Abstract
The early symptoms of pancreatic cancer are often very vague. They may precede the diagnosis by years and go unrecognized. This makes pancreatic cancer one of the cancers with the worst survival rates. The progression rate of the early phase might be slower than previously thought. Here, we report a case where symptoms, including thromboembolism and new-onset diabetes mellitus, preceded the diagnosis of pancreatic cancer by 6 years or longer. The awareness of the early symptoms of pancreatic cancer is required for being vigilant and further diagnostic tests. A simple clinical model utilizing certain risk factors and symptoms for pancreatic cancer will help stratify the patients for further screening tests.
Key Words: Pancreatic cancer, Early symptoms, Screening, Thromboembolism, Diabetes mellitus
Introduction
Pancreatic cancer is one of the few cancers with increasing incidence. It accounts for 2.8% of new cancers but 6.2% of all cancer deaths as the fourth leading cause of cancer mortality. The 5-year relative survival rate is 6%, with the least improvements in survival over the past three decades compared to most other cancers [1]. The dismal prognosis of pancreatic cancer is mainly associated with late diagnosis, in addition to low resection rate, aggressiveness of the cancer, and lack of effective treatment. Most patients are diagnosed with metastatic disease that showed less sustained response to therapies. Therefore, pancreatic cancer must be discovered early to increase the chance of being cured by surgery [2]. A simple model including early clinical symptoms may facilitate the recognition of this almost always fatal disease.
Case Presentation
A 50-year-old male presented in 2006 with chest pain, productive cough for 1 week, and progressive lower extremity edema for 1 year.
The past medical history included hypertension, hypothyroid, dyslipidemia, dilated cardiomyopathy, hypothyroidism, chronic obstructive pulmonary disease, morbid obesity, sleep apnea, gastroesophageal reflux disease, depression, and cryptorchidism treated with orchidopexy. He had a 45-pack-year history of smoking and heroin abuse, and was on methadone maintenance.
Physical examination revealed the following: blood pressure 120/72, heart rate 64, respiration rate 18, temperature 97.0°F, bilateral lower extremity edema 2+. Physical examination was otherwise unremarkable. Hemoglobin was 12.6, hematocrit 35.5, white cell count 7.8, platelet 289. Sodium was 135, potassium 3.8, chloride 103, bicarbonate 25, blood urea nitrogen 17, creatinine 1.1, and glucose 103. Hepatitis B and C tests were negative. Imaging studies and stress test showed no deep venous thrombosis, pulmonary embolism (PE), or myocardial infarction. He was discharged after stabilization with the primary diagnosis of unstable angina.
In 2007, he presented with severe dyspnea, hemoptysis, and retrosternal chest pain radiating towards back, recent weight loss of 20 pounds without alternation in appetite or bowel habits. The patient also developed new-onset diabetes mellitus (DM). CT angiogram revealed segmental PE, and the patient was started on warfarin.
Gradually, the patient became weaker. In 2012, he developed exertional dyspnea, gait unsteadiness, tendency to fall, and loss of more than 80 pounds over 6 months, which was attributed to dental and recurrent periodontal diseases. Eventually, he became unable to work.
In 2013, the patient was admitted for recurrent abdominal pain and persistent weight loss. The CT scan showed abnormalities suggesting malignancy in the distal body and tail of the pancreas, retroperitoneal adenopathy, multiple hepatic masses, as well as thrombi in hepatic, splenic, left renal, and portal veins (fig. 1). Liver biopsy revealed adenocarcinoma of the pancreatic primary with tumor cells positive for CK 7, CK 19, CA19.9, and negative for CD 56, s100, chromogranin, synaptophysin, CK 20, and Hep Par 1. Chemotherapy was subsequently started.
Fig. 1.
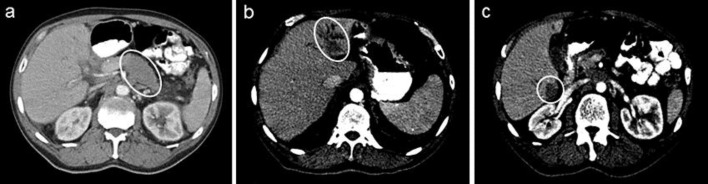
CT of the abdomen showing the mass in the pancreatic body and tail and possible metastasis in the liver. a Hypodensity in the body and tail of the pancreas, with the pancreas appearing prominent in size in this region, measuring up to 4.9 cm (circle). No obvious pancreatic ductal dilatation is noted. b, c Hypodense mass in the left lobe of the liver, measuring up to approximately 4.6 cm. Smaller hypodense lesions in the right lobe of the liver measuring up to 2.6 cm in diameter (circles). There is associated confluent soft tissue density in the peripancreatic region posteriorly, with apparent extension to the para-aortic region.
Discussion
Although it is generally believed to be rapid, the rate of early pancreatic cancer progression might be slower than what it was thought, as estimated in other types of solid cancers [3]. A recent study illustrated that it takes about 12 years for the initiating mutation to result in the nonmetastatic founder cell; another 7 years to acquire the metastatic ability, and then 3 more years to cause death [4]. In other studies, the small pancreatic cancer was demonstrated to have a slow progressive rate. It takes over 29 months for this cancer to grow into an invasive carcinoma measuring 1 cm in diameter [5]. In small invasive pancreatic carcinoma, the tumor volume doubling time was reported to be 252 days [6]. These findings make it possible to detect the small pancreatic cancer during the relatively long, silent, and most importantly curable period using certain screening methods [7] and provide a significant extension of time to perform effective treatment.
However, owing to the low prevalence and absence of promising treatment approaches, even in early stages, it is currently infeasible to do mass screening for asymptomatic pancreatic cancer in the general population [8]. Still, those limitations do not mitigate the necessity of striving for early diagnosis of pancreatic cancer.
Statistical models have been developed to estimate the pretest probability of developing pancreatic cancer [9]. A prediction rule was developed to stratify the risk for pancreatic cancer in patients with chronic pancreatitis, where gender, mass location, mass number, direct bilirubin, and CA 19-9 were included in the calculation [10]. However, a pretest clinical model employing risk factors and symptoms would be plausible to help clinicians to stratify the patients into different risk categories and determine whether further investigation is needed. Weighted noninherited factors including obesity, cigarette smoking, carcinogen exposure, DM, chronic pancreatitis, liver disease, as well as inherited factors such as hereditary pancreatitis and Peutz-Jeghers syndrome might be used to construct such a model [8, 11].
The findings in this report reinforce the feasibility of a clinical model. The thromboembolism, late onset of DM, weight loss, and other changes preceded the diagnosis of pancreatic cancer by 6–7 years in this case. Thromboembolism is a common complication of cancer, it occurs in 17–57% of pancreatic cancer patients. Pancreatic cancer cells can activate platelets, release various procoagulant factors, induce venous or arterial thrombosis, or disseminated intravascular coagulation [12]. In the absence of heart failure, renal insufficiency, or liver disease, the initial pedal edema in the patient was likely due to iliac deep venous thrombosis, which might be missed by the lower extremity venous duplex. As a matter of fact, diffuse intra-abdominal venous thrombosis was detected by the subsequent CT scan. However, even with the knowledge of the association between cancer and thrombosis, the existence of some known PE risk factors, as in this case morbid obesity and sleep apnea, may discourage the physician from further investigation for other possible etiologies.
DM is a risk factor for (especially chronic DM >5 years) [8, 13] or a complication of pancreatic cancer. DM was found in 50% of pancreatic cancer patients, and in over 20% DM occurs during the asymptomatic phase [14]. About 1% of patients aged 50 years or older who had new-onset DM will develop pancreatic cancer within 3 years after the diagnosis of DM [14]. Patients with pancreatic cancer diagnosed in association with DM had better survival than symptomatic patients, and the cancer was generally resectable if found at the onset of DM [15]. The new-onset DM in adults requires careful evaluation to exclude malignancy. With a high prevalence of type 2 DM in the general population, the discovery of a biomarker to distinguish the cancer-related DM and type 2 DM would markedly facilitate this process.
Knowledge and recognition of certain early manifestations may lead to further investigation and diagnosis of presymptomatic pancreatic cancer before it becomes incurable. Furthermore, a standardized clinical scoring system based on risk factors and clinical findings would help objectify pretest probability of pancreatic cancer and facilitate the decision making regarding whether and which diagnostic test, for example endoscopic ultrasonography, is needed for individuals with increased risk of pancreatic cancer [7]. Further retrospective and prospective studies are warranted for this purpose.
References
- 1.Siegel R, Ma JM, Zou ZH, Jemal A. Cancer statistics, 2014. Cancer J Clin. 2014;64:9–29. doi: 10.3322/caac.21208. [DOI] [PubMed] [Google Scholar]
- 2.Hackert T, Büchler MW. Pancreatic cancer: advances in treatment, results and limitations. Dig Dis. 2013;31:51–56. doi: 10.1159/000347178. [DOI] [PubMed] [Google Scholar]
- 3.Patrone MV, Hubbs JL, Bailey JE, Marks LB. How long have I had my cancer, doctor? Estimating tumor age via Collins’ law. Oncology (Williston Park) 2011;25:38–43, 46. [PubMed] [Google Scholar]
- 4.Yachida S, Jones S, Bozic I, Antal T, Leary R, Fu B, Kamiyama M, Hruban RH, Eshleman JR, Nowak MA, Velculescu VE, Kinzler KW, Vogelstein B, Iacobuzio-Donahue CA. Distant metastasis occurs late during the genetic evolution of pancreatic cancer. Nature. 2010;467:1114–1117. doi: 10.1038/nature09515. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Nakamura T, Masuda K, Harada S, Akioka K, Sako H. Pancreatic cancer: slow progression in the early stages. Int J Surg Case Rep. 2013;4:693–696. doi: 10.1016/j.ijscr.2013.04.040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Hisa T, Ohkubo H, Shiozawa S, Ishigame H, Takamatsu M, Furutake M, Nobukawa B, Suda K. Growth process of small pancreatic carcinoma: a case report with imaging observation for 22 months. World J Gastroenterol. 2008;14:1958–1960. doi: 10.3748/wjg.14.1958. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Gemmel C, Eickhoff A, Helmstädter L, Riemann JF. Pancreatic cancer screening: state of the art. Expert Rev Gastroenterol Hepatol. 2009;3:89–96. doi: 10.1586/17474124.3.1.89. [DOI] [PubMed] [Google Scholar]
- 8.Decker GA, Batheja MJ, Collins JM, Silva AC, Mekeel KL, Moss AA, Nguyen CC, Lake DF, Miller LJ. Risk factors for pancreatic adenocarcinoma and prospects for screening. Gastroenterol Hepatol (NY) 2010;6:246–254. [PMC free article] [PubMed] [Google Scholar]
- 9.Zhao D, Weng C. Combining PubMed knowledge and EHR data to develop a weighted bayesian network for pancreatic cancer prediction. J Biomed Inform. 2011;44:859–868. doi: 10.1016/j.jbi.2011.05.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Cai QC, Chen Y, Xiao Y, Zhu W, Xu QF, Zhong L, Chen SY, Zhang MM, Wang LW, Li ZS. A prediction rule for estimating pancreatic cancer risk in chronic pancreatitis patients with focal pancreatic mass lesions with prior negative EUS-FNA cytology. Scand J Gastroenterol. 2011;46:464–470. doi: 10.3109/00365521.2010.539256. [DOI] [PubMed] [Google Scholar]
- 11.Tanţ˘u A, Negrean V, Alexescu T, Para I, T˘rmure S, Casoinic F, Sâmpelean D, Mocanu T, Tanţ˘u M. Two different types of diabetes mellitus in pancreatic cancer population. Comparative study between new onset and long standing diabetes mellitus on 76 patients with pancreatic cancer. Rom J Intern Med. 2014;52:18–23. [PubMed] [Google Scholar]
- 12.Khorana AA, Fine RL. Pancreatic cancer and thromboembolic disease. Lancet Oncol. 2004;5:655–663. doi: 10.1016/S1470-2045(04)01606-7. [DOI] [PubMed] [Google Scholar]
- 13.Chari ST, Leibson CL, Rabe KG, Ransom J, de Andrade M, Petersen GM. Probability of pancreatic cancer following diabetes: a population-based study. Gastroenterology. 2005;129:504–511. doi: 10.1053/j.gastro.2005.05.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Pannala R, Basu A, Petersen GM, Chari ST. New-onset diabetes: a potential clue to the early diagnosis of pancreatic cancer. Lancet Oncol. 2009;10:88–95. doi: 10.1016/S1470-2045(08)70337-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Pelaez-Luna M, Takahashi N, Fletcher JG, Chari ST. Resectability of presymptomatic pancreatic cancer and its relationship to onset of diabetes: a retrospective review of CT scans and fasting glucose values prior to diagnosis. Am J Gastroenterol. 2007;102:2157–2163. doi: 10.1111/j.1572-0241.2007.01480.x. [DOI] [PubMed] [Google Scholar]


