Abstract
Objective
Polycystic liver diseases (PCLDs) are genetic disorders characterised by progressive bile duct dilatation and/or cyst development. Their pathogenesis is a consequence of hyperproliferation, hypersecretion and microRNA alterations in cholangiocytes. Here we evaluate the role of matrix metalloproteases (MMPs) in the hepatic cystogenesis of PCLDs.
Design
Metalloprotease activity was measured by microfluorimetric assays in normal and polycystic cholangiocyte cultures from humans and rats, and gene expression by real time quantitative PCR. The role of cytokines, oestrogens and growth factors present in the cystic fluid of PCLD patients was evaluated for MMP activity. The MMP inhibitor marimastat was examined for cystic expansion in vitro and in polycystic kidney (PCK) rats.
Results
Polycystic human and rat cholangiocytes displayed increased MMP activity, which was associated with increased mRNA levels of different MMPs. Interleukin (IL)-6 and IL-8, and 17β-oestradiol, all stimulated MMP activity in human cholangiocytes. The presence of antibodies against IL-6 and/or IL-8 receptor/s inhibited baseline MMP hyperactivity of polycystic human cholangiocytes but had no effect on normal human cholangiocytes. MMP-3 was overexpressed in cystic cholangiocytes from PCLD human and PCK rat livers by immunohistochemistry. Marimastat reduced MMP hyperactivity of polycystic human and rat cholangiocytes and blocked the cystic expansion of PCK cholangiocytes cultured in three-dimensions. Chronic treatment of 8-week-old PCK rats with marimastat inhibited hepatic cystogenesis and fibrosis.
Conclusions
PCLDs are associated with cholangiocyte MMP hyperactivity resulting from autocrine/paracrine stimulation by IL-6 and IL-8. Inhibition of this MMP hyperactivity with marimastat decreased hepatic cystogenesis in vitro and in an animal model of PCLD, offering a potential therapeutic tool.
INTRODUCTION
Polycystic liver diseases (PCLDs) are genetic disorders characterised by bile duct dilatation and/or cyst development, which become progressively more severe, resulting in significant morbidity and mortality.1–3 They are inherited in a dominant or recessive fashion, and develop alone or in association with polycystic kidney diseases (PKDs).1–3 One form of PCLDs, autosomal dominant polycystic liver disease (ADPLD; ~1: 100 000 prevalence), is characterised by the presence of cysts mainly in the liver.1 PCLDs with renal involvement (PKD) include both autosomal dominant PKD (ADPKD; ~1: 400 prevalence) and autosomal recessive PKD (ARPKD; ~1: 20 000 prevalence).2,3 Currently, there is no standard treatment for PCLDs. Pharmacological approaches include somatostatin analogues and mTOR inhibitors, but most clinical trials have only shown a small reduction in liver volume.1–3 On the other hand, surgical procedures, such as aspiration and sclerotherapy, fenestration, segmental hepatic resection and liver transplantation, show better short term effects but high recurrence and complication rates.1–3
In general, the development of PCLDs starts at puberty as a heterogeneous process with significant intrafamilial variability.1–3 However, for each patient, hepatic cysts grow steadily with age, both in number and size. Although both men and women can develop PCLDs, women usually present a stronger phenotype.1,2,4 Several cytokines, such as interleukin (IL)-6 and IL-8, oestrogens and growth factors (ie, vascular endothelial growth factor (VEGF), hepatocyte growth factor (HGF), epidermal growth factor (EGF), epithelial derived neutrophil activating peptide 78 (ENA78) and growth regulated oncogene α (GROα)) may be secreted by cholangiocytes lining the hepatic cysts and are present in high levels in cystic fluid.4–7 These molecules participate in autocrine/paracrine processes (such as proliferation, secretion and/or angiogenesis), promoting hepatic cystogenesis and representing potential therapeutic targets.4–7 The mechanisms of hepatic cystogenesis result from defects in the ductal plate and involve processes of hyperproliferation, hypersecretion and microRNA alterations in cholangiocytes.1–3 However, there is evidence suggesting that alterations in cholangiocyte–extracellular matrix (ECM) interactions could also have an important role in the development of PCLDs.8–10
Cell matrix interactions involve dynamic events influenced by many physiological and pathological processes. These interactions play a key role in embryogenesis and regeneration, but also in cancer and other diseases,11,12 and as such are potential targets for diagnosis and therapy. The ECM is a complex structure consisting of collagen, proteoglycans, glycosaminoglycans and glycoproteins. ECM is produced by cells, and its remodelling is modulated by the action of different proteases (ie, matrix metalloproteases (MMPs)), natural MMP inhibitors (ie, tissue inhibitor of metalloproteases (TIMPs)) and hormones.11,12 Increasing evidence indicates that patients with hepatorenal polycystic diseases may develop abnormalities of the ECM within the liver and kidney.8,10,13 Although various enzymes can degrade matrix components, MMPs are considered one of the main families of proteases involved in ECM degradation and remodelling.14 MMPs are present in all types of organisms, from non-vertebrates and plants, to the 28 members characterised so far in humans. Most MMP members share a basic common structure and are secreted extracellularly, while other members are anchored to the plasma membrane.14 They are strictly regulated under physiological conditions and are only expressed and active in specific cell types and at particular times and places.
