study (1986–90) and in the nationwide immunisation program. BMC Infect Dis 14: 7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shiraki K, Yoshihara N, Sakurai M, Eto T, Kawana T 1980. Acute hepatitis B in infants born to carrier mother with the antibody to hepatitis B e antigen. J Pediatr 97: 768–770. [DOI] [PubMed] [Google Scholar]
- Stevens CE, Beasley RP, Tsui J, Lee WC 1975. Vertical transmission of hepatitis B antigen in Taiwan. N Engl J Med 292: 771–774. [DOI] [PubMed] [Google Scholar]
- Stevens CE, Taylor PE, Tong MJ, Toy PT, Vyas GN, Nair PV, Weissman JY, Krugman S 1987. Yeast-recombinant hepatitis B vaccine. Efficacy with hepatitis B immune globulin in prevention of perinatal hepatitis B virus transmission. JAMA 257: 2612–2616. [DOI] [PubMed] [Google Scholar]
- Su WJ, Liu CC, Liu DP, Chen SF, Huang JJ, Chan TC, Chang MH 2012. Effect of age on the incidence of acute hepatitis B after 25 years of a universal newborn hepatitis B immunization program in Taiwan. J Infect Dis 205: 757–62. [DOI] [PubMed] [Google Scholar]
- Szmuness W, Stevens CE, Harley EJ, Zang EA, Oleszko WR, William DC, Sadovsky R, Morrison JM, Kellner A 1980. Hepatitis B vaccine: Demonstration of efficacy in a controlled clinical trial in a high-risk population in the United States. N Engl J Med 303: 833–841. [DOI] [PubMed] [Google Scholar]
- Tang JR, Hsu HY, Lin HH, Ni YH, Chang MH 1998. Hepatitis B surface antigenemia at birth: A long-term follow-up study. J Pediatr 133: 374–377. [DOI] [PubMed] [Google Scholar]
- Tsai YT, Lo KJ, Lee SD, Wang JY, Wu TC 1984. Immunoprophylaxis against hepatitis B virus infection: A controlled trial in infants born to HBeAg-negative HBsAg carrier mothers in Taiwan. Chinese J Gastroenterol 1: 181–185. [Google Scholar]
- Valenzuela P, Medina A, Rutter WJ, Ammerer G, Hall BD 1982. Synthesis and assembly of hepatitis B virus surface antigen particles in yeast. Nature 298: 347–350. [DOI] [PubMed] [Google Scholar]
- van Herck K, Vorsters A, Van Damme P 2008. Prevention of viral hepatitis (B and C) reassessed. Best Pract Res Clin Gastroenterol 22: 1009–1029. [DOI] [PubMed] [Google Scholar]
- van Zonneveld M, van Nunen AB, Niesters HG, de Man RA, Schalm SW, Janssen HL 2003. Lamivudine treatment during pregnancy to prevent perinatal transmission of hepatitis B virus infection. J Viral Hepat 10: 294–297. [DOI] [PubMed] [Google Scholar]
- Wang CW, Wang LC, Chang MH, Ni YH, Chen HL, Hsu HY, Chen DS 2005. Long-term follow-up of hepatitis B surface antibody levels in subjects receiving universal hepatitis B vaccination in infancy in an area of hyperendemicity: Correlation between radioimmunoassay and enzyme immunoassay. Clin Diagn Lab Immunol 12: 1442–1447. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wasley A, Miller JT, Finelli L, Centers for Disease Control and Prevention (CDC). 2007. Surveillance for acute viral hepatitis—United States, 2005. MMWR Surveill Summary 56: 1–24. [PubMed] [Google Scholar]
- Wen WH, Chang MH, Zhao LL, Ni YH, Hsu HY, Wu JF, Chen PJ, Chen DS, Chen HL 2013. Mother-to-infant transmission of hepatitis B virus infection: Significance of maternal viral load and strategies for intervention. J Hepatol 59: 24–30. [DOI] [PubMed] [Google Scholar]
- Whittle HC, Maine N, Pilkington J, Mendy M, Fortuin M, Bunn J, Allison L, Howard C, Hall A 1995. Long-term efficacy of continuing hepatitis B vaccination in infancy in two Gambian villages. Lancet 345: 1089–92. [DOI] [PubMed] [Google Scholar]
- Wichajarn K, Kosalaraksa P, Wiangnon S 2008. Incidence of HCC in children in Khon Kaen before and after national hepatitis B vaccine program. Asian Pac J Cancer Prev 9: 507–509. [PubMed] [Google Scholar]
- Wu TW, Lin HH, Wang LY 2013. Chronic hepatitis B infection in adolescents who received primary infantile vaccination. Hepatology 57: 37–45. [DOI] [PubMed] [Google Scholar]
- Xu WM, Cui YT, Wang L, Yang H, Liang ZQ, Li XM, Zhang SL, Qiao FY, Campbell F, Chang CN, et al. 2009. Lamivudine in late pregnancy to prevent perinatal transmission of hepatitis B virus infection: A multicentre, randomized, double-blind, placebo-controlled study. J Viral Hepat 16: 94–103. [DOI] [PubMed] [Google Scholar]
- Yang JJ, Liu CC, Chen TJ, Lee MF, Chen SH, Shih HH, Chang MH 2003. Role of hepatitis B immunoglobulin in infants born to hepatitis B e antigen-negative carrier mothers in Taiwan. Pediatr Infect Dis J 22: 584–588. [DOI] [PubMed] [Google Scholar]