Abstract
Background
Complete blood count (CBC) reference intervals are important to diagnose diseases, screen blood donors, and assess overall health. However, current reference intervals established by older instruments and technologies and those from American and European populations are not suitable for Chinese samples due to ethnic, dietary, and lifestyle differences. The aim of this multicenter collaborative study was to establish CBC reference intervals for healthy Han Chinese adults.
Methods
A total of 4,642 healthy individuals (2,136 males and 2,506 females) were recruited from six clinical centers in China (Shenyang, Beijing, Shanghai, Guangzhou, Chengdu, and Xi’an). Blood samples collected in K2EDTA anticoagulant tubes were analyzed. Analysis of variance was performed to determine differences in consensus intervals according to the use of data from the combined sample and selected samples.
Results
Median and mean platelet counts from the Chengdu center were significantly lower than those from other centers. Red blood cell count (RBC), hemoglobin (HGB), and hematocrit (HCT) values were higher in males than in females at all ages. Other CBC parameters showed no significant instrument-, region-, age-, or sex-dependent difference. Thalassemia carriers were found to affect the lower or upper limit of different RBC profiles.
Conclusion
We were able to establish consensus intervals for CBC parameters in healthy Han Chinese adults. RBC, HGB, and HCT intervals were established for each sex. The reference interval for platelets for the Chengdu center should be established independently.
INTRODUCTION
Complete blood count (CBC) reference intervals are essential for effectively diagnosing diseases, screening blood donors, and assessing overall health. However, critical accuracy gaps exist in the reference intervals that are currently used in China. Most available reference intervals for laboratory tests are outdated or adopted from the manufacturers of the diagnostic tests. Over the past 30 years, the nutritional status and lifestyles of Chinese people have undergone dramatic changes. Thus, the reference intervals established using older instruments and technologies might no longer be relevant. In addition, manufacturers’ reference intervals, which were predominantly established for North American and European populations, may not account for variations due to race, diet, and lifestyle. Therefore, there is an urgent need to establish accurate CBC reference intervals for Chinese people.
The use of different methods, instruments, and populations can lead to significant differences in CBC parameters, such as hemoglobin (HGB), hematocrit (HCT), red blood cell count (RBC), and white blood cell count (WBC) [1, 2]. Advances in standardizing methods and laboratory techniques have minimized the effects of site-to-site differences in many analytical methods [3]. Population is one of the key remaining variables that can influence a reference interval. The ethnic Han population is the predominant population in Mainland China. Therefore, a common reference interval for CBC parameters in Han Chinese adults should be established via multicenter collaborative studies using uniform standards.
MATERIALS AND METHODS
Ethical considerations
The Ministry of Health and Clinical Laboratory Branch of the Chinese Medical Association supported a multicenter research study to establish reference intervals for major clinical laboratory items in the Han Chinese population. Six centers were selected to participate in this research: the Second Affiliated Hospital of Guangzhou University of Chinese Medicine, First Affiliated Hospital of China Medical University, Zhongshan Hospital of Fudan University, Peking University Third Hospital, West China Hospital of Sichuan University, and Xijing Hospital of the Fourth Military Medical University. Each hospital was responsible for recruiting healthy local candidates, collecting samples, and performing tests. The National Center of Clinical Laboratories (NCCL) was responsible for project quality assurance.
This study was approved by the Research and Ethics committees of the six participating hospitals. Each participant signed consent forms before undertaking any study-related activities.
Subject enrollment
Healthy ethnic Han male and female candidates between 20 and 79 years of age were screened and enrolled across the six clinical research centers, representing the six official geographical regions in China (Shenyang, Beijing, Shanghai, Guangzhou, Chengdu, and Xi’an). At enrollment, demographic information and medical history were collected, a physical examination was performed, and blood and urine samples were obtained. After prescreening, potentially eligible subjects with positive laboratory screening results were excluded based on the following criteria:
Diagnosed disease, including acute or chronic infection, atherosclerosis, empyrosis, renal failure, hyper- or hypothyroidism, diabetes, tumor, or hepatic, heart, allergic, hematopoietic, or respiratory disease;
Surgery within 6 months after enrollment;
Blood donation or transfusion within 4 months of enrollment;
Body mass index exceeding 28 kg/m2;
Pregnancy;
Hypertension;
Use of alcohol, tobacco, or oral contraceptives;
Positivity for hepatitis B surface antigen (HBsAg), hepatitis C antibody (anti-HCV), or human immunodeficiency virus antibody (anti-HIV);
Abnormal urinalysis results, including the presence of erythrocytes, granulocytes, glucose, protein, or nitrite;
Plasma fasting blood glucose (FBG) level exceeding 7.0 mmol/L;
Serum iron level lower than 6 μmol/L;
HGB concentration less than 90 g/L;
WBC less than 3.0×109/L or more than 12.5×109/L; and
Thalassemia trait (TT) or carrier (Guangzhou and Chengdu centers only).
From September 2010 to January 2011, 18,000 volunteers were recruited. Of them, 7,612 volunteers (42%) were enrolled, but 4,642 volunteers (26%; 2,136 males and 2,506 females) were included in the final analysis to create the CBC reference intervals. A large number of volunteers were excluded because of abnormal physical examination results or positive laboratory screening results. Reference subjects were divided into six age groups: 20–29, 30–39, 40–49, 50–59, 60–69, and 70–79 years (Table 1). The ratio of subjects from the city and surrounding rural area was 7:3.