In the present study, we investigated the role of MMPs in hepatic cystogenesis and their potential regulation by cystic fluid components. Moreover, the potential therapeutic impact of MMP inhibition in PCLDs was evaluated both in vitro and in vivo.
MATERIALS AND METHODS
Isolation and culture of normal and polycystic cholangiocytes
Normal and polycystic human cholangiocytes were isolated according to a novel protocol recently described by our group.15 Normal cholangiocytes were isolated from bordering tissue samples obtained during surgical dissection of a local hepatic adenoma in a female patient; only tissue pieces identified as normal by an experienced pathologist were employed.15 In contrast, polycystic human cholangiocytes were isolated from a female patient who had undergone hepatorenal transplant for advanced ADPKD at the Clinic University of Navarra.
Cholangiocyte primary cultures from normal and polycystic kidney (PCK) male rats were also employed.16,17
RT-PCR and quantitative PCR
mRNA levels of cholangiocyte markers (ie, cytokeratin 7 (CK7), cytokeratin 19 (CK19), anion exchanger 2a (AE2a), aquaporin 1 (AQP1)), MMPs, TIMPs, profibrotic and proinflammatory genes were analysed by RT-PCR or quantitative PCR in normal and polycystic cholangiocytes in culture, or in liver tissue from normal and PCK rats. These data are described in the online supplementary materials and methods section.
Immunofluorescence
The presence of the primary cilium in normal and polycystic human cholangiocyte cultures was analysed by immunofluorescence using a specific antibody against acetylated α-tubulin (figure 1), as described in the online supplementary materials and methods section.
Figure 1.

Polycystic human and rat cholangiocytes show increased metalloprotease activity in culture, associated with alterations in the expression levels of matrix metalloproteases (MMPs) and tissue inhibitor of metalloproteases (TIMPs). (A) Microfluorometric assays for the analysis of MMP activity revealed that polycystic human cholangiocytes possess increased activity compared with normal human cholangiocytes in culture. GM6001 (MMP inhibitor) and recombinant MMP-10 were employed as controls in the assay. (B, C) In addition, changes in mRNA expression levels of different MMPs and TIMPs were found in polycystic human cholangiocytes compared with normal cholangiocyte cultures. Metalloproteases were grouped according to their substrate specificity into collagenases, gelatinases, stromelysins, matrilysins, membrane associated metalloproteases and others, showing global increased expression values in most MMP clusters. n, number of wells analysed in each group.
Cytokine detection by ELISA
Protein levels of IL-6 and IL-8 were quantitatively determined in the supernatant of normal and polycystic human cholangiocyte cultures following the manufacturer’s instructions (human IL-6 and IL-8 ELISA Kits: Refs. KHC0061 and KHC0081, respectively; Invitrogen).
Metalloprotease activity
Metalloprotease activity was measured in the supernatants of normal and polycystic cholangiocyte cultures from humans and rats using an assay based on cleavage of the fluorogenic synthetic peptide ES001 (R&D Systems Minneapolis, Minnesota, USA). Details are described in the online supplementary materials and methods section.
Immunohistochemistry
Analysis of MMP-3 protein expression was carried out in liver samples from: (i) normal human individuals, (ii) ADPKD patients, (iii) ARPKD patients, (iv) normal rats and (v) PCK (animal model of ARPKD) rats, as described in the online supplementary materials and methods section.
Three-dimensional culture of rat cholangiocytes
Cystic expansion of three-dimensional cultured PCK bile ducts was monitored for 24 h by light microscopy in the presence or absence of marimastat (200 μM), as described in the online supplementary materials and methods section.
Treatment of normal and PCK rats with marimastat
PCK rats (8 weeks or 18 weeks old) were orally administered marimastat (twice a day, 0.2856 mg/kg/day) for 8 weeks to determine its potential inhibitory effect on hepatic cystogenesis. Additionally, as a control, 8-week-old normal rats were also orally administered marimastat for 1 week. Details are described in the online supplementary materials and methods section.
All in vivo experimental procedures were approved by the ethics committee of the Biodonostia Research Institute (Donostia University Hospital).
Western blot
Changes in Ck19 protein expression were detected by immunoblotting in liver tissue samples from PCK rats, as described in the online supplementary materials and methods section.
