Abstract
Background:
During adolescence, sex hormones play an important role in regulating proliferation, differentiation, maturation, and the scheduled death of chondrocytes. Although some studies have reported the regulatory role of estrogen in the development and progression of cartilage, some of the mechanisms still remain unclear, including the role of estrogen in the expression of cartilage-specific genes in chondrogenesis process, which we cover in this study.
Materials and Methods:
In the present study, we used adipose-derived stem cells (ADSCs) to differentiate into cartilage. Differentiated cartilage cells were used in the control (without estrogen E2 in the culture medium) and experimental (with estrogen in the culture medium) groups to evaluate the expression of type II collagen and aggrecan as chondrogenic genes markers, with -real-time polymerase chain reaction technique.
Results:
Our results indicated that estrogen leads to inhibition of type II collagen gene expression and reduction of aggrecan gene expression.
Conclusion:
Therefore, estrogen probably has negative effects on chondrogenesis process of ADSCs.
Keywords: Adipose-derived stem cells, aggrecan, chondrogenesis, estrogen, type II collagen
INTRODUCTION
Cartilage is a specific connective tissue, and it is composed of chondrocytes and extracellular matrix. Chondrocytes derive from mesenchymal cells and occupy less than 5% of the total volume of cartilage.[1] Their main function is to maintain the extracellular matrix (ECM); the component of articular cartilage that provides a substrate for cell anchorage. The ECM consists of water (65%-80% of the total weight), proteoglycans (PG), collagens, and a variety of other proteins and glycoproteins. There are different types of collagen in articular cartilage such as types II (90%-95%), V, VI, IX, X, and XI.[2] The interfibrillar space of the cartilage matrix is occupied by aggrecan proteoglycan.[3,4]
Collagen as a sturdy fibrous protein prepares the main framework of cartilage, whereas proteoglycans serve as shock absorbers in the joint. Collagen forms a net-like structure in the joint to hold the proteoglycans and water. The shape and tensile strength of cartilage are due to this net. Spread of proteoglycans throughout the collagen net makes the cartilage a flexible and stretchable structure with movement.[2]
Under normal conditions, different components of the cartilage matrix continuously are turned over by chondrocytes in a lifetime.[5] Despite low metabolic activity and relatively poor ability to repair of articular cartilage, it is a dynamic and responsive tissue. The articular cartilage provides a wear-resistant and extremely smooth bearing surface for the joints to support and distribute loads and absorb shocks.[6]
Osteoarthritis is a joint inflammation that results from degeneration or loss of the articular cartilage as cushion between the joints. Osteoarthritis as a form of arthritis occurs when articular cartilage becomes damaged and useless and therefore size or volume of worn out cartilage decreases and it does not work efficiently. When the bones lose the articular cartilage in the joints in fact they lose their shock-absorbing buffers and start to rub against each other.[7]
The most appropriate treatment for this disease still is controversial. However, according to outcomes of increasing clinical experience, it has been demonstrated that implantation of autologous chondrocytes with or without a scaffold is a more effective choice to repair damaged articular cartilage.
Estrogen has been widely shown to acutely contribute to the regulation of bone growth and development. Estrogen directly affects chondrocyte proliferation, differentiation, and extracellular matrix (ECM) synthesis[8,9,10,11] and indirectly other hormones and local factors secreted by cells in response to estrogen stimulation.[12,13] The importance of understanding the effects of estrogen supplementation on skeletal mature tissues is evident. A change in estrogen concentrations during late maturation of an organism may have a significant effect on cartilage ECM, thereby influencing the load tolerance of articular cartilage during activity and its potential for degeneration.[14] Previously, researchers have reported that 17-β estradiol (E2) increased the type X collagen, inhibited the type II collagen, and reduced the proteoglycan content of the cartilage.[15,16]
Ray and coworkers have shown that estrogen receptors are present on stem cells.[17] Lanzl showed estradiol inhibits chondrogenic differentiation of mesenchymal stem cells via nonclassic signaling.[18] But so far, the direct effect of estrogen on chondrogenesis of adipose-derived stem cells (ADSCs) has not been investigated. For this purpose, in present study, after separation of ADSCs, these cells were cultured and differentiated to cartilage in the presence or the absence of estrogen and eventually the cartilage-specific gene expression was determined by real-time polymerase chain reaction (RT-PCR) method.