Table 1. Population Demographics for Establishing the Reference Range by Center.
| ClinicalCenter | Number | Sex | Age (years) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| M | F | Mean | 20–29 | 30–39 | 40–49 | 50–59 | 60–69 | 70–79 | |||
| Shenyang | 861 | 368 | 493 | 43.9 | 186 | 145 | 203 | 188 | 97 | 42 | |
| Beijing | 760 | 340 | 420 | 43.7 | 177 | 155 | 136 | 148 | 83 | 61 | |
| Shanghai | 736 | 292 | 444 | 44.0 | 156 | 146 | 129 | 188 | 101 | 16 | |
| Guangzhou | 945 | 446 | 499 | 47.8 | 179 | 162 | 159 | 162 | 149 | 134 | |
| Chengdu | 644 | 331 | 313 | 41.4 | 157 | 150 | 157 | 114 | 53 | 13 | |
| Xi’an | 696 | 359 | 337 | 38.4 | 228 | 162 | 128 | 121 | 44 | 13 | |
| Total | 4 642 | 2 136 | 2 506 | 43.5 | 1 083 | 920 | 912 | 921 | 527 | 279 | |
Instruments and measurement parameters
All six centers measured CBC parameters with XE-2100 (Sysmex Corp., Kobe, Japan) and BC-5800 (Mindray Medical Electronics Co., Shenzhen, China) hematology analyzers. Measured CBC parameters included RBC, HGB, HCT, mean corpuscular volume (MCV), mean corpuscular hemoglobin (MCH), MCH concentration (MCHC), platelet count (PLT), WBC, and WBC differentials, including percentages and absolute counts of neutrophils (NEUT% and NEUT), lymphocytes (LYM% and LYM), monocytes (MONO% and MONO), basophils (BASO% and BASO), and eosinophils (EO% and EO).
Sample collection and analysis
Each participant fasted from food and water for at least 8 hours but no more than 14 hours. Blood from each participant was drawn from the cubital vein into appropriate blood collection tubes using vacuum tube needles (Becton Dickinson Medical Devices Co. Ltd., Franklin Lakes, USA). K2EDTA tubes were used for CBC and DNA analyses. Fluoride oxalate tubes were used for FBG analysis. Plain tubes were used for estimating serum iron, anti-HCV, anti-HIV, and HBsAg levels. Samples collected in fluoride oxalate or plain tubes were separated by centrifugation for 10 minutes at 3,000 rpm. All enrolled subjects provided urine samples for urinalysis. Samples were transported and tested within 4 hours after collection.
Among the six centers, the prevalence of thalassemia was high in Guangzhou and Chengdu [4]. At these centers, an MCV value of less than 82 femtoliters (fL) promoted DNA analysis to screen for thalassemia carriers. Gap-polymerase chain reaction (PCR) was used to detect three routine gene deletion types in the α-thalassemia gene (-α3.7, -α4.2, and —SEA). Seventeen common mutants of β-thalassemia in the Chinese population were detected by PCR reverse dot-blot hybridization (PCR-RDH; YANENG Bioscience Co. Ltd., Shenzhen, China) [5,6].
Quality assurance
According to Clinical and Laboratory Standards Institute (CLSI) guideline C28-A3 [3], the compatibility of different measurement systems must be established when a multicenter reference interval study is performed. Before collecting samples for final analysis, the performance of the automated hematology analyzers was evaluated. After calibration by a field service representative, the imprecision, carry over, and linearity of the analyzers were evaluated according to the International Council for Standardization in Haematology (ICSH) guidelines [7]. Accuracy tests were performed monthly throughout the entire study, in accordance with a standard protocol. Fresh K2EDTA anticoagulant specimens were prepared by the NCCL and tested within 8 hours after sample collection. Test results were compared with reference values defined by the NCCL. The acceptable range of bias for WBC, RBC, HCT, HGB, and PLT was half of the allowable error described by the Clinical Laboratory Improvement Amendments of 1988 (CLIA 88) [8].
To ensure standardization, all laboratory staff received equipment training and were required to pass an independent quality review before enrolling volunteers. The procedure and timing of blood sample collection were standardized to minimize the preanalytical variables. Commercial control samples at three different levels (high, medium, and low) were measured before and after every analytical run.
Statistical analysis
Data were analyzed with the Statistical Package for Social Sciences (SPSS version 17.0) and Microsoft Excel 2003. Outliers were excluded by the one-third rule for the D/R ratio, where D is the absolute difference between an extreme observation (large or small) and the next largest (or smallest) observation, and R is the range of all observations, including extremes [3]. Multiple linear regression was performed to explore impact factors. Step-wise regression was used to screen variables. Nested analysis of variance (ANOVA) was performed to analyze data by instrument, clinical center, age, and sex. For parameters with a skewed distribution, all ANOVA tests were performed after a log transformation, and the geometric means were compared instead of the arithmetic means.
If the overall F-test from an ANOVA on mean values was statistically significant (P < 0.05), then a step-wise procedure was used to determine which intervals were close enough to combine into a “consensus interval” [9]. First, we compared the intervals between the two most similar sites based on the P-values from the overall ANOVA adjusted for multiple comparisons by the Tukey method. Data from the two sites were combined if: 1) the difference was not statistically significant, 2) the difference between the means was significant, but was less than 25% of the width of the 95% reference interval estimated from the combined sample, and the ratio of standard deviations (SDs) was less than 1.5. The combined data were compared to each of the remaining sites as described above, using a new ANOVA. These steps were repeated until all of the sites were combined into or excluded from the consensus interval. Finally, the consensus intervals for men and women were compared and combined, if appropriate, in the same manner. Nonparametric methods were used to establish the CBC consensus intervals, which were calculated using CBC values between the 2.5th and 97.5th percentiles that included 95% of the reference sample group data.