Statistical analysis
Data are shown as mean±SEM. Once normality was assessed using D’Agostino–Pearson or Shapiro–Wilks tests, we used the Student’s t test for statistical comparisons between two groups of normally distributed variables, and one way analysis of variance and a subsequent Bonferroni post hoc test for comparisons between more than two groups. When non-parametric methods were required, we used Mann–Whitney tests. Analyses were carried out with GraphPad Prism 5 statistical software. Two tailed p values<0.05 were considered statistically significant.
RESULTS
Isolated and cultured human cholangiocytes show markers of differentiated biliary cells
Normal and polycystic cholangiocytes were isolated from human liver tissues (see online supplementary figure S1A). Analysis of the cellular phenotype by phase contrast microscopy showed that both cell types exhibited a polygonal morphology typical of cholangiocytes in culture (see online supplementary figure S1B). The study of differentiated cholangiocyte markers at the mRNA level showed that both human cell cultures (normal and polycystic) expressed CK7 and CK19 (ie, cytoskeletal proteins that are only expressed by differentiated cholangiocytes within the liver), AE2a and AQP1 (see online supplementary figure S1C). In addition, both normal and polycystic human cholangiocytes in culture possessed primary cilium, a cholangiocyte sensory organelle (see online supplementary figure S1D). Normal and PCK rat cholangiocytes previously characterised were also employed in the following studies.16,17
Polycystic human and rat cholangiocytes showed increased baseline MMP activity
Microfluorometric assays were carried out to analyse baseline MMP activity of the cell cultures. Our data showed that polycystic human cholangiocytes had increased MMP activity compared with normal human cholangiocytes in culture (p<0.001, figure 1A), which was maintained over at least 10 passages of culture (see online supplementary figure S2A). This effect was specific as the MMP activity of both cell types was inhibited in the presence of the MMP inhibitor GM6001 (p < 0.001 for both cell types; figure 1A). In addition, recombinant MMP-10 was used as a positive control for the assay, which was also inhibited by GM6001 (figure 1A). Similarly, PCK rat cholangiocytes also showed increased MMP activity compared with normal rat cholangiocytes in culture (p<0.0001, see online supplementary figure S3A).
The aforementioned baseline MMP hyperactivity of both polycystic human and rat cholangiocytes was associated with changes in mRNA expression levels of MMPs and their natural inhibitors TIMPs. Thus global upregulation of MMPs was found in both polycystic human and rat cholangiocytes compared with normal cells (figure 1B and see online supplementary figure S3B, respectively). Likewise, TIMP1–3 mRNA levels were increased in both polycystic human and rat cholangiocytes (figure 1C and see online supplementary figure S3C, respectively), while TIMP4 expression was decreased in polycystic human cholangiocytes compared with normal cells (figure 1C).
IL-6 and IL-8, and 17β-oestradiol, increase MMP activity in both normal and polycystic human cholangiocytes in culture
The role of cytokines, oestrogens and growth factors present at high levels in the cystic fluid of patients with PCLDs4–7 was tested on MMP activity of both normal and polycystic human cholangiocyte cultures. Our data showed that the presence of IL-6, IL-8 or 17β-oestradiol increased MMP activity of both normal and polycystic human cholangiocytes (figure 2A, B), and no differences in the magnitude of increase were found between the two cell types. These effects were associated with upregulation of different MMPs at the mRNA level (see online supplementary figures S4A–E). The IL-6–IL-8 combination did not induce a synergic effect on MMP activity (data not shown). In contrast, growth factors such as VEGF, HGF, EGF, ENA78 and GROα did not affect MMP activity in any cell type (figure 2A, B) under the experimental conditions employed.
Figure 2.
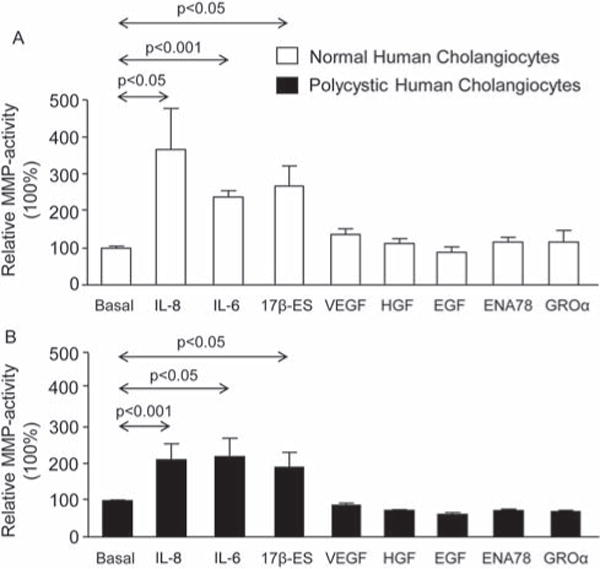
Interleukin (IL)-6, IL-8 and 17β-oestradiol stimulate matrix metalloprotease (MMP) activity of normal and polycystic human cholangiocytes. IL-6, IL-8 and oestrogens—present in high levels in the cystic fluid of patients with polycystic liver diseases (PCLDs)—increased MMP activity of both (A) normal and (B) polycystic human cholangiocytes in culture. On the other hand, growth factors present at high levels in the cystic fluid of patients with PCLDs (ie, vascular endothelial growth factor (VEGF), hepatocyte growth factor (HGF), epidermal growth factor (EGF), epithelial derived neutrophil activating peptide 78 (ENA78) and growth regulated oncogene α (GROα)) did not affect MMP activity of both human cell cultures (A, B). n=5 in each group (number of wells analysed).