MATERIALS AND METHODS
Cell culture method
Mesenchymal stem cells were isolated from human subcutaneous adipose tissue (N = 4, age = 25-55 years) by enzymatic digestion 1 mg collagenase IA (Sigma, USA) per 1 g adipose tissue, at 37°C for 40 min. The cells were grown in DMEM (Dulbecco's Modified Essential Medium) (Sigma, USA) medium supplemented with 10% FBS (Fetal Bovine Serum) (Gibco, USA) and 1% penicillin/streptomycin (Gibco, USA), and were incubated at 37°C, and 5% CO2. The medium was changed twice a week.
Pellet culture system
To form a pellet, 2 × 105 cells/mL centrifuged in 15 mL polypropylene conical tube at 1500 rpm for 10 min. Then cells were suspended in chondrogenic medium, which consists of DMEM-high glucose (Gibco, USA), 1% insulin-transferrin-selenium (Sigma), 1.25 mg/mL bovine serum albumin (Sigma), 5 μg/mL linoleic acid (Sigma), 50 μg/mL ascorbate-2-phosphate (Sigma), 1% penicillin-streptomycin (Gibco), and 10 ng/mL transforming growth factor-β3 (Sigma). Pellets were incubated at 37°C, 5% CO2 for 14 days. The experimental group had 10−8 M estrogen E2 in medium. The culture medium was changed every 3 days.
Reverse transcription polymerase chain reaction method
RT-PCR method was used for assessing gene expression of cartilage-specific markers. For this purpose, the total inventory of cellular RNA was extracted by using RNX-plus kit according to the manufacturer's protocol (CinnaGen, Tehran, Iran). DNase I was used to remove contaminating genomic DNA. The RNA concentration was determined with a spectrophotometer at 260 nm wave length. cDNA was synthesized from total RNA using RevertAid First strand cDNA synthesis kit (Fermentase, Germany). The reaction mix and protocol were performed following the manufacturer's recommendations.
The product cDNA was used as template for PCR, which contains 2 μL cDNA, 1 × PCR buffer (10×), 0.75 Mm MgCl2, 0.5 Mm dNTPs, 1 μM of each primer pair, and 1 unit Taq DNA polymerase. Amplification conditions consisted of 95°C for 5 min followed by 29 cycles of 95°C 30s, 56°C 30 s, 72°C 45 s, followed by 5 min incubation at 72°C for final extension. Then PCR products visualized on 1.5% agarose gel. (The sequence of primers: Aggrecan: F -AGGAGGGCTGGAACAAGTACC, R - GGAGGTGGTAATTCCAGGGAACA; Collagen II: F - CAGGTCAAGATGGTC, R – TTCAGCACCTGTCTCACCA; GAPDH (Glyceraldehyde 3 Phosphate Dehydrogenase): F - GGGCTGCTTTTAACTCTGGT, R - GCAGGTTTTTCTAGACGG.)
RESULTS
Cell culture
Homogeneous ADSCs were obtained in the third passage with fibroblastic-like morphology [Figure 1].
Figure 1.