Results
Region-related findings
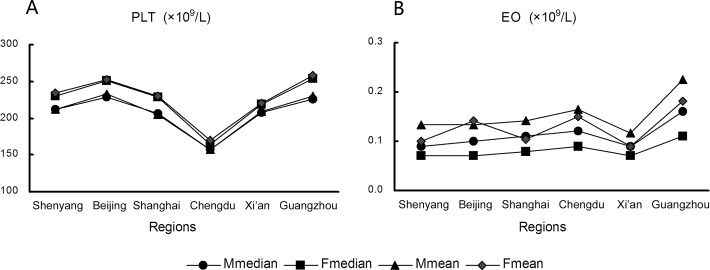
Median and mean values of the PLT parameter in samples from the Chengdu center were significantly lower than values from other centers (Fig. 1A; P < 0.05). The difference between means was greater than 25% of the width of the 95% reference interval estimated from the combined sample data; therefore, data from the Chengdu center were excluded from the consensus interval for PLT. Median and mean EO values were significantly greater in samples from the Guangzhou center than in those from other centers (Fig. 1B; P < 0.05). However, the difference between means was minor, and EO data from all centers could be combined to calculate the consensus interval.
Fig 1. Median and mean platelet counts (PLT, A) and eosinophil (EO, B) values for different regions and sexes.
Instrument-related findings
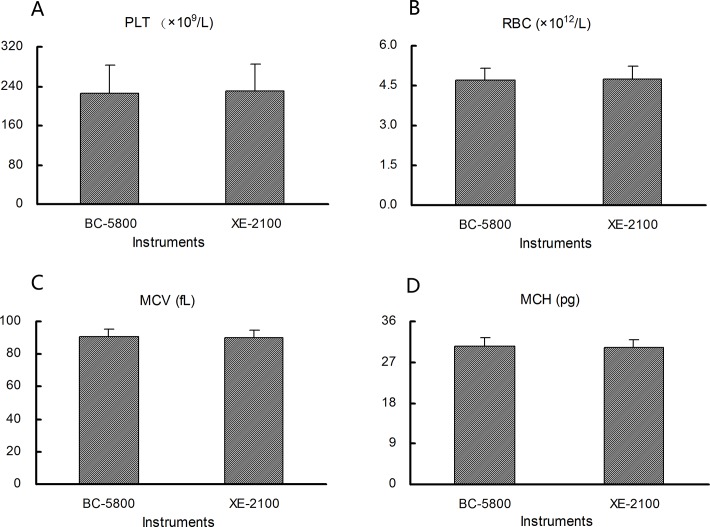
No statistically significant differences in the RBC, MCV, MCH, and PLT parameters were observed between the Mindray BC-5800 and SYSMEX XE-5000 analyzers (Fig. 2A-2D; P > 0.05). Similar results were obtained for HGB, HCT, MCHC, WBC, and WBC differentials between the two instruments (data not shown; P > 0.05). Thus, the data obtained for each parameter from each instrument could be pooled to determine the consensus intervals.
Fig 2. Mean platelet count (PLT, A), red blood cell count (RBC, B), mean corpuscular volume (MCV, C), and mean corpuscular hemoglobin (MCH, D) value according to the use of different instruments.
Age- and sex-related findings
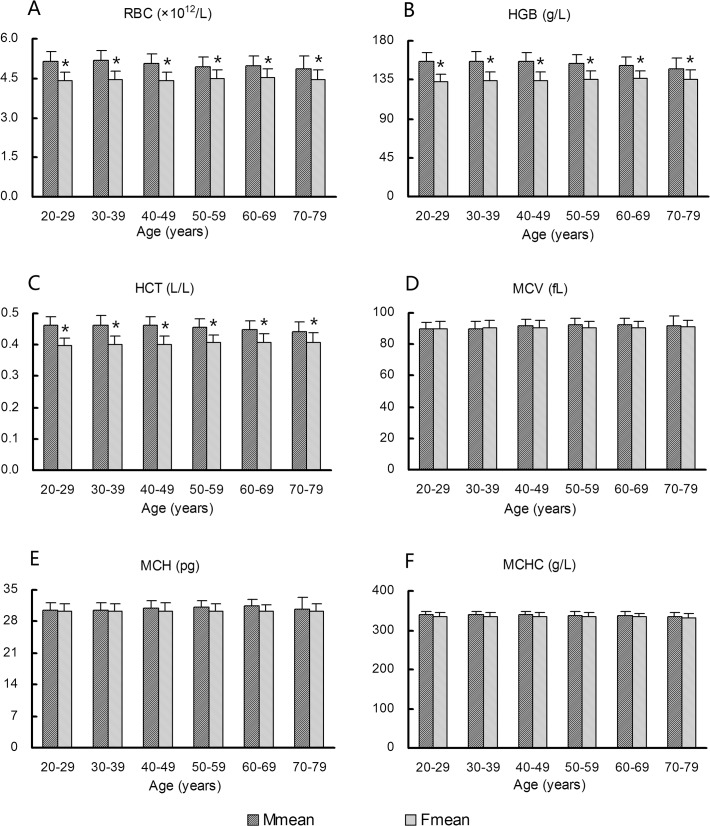
Fig. 3A-3C display mean values of RBC, HGB, and HCT according to age and sex. A significant difference between sexes (P < 0.05) and a minor difference among age groups were observed. Thus, RBC, HGB, and HCT data from different age groups were pooled, but the sex distinction was preserved when establishing reference intervals. RBC, HGB, and HCT values tended to decrease with age in males and were obviously reduced in the 70–79 age group, and these values tended to increase slightly with age in females and were lower than values in males in all age groups. Mean values of MCV, MCH, and MCHC did not differ according to age or sex in the combined analysis of all data (Fig. 3D-3F).