Presence of antibodies against IL-6 and/or IL-8 receptors decreased baseline MMP hyperactivity of polycystic human cholangiocytes
The autocrine/paracrine role of IL-6 and IL-8 on baseline MMP hyperactivity of polycystic human cholangiocytes was evaluated by blocking their receptors with specific antibodies (ie, against IL-6R and IL-8R/CXCR1, respectively).
First we determined mRNA levels of IL-6 and IL-8 and found that polycystic human cholangiocytes have increased levels compared with normal human cholangiocyte cultures (p < 0.0001; figure 3A). In addition, IL-6 and IL-8 protein levels were found to be upregulated in the supernatant of polycystic compared with normal cells (p < 0.001 and p < 0.0001, respectively; figure 3B). The presence of antibodies against IL-6R and/or IL-8R decreased baseline MMP hyperactivity of polycystic human cholangiocytes compared with this cell type incubated with a negative control antibody (p < 0.05; figure 3C). Importantly, baseline MMP activity of normal human cholangiocytes was unaffected by the presence of anti-IL-6R and/or anti-IL-8R antibodies in the culture medium compared with this cell type incubated with a negative control antibody (figure 3D).
Figure 3.

Baseline matrix metalloprotease (MMP) hyperactivity of polycystic human cholangiocytes is autocrine/paracrine stimulated by interleukin (IL)-6 and/or IL-8. (A) mRNA levels of IL-6 and IL-8 are overexpressed in polycystic versus normal human cholangiocytes in culture. (B) Extracellular secretion of both IL-6 and IL-8 proteins is increased in polycystic versus normal human cholangiocytes in culture. (C) The presence of antibodies against IL-6 receptor (IL-6R) and/or IL-8 receptor (IL-8R) in the culture medium inhibited baseline MMP hyperactivity of polycystic human cholangiocytes compared with cells incubated with a negative control antibody. (D) In contrast, the presence of antibodies against IL-6R and/or IL-8R did not affect baseline MMP activity of normal human cholangiocytes in culture. n, number of wells analysed in each.
MMP-3 is overexpressed in cystic cholangiocytes within the liver of PCLD patients and PCK rats
The fact that PCLDs may progress through processes of survival, neovascularisation and loss of cell adhesion1–3,10 prompted us to study in vivo the expression of MMP-3, a metalloprotease reported to participate in all of these mechanisms.18 MMP-3 was found to be ~4.5-fold and ~35-fold overexpressed in polycystic human and rat cholangiocyte cultures, respectively (p < 0.0001; figure 1B and see online supplementary figure S3B), and is stimulated by both IL-8 and 17β-oestradiol in normal human cholangiocytes (see online supplementary figure S4B and S4D, respectively).
As shown by immunohistochemistry, MMP-3 protein was overexpressed in cystic cholangiocytes from liver biopsies of ARPKD and ADPKD patients compared with normal individuals (figure 4A). Moreover, MMP-3 was also found to be overexpressed in cystic cholangiocytes (as well as in hepatocytes) from PCK rat livers compared with normal rats (figure 4B). This protein overexpression was mainly localised in the cytoplasm of polycystic human and rat cholangiocytes.
Figure 4.

Matrix metalloprotease 3 (MMP-3) is overexpressed in cystic cholangiocytes of polycystic liver diseases. Immunohistochemical staining showed that MMP-3 protein expression is increased in the cytoplasm of cystic cholangiocytes from both (A) autosomal recessive polycystic kidney disease (ARPKD) and autosomal dominant PKD (ADPKD) patients, and (B) in polycystic kidney (PCK) rats compared with their matched normal liver tissues. Representative images are shown. Magnification 20×. Arrows denote positive staining for MMP-3 in cholangiocytes.