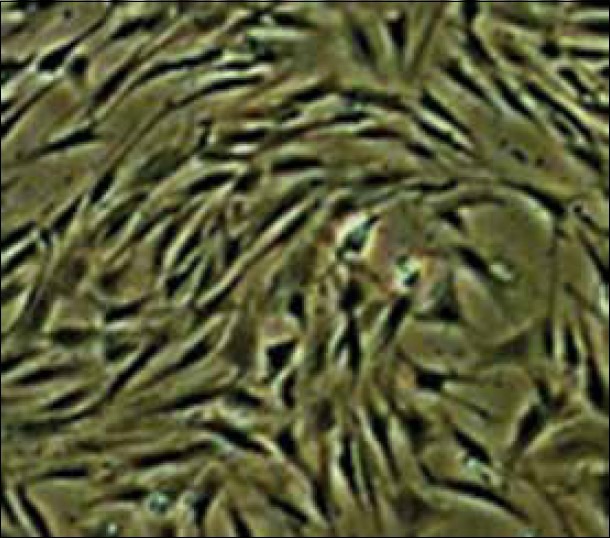
Adipose-derived stem cells indicated fibroblastic-like shape in third passage
Differentiated pellet masse after 2 weeks showed relative oval or round shape with less than 1 mm diameter [Figure 2].
Figure 2.
Tissue mass after chondrogenesis of adipose-derived stem cells in pellet culture system in conical tube
Review of the results of RT-PCR
In this study, we used ADSCs to differentiate into cartilage by differentiation factors. Cartilage differentiated cells in the control group (group without estrogen E2 in the culture medium) and experimental group (with estrogen E2 in the culture medium) were held in the freezer at − 70 to assess with RT-PCR method. By this method, the effects of estrogen on the expression of cartilage-specific genes in the process of chondrogenesis were evaluated.
For this aim, when RNA was extracted from differentiated cells, cDNA was synthesized and after PCR and electrophoresis the results obtained are shown in Figure 3. GAPDH was used as a house-keeping gene that expressed in both experimental and control groups.
Figure 3.
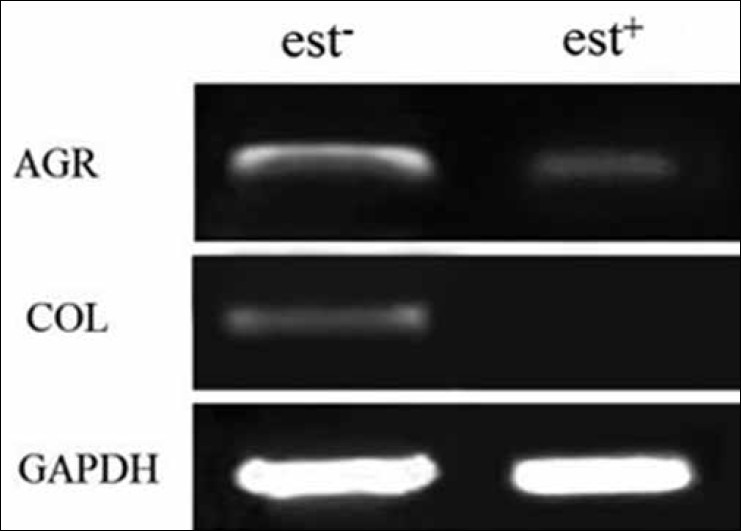
Polymerase chain reaction results: Results showed the aggrecan gene expression in the experimental group (with estrogen) was significantly lower than control (without estrogen) and collagen II was not expressed in experimental group. GAPDH was considered as house-keeping gene
Type II collagen expressed in the control group, but no band was observed in the experimental group. The expression of aggrecan was detected in both experimental and control groups.
By comparing the band intensity resulted from RT-PCR through Image J software and their relative mean with statistical t test, there was a significant difference of aggrecan expression between groups with estrogen and without it (P < 0.05).
Our data showed more expression of aggrecan gene in group without estrogen compared with experimental group [Figure 4].
Figure 4.