Fig 3. Variations in red blood cell (RBC) profile according to age and sex.
(A) RBC, (B) hemoglobin (HGB), (C) hematocrit (HCT), (D) mean corpuscular volume (MCV), (E) mean corpuscular hemoglobin (MCH), (F) MCH concentration (MCHC). *Females show significantly lower values (P < 0.05) than males in the same age group.
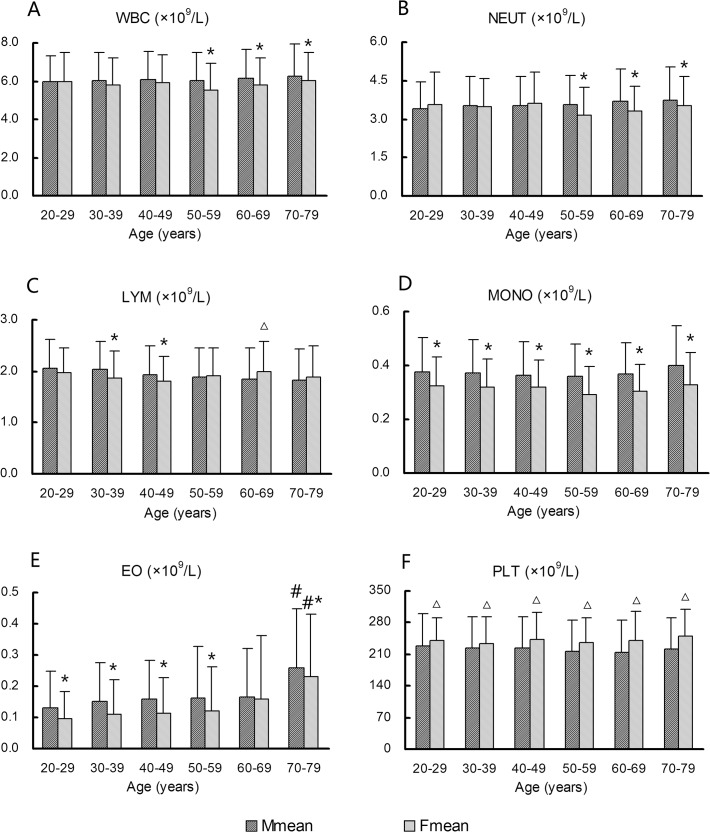
Mean values for the WBC and NEUT parameters were slightly increased in males over 60 years, and were higher in males than in females over the age of 50 years (Fig. 4A and B, P < 0.05). Conversely, LYM values showed a decreasing trend with age in males (Fig. 4C). EO values in males and females tended to increase with age, with an apparent increase in the 70–79-year age group (P < 0.05). EO and MONO counts appeared to be consistently lower in females than in males across all age groups (Fig. 4D and 4E, P < 0.05). In contrast, mean platelet counts were significantly higher in females than in males in all age groups (Fig. 4F, P < 0.05). However, these differences were not sufficiently significant to prompt exclusion from study consensus intervals. Apart from RBC, HGB, and HCT, data for other CBC parameters could be combined to calculate consensus intervals, regardless of age or sex.
Fig 4. Variations in white blood cell (WBC) profile and platelet count (PLT) according to age and sex.
(A) WBC, (B) neutrophils (NEUT), (C) lymphocytes (LYM), (D) monocytes (MONO), (E) eosinophils (EO), (F) PLT. *Females show significantly lower values (P < 0.05) than males in the same age group. △Females show significantly higher values (P < 0.05) than males in the same age group. #EO values are significantly higher (P < 0.05) in the 70–79-year age group than in other age groups in males and females, respectively.
Relationship between TT and reference intervals for RBC parameters
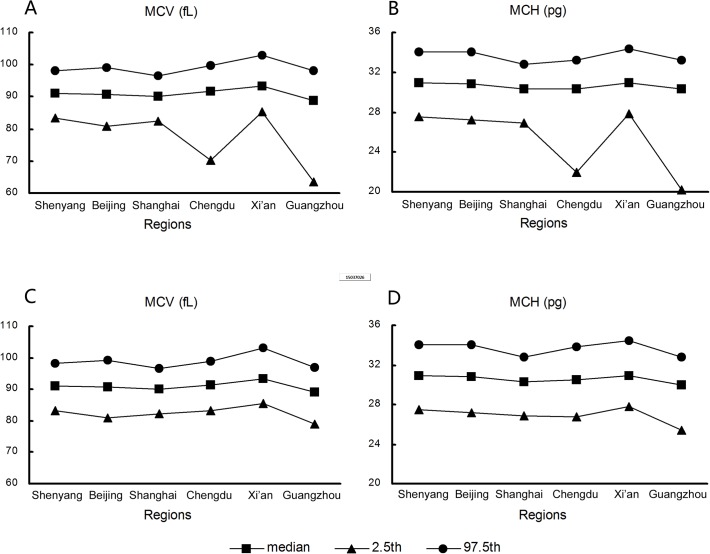
At the Guangzhou and Chengdu centers, TT carriers were identified based on MCV values and thalassemia gene DNA analysis. TT carriers accounted for 10.5% (170/1,622) and 4.3% (39/912) of subjects from these centers, respectively. Inclusion of TT carriers from these centers in the final analysis clearly affected the lower limits of the MCV, HGB, and MCH parameters and the upper limit of the RBC parameter (Table 2, Fig. 5A and 5B), but had little or no impact on the HCT and MCHC intervals. After the exclusion of TT carriers, differences in RBC, HGB, MCV, and MCH percentiles among the six centers were minor, and all data could be combined to determine the consensus intervals for each parameter (Fig. 5C and 5D). TT carriers had an obvious effect on the 2.5th percentiles, but negligible impacts on the median and 97.5th percentiles of the MCV and MCH intervals.
Table 2. Reference Intervals for RBC Profiles with and without TT Carriers.
| Parameters | Sex | Chengdu | Guangzhou | |||
|---|---|---|---|---|---|---|
| With TT | Without TT | With TT | Without TT | |||
| RBC (×1012/L) | M | 4.29–6.20 | 4.29–5.66 | 4.34–6.88 | 4.31–5.94 | |
| RBC (×1012/L) | F | 3.83–5.02 | 3.83–4.95 | 3.92–5.83 | 3.91–5.24 | |
| HGB (g/L) | M | 129–171 | 133–171 | 125–176 | 133–179 | |
| HGB (g/L) | F | 109–147 | 110–147 | 111–153 | 115–153 | |
| HCT (L/L) | M | 0.40–0.51 | 0.41–0.51 | 0.39–0.51 | 0.40–0.51 | |
| HCT (L/L) | F | 0.35–0.45 | 0.35–0.45 | 0.34–0.45 | 0.35–0.45 | |
| MCV (fL) | M/F | 74.0–98.9 | 83.6–99.0 | 64.0–96.6 | 78.6–96.8 | |
| MCH (pg) | M/F | 22.7–32.7 | 26.5–32.7 | 20.3–32.8 | 25.4–32.9 | |
| MCHC (g/L) | M/F | 308–344 | 310–345 | 311–356 | 317–356 | |
Fig 5. Mean corpuscular volume (MCV) and mean corpuscular hemoglobin (MCH) reference intervals for samples with and without thalassemia trait (TT).
Percentiles for (A) MCV with TT, (B) MCH with TT, (C) MCV without TT, (D) MCH without TT according to regions.
Establishing final consensus intervals
Apart from RBC, HGB, and HCT, the consensus CBC parameters did not differ by sex. Consensus intervals obtained by using the 2.5th and 97.5th percentiles as the lower and upper limits, respectively, are shown in Table 3. According to the CLSI C28-A3 guideline [3], CBC consensus reference intervals may be required to validate the transference. For the previously described screening standards, data from at least 20 new male and female subjects were collected from each of the six centers to verify the consensus intervals. A total of 309 individuals (149 males and 160 females) were selected to verify the consensus intervals. The qualified rate of validation for all of the analytes was not less than 95%. The analytes that most commonly exceeded the limits of the consensus intervals were HCT and MCHC, which were the two calculated parameters. Other previously published consensus intervals are shown for comparison in Table 3.
Table 3. Consensus intervals, validation rates, and comparative interval.
| Parameter | Sex | Consensus interval | Qualified rate of validation | Current interval used in China | American | Malaysian | African |
|---|---|---|---|---|---|---|---|
| RBC (×1012/L) | M | 4.28–5.81 | 97.3% | 4.09–5.74 | 4.5–5.9 | 4.18–6.06 | 4.0–6.4 |
| RBC (×1012/L) | F | 3.81–5.13 | 96.3% | 3.68–5.13 | 4.0–5.2 | 3.52–5.16 | 3.8–5.6 |
| HGB (g/L) | M | 133–175 | 99.3% | 131–172 | 135–175 | 120–165 | 122–177 |
| HGB (g/L) | F | 115–152 | 96.9% | 113–151 | 120–160 | 98–138 | 95–158 |
| HCT (L/L) | M | 0.40–0.51 | 95.3% | 0.380–0.508 | 0.41–0.53 | 0.375–0.498 | 0.35–0.51 |
| HCT (L/L) | F | 0.35–0.46 | 96.3% | 0.335–0.450 | 0.36–0.46 | 0.318–0.424 | 0.29–0.45 |
| MCV (fL) | M/F | 82.3–99.2 | 98.1% | M:83.9–99.1/F:82.6–99.1 | 80–100 | M:78.9–95.7/F:77.5–94.5 | 68–98 |
| MCH (pg) | M/F | 27.0–33.7 | 98.4% | M:27.8–33.8/F:26.9–33.3 | - | M:25.4–31.1/F:24.8–31.2 | - |
| MCHC (g/L) | M/F | 316–354 | 95.1% | M:320–355/F:322–362 | - | M:30.6–34.8/F:29.4–64.4 | - |
| WBC (×109/L) | M/F | 3.64–9.39 | 98.7% | M:3.97–9.15/F:3.69–9.16 | 4.5–11.0 | M:3.8–9.7/F:3.4–10.1 | 3.1–9.1 |
| NEUT (%) | M/F | 41.5–73.8 | 99.0% | 50–70 | 40–70 | M:42.8–69.2/F:43.2–70.6 | 25–66 |
| LYM (%) | M/F | 18.6–48.7 | 98.7% | 20–40 | 22–44 | M:18.5–47.7/F:19.2–47.5 | 23–59 |
| MONO (%) | M/F | 3.2–9.5 | 97.1% | 3–10 | 4–11 | - | 4.5–13.1 |
| EO (%) | M/F | 0.4–8.1 | 97.4% | 0.5–5 | 0–8 | - | 0.8–21.8 |
| BASO (%) | M/F | 0.1–1.1 | 99.7% | 0–1 | 0–3 | - | 0.4–2.5 |
| NEUT (×109/L) | M/F | 1.80–6.30 | 99.0% | 2.0–7.0 | 1.8–7.7 | M:1.58–5.94/F:1.55–6.07 | 1.0–5.3 |
| LYM (×109/L) | M/F | 1.06–3.20 | 98.4% | 0.8–4.0 | 1.0–4.8 | M:1.14–3.22/F:1.05–3.29 | 1.2–3.7 |
| MONO (×109/L) | M/F | 0.16–0.62 | 98.1% | 0.12–1.00 | 0–0.8 | M:0.15–0.67/F:0.1–0.74 | 0.20–0.78 |
| EO (×109/L) | M/F | 0.02–0.52 | 98.1% | 0.02–0.50 | 0–0.45 | M:0.08–0.28/F:0.03–0.27 | 0.04–1.53 |
| BASO (×109/L) | M/F | 0.00–0.06 | 99.4% | 0–1 | 0–0.2 | M:0.01–0.05/F:0.01–0.05 | 0.01–0.15 |
| PLT (×109/L)* | M/F | 127–350 | 97.4% | M:85–303/F:101–320 | 150–350 | M:167–376/F:158–410 | 126–438 |
| PLT (×109/L) # | M/F | 67–287 | 100% |
*: Five centers after exclusion of Chengdu,
#: only Chengdu center.
Discussion
Reference methods for measuring the RBC, WBC, HGB, HCT and PLT parameters have been previously published by the ICSH [10–13]. The published values are comparable regardless of analyzer, reagent, or analytical principle. Consequently, the values from different laboratories or instruments are considered interchangeable, and it is possible to combine data from multiple centers to establish consensus reference intervals. Once the consensus reference intervals are established, receiving laboratories must only validate them by examining a small number (e.g., 20) of reference individuals from their subject populations [3].
There is relatively little published data available describing the CBC reference intervals for people in China. Following the CLSI guidelines and standardization protocol, we established consensus reference intervals for the ethnic Han population, which represents 98% of the population in China. To ensure the accuracy and validity of the results, quality assurance steps were taken in the preanalytical and analytical stages. We established CBC consensus intervals using statistical calculations and verified these intervals with a smaller sample of reference subjects.
CBC values are influenced by several factors, including race, age, sex, altitude, and smoking [14–16]. In this study, the influences of instrument type, age, sex, and region on CBC reference intervals were analyzed. Of the CBC parameters, only PLT showed significant regional variability. PLT values from male and female subjects in Chengdu were significantly lower than values from subjects at other sites and, therefore, were not included in the consensus intervals. The reason for the lower PLT values remains unclear, but could be partially attributable to the humid climate and lack of sunshine in the Sichuan basin. Significant sex-based differences in platelet counts were also observed in African studies [17,18]. The reasons for these differences are still unclear. The lower limit for the PLT parameter in our consensus intervals is slightly lower than what has been previously established for Americans [19] and Malaysians [20], but is higher than the current interval used in China [21].
The WBC and NEUT values increased slightly with age, which might be related to chronic infection in elderly people that were not excluded according to our criteria. Differences in EO values according to region, sex, and age group were identified, and may be attributable to the presence of allergic and parasitic diseases in the apparently healthy elderly subjects. For example, mites living in tropical and humid conditions [22] or pathogenic protozoa consumed in contaminated water and foods in Guangdong Province [23, 24] may have caused such diseases. The apparent increase in EO values in males and females aged 70–79 years is consistent with data from the Guangzhou center, from which almost 50% subjects were aged more than 70 years. We observed no significant sex-based difference in WBC values across our study populations. This finding was similar to data reported in the 2nd National Guide to Clinical Laboratory Procedures, but different from those reported in the 3rd National Guide [21, 25]. Compared with other published data, the reference intervals for WBC and NEUT were similar to intervals for Malaysians, lower than those for Americans, and higher than those for Africans [9].
The lower limits of the RBC, HGB, and HCT parameters decreased slightly with age in male subjects. This decrease could be due to the gradual loss of androgens, which stimulate increased production of erythrocytes [26, 27]. In contrast, RBC, HGB, and HCT values increased slightly with age in females, consistent with the lower levels of these parameters before menopause and higher levels after menopause [28]. The average RBC, HGB, and HCT values in males decreased at 70–79 years of age, which could contribute to nutritional deficiency, occult malignancy, and chronic anemia in the aging population. The same results were observed in North American populations [15]. In addition, the RBC, HGB, and HCT values were higher in males than in females at all ages due to chronic menstrual blood loss in females and a higher androgen level in males.
We identified regional variations in the MCV and MCH parameters that could be related to the high prevalence of TT in Guangzhou and Chengdu (Southern China). The prevalence of TT carriers we observed in Guangzhou (10.5%) and Chengdu (4.3%) were similar to previously published data [29, 30]. When TT carriers were excluded from analysis, the MCV and MCH parameters were not significantly different among the six centers. However, because thalassemia gene DNA analysis was only performed on enrolled individuals whose MCV was 82 fL or less, and as only 20 of the most typical DNA deletions or mutations were detected, there could be a small population with TT that were not excluded in our study. Nevertheless, when TT carriers with microcytic characteristics were excluded, the influence of TT carriers with MCV greater than 82 fL on the RBC reference intervals was negligible. Furthermore, the CLSI guidelines do not provide clear protocols for how to proceed when apparently healthy subjects have asymptomatic illnesses that impact the reference intervals. Our results suggest that in regions with a high prevalence of thalassemia, such as the Mediterranean, the Middle East, Africa, and Southeast Asia [9, 20, 31, 32], it is critical to exclude TT carriers from healthy candidates to establish RBC reference intervals. Different methods, such as hemoglobin electrophoresis, RBC indices, blood film, HPLC analysis, or DNA analysis, can be used to screen reference individuals [33–35].
The lower limit of MCV in our consensus intervals was slightly higher than those in America [19] and Malaysia [20], and substantially higher than that in Africa [9]. The MCV consensus interval was similar to American populations with a low prevalence of thalassemia, and similar to the lower limit of the Malaysian population after excluding TT carriers with the Biorad Variant II hemoglobin analyzer [20]. Without exclusion of TT carriers, the decreased lower limit of MCV is more obvious in CBC reference intervals of the African population [9]. The lower limit of the HGB reference interval has substantially increased in the Chinese population over the past 30 years compared to previous studies [25].
With a sufficient sample size, standard statistical analyses can detect significant differences, but that does not necessarily mean that the differences are clinically significant for the subjects. In our study, most parameters (e.g., MCV, RBC, MCH, WBC, NEUT, and PLT) had some variations across instruments, regions, age, or sex. However, the variations were not significant, and all of the data could be combined to calculate the consensus intervals. Reference intervals derived from different regions, instruments, and ages might appear to be more conservative in clinical application. The present study is the largest published study to define CBC reference intervals in China. As China is a vast country with a large population, it is reasonable to assume that CBC parameters could vary regionally and in a population-dependent manner. Our multicenter study addresses this possible variation and establishes consensus CBC reference intervals (except for PLT) for healthy Han Chinese adults. The established laboratory intervals will aid in clinical diagnosis and prognosis analyses.
Acknowledgments
We acknowledge and thank all of the participants for their cooperation and sample contributions. We thank all of the staff of research centers involved in this study. This study was supported by the Ministry of Health, the People's Republic of China.
Funding Statement
This work was supported by National Key Technologies R&D Program of China (2012BAI37B01). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1. Cong YL, Jin DM, Wang HL, Okada T, Peng ZH. Establishing the reference range of venous blood measured by automated haematology analyzer in Chinese adults. Zhonghua Yi Xue Za Zhi. 2003; 83: 1201–1205. [PubMed] [Google Scholar]
- 2. Ge M, Zhang Y, He J, Yan Y, Wang X, Cao L, et al. Normal red blood cell count reference values in Chinese presenile women given by geographical area. J Formos Med Assoc. 2010; 109: 656–662. 10.1016/S0929-6646(10)60106-4 [DOI] [PubMed] [Google Scholar]
- 3.CLSI. Defining establishing and verifying reference intervals in the clinical laboratory; approved guideline—third edition. CLSI document C28-A3c. Horowitz GL: Clinical and Laboratory Standards Institute; 2010.
- 4. Zhang JW, Long GF. Hemoglobin and hemoglobinopathies Guangxi Science and technology Press; 2003: 215–218. [Google Scholar]
- 5. Lin M, Wang Q, Zheng L, Huang Y, Lin F, Lin CP, et al. Prevalence and molecular characterization of abnormal hemoglobin in eastern Guangdong of southern China. Clin Genet. 2012; 81: 165–171. 10.1111/j.1399-0004.2011.01627.x [DOI] [PubMed] [Google Scholar]
- 6. Cai SP, Wall J, Kan YW, Chehab FF. Reverse dot blot probes for the screening of beta- thalassemia mutations in Asians and American blacks. Hum. Mutat. 1994; 3: 59–63. [DOI] [PubMed] [Google Scholar]
- 7. England JM, Rowan RM, van Assendelft OW, Coulter WH, Groner W, Jones AR, et al. Protocol for evaluation of automated blood cell counters. International Committee for Standardization in Haematology (ICSH). Clin Lab Haematol. 1984; 6: 69–84. [DOI] [PubMed] [Google Scholar]
- 8.Department of Health and Human Services, Centers for Medicare and Medicaid Services. Clinical laboratory Improvement Amendments of 1988; Final Rule. 42 CFR 493.941(c) (2). Washington. DC: US Government Printing Office; Published annually.
- 9. Karita E, Ketter N, Price MA, Kayitenkore K, Kaleebu P, Nanvubya A, et al. CLSI- derived hematology and biochemistry reference intervals for healthy adults in eastern and southern Africa. PLoS One. 2009; 4: e4401 10.1371/journal.pone.0004401 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Zwart A, van Assendelft OW, Bull BS, England JM, Lewis SM, Zijlstra WG. et al. Recommendations for reference method for haemoglobinometry in human blood (ICSH standard 1995) and specifications for international haemiglobincyanide reference preparation (4th edition). J Clin Pathol. 1996; 49: 271–274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. International Council for Standardization in Haematology Expert Panel on Blood Cell Sizing. Recommendation for reference method for determination by centrifugation of packed cell volume of blood. J Clin Pathol. 1980; 33:1–2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. International Council for Standardization in Haematology Expert Panel on Cytometry. Reference method for the enumeration of erythrocytes and leucocytes. Clin Lab Haematol. 1994; 16: 131–138. [PubMed] [Google Scholar]
- 13. International Council for Standardization in Haematology Expert Panel on Cytometry; International Society of Laboratory Hematology Task Force on Platelet Counting. Platelet counting by the RBC/platelet ratio method. A reference method. Am J Clin Pathol. 2001; 115: 460–464. [DOI] [PubMed] [Google Scholar]
- 14. Sirdah MM, Tarazi IS, El Jeadi H, Al Haddad RM. Normal Blood Cells Reference Intervals of Healthy Adults at the Gaza Strip-Palestine. Journal of Clinical Laboratory Analysis. 2008; 22: 353–361. 10.1002/jcla.20265 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Cheng CK, Chan J, Cembrowski GS, van Assendelft OW. Complete Blood Count Reference Interval Diagrams Derived from NHANES III: Stratification by Age, Sex, and Race. Laboratory Hematology. 2004; 10: 42–53. [DOI] [PubMed] [Google Scholar]
- 16. Ge M, Liu XP, Fu HY, Wang ZL, Zhang YF, Zhang J, et al. Normal reference value of hemoglobin of middleaged women and altitude. Yale J Biol Med. 2004; 77: 117–123. [PMC free article] [PubMed] [Google Scholar]
- 17. Adetifa IM, Hill PC, Jeffries DJ, Jackson-Sillah D, Ibanga HB, Bah G, Donkor S, et al. Haematological values from a Gambian cohort—Possible reference range for a West African population. Int J Lab Hematol. 2009; 31: 615–622. 10.1111/j.1751-553X.2008.01087.x [DOI] [PubMed] [Google Scholar]
- 18. Kibaya RS, Bautista CT, Sawe FK, Shaffer DN, Sateren WB, Scott PT, et al. Reference ranges for the clinical laboratory derived from a rural population in Kericho, Kenya. PLoS One. 2008; 3: e3327–13.. 10.1371/journal.pone.0003327 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Kratz A, Ferraro M, Sluss PM, Lewandrowski KB. Case records of the Massachusetts General Hospital. Weekly clinicopathological exercises. Laboratory reference values. N Engl J Med. 2004; 351:1548–1563. [DOI] [PubMed] [Google Scholar]
- 20. Roshan TM, Rosline H, Ahmed SA, Rapiaah M, Wan Zaidah A, Khattak MN. Hematological reference values of healthy Malaysian population. Int J Lab Hematol. 2009; 31: 505–512. 10.1111/j.1751-553X.2008.01068.x [DOI] [PubMed] [Google Scholar]
- 21. Hong H, Fan Y, Yang Q, Li X, Guo J, Chen X. A cohort investigation of the allergen and pathogenesis in the patients with allergic rhinitis from three hospital of Guangdong Province. Lin Chung Er Bi Yan Hou Tou Jing Wai Ke Za Zhi. 2011; 25: 1064–1067. [PubMed] [Google Scholar]
- 22. Ye YW, Wang YS, Shen ZY. National guide to clinical laboratory procedures 3rd ed. Southeast University Press; 2006: 1017. [Google Scholar]
- 23. Lun ZR, Gasser RB, Lai DH, Li AX, Zhu XQ, Yu XB, et al. Clonorchiasis: a key foodborne zoonosis in China. Lancet Infect Dis. 2005; 5: 31–41. [DOI] [PubMed] [Google Scholar]
- 24. Yoo WG, Kim DW, Ju JW, Cho PY, Kim TI, Cho SH, et al. Developmental transcriptomic features of the carcinogenic liver fluke, Clonorchis sinensis. PLoS Negl Trop Dis. 2011; 5: e1208 10.1371/journal.pntd.0001208 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Ye YW, Wang YS. National guide to clinical laboratory procedures 2nd ed. Southeast University Press; 1997: 613. [Google Scholar]
- 26. Cui YG, Tong JS, Pan QQ, Di FS, Jia Y, Feng T, et al. Effect of androgen on erythropoietin in patients with hypogonadism. Zhonghua Nan Ke Xue. 2003; 9: 248–251. [PubMed] [Google Scholar]
- 27. Paul AK, Latif ZA, Iqbal S, Amin F, Shefin SM, Ashrafuzzaman SM. Androgen versus erythropoietin for the treatment of anaemia of pre-dialysis chronic kidney disease. Mymensingh Med J. 2012; 21:125–128. [PubMed] [Google Scholar]
- 28. Saxena S, Wong ET. Heterogeneity of common hematologic parameters among racial, ethnic, and gender subgroups. Arch Pathol Lab Med. 1990,114: 715–719. [PubMed] [Google Scholar]
- 29. Xu XM, Zhou YQ, Luo GX, Liao C, Zhou M, Chen PY, et al. The prevalence and spectrum of alpha and beta thalassaemia in Guangdong Province: implications for the future health burden and population screening. J Clin Pathol. 2004; 57: 517–522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Wang X, Jiang H, Jia J, Zhou J, Liao J, Zuo C. Screening and genetic analysis of thalassemia in Sichuan District. Sheng Wu Yi Xue Gong Cheng Xue Za Zhi. 2011; 28: 135–137. [PubMed] [Google Scholar]
- 31. Abolghasemi H, Amid A, Zeinali S, Radfar MH, Eshghi P, Rahiminejad MS, et al. Thalassemia in Iran: epidemiology, prevention, and management. J Pediatr Hematol Oncol. 2007; 29: 233–238. [DOI] [PubMed] [Google Scholar]
- 32. Birgens H, Ljung R. The thalassaemia syndromes. Scand J Clin Lab Invest. 2007; 67: 11–26. [DOI] [PubMed] [Google Scholar]
- 33. Ryan K, Bain BJ, Worthington D, James J, Plews D, Mason A, et al. British Committee for Standards in Haematology. Significant haemoglobinopathies: guidelines for screening and diagnosis. Br J Haematol. 2010; 149: 35–49. 10.1111/j.1365-2141.2009.08054.x [DOI] [PubMed] [Google Scholar]
- 34. Trent RJA. Diagnosis of the Haemoglobinopathies. Clin Biochem Rev. 2006; 27: 27–38. [PMC free article] [PubMed] [Google Scholar]
- 35. Galanello R, Eleftheriou A, Traeger-Synodinos J, Old J, Petrou M, Angastiniotis M. Prevention of Thalassemias and other haemoglobin disorders—Volume 1 Thalassemia international federation Publications; 2003: 35. [PubMed] [Google Scholar]