Abstract
Aims
Ageing-related cardiac disorders such as heart failure and atrial fibrillation often present with intracellular calcium homeostasis dysfunction. However, knowledge of the intrinsic effects of ageing on cellular calcium handling in the human heart is sparse. Therefore, this study aimed to analyse how ageing affects key mechanisms that regulate intracellular calcium in human atrial myocytes.
Methods and results
Whole membrane currents and intracellular calcium transients were measured in isolated human right atrial myocytes from 80 patients with normal left atrial dimensions and no history of atrial fibrillation. Patients were categorized as young (<55 years, n = 21), middle aged (55–74 years, n = 42), and old (≥75 years, n = 17). Protein levels were determined by western blot. Ageing was associated with the following electrophysiological changes: (i) a 3.2-fold decrease in the calcium transient (P < 0.01); (ii) reduction of the L-type calcium current (ICa) amplitude (2.4 ± 0.3 pA/pF vs. 1.4 ± 0.2 pA/pF, P < 0.01); (iii) lower levels of L-type calcium channel alpha-subunit (P < 0.05); (iv) lower rates of both fast (14.5 ± 0.9 ms vs. 20.9 ± 1.9, P < 0.01) and slow (73 ± 3 vs. 120 ± 12 ms, P < 0.001) ICa inactivation; and (v) a decrease in the sarcoplasmic reticulum calcium content (10.1 ± 0.8 vs. 6.4 ± 0.6 amol/pF, P < 0.005) associated with a significant decrease in both SERCA2 (P < 0.05) and calsequestrin-2 (P < 0.05) protein levels. In contrast, ageing did not affect spontaneous sarcoplasmic reticulum calcium release.
Conclusion
Ageing is associated with depression of SR calcium content, L-type calcium current, and calcium transient amplitude that may favour a progressive decline in right atrial contractile function with age.
Keywords: L-type calcium current, SERCA2, Calsequestrin-2, Sarcoplasmic reticulum calcium release, Atrial fibrillation
1. Introduction
As a result of the progressive increase in life span in highly developed countries, knowledge of the effects of ageing on human pathophysiology is of great relevance. Coronary atherosclerosis, heart failure, and atrial fibrillation (AF) are, among others, prevalent cardiovascular diseases closely linked to ageing.1
Left atrial enlargement with impaired mechanical function is often observed in healthy humans after the eighth decade.2–4 Since older age is frequently associated with diastolic left-ventricular (LV) dysfunction,2 enlargement of the left atrium in older subjects could merely result from the increased LV filling pressure. However, an intrinsic derangement of atrial cellular calcium homeostasis induced by age could theoretically impair the mechanical atrial function in subjects with otherwise normal LV function. Moreover, the propensity of older subjects to develop episodes of AF5 could also be favoured by an age-related intrinsic dysfunction of atrial intracellular calcium homeostasis. Nevertheless, knowledge of the intrinsic effects of ageing on atrial intracellular calcium handling in humans is lacking and data from experimental animal models are sparse and often inconsistent.6
Therefore, this study aimed to analyse the effects of ageing on calcium handling in isolated human right atrial myocytes to gain insight into the pathophysiology of prevalent age-associated derangements of atrial function and atrial disease in humans.
2. Methods
2.1. Human atrial tissue
A total of 159 isolated right atrial myocytes obtained from 80 patients submitted to elective cardiac surgery were analysed. The tissue samples were collected just prior to atrial cannulation for extracorporeal circulatory bypass, were stored in cold Tyrode solution, and immediately (within 5–10 min.) brought to the laboratory for cell isolation, and further analysis within 8 h after cell isolation. Although this atrial tissue is currently discarded during cardiac surgery, all patients gave written consent to obtain and use their specimens. The study was approved by the Ethical Committee of our institution and was conducted in accordance with the Declaration of Helsinki.
None of the patients had a previous history of AF and all presented with normal left-atrial size at echocardiography (reference range for normal indexed left-atrial diameter: <2.3 cm/m2). Patients were divided into three age categories: (i) young (<55 years, 49 myocytes, n = 21); (ii) middle aged (55–74 years, 60 myocytes, n = 42); and (iii) old (≥75 years, 50 myocytes, n = 17).
2.2. Patch-clamp technique
The experimental solutions used for this study had the following composition. Extracellular solution (mM): NaCl 127, TEA 5, HEPES 10, NaHCO3 4, NaH2PO4 0.33, glucose 10, pyruvic acid 5, CaCl2 2, MgCl2 1.8 (pH 7.4). The pipette solution contained (mM): aspartatic acid 109, CsCl 47, Mg2ATP 3, MgCl2 1, Na2-phosphocreatine 5, Li2GTP 0.42, HEPES 10 (pH 7.2 with CsOH). Amphotericin (250 µg/mL) was added to the pipette solution before starting the experiment. Chemicals were from Sigma-Aldrich.
Whole membrane currents were measured in the perforated patch configuration with an EPC-10 amplifier (HEKA Elektronik). The L-type calcium current (ICa) was measured using a 50 ms prepulse from −80 to −45 mV (used to inactivate INa), followed by a 200 ms depolarization to 0 mV. The ICa amplitude was determined as the difference between the peak inward current and the current at the end of the depolarization. The current–voltage relationship for ICa and the voltage-dependent inactivation were obtained using test potentials between −40 and +50 mV. The time constants for fast (tau-1) and slow (tau-2) steady-state ICa inactivation were determined from a double exponential fit of the decaying phase of ICa. Recovery of ICa from inactivation was assessed using a two-pulse protocol with increasing intervals between the first and the second pulse used to elicit ICa.
The SR calcium content was measured as the time integral of the current elicited by rapid exposure to 10 mM caffeine and was converted to amoles (10−18 mol) of calcium released from the SR, assuming a stoichiometry of 3 Na+:1 Ca2+ for the Na+-Ca2+ exchanger.7 Spontaneous calcium release was examined with the membrane potential clamped at −80 mV and detected by measuring the transient inward currents (ITI) induced by spontaneous calcium release as previously described.8
2.3. Confocal microscopy and calcium imaging
A resonance-scanning confocal microscope (Leica TCS SP5 AOBS) was used to visualize intracellular calcium levels in the frame-scanning mode as we previously described.9 The cross-sectional area of the cells was measured from the transmission images using Image J software. For calcium imaging, myocytes were loaded with 2.5 µmol/L fluo-4AM (Invitrogen) for 20 min at room temperature, followed by wash and de-esterification for at least 30 min. Fluo-4 was excited at 488 nm with the laser power set to 20% of maximum and attenuation to 4%. Fluorescence emission was collected between 500 and 650 nm. Synchronization of confocal images and ionic current recordings was achieved using a Leica DAQ box and HEKA patch-master software (HEKA Elektronik), which delivered the triggers for confocal image acquisition. Calcium transients and calcium waves were measured from the whole cell fluorescence (see Supplementary material online, Figure S1). For comparison of the maximal and mean fluorescence intensities during a calcium wave, the maximal fluorescence was measured in a 5 × 10 µm rectangle covering the peak of the calcium transient and the mean fluorescence was measured in the whole cell area.
The presence of T-tubular structures was assessed by incubating isolated myocytes with one of the fluorescent dyes Di-4-ANEPPS or RH-237 (2 µM) for 5 min followed by a 30 min wash. Di-4-ANEPPS was excited at 488 nm and fluorescence emission collected between 500 and 650 nm. RH-237 was excited at 543 nm and fluorescence emission collected between 580 and 750 nm.
To visualize RyR2 clusters, human atrial myocytes were immunofluorescently labelled using a primary anti-RyR2 antibody (Calbiochem). Clusters were detected automatically using a custom-made algorithm that first enhanced the contrast of images using a histogram stretching intensity transformation. Subsequently, it removed background noise by using an adaptive median filter that estimated the noise level, and enhanced the location of all labelled RyRs with a 2D Gaussian filter with a standard deviation of 0.5 µm followed by segmentation using a multilevel watershed algorithm. Non-specific staining was eliminated by setting the maximal RyR diameter to 1.2 µm.
2.4. Analysis of protein levels
Right-atrial tissue samples of about 20 mg were pulverized in liquid nitrogen and ‘homogenized’ in 200 µL of ice-cold lysis buffer containing 50 mM HEPES pH 7.4, 0.1% (v/v) Tween 20, 100 mM NaCl, 2.5 mM EGTA, 10 mM glycerol-2-phosphate, 10% (v/v) glycerol, and 1 mM DTT supplemented with a cocktail of protease inhibitors (Roche). Proteins were separated by SDS–PAGE (10% acrylamide: bisacrylamide) and electrotransferred onto Immobilon polyvinylidene diflouride membranes (Millipore). Membranes were incubated with primary and secondary antibodies diluted in 5% non-fat dry milk except for DHPR blots, for which SuperBlock™ Blocking Buffer (Thermo Scientific) was used. Antibodies against SERCA (#9580, Cell Signaling Technology), calsequestrin-2 (ab3516, Abcam), DHPR (ab81980, Abcam), and NCX1 (ab135735, Abcam) were used. After a standard washing protocol, detection was performed using the appropriate horseradish peroxidase-labelled IgG and the Supersignal™ detection system (Supersignal West Dura™, Pierce). Molecular-mass standards (Bioline) were used to estimate protein size and glyceraldehyde-3-phosphate dehydrogenase (GAPDH; MAB374, Millipore) was used as a loading control. Immunoblots were digitized (GS-800 Calibrated Densitometer; Bio-Rad) and analysed with the Quantity One 4.6.3 software (Bio-Rad).
2.5. Data analysis
Statistical analysis was performed using SPSS software. Unless otherwise stated, values are expressed as mean ± SEM. Data sets were tested for normality. Statistical significance was evaluated using a Student's t-test and ANOVA was used for comparison of multiple effects. Age, sex, LV ejection fraction, valvular heart disease, ischaemic heart disease, and treatment with ACE-inhibitors, beta-blockers, angiotensin receptor blockers, and calcium channel antagonists were evaluated and taken into account as potential confounding factors using a general linear model. Bonferroni post-test was used to evaluate the significance of specific effects. Data were also analysed as a continuum and correlations were reported using the Pearson Correlation (r), the slope and the significance (P) for a two-tailed analysis. Differences were considered statistically significant when P < 0.05.
3. Results
3.1. Study population
Table 1 summarizes the clinical characteristics of the 80 patients included in the study. Older patients had a higher incidence of combined valvular and ischaemic heart disease and greater percentage of coronary bypass surgery than young and middle-aged patients. There were no statistically significant differences in sex, left-atrial size, and LV ejection fraction among the three age groups. ACE-inhibitors and beta-blockers were administered in nearly 30% of instances and only about 17.5% of patients were on angiotensin receptor blockers.
Table 1.
Clinical data of the study patients
| Total (n = 80) | <55 years (n = 21) | 55–75 years (n = 42) | ≥75 years (n = 17) | P-value* | |
|---|---|---|---|---|---|
| Anthropometrics characteristics | |||||
| Male, n (%) | 59 (73.8) | 16 (76.2) | 33 (78.6) | 10 (58.8) | ns |
| BMI, kg/m2 | 28.3 ± 5.7 | 24.9 ± 4.0 | 29.1 ± 5.0 | 28.2 ± 4.1 | 0.004 |
| Echocardiographic characteristics | |||||
| LA diameter, mm | 37 ± 5 | 36 ± 5 | 38 ± 5 | 38 ± 4 | ns |
| Indexed LA, cm/m2 | 2.0 ± 0.2 | 1.9 ± 0.2 | 2.0 ± 0.2 | 2.1 ± 0.2 | ns |
| LVEF, % | 59 ± 13 | 60 ± 13 | 59 ± 12 | 53 ± 16 | ns |
| Heart disease | |||||
| Valvular heart disease, n (%) | 33 (41.3) | 10 (47.6) | 17 (40.5) | 6 (35.3) | ns |
| Ischaemic heart disease, n (%) | 23 (28.8) | 4 (19.0) | 13 (31) | 6 (35.3) | ns |
| Valvular + ischaemic heart disease, n (%) | 11 (13.8) | 2 (9.5) | 5 (11.9) | 4 (23.5) | 0.023 |
| Surgical treatment | |||||
| Aortic valve replacement, n (%) | 40 (50) | 10 (47.6) | 20 (47.6) | 10 (58.8) | ns |
| Mitral valve replacement, n (%) | 3 (3.8) | 1 (4.8) | 2 (4.8) | 0 | ns |
| Tricuspid valve surgery, n (%) | 6 (8.1) | 4 (19.0) | 2 (4.8) | 0 | ns |
| CABG, n (%) | 35 (43.8) | 3 (14.3) | 20 (47.6) | 12 (70.6) | 0.002 |
| CABG + valve replacement, n (%) | 16 (20.0) | 1 (4.8) | 8 (19) | 7 (41.2) | 0.020 |
| Pharmacological treatment | |||||
| ACE-inhibitors, n (%) | 24 (30.0) | 6 (28.6) | 11 (26.2) | 7 (41.2) | ns |
| Angiotensin receptor blocker, n (%) | 14 (17.5) | 1 (4.8) | 9 (21.4) | 4 (23.5) | ns |
| Beta-blockers, n (%) | 23 (28.8) | 6 (28.6) | 11 (26.2) | 6 (35.3) | ns |
| Calcium channels antagonists, n (%) | 15 (18.8) | 1 (4.8) | 9 (21.4) | 5 (29.4) | ns |
BMI, body mass index; LA, left atrium; LVEF, left-ventricular ejection fraction; CABG, coronary artery bypass grafting; ACE, angiotensin conveter enzyme; ns, not significant.
*P-value corresponding from ANOVA analysis.
Measurements of a cross-sectional area of the myocytes (see Supplementary material online, Figure S2A) and cell membrane capacitance (see Supplementary material online, Figure S2B) revealed no significant differences between the three age groups, suggesting that ageing does not affect cell-surface area or cell size. Moreover, T-tubular structures were not detectable in myocytes from any of the three age groups (see Supplementary material online, Figure S2C).
3.2. Ageing reduces the intracellular calcium transient amplitude and decay
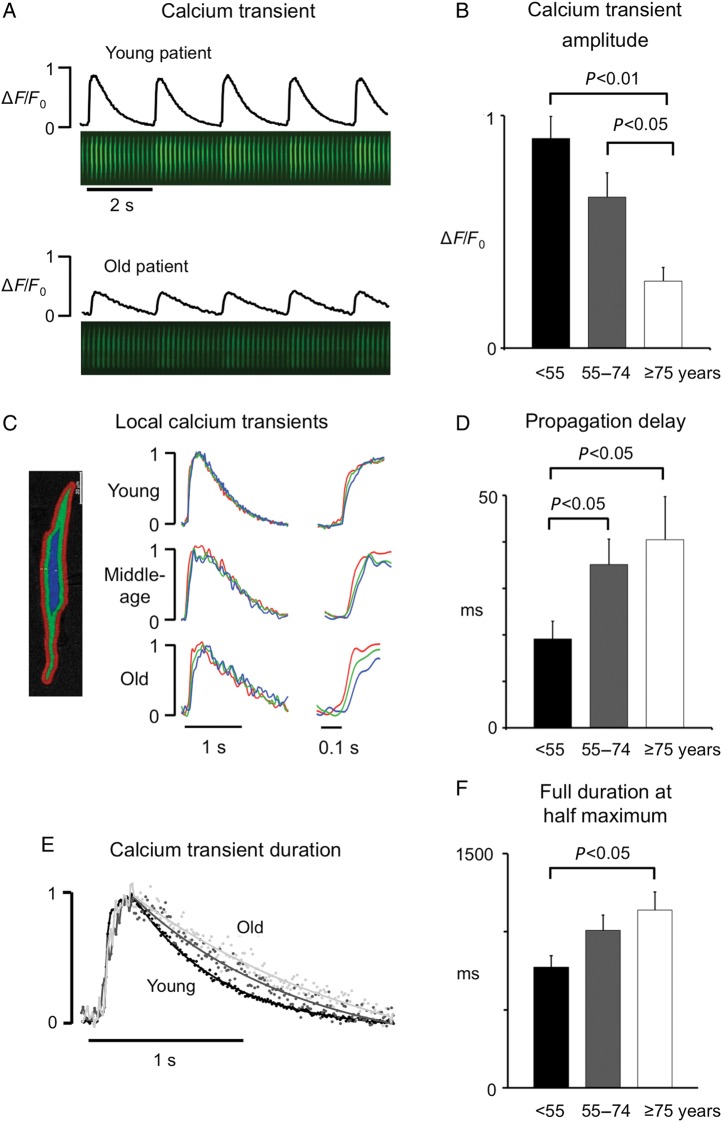
Confocal calcium imaging was used to compare the amplitude and kinetics of intracellular calcium transients between the three age groups. Figure 1A shows contact sheets of consecutive time-averaged calcium images and the resulting calcium transient recorded in right-atrial myocytes from a young (top panel) and an old patient (bottom panel). As shown in Figure 1B, the calcium transient amplitude decreased gradually with age and was 3.2-fold smaller in myocytes from patients 75 years or older than in those younger than 55 years (P < 0.01). Segmentation of the cells in three concentric layers, extending from the sarcolemma to the centre of the cell (Figure 1C), also revealed that there was an age-related delay in the propagation of the calcium transient from the sarcolemma to the cell centre (Figure 1D). This delay was apparently not attributable to the presence of T-tubular structures, as they were not detected in any of the human atrial myocytes analysed (see Supplementary material online, Figure S2C).
Figure 1.
Effects of ageing on the intracellular calcium transient. (A) Calcium transient traces and the corresponding sequence of 68 consecutive time-averaged calcium images, recorded in myocytes from a young and an old patient. (B) Average calcium transient amplitude in myocytes from young (8 cells; n = 7) middle age (10 cells; n = 7), and old (6 cells; n = 5) patients. (C) Calcium transients measured in three concentric rings (shown on the left). Transients were normalized to their peak values in a young, a middle aged, and an old patient. The upstroke of the calcium transient is amplified on the right. Notice the delay between the calcium transient near the sarcolemma (SL; red) and the cell centre (CC; blue) in the old patient. (D) Average time-delay between the calcium transient in the SL and CC for the three patient groups. (E) Superimposed calcium transients normalized to their peak amplitude. (F) Average duration of the calcium transient at half maximal amplitude. P-values for significant differences are indicated above the corresponding bars.
The decay of the calcium transient also became slower with increasing age (Figure 1E), as reflected by a significant longer duration at the half maximal amplitude (FDHM) in the oldest patient group (Figure 1F).
3.3. Ageing reduces the L-type calcium current (ICa)
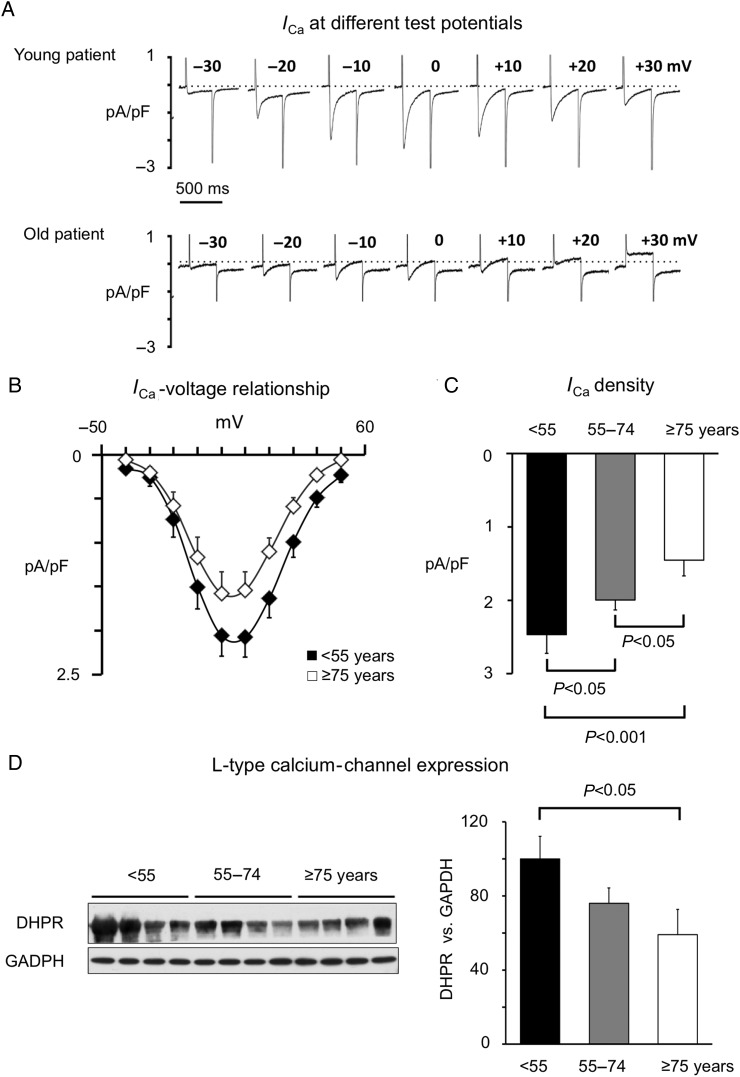
To identify the mechanisms underlying the age-related depression of the calcium transient, we examined the effects of age on sarcolemmal calcium entry. Figure 2A shows recordings of ICa elicited at different membrane potentials in a young and an old patient demonstrating that ageing was associated with depressed ICa amplitude without changes in the shape of the current–voltage relationship (Figure 2B). Accordingly, continuous stimulation at 0.5 Hz revealed an age-dependent decrease in the ICa amplitude at steady state from 2.4 ± 0.3 pA/pF in young to 2.1 ± 0.2 pA/pF in middle aged and 1.4 ± 0.2 pA/pF in old patients (P < 0.001, young vs. old patients, Figure 2C). This effect was independent of confounding clinical factors included in the statistical analysis. Moreover, analysis of the data on a continuum confirmed that there is a significant correlation between age and the ICa density (r = 0.419; P < 0.001; see Supplementary material online, Figure S3). Western blot analysis revealed a concurrent age-related reduction in the expression of the L-type calcium channel alpha-subunits in patients (Figure 2D).
Figure 2.
Effects of ageing on L-type calcium channel expression and current. (A) Representative L-type calcium currents elicited by depolarization to different membrane potentials (indicated above traces) in a young (20 years) and an old patient (77 years). (B) The ICa-voltage relationship in 21 young (<55) and 17 old (≥75) patients. (C) Peak ICa density in 21 young (<55), 42 middle aged (55–74), and 17 old (≥75) patients. (D) Western blot of the L-type calcium channel alpha subunit (DHPR) in seven young (<55 years), eight middle aged, and seven old (≥75 years) patients. Average DHPR levels are expressed as a percentage of the GAPDH level in the right panel. P-values for significant differences are indicated above the corresponding bars.
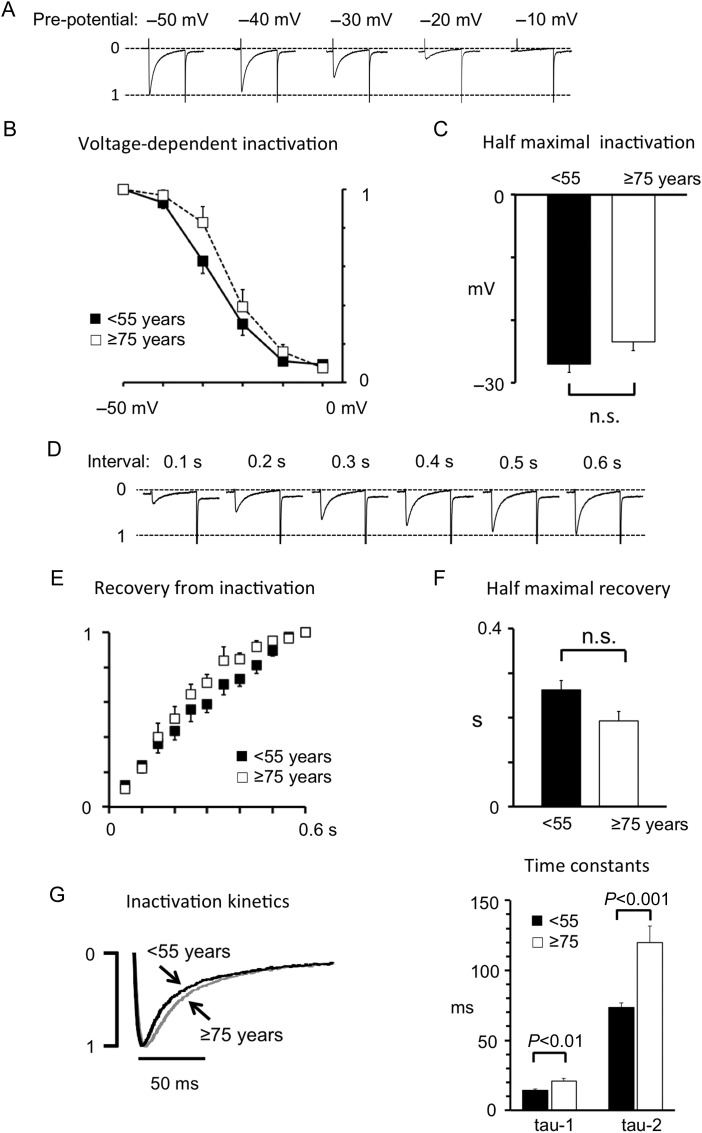
Analysis of intrinsic ICa properties in the older and younger patients showed no significant differences in voltage-dependent inactivation (Figure 3A–C) or in recovery of ICa from inactivation (Figure 3D–F). However, both fast and slow steady-state ICa-inactivation were significantly slower in patients 75 years or older when compared with patients younger than 55 years (Figure 3G). Thus, the time constant (tau-1) for fast ICa inactivation increased by 44% from 14.5 ± 0.9 ms in young to 20.9 ± 1.9 ms in old patients (P < 0.01). Similarly the tau-2 for slow ICa inactivation was 73 ± 3 ms in young vs. 120 ± 12 ms in old patients (P < 0.001).
Figure 3.
Effects of ageing on intrinsic L-type calcium channel properties. (A) Representative ICa traces obtained after a prepulse to different membrane potentials (given above traces). Current traces are normalized to the ICa amplitude at −50 mV. (B) Relationship between the prepulse potential and the ICa amplitude. (C) Bar diagram of the voltage for half-maximal ICa inactivation. Values were obtained by fitting data in (B) with a Boltzmann equation. (D) Representative ICa traces elicited at different times (given above traces) after the preceding stimulation pulse. (E) Relationship between pulse interval and the recovery of the ICa amplitude. Values were normalized to the ICa amplitude at 0.6 s. (F) Bar diagram of the time for half-maximal recovery of ICa. Values were obtained by fitting data in (E) with an exponentially decaying function. (G) Normalized ICa traces from a young and an old patient. Average fast (tau-1) and slow (tau-2) time constants are shown in the right panel. P-values for significant differences are indicated for corresponding bars. All values are from 21 young (<55) and 17 old (≥75) patients (n.s. indicates a non-significant difference).
3.4. Ageing reduces sarcoplasmic reticulum calcium content
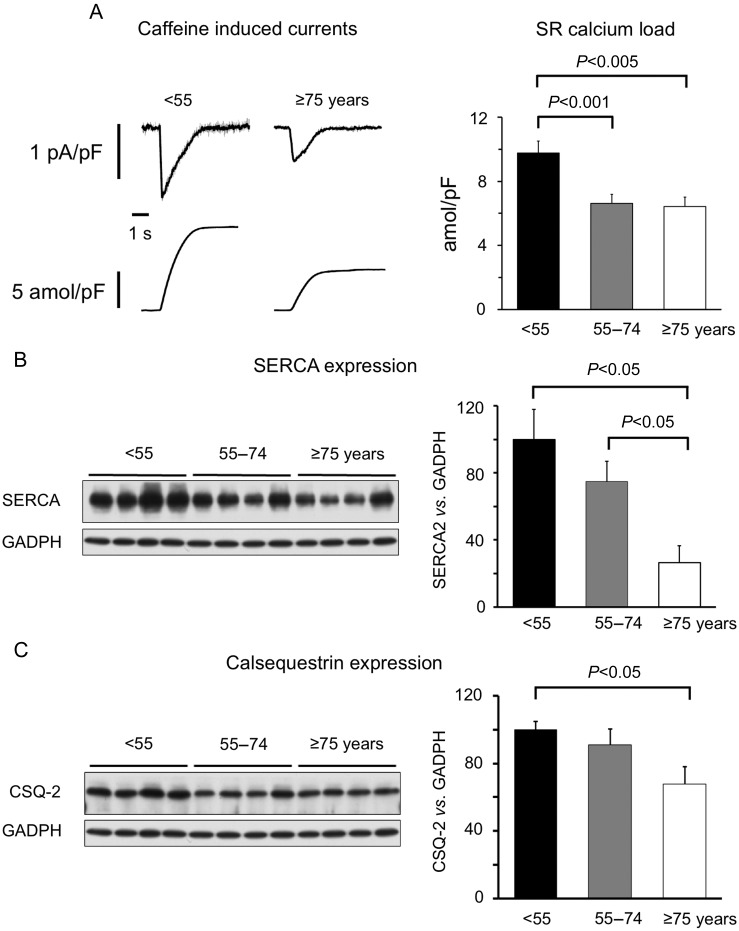
Since ageing concurred with a slowing of the decay of the calcium transient and with down-regulation of sarcolemmal calcium entry through the L-type calcium channel, we also examined whether age affected removal of calcium from the cytosol by the SR during cell relaxation. Indeed, as shown in Figure 4A, we observed an age-related decrease in the caffeine releasable SR calcium content [from 10.1 ± 0.8 amol/pF in the younger patients to 6.5 ± 0.4 amol/pF in the middle aged patients (P < 0.01) and 6.4 ± 0.6 amol/pF in the older group (P < 0.05)]. This effect was independent of confounding clinical factors, and analysis of the data on a continuum confirmed a significant correlation between age and SR calcium content (r = −0.366; P < 0.001; see Supplementary material online, Figure S3). Western blot analysis revealed that not only SERCA2 (Figure 4B) but also Calsequestrin-2 (CSQ-2, Figure 4C) protein levels decreased significantly with age.
Figure 4.
Effects of ageing on sarcoplasmic reticulum calcium uptake. (A) Traces of the transient inward current elicited by caffeine (top traces) and its time integral (bottom traces) in a young and an old patient. Average time integrals are shown on the right. (B) Western blot of SERCA2 in seven young (<55 years), eight middle aged, and seven old (≥75 years) patients. Average SERCA2 levels are expressed as percentage of the GAPDH level on the right. (C) Western blot of CSQ-2 in seven young, eight middle-aged, and seven old patients. Average CSQ-2 levels are expressed as percentage of the GAPDH level on the right.
In contrast, ageing was not associated with significant changes in the expression of the cardiac Na–Ca exchanger nor with its ability to extrude calcium across the sarcolemma (see Supplementary material online, Figure S4).
3.5. Ageing does not alter spontaneous calcium release
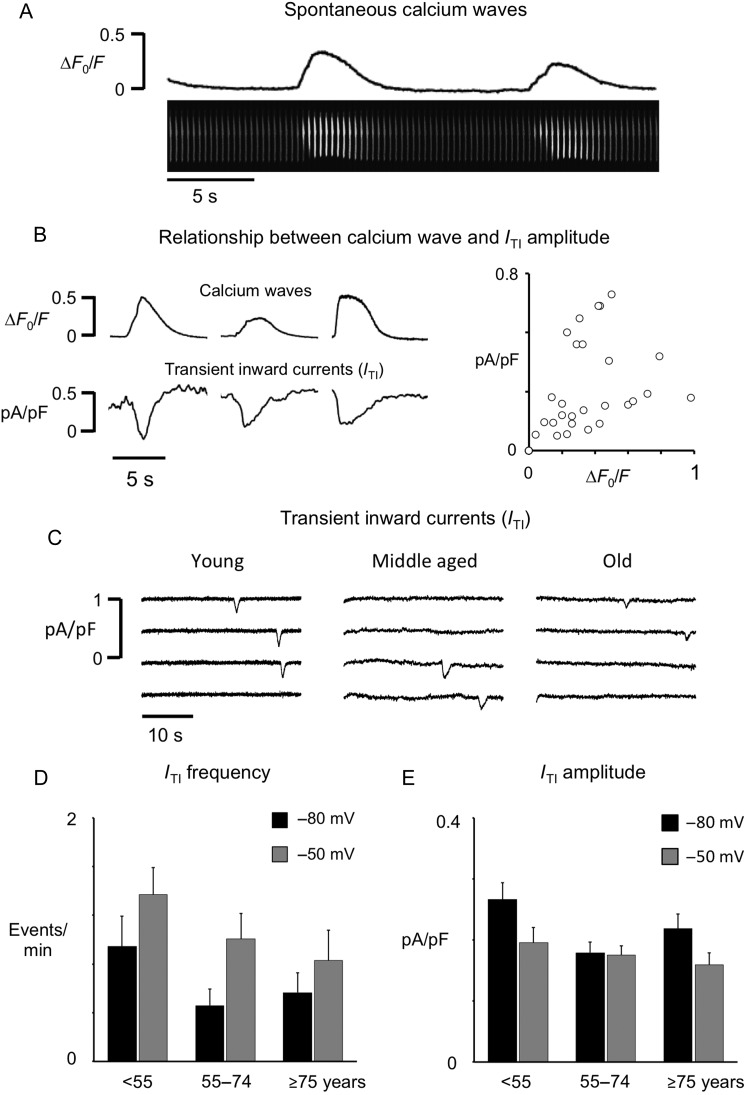
To assess whether ageing could favour a potentially arrhythmogenic release of calcium from the SR, we measured the frequency of spontaneous calcium waves (Figure 5A) and/or the associated transient inward currents (ITI) in myocytes from the three age groups. Figure 5B demonstrates that the ITI amplitude was proportional to the amplitude of the calcium wave (slope = 0.48; r = 0.54, P < 0.05). Similarly, 2D analysis of the calcium image sequences showed that there was a linear relationship between the maximal amplitude of the calcium wave with the mean calcium wave amplitude (slope = 1.6; r = 0.86, P < 0.05). However, no age-related differences were observed in these two parameters. The ITI frequency and the ITI amplitude were comparable among the three age groups (Figure 5C–E) at holding potentials of −80 or −50 mV. Analysis of these data on a continuum confirmed the lack of linear correlation between age and ITI frequency (r = −0.118; P = n.s.) or amplitude (r = −0.237; P = n.s. see Supplementary material online, Figure S3). In line with this, the RyR2 density was not different in a total of 96 myocytes from the three patient groups (see Supplementary material online, Figure S5).
Figure 5.
Effects of ageing on spontaneous SR calcium release. (A) Sequence of consecutive time-averaged calcium images with two spontaneous calcium waves. The whole cell calcium signal is shown above images. (B) Simultaneous recordings of calcium waves and concurrent ITI. The relationship between the ITI amplitude and the calcium wave amplitude is shown on the right. (C) Spontaneous inward currents recorded in myocytes from a young (<55 years), a middle aged (55–74 years), and an old (≥75 years) patient. (D) Average transient inward current (ITI) frequency. (E) Average ITI amplitude. Values are from 21 young, 42 middle aged, and 17 old patients.
4. Discussion
4.1. Main findings
To our knowledge, this is the first study reporting age-related changes in intracellular calcium homeostasis in isolated human right atrial myocytes. We observed a reduction in the expression of several key calcium regulatory proteins with age that lead to coherent concurrent functional alterations in intracellular calcium homeostasis. Specifically we found: (i) reduction in the expression of the alpha sub unit of the L-type calcium channel and decreased ICa density; (ii) reduction in SERCA2 and CSQ-2 expression associated with lower caffeine releasable SR calcium content; and (iii) slower propagation of calcium transient towards the cell centre.
These observations, likely account for the observed three-fold reduction of the calcium transient amplitude and slowing of its decay in patients older than 75 years, which may favour a progressive decline in atrial contractile function with age.
4.2. Effects of ageing on intracellular calcium homeostasis
Most of the studies analysing the effects of ageing on cardiomyocyte function have been performed in ventricular myocytes from small rodents and have provided inconsistent results. Indeed, some studies found increased calcium transients and high ICa density in rodents6 and in sheep,10 whereas others failed to detect changes in ICa or SR calcium handling proteins11 or even found depressed levels of ICa density or SR calcium handling proteins.12–15
Here we report for the first time on age-dependent changes in humans using isolated right-atrial myocytes and tissue samples. The findings revealed a gradual decay in the peak ICa density with age, which is linked to a reduction in the expression of the alpha subunit of the L-type calcium channel. Ageing has also been associated with increased levels of reactive oxygen species (ROS) in rabbit hearts,16 and in humans with AF, elevation of ROS caused by reduced glutathione levels has been proposed to underlie reduced ICa.17 Thus, increased ROS could be a possible mechanism underlying age-related ICa reduction.
Similarly, the SERCA2 and CSQ-2 expression was significantly reduced in patients aged 75 years or older and concurred with significantly lower caffeine releasable SR calcium content and a reduction in the calcium release-induced ICa inactivation. These observations, likely account for the observation of a three-fold smaller calcium transient amplitude and a 50% slowing of its decay in the older patients. However, we cannot rule out additional modulation of SR calcium homeostasis caused by concurrent age-dependent changes in phosphlamban expression and/or phosphorylation. The lower SR calcium content in the old age group might also contribute to the observed slowing of the propagation of the calcium transient from the sarcolemma to the cell centre, especially since t-tubular structures were not detected in any of the three age groups.
While the reduction in SERCA and CSQ-2 expression is expected to contribute to the observed reduction of SR calcium loading in the oldest patients, low CSQ-levels have also been reported to favour spontaneous calcium release and arrhythmia in transgenic mouse models.18–20 However, the ITI frequency was not different among the three age groups examined suggesting that the concurrent lowering of SERCA and L-type calcium channels may reduce the amount of calcium available for binding to CSQ-2 sufficiently to prevent an increase in spontaneous SR calcium release events.21 The lack of significant age-dependent changes in the amplitude of the calcium-wave front or the global calcium wave, and in the ITI frequency also suggests that ageing per se is likely not responsible for a higher rate of spontaneous calcium release events reported in myocytes from AF-patients.8,22,23
4.3. Considerations on the model
Human right-atrial tissue is currently accessible in vivo during pump-on cardiac surgery because cannulation of the right atrium is always required to set-up the extracorporeal circulation. In contrast, extraction of left-atrial tissue samples would only be justifiable in patients undergoing mitral valve surgery, but in these cases usually the left atrium is diseased and the cavity dilated. Thus, analysis of atrial cellular electrophysiology in nearly normal human atrial myocytes is more feasible in the right than in the left atrium.
Cell viability is a common challenge when using isolated human cardiomyocytes. Therefore, only elongated cells with a clear striation were included in this study. Moreover, myocytes with spontaneous ITI were only accepted for further study if the ITI frequency remained stable for at least 5 min in control conditions. Finally, experiments were performed in the amphotericin perforated patch configuration. This technique avoids dialysis of the cytosol through the patch pipette, renders the current recordings stable for more than 30 min, and has yielded reproducible recordings of ICa, SR calcium loading, and ITI in human atrial myocytes.7–9,22,24,25
Another source of potential concern is the heterogeneous clinical profile of patients undergoing cardiac surgery and their individualized pharmacological treatments. To deal with this issue, we included only patients with normal left-atrial size and no previous history of AF. Moreover, possible confounding effects of sex, the presence of valvular or ischaemic heart disease, the treatment of patients with ACE-inhibitors, beta-blockers, angiotensin receptor blockers, calcium antagonists as well as the LV ejection fraction were taken into account in the statistical analysis of the electrophysiological data.
Interestingly, we did not observe any differences in the cross-sectional area or myocyte capacitance among the three patient groups, suggesting that the observed effects of ageing on calcium handling are not secondary to myocyte hypertrophy.
4.4. Clinical implications
The age-related changes in cellular calcium homeostasis observed in our patients may have mechanical and electrophysiological implications.
First, as a result of the reduction of ICa, the SR calcium content and the calcium transient amplitude, it is conceivable that the mechanical efficiency of the atria will decline over age. In this regard, information on the intrinsic atrial contractility in elderly subjects based on invasive atrial haemodynamic recordings is not available. However, echocardiographic studies analysing atrial dynamics and volumes have found left-atrial dilation and reduced atrial mechanical function with ageing, especially in subjects older than 80.2–4,26 Likewise, older age has been associated with reduced atrial functional response to pharmacological adrenergic stress using 3D cardiovascular magnetic resonance.27 This indirect evidence combined with the present data, suggest that further invasive assessment of the intrinsic atrial contractile properties is warranted, to establish the concept that ageing can directly blunt atrial mechanical function.
Secondly, the present study shows that ageing per se causes changes in intracellular calcium homeostasis that have previously been related to the genesis of AF.5 Specifically, the reduction of the ICa density and the calcium transient observed in our old patients are hallmarks of myocytes from patients with AF22,28 or from patients at high risk for this arrhythmia29; suggesting that these age-dependent changes could contribute to increase the risk of AF in the elderly. Possibly, the electrophysiological derangements reported here work in concert with concurrent age-related structural alterations such as increased atrial fibrosis and amyloid deposition,30 or arrhythmogenic conduction abnormalities and local microreentry linked to non-uniform cellular connectivity of structural origin.31 On the other hand, an increased frequency of spontaneous calcium release, another characteristic feature of myocytes from patients with AF,8,22,23 was not observed in the old patient group. This lack of frequent SR calcium release could in part be interpreted as a protective effect of the reduced ICa density and SR calcium content in the elderly.
In summary, we report an ageing-associated depression of L-type calcium channel expression and current, SERCA-2, CSQ-2, and SR calcium content. These electrophysiological derangements likely underlie a 3.5-fold reduction of the calcium transient amplitude and slowing of its decay in myocytes from the older patients and may blunt contraction and relaxation in the ageing human atria. Moreover, reduced ICa and calcium transient are hallmarks of atrial myocytes from patients with AF, which could contribute to increase the risk of this arrhythmia in the elderly.
Supplementary material
Supplementary material is available at Cardiovascular Research online.
Funding
Spanish Ministry of Science and Innovation (SAF2011–30312) and (CNIC-2009-08) to L.H.M.; (DPI2013-44584-R) to R.B., and (SAF2012-40127) to J.M.G.; Instituto de Salud Carlos III (Red de Investigación Cardiovascular RD12/0042/0002) and Fondo Europeo de Desarrollo Regional (FEDER) to J.C. and (RD12/0042/0053) to J.M.G.; and Marie Curie Intra-European Fellowship for Career Development (PIEF-GA-331241) to C.E.M. Funding to pay the Open Access publication charges for this article was provided by Red de Investigación Cardiovascular (RIC) RD12/0042/0002; Ministerio de Economía y Competitividad, Redes Temáticas de Investigación Cooperativa en Salud (RETICS) del Instituto de Salud Carlos III and Fondo Europeo de Desarollo Regional (FEDER).
Acknowledgements
The collaboration of the Cardiac Surgery Team at Hospital de la Santa Creu i Sant Pau is greatly appreciated.
Conflict of interest: none declared.
References
- 1.World Health Statistics (WHS) 2013. http://www.who.int.
- 2.Boyd AC, Richards DA, Marwick T, Thomas L. Atrial strain rate is a sensitive measure of alterations in atrial phasic function in healthy ageing. Heart (British Cardiac Society) 2011;97:1513–1519. [DOI] [PubMed] [Google Scholar]
- 3.Boyd AC, Schiller NB, Leung D, Ross DL, Thomas L. Atrial dilation and altered function are mediated by age and diastolic function but not before the eighth decade. JACC Cardiovasc Imaging 2011;4:234–242. [DOI] [PubMed] [Google Scholar]
- 4.Gardin JM, Henry WL, Savage DD, Ware JH, Burn C, Borer JS. Echocardiographic measurements in normal subjects: evaluation of an adult population without clinically apparent heart disease. J Clin Ultrasound 1979;7:439–447. [DOI] [PubMed] [Google Scholar]
- 5.Kannel WB, Benjamin EJ. Current perceptions of the epidemiology of atrial fibrillation. Cardiol Clin 2009;27:13–24, vii. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Janczewski AM, Lakatta EG. Modulation of sarcoplasmic reticulum Ca(2+) cycling in systolic and diastolic heart failure associated with aging. Heart Fail Rev 2010;15:431–445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Hove-Madsen L, Llach A, Molina CE, Prat-Vidal C, Farre J, Roura S, Cinca J. The proarrhythmic antihistaminic drug terfenadine increases spontaneous calcium release in human atrial myocytes. Eur J Pharmacol 2006;553:215–221. [DOI] [PubMed] [Google Scholar]
- 8.Hove-Madsen L, Llach A, Bayes-Genis A, Roura S, Rodriguez Font E, Aris A, Cinca J. Atrial fibrillation is associated with increased spontaneous calcium release from the sarcoplasmic reticulum in human atrial myocytes. Circulation 2004;110:1358–1363. [DOI] [PubMed] [Google Scholar]
- 9.Llach A, Molina CE, Fernandes J, Padro J, Cinca J, Hove-Madsen L. Sarcoplasmic reticulum and l-type Ca2+ channel activity regulate the beat-to-beat stability of calcium handling in human atrial myocytes. J Physiol 2011;589:3247–3262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Dibb KM, Rueckschloss U, Eisner DA, Isenberg G, Trafford AW. Mechanisms underlying enhanced cardiac excitation contraction coupling observed in the senescent sheep myocardium. J Mol Cell Cardiol 2004;37:1171–1181. [DOI] [PubMed] [Google Scholar]
- 11.Xu A, Narayanan N. Effects of aging on sarcoplasmic reticulum Ca2+-cycling proteins and their phosphorylation in rat myocardium. Am J Physiol 1998;275:H2087–H2094. [DOI] [PubMed] [Google Scholar]
- 12.Dun W, Yagi T, Rosen MR, Boyden PA. Calcium and potassium currents in cells from adult and aged canine right atria. Cardiovasc Res 2003;58:526–534. [DOI] [PubMed] [Google Scholar]
- 13.Guo KK, Ren J. Cardiac overexpression of alcohol dehydrogenase (adh) alleviates aging-associated cardiomyocyte contractile dysfunction: role of intracellular Ca2+ cycling proteins. Aging Cell 2006;5:259–265. [DOI] [PubMed] [Google Scholar]
- 14.Lompre AM, Lambert F, Lakatta EG, Schwartz K. Expression of sarcoplasmic reticulum Ca(2+)-atpase and calsequestrin genes in rat heart during ontogenic development and aging. Circ Res 1991;69:1380–1388. [DOI] [PubMed] [Google Scholar]
- 15.Ren J, Li Q, Wu S, Li SY, Babcock SA. Cardiac overexpression of antioxidant catalase attenuates aging-induced cardiomyocyte relaxation dysfunction. Mech Ageing Dev 2007;128:276–285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Cooper LL, Li W, Lu Y, Centracchio J, Terentyeva R, Koren G, Terentyev D. Redox modification of ryanodine receptors by mitochondria-derived reactive oxygen species contributes to aberrant Ca2+ handling in ageing rabbit hearts. J Physiol 2013;591:5895–5911. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Carnes CA, Janssen PM, Ruehr ML, Nakayama H, Nakayama T, Haase H, Bauer JA, Chung MK, Fearon IM, Gillinov AM, Hamlin RL, Van Wagoner DR. Atrial glutathione content, calcium current, and contractility. J Biol Chem 2007;282:28063–28073. [DOI] [PubMed] [Google Scholar]
- 18.Chopra N, Kannankeril PJ, Yang T, Hlaing T, Holinstat I, Ettensohn K, Pfeifer K, Akin B, Jones LR, Franzini-Armstrong C, Knollmann BC. Modest reductions of cardiac calsequestrin increase sarcoplasmic reticulum Ca2+ leak independent of luminal Ca2+ and trigger ventricular arrhythmias in mice. Circ Res 2007;101:617–626. [DOI] [PubMed] [Google Scholar]
- 19.Postma AV, Denjoy I, Hoorntje TM, Lupoglazoff JM, Da Costa A, Sebillon P, Mannens MM, Wilde AA, Guicheney P. Absence of calsequestrin 2 causes severe forms of catecholaminergic polymorphic ventricular tachycardia. Circ Res 2002;91:e21–e26. [DOI] [PubMed] [Google Scholar]
- 20.Terentyev D, Viatchenko-Karpinski S, Gyorke I, Volpe P, Williams SC, Gyorke S. Calsequestrin determines the functional size and stability of cardiac intracellular calcium stores: mechanism for hereditary arrhythmia. Proc Natl Acad Sci USA 2003;100:11759–11764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Alcalai R, Wakimoto H, Arad M, Planer D, Konno T, Wang L, Seidman JG, Seidman CE, Berul CI. Prevention of ventricular arrhythmia and calcium dysregulation in a catecholaminergic polymorphic ventricular tachycardia mouse model carrying calsequestrin-2 mutation. J Cardiovasc Electrophysiol 2011;22:316–324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Llach A, Molina CE, Prat-Vidal C, Fernandes J, Casado V, Ciruela F, Lluis C, Franco R, Cinca J, Hove-Madsen L. Abnormal calcium handling in atrial fibrillation is linked to up-regulation of adenosine a2a receptors. Eur Heart J 2011;32:721–729. [DOI] [PubMed] [Google Scholar]
- 23.Voigt N, Li N, Wang Q, Wang W, Trafford AW, Abu-Taha I, Sun Q, Wieland T, Ravens U, Nattel S, Wehrens XH, Dobrev D. Enhanced sarcoplasmic reticulum Ca2+ leak and increased Na+–Ca2+ exchanger function underlie delayed afterdepolarizations in patients with chronic atrial fibrillation. Circulation 2012;125:2059–2070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Hove-Madsen L, Prat-Vidal C, Llach A, Ciruela F, Casado V, Lluis C, Bayes-Genis A, Cinca J, Franco R. Adenosine a2a receptors are expressed in human atrial myocytes and modulate spontaneous sarcoplasmic reticulum calcium release. Cardiovasc Res 2006;72:292–302. [DOI] [PubMed] [Google Scholar]
- 25.Molina CE, Leroy J, Richter W, Xie M, Scheitrum C, Lee IO, Maack C, Rucker-Martin C, Donzeau-Gouge P, Verde I, Llach A, Hove-Madsen L, Conti M, Vandecasteele G, Fischmeister R. Cyclic adenosine monophosphate phosphodiesterase type 4 protects against atrial arrhythmias. J Am Coll Cardiol 2012;59:2182–2190. [DOI] [PubMed] [Google Scholar]
- 26.Spencer KT, Mor-Avi V, Gorcsan J, III, DeMaria AN, Kimball TR, Monaghan MJ, Perez JE, Weinert L, Bednarz J, Edelman K, Kwan OL, Glascock B, Hancock J, Baumann C, Lang RM. Effects of aging on left atrial reservoir, conduit, and booster pump function: A multi-institution acoustic quantification study. Heart (British Cardiac Society) 2001;85:272–277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Jeevanantham V, Chughtai H, Little WC, Morgan T, Kitzman DW, Hamilton CA, Hundley WG. Aging reduces left atrial performance during adrenergic stress in middle aged and older patients. Cardiol J 2012;19:45–52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Van Wagoner DR, Pond AL, Lamorgese M, Rossie SS, McCarthy PM, Nerbonne JM. Atrial l-type Ca2+ currents and human atrial fibrillation. Circ Res 1999;85:428–436. [DOI] [PubMed] [Google Scholar]
- 29.Dinanian S, Boixel C, Juin C, Hulot JS, Coulombe A, Rucker-Martin C, Bonnet N, Le Grand B, Slama M, Mercadier JJ, Hatem SN. Downregulation of the calcium current in human right atrial myocytes from patients in sinus rhythm but with a high risk of atrial fibrillation. Eur Heart J 2008;29:1190–1197. [DOI] [PubMed] [Google Scholar]
- 30.Goudis CA, Kallergis EM, Vardas PE. Extracellular matrix alterations in the atria: Insights into the mechanisms and perpetuation of atrial fibrillation. Europace 2012;14:623–630. [DOI] [PubMed] [Google Scholar]
- 31.Spach MS, Heidlage JF, Dolber PC, Barr RC. Mechanism of origin of conduction disturbances in aging human atrial bundles: Experimental and model study. Heart Rhythm 2007;4:175–185. [DOI] [PMC free article] [PubMed] [Google Scholar]