Comparing reverse transcription polymerase chain reaction results in the groups with and without estrogen. Aggracan gene expression in the group without estrogen was significantly higher than that in the group with estrogen, and this difference is statistically significant (P < 0.05)
DISCUSSION
For the first time in 1999, scientists examined the gene expression of the two estrogen receptors (ERα and ERβ) in the human cartilage.[19] Since then, numerous studies about the presence of estrogen receptors in human articular cartilage and cartilage growth plate have taken place.[20,21] Besides, the presence of estrogen receptors on the stem cells has been studied, MSCs are novel therapeutic agents for tissue engineering and by higher or lower production rate of cytokines and growth factors, estrogen may regulate the function of these cells.[18] There are evidences of estrogen role in the differentiation of MSCs to bone and that 17-β estradiol mediates growth and differentiation of the bone, particularly through the ERα receptor.[18] The effect of productive status on the proliferation rate and differentiation of stem cells has been investigated.[11,22,23] These primary studies have led to the necessity of further researches on the effect of estrogen on the process of the chondrogenesis of the ADSCs. Fat tissue is a very important source for the MSCs.[24,25,26,27] These multipotent stem cells are called ADSCs. Many factors may affect the proliferation rate and differentiation capacity of ADSCs, such as age of the cell donor; type of the used fat tissue (white or brown); the location of the fat tissue (subcutaneous adipose tissue or visceral fat); the process of tissue removal, isolation, and separation method; culture condition; and the density and formula of the medium.[20,21] In addition to these factors, some of the hormones such as estrogen can stimulate proliferation of the mouse ESC and influence the differentiation capacity of MSCs.[26]
To achieve the best chondrogenic medium for the stem cell differentiation into cartilage it is necessary to fully investigate the factors inducing the differentiation of these cells to cartilage. Considering that the estrogen is a factor that plays a very important role in the skeleton system during puberty, the effect of estrogen on the expression of some of the cartilage-specific genes in the process of chondrogenesis was studied in this article and the expression rate of these genes was compared with the medium lacking estrogen.
From the results of RT-PCR, the expression of specialized chondrogenic genes was clearly obvious in the samples without E2, whereas the expression of type II collagen and aggrecan genes in the presence of estrogen was different, meaning that aggrecan is expressed in the presence of estrogen but type II collagen in the samples treated with estrogen had no expression.
Unexpression of type II collagen, which is a specialized chondrogenic gene, may be directly related to the inhibitory effects of estrogen on the chondrogenesis. Consistent with our results, the inhibitory action of E2 on the chondrogenesis has been reported by other researchers.[18,23] However, the effect of E2 on specialized cartilage genes has not been investigated yet. Previous studies showed that estradiol inhibits the chondrogenic differentiation of the MSCs. Inhibitory effects of E2 are not only mediated by the classical cytoplasmic receptors, but also E2 can mediate the inhibition of the chondrogenic differentiation, mainly by the estrogen receptors coupled with GPR30 membrane. At lower concentration of E2 (less than 10−8), the membrane effects of E2 are replaced by ERα and ERβ intracellular classical pathways. So the inhibitory effect of estradiol at higher concentrations is mainly mediated by GPR30 membrane receptors. Therefore, the sensitivity of estrogen receptor is dose dependent.[23,28] In addition, the effects of E2 depend on the exposure time of the sample to the estrogen, gender, and chondrocytes maturation situation.[11] As a result, unexpression of type II collagen and decreased expression of aggrecan cause reduction of cartilage resistance and may lead to increased potential of cartilage damage.
In conclusion, in this study we investigated the influence of 17-β estradiol (E2) on chondrogenic differentiation of ADSCs and proved that the estrogen had inhibitory effect on the expression of type II collagen and also led to a reduction of the expression of aggrecan gene. Therefore, by inhibition of type II collagen and reduction of the expression of aggrecan gene, which are the most important cartilage maker genes, estrogen can inhibit the chondrogenic differentiation of the ADSCs. Thus it should not be considered as a suitable factor for chondrogenic differentiation of the stem cells.
Footnotes
Source of Support: Nil
Conflict of Interest: None declared.
REFERENCES
- 1.Mansour J, Mow VC. The permeability of articular cartilage under compressive strain and at high pressures. J Bone Joint Surg Am. 1976;58:509–16. [PubMed] [Google Scholar]
- 2.Junquera L, Carneiro J. New York: McGraw-Hill End II; 2005. Basic histology text and atlas; pp. 120–33. [Google Scholar]
- 3.Heinegård D, Franzén A, Hedbom E, Sommarin Y. Common structures of the core proteins of interstitial proteoglycans. Ciba Found Symp. 1986;124:69–88. doi: 10.1002/9780470513385.ch5. [DOI] [PubMed] [Google Scholar]
- 4.Buckwalter JA, Rosenberg LA, Hunziker EB. New York: Raven Press; 1990. Articular cartilage and knee joint function: Basic science and arthroscopy; p. 214. [Google Scholar]
- 5.Hardingham T, Tew S, Murdoch A. Tissue engineering: Chondrocytes and cartilage. Arthritis Res. 2002;4(Suppl 3):63–8. doi: 10.1186/ar561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Mitchell N, Shepard N. The resurfacing of adult rabbit articular cartilage by multiple perforations through the subchondral bone. J Bone Joint Surg Am. 1976;58:230–3. [PubMed] [Google Scholar]
- 7.Kuo CK, Li WJ, Mauck RL, Tuan RS. Cartilage tissue engineering: Its potential and uses. Curr Opin Rheumatol. 2006;18:64–73. doi: 10.1097/01.bor.0000198005.88568.df. [DOI] [PubMed] [Google Scholar]
- 8.Ross JL, Cassorla FG, Skerda MC, Valk IM, Loriaux DL, Cutler GB., Jr A preliminary study of the effect of estrogen dose on growth in Turner's syndrome. N Engl J Med. 1983;309:1104–6. doi: 10.1056/NEJM198311033091806. [DOI] [PubMed] [Google Scholar]
- 9.Cassorla FG, Skerda MC, Valk IM, Hung W, Cutler GB, Jr, Loriaux DL. The effects of sex steroids on ulnar growth during adolescence. J Clin Endocrinol Metab. 58:717–20. doi: 10.1210/jcem-58-4-717. [DOI] [PubMed] [Google Scholar]
- 10.Takahashi MM, Noumura T. Sexually dimorphic and laterally asymmetric development of the embryonic duck syrinx: Effects of estrogen on in vitro cell proliferation and chondrogenesis. Dev Biol. 1987;121:417–22. doi: 10.1016/0012-1606(87)90178-3. [DOI] [PubMed] [Google Scholar]
- 11.Nasatzky E, Schwartz Z, Boyan BD, Soskolne WA, Ornoy A. Sex-dependent effects of 17-beta-estradiol on chondrocyte differentiation in culture. J Cell Physiol. 1993;154:359–67. doi: 10.1002/jcp.1041540220. [DOI] [PubMed] [Google Scholar]
- 12.Gray TK, Flynn TC, Gray KM, Nabell LM. 17 beta-estradiol acts directly on the clonal osteoblastic cell line UMR106. Proc Natl Acad Sci USA. 1987;84:6267–71. doi: 10.1073/pnas.84.17.6267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Gray TK. Estrogens and the skeleton: Cellular and molecular mechanisms. J Steroid Biochem. 1989;34:285–7. doi: 10.1016/0022-4731(89)90093-9. [DOI] [PubMed] [Google Scholar]
- 14.Räsänen T, Messner K. Articular cartilage compressive stiffness following oophorectomy or treatment with 17 beta-estradiol in young postpubertal rabbits. Acta Obstet Gynecol Scand. 1999;78:357–62. [PubMed] [Google Scholar]
- 15.Talwar RM, Wong BS, Svoboda K, Harper RP. Effects of estrogen on chondrocyte proliferation and collagen synthesis in skeletally mature articular cartilage. J Oral Maxillofac Surg. 2006;64:600–9. doi: 10.1016/j.joms.2005.12.006. [DOI] [PubMed] [Google Scholar]
- 16.Hashem G, Zhang Q, Hayami T, Chen J, Wang W, Kapila S. Relaxin and beta-estradiol modulate targeted matrix degradation in specific synovial joint fibrocartilages: Progesterone prevents matrix loss. Arthritis Res Ther. 2006;8:R98. doi: 10.1186/ar1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ray R, Novotny NM, Crisostomo PR, Lahm T, Abarbanell A, Meldrum DR. Sex steroids and stem cell function. Mol Med. 2008;14:493–501. doi: 10.2119/2008-00004.Ray. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Jenei-Lanzl Z, Straub RH, Dienstknecht T, Huber M, Hager M, Grässel S, et al. Estradiol inhibits chondrogenic differentiation of mesenchymal stem cells via nonclassic signaling. Arthritis Rheum. 2010;62:1088–96. doi: 10.1002/art.27328. [DOI] [PubMed] [Google Scholar]
- 19.Ushiyama T, Ueyama H, Inoue K, Ohkubo I, Hukuda S. Expression of genes for estrogen receptors alpha and beta in human articular chondrocytes. Osteoarthritis Cartilage. 1999;7:560–6. doi: 10.1053/joca.1999.0260. [DOI] [PubMed] [Google Scholar]
- 20.Den F, Ola N. The role of estrogen in growth plate chondrogenesis. Karolinska Institutet. 2002;91:7349–410. [Google Scholar]
- 21.Richmond RS, Carlson CS, Register TC, Shanker G, Loeser RF. Functional estrogen receptors in adult articular cartilage: Estrogen replacement therapy increases chondrocyte synthesis of proteoglycans and insulin-like growth factor binding protein 2. Arthritis Rheum. 2000;43:2081–90. doi: 10.1002/1529-0131(200009)43:9<2081::AID-ANR20>3.0.CO;2-I. [DOI] [PubMed] [Google Scholar]
- 22.Hong SH, Nah HY, Lee Y, Lee JW, Park JH, Kim SJ, et al. Expression of estrogen receptor-alpha and -beta, glucocorticoid receptor, and progesterone receptor genes in human embryonic stem cells and embryoid bodies. Mol Cells. 2004;18:320–5. [PubMed] [Google Scholar]
- 23.Ng LW, Yip SK, Wong HK, Yam GH, Liu YM, Lui WT, et al. Adipose-derived stem cells from pregnant women show higher proliferation rate unrelated to estrogen. Hum Reprod. 2009;24:1164–70. doi: 10.1093/humrep/dep001. [DOI] [PubMed] [Google Scholar]
- 24.Fushimi S, Wada N, Nohno T, Tomita M, Saijoh K, Sunami S, et al. 17beta-estradiol inhibits chondrogenesis in the skull development of zebrafish embryos. Aquat Toxicol. 2009;95:292–8. doi: 10.1016/j.aquatox.2009.03.004. [DOI] [PubMed] [Google Scholar]
- 25.Hashemibeni B, Razavi SH, Esfandiary E, Karbasi S, Sadeghi F, Mardani M, et al. Effect of TGF-β3 and BMP-6 growth factors on chondrogenic differentiation of adipose-derived stem cells in alginate scaffold. JIMS. 2010;28:607–20. [Persian] [Google Scholar]
- 26.Hashemibeni B, Razavi SH, Esfandiarym E, Karbasi S, Mardani M, Nadali F, et al. Human cartilage tissue engineering from adipose-derived stem cells with BMP-6 in alginate scaffold. J Iran Anatomical Sci. 2010;31:126–36. [Persian] [Google Scholar]
- 27.Zuk P, Zhu M, Ashjian P, De Ugarte DA, Huang JI, Mizuno H, et al. Human adipose tissue is a source of multipotent stem cells. Mol Biol Cell. 2002;13:4279–95. doi: 10.1091/mbc.E02-02-0105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Takano H, Aizawa T, Irie T, Itoi E, Kokubun S, Roach HI. Normal bone growth requires optimal estrogen levels: Negative effects of both high and low dose estrogen on the number of growth plate chondrocytes. Tohoku J Exp Med. 2008;214:269–80. doi: 10.1620/tjem.214.269. [DOI] [PubMed] [Google Scholar]