Marimastat decreases baseline MMP activity of human and rat cholangiocytes, and inhibits cystic growth of three-dimensional cultured PCK cholangiocytes
We studied the effect of marimastat, an MMP inhibitor,19–22 on baseline MMP activity of normal and polycystic cholangiocyte cultures from humans and rats. The presence of marimastat (200 μM) in the culture medium inhibited this MMP activity in both normal and polycystic human cholangiocytes (p < 0.001; figure 5A), as well as baseline MMP hyperactivity of PCK rat cholangiocyte cultures (p < 0.01; figure 5B).
Figure 5.

Marimastat inhibits matrix metalloprotease (MMP) activity of human and rat cholangiocytes and suppresses growth of hepatic cysts in three-dimensional cultures. (A) Marimastat (200 μM), a pseudopeptide synthetic MMP inhibitor, is able to inhibit baseline MMP activity of both normal and polycystic human cholangiocytes in culture. (B) Similarly, marimastat inhibits baseline MMP hyperactivity of polycystic kidney (PCK) rat cholangiocytes in culture. n, number of wells analysed in each condition. (C) Representative images of cystic cholangiocytes derived from PCK rats in the presence or absence of marimastat (200 μM) in the culture medium for 24 h. Magnification 4×. Graph shows the percentage of cystic expansion in each condition. The circumferential area of each cyst was measured using Image J software (see material and methods section). n, number of cysts analysed in each condition.
We next investigated the effect of marimastat on the growth of PCK cystic structures using the model of three-dimensional cultured cholangiocytes. The presence of marimastat in the culture media halted the growth of the PCK cholangiocyte cystic structures (p < 0.0001; figure 5C).
Chronic treatment of 8-week-old PCK rats with marimastat halts hepatic cystogenesis
To further test the role of MMPs in the hepatic cystogenesis of PCLDs, marimastat was administered to PCK rats (8 weeks or 18 weeks old) for 8 weeks. Treatment of 8-week-old PCK rats with marimastat decreased hepatic cystogenesis (ie, area of the intrahepatic biliary cysts) compared with matched controls (p < 0.05, figure 6A); this event was associated with hepatic downregulation of both the cholangiocyte marker Ck19 (p < 0.001, figure 6B) and collagen-1a1 (p < 0.05, figure 6C). Interestingly, 8-week-old PCK rats treated with marimastat showed an almost statistically significant downregulation of both IL-6 and Cxcl1 (IL-8 homologue) mRNA levels (p=0.1 and p=0.08; figure 6D). On the other hand, no changes in mRNA expression levels of other profibrotic (ie, transforming growth factor beta (Tgfβ1), α-smooth muscle actin (α-Sma) and connective tissue growth factor (Ctgf)) and proinflammatory (ie, IL-1β) genes were observed (see online supplementary figure S5). In contrast, treatment of 18-week-old PCK rats with marimastat did not affect hepatic cystogenesis (figure 6E) or fibrosis (data not shown). Eighteen-week-old PCK rats show a fully developed liver cystic phenotype (see online supplementary figure S6). Marimastat did not affect renal cystogenesis, also developed by PCK rats (see online supplementary figure S7A,B).
Figure 6.

Chronic treatment of polycystic kidney (PCK) rats with marimastat decreases liver cystogenesis in 8-week-old, but not 18-week-old, animals. (A) Representative images and dot plot showing that hepatic cystic areas are decreased in 8-week-old marimastat treated PCK rats compared with PCK matched controls. Livers were stained with haematoxylin–eosin. (B) This event was associated with hepatic downregulation of the cholangiocyte marker cytokeratin 19 (Ck19) at the protein level. The western blot shows Ck19 protein expression of three representative PCK rats treated with marimastat or placebo. Bar graph shows Ck19 quantification (n=6 in each condition). (C) Representative images of sirius red staining and collagen-1a1 (Col1a1) mRNA levels (bar graph), showing that hepatic collagen levels are decreased in 8-week-old PCK rats treated with marimastat compared with non-treated matched PCK rats. Interestingly, 8-week-old PCK rats treated with marimastat showed an almost statistically significant downregulation of both (D) IL-6 and IL-8 homologue Cxcl1 mRNA levels. (E) Representative haematoxylin–eosin stained images and dot plot showing that hepatic cystic areas are not altered in 18-week-old marimastat treated PCK rats compared with PCK matched controls. n, number of animals analysed in each group.
Finally, treatment of normal rats with marimastat for 1 week did not affect the histology of the liver or kidneys (see online supplementary figure S8), or mRNA expression of the aforementioned profibrotic and proinflammatory genes (data not shown).
No apparent toxic effects were found in any of the treated animals (see online supporting information).
DISCUSSION
The key findings reported here relate to the cellular mechanisms involved in hepatic cystogenesis of PCLDs. We found that cystic cholangiocytes showed MMP hyperactivity, and performed experiments to determine the role of MMP in the development of PCLDs. Our data indicated that: (i) polycystic human and rat cholangiocytes show MMP hyperactivity compared with normal cholangiocyte cultures and (ii) that this event is associated with global overexpression of metalloproteases; (iii) IL-6, IL-8 and 17β-oestradiol all increase MMP activity of human cholangiocyte cultures via overexpression of different MMPs while (iv) the growth factors VEGF, HGF, EGF, ENA78 and GROα show no effect; (v) mRNA and protein levels of IL-6 and IL-8 are increased in polycystic human cholangiocytes, and the presence of antibodies against their receptors inhibits their baseline MMP hyperactivity but does not affect MMP activity of normal human cholangiocyte cultures; (vi) MMP-3, found to be ~4.5-fold and ~35-fold overexpressed in polycystic human and rat cholangiocyte cultures, respectively, is also upregulated in cystic cholangiocytes from both ARPKD and ADPKD livers compared with bile ducts from normal individuals; a similar effect was observed in PCK rat livers compared with normal rats; (vii) marimastat inhibits baseline MMP activity of both human and rat cholangiocyte cultures, and the growth of PCK cystic structures cultured in three-dimensions; and (viii) treatment of 8-week-old PCK rats with marimastat for 8 weeks decreases hepatic cystogenesis and fibrosis. These results are consistent with the notion that polycystic cholangiocytes display MMP hyperactivity as a result of autocrine/paracrine stimulation by cytokines and oestrogens found to be overexpressed in cystic fluid, and that MMPs participate in the hepatic cystogenesis of PCLDs, which makes them potential therapeutic targets.
We had previously reported that hepatic cystogenesis in PCLDs is a consequence of hypersecretion, hyperproliferation and microRNA alterations in cholangiocytes.16,20,23,24 However, there is also evidence suggesting that alterations in the cholangiocyte–ECM interaction could participate in this pathological process.8–10 Several studies have indicated that patients with hepatorenal polycystic diseases develop abnormalities of the ECM in liver and kidneys.8,10,13 Although various enzymes can degrade the matrix components, MMPs are considered one of the leading families of proteases responsible for this remodelling. Therefore, we analysed the role of cholangiocyte produced MMPs on hepatic cystogenesis of PCLDs. Our data indicate that polycystic human and rat cholangiocytes show increased MMP activity compared with normal cholangiocyte cultures, which is associated with global upregulation of MMPs at the mRNA level. MMPs are regulated by complex and varied mechanisms. Their expression levels are generally low in normal adult tissues, but dynamic changes in MMP expression are characteristic of several pathological processes.14 MMP activity is regulated by the action of natural inhibitors called TIMPs, which exert their inhibitory effect by binding covalently to the catalytic site of MMPs.25 Our results showed that mRNA levels of three TIMP members (ie, TIMP1–3) were upregulated in polycystic human and rat cholangiocytes, and expression levels of TIMP4 downregulated in polycystic human cholangiocyte cultures. These data suggest that overexpression of TIMP1–3 could be a compensatory mechanism of polycystic cholangiocytes trying to counterbalance their MMP hyperactivity.
In addition, we tested the role of cytokines (IL-6 and IL-8), oestrogens and growth factors (EGF, HGF, VEGF, ENA78, GROα) present in high levels in the cystic fluid of PCLD patients5,7 on MMP activity of both normal and polycystic human cholangiocytes. We found that IL-6, IL-8 and 17β-oestradiol all stimulated MMP activity of both normal and polycystic human cholangiocyte cultures, and these effects were associated with changes in the gene expression of different MMPs. Interestingly, these effects were not observed in both cell types in the presence of the aforementioned growth factors. Our data support the hypothesis that different molecules abnormally present in the cystic fluid of PCLD patients can regulate certain cholangiocyte functions, in particular MMP activity and the ECM.
As polycystic human cholangiocytes show higher baseline MMP activity than normal human cholangiocyte cultures, and both IL-6 and/or IL-8 increase MMP activity of human cholangiocyte cultures, we tested the potential autocrine/paracrine role of these two cytokines on baseline MMP hyperactivity of polycystic human cholangiocytes. For this purpose, we first evaluated both mRNA expression and protein secretion of IL-6 and IL-8 in normal and polycystic human cholangiocyte cultures. Data showed that both cytokines were overexpressed at the mRNA level and oversecreted in the latter. Subsequently, we incubated both cells types under baseline conditions with antibodies against IL-6 and/or IL-8 receptors, and found that MMP hyperactivity of polycystic cholangiocytes was inhibited compared with these cells in the presence of control IgG antibodies. Interestingly, such effects were not observed in normal human cholangiocytes incubated with the aforementioned antibodies, indicating that baseline MMP activity of normal human cholangiocytes is not associated with an autocrine/paracrine effect of IL-6 and/or IL-8. In addition, the combination of IL-6R and IL-8R antibodies did not have an additive or synergic effect on baseline MMP hyperactivity of polycystic human cholangiocytes, suggesting that both cytokines might modulate MMPs through a common signalling pathway; in this regard, it was reported that both cytokines can regulate MMP expression through activation of the STAT3 signalling pathway.26,27 Moreover, analysis of the role of IL-6 and IL-8 in the regulation of cholangiocyte MMP activity could also be important in the pathogenesis of other biliary diseases associated with inflammation, such as primary biliary cirrhosis, primary sclerosis cholangitis or cholangiocarcinoma, which need further research.
Both men and women can develop PCLDs, but women are commonly associated with a worse prognosis.1,2,4 It was recently reported that oestrogen levels are upregulated in the serum and hepatic cystic fluid of ADPKD female patients, and that 17β-oestradiol stimulates the proliferation of ADPKD human cystic cholangiocytes; moreover, these events were associated with baseline overexpression of the oestrogen receptors (ER-α and ER-β) in cystic cholangiocytes.4 Here we found that oestrogens can also stimulate hepatic cystogenesis through activation of MMP activity in cystic cholangiocytes, providing new evidence for the involvement of oestrogens in PCLDs.
PCLDs are characterised by processes of survival, neovascularisation and loss of cell adhesion.1–3 Thus we wanted to analyse in vitro and in vivo the expression of MMP-3, a metalloprotease reported to participate in all of these mechanisms.18 As mentioned above, MMP-3 mRNA expression is upregulated in polycystic human and rat cholangiocytes, and is stimulated by IL-8 and 17β-oestradiol in normal human cholangiocyte cultures. MMP-3 was found to be overexpressed in biliary cells lining the hepatic cyst of ARPKD and ADPKD patients (and PCK rats) compared with liver biopsies from normal human (and rat) individuals; this overexpression was found in the cytoplasm of the cells. These immunohistochemical experiments, together with our aforementioned in vitro data, strongly support the fact that cholangiocytes from PCLD patients exhibit increased MMP activity compared with normal controls, which prompted us to study the potential therapeutic role of MMP inhibition in PCLDs. First, we tested the role of the MMP inhibitor marimastat on MMP activity of normal and polycystic cholangiocyte cultures. Marimastat is a pseudopeptide inhibitor belonging to the family of hydroxamates, which have a structure mimicking the substrate binding site of MMPs.19–22 Thus marimastat binds to the catalytic site of MMPs acting as a Zn2+ chelator. It inhibits a wide spectrum of MMPs, but particularly MMP-3.28 Marimastat is currently being used in clinical trials for the treatment of various types of cancers.20,22 Our data showed that marimastat can inhibit MMP activity of both normal and polycystic human cholangiocytes, as well as MMP hyperactivity of PCK rat cholangiocytes. Based on these results, we decided to analyse the potential therapeutic role of marimastat in inhibiting the hepatic cystogenesis of PCLDs. Thus we tested the effect of marimastat on the growth of PCK cystic structures cultured in three-dimensions and on the hepatic cystogenesis developed by PCK rats. Our data demonstrated that marimastat blocked the growth of the PCK cystic structures cultured in three-dimensions compared with controls, and that long term treatment of 8-week-old PCK rats with marimastat halted the development and progression of liver disease compared with PCK matched controls, without affecting renal cystogenesis. Moreover, marimastat did not affect body weight, or liver or kidneys weight at the moment of sacrifice, or serological levels of biochemical markers. As marimastat was orally administered to have a direct effect on the liver through the enterohepatic circulation and to reduce potential systemic side effects, we cannot exclude the possibility that parenteral administration could also have an effect on renal cystogenesis. Various studies have provided evidence for the importance of certain MMPs and TIMPs in the pathogenesis of PCK diseases.8 In this regard, it was reported that daily intraperitoneal injections of batimastat (another synthetic MMP inhibitor with less inhibitory effect on MMPs, poor solubility and toxic features)28 to Cy/+ rats (animal model of PCK diseases) resulted in reduction of renal cystogenesis.29 On the other hand, chronic treatment of 18-week-old PCK rats with marimastat did not affect their hepatorenal cystogenesis, suggesting that cholangiocyte MMP hyperactivity may be key for the development of PCLDs, but may not have a role when the disease is fully developed. Therefore, future therapeutic strategies targeting cholangiocyte MMP hyperactivity should focus on PCLD patients under disease progression. These data, together with previous reports in PCK diseases, emphasise the importance of MMPs in cystogenesis.
In summary, our results identify MMP hyperactivity of polycystic cholangiocytes as a molecular mechanism involved in the hepatic cystogenesis of PCLDs, and provide direct evidence that this process is autocrine/paracrine regulated by cytokines and oestrogens present in abundance in the cystic fluid of these patients. Moreover, our data introduce the concept that targeting of MMP hyperactivity in cystic cholangiocytes with marimastat could offer a potential therapy for these diseases.
Supplementary Material
Significance of this study.
What is already known on this subject?
-
►
Polycystic liver diseases (PCLDs) are genetic disorders characterised by bile duct dilatation and/or cyst development, resulting in significant morbidity and mortality due to the lack of effective therapeutic approaches.
-
►
Several cytokines, oestrogens and growth factors present at high levels in the cystic fluid of PCLDs patients promote hepatic cystogenesis via stimulation of proliferation, secretion and/or angiogenesis.
-
►
Remodelling of the extracellular matrix by proteases, such as matrix metalloproteases (MMPs), could have an important role in the development of PCLDs.
What are the new findings?
-
►
Polycystic human and rat cholangiocytes present increased MMP activity, associated with alterations in the expression patterns of MMPs, which are found to be globally upregulated.
-
►
MMP hyperactivity of polycystic human cholangiocytes is autocrine/paracrine regulated by interleukin 6 and interleukin 8, and is also stimulated by 17β-oestradiol.
-
►
Targeting of MMP hyperactivity with marimastat inhibited the cystic expansion of polycystic kidney rat (PCK) cholangiocytes in vitro, and the hepatic cystogenesis and fibrosis of treated 8-week-old PCK rats.
How might it impact on clinical practice in the foreseeable future?
-
►
New strategies are needed to successfully inhibit the progression of PCLDs.
-
►
MMPs and cystic fluid components represent potential diagnostic and prognostic markers of PCLDs, as well as potential therapeutic targets to improve the pathogenesis of these disorders.
-
►
Targeting of MMP hyperactivity in cystic cholangiocytes with marimastat is a potential therapy for PCLDs.
Acknowledgments
The authors thank Dr José Antonio Rodríguez García for the gift of the recombinant MMP-10 protein.
Funding This work was supported by grants from: the Spanish Ministries of Economy and Competitiveness (JMB: FIS PI12/00380), Science and Technology (JJGM: SAF2010-15517) and the ‘Instituto de Salud Carlos III’ (JMB, LB, JJGM and JP: Ciberehd), Spain; the Department of Industry of the Basque Country (JMB: SAIO12-PE12BN002), the Spanish UTE for CIMA Project (JMB and JP) and by the NIH, USA (NFL: DK24031 grant). JMB and OE are funded by the ‘Asociación Española Contra el Cancer (AECC)’.
Footnotes
Additional material is published online only. To view please visit the journal online (http://dx.doi.org/10.1136/gutjnl-2013-305281).
Contributors ADU, PM-G, MJP, OE, MM-A, AA, EL, EH, JPJ and JMB performed the experiments. ADU, PM-G, MJP, OE, MM-A, AA, RJ-A, EL, EH, JPJ, MM, JJGM, MGF-B, TVM, NFL, JP, LB and JMB discussed and interpreted the data, and drafted the manuscript.
Competing interests None.
Ethics approval All in vivo experimental procedures were approved by the ethics committee of the Biodonostia Research Institute (Donostia University Hospital).
Provenance and peer review Not commissioned; externally peer reviewed.
References
- 1.Gevers TJ, Drenth JP. Diagnosis and management of polycystic liver disease. Nat Rev Gastroenterol Hepatol. 2013;10:101–8. doi: 10.1038/nrgastro.2012.254. [DOI] [PubMed] [Google Scholar]
- 2.Masyuk T, LaRusso N. Polycystic liver disease: new insights into disease pathogenesis. Hepatology. 2006;43:906–8. doi: 10.1002/hep.21199. [DOI] [PubMed] [Google Scholar]
- 3.Strazzabosco M, Somlo S. Polycystic liver diseases: congenital disorders of cholangiocyte signaling. Gastroenterology. 2011;140:1855–9. doi: 10.1053/j.gastro.2011.04.030. 9 e1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Alvaro D, Onori P, Alpini G, et al. Morphological and functional features of hepatic cyst epithelium in autosomal dominant polycystic kidney disease. Am J Pathol. 2008;172:321–32. doi: 10.2353/ajpath.2008.070293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Amura CR, Brodsky KS, Gitomer B, et al. CXCR2 agonists in ADPKD liver cyst fluids promote cell proliferation. Am J Physiol Cell Physiol. 2008;294:C786–96. doi: 10.1152/ajpcell.00457.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Fabris L, Cadamuro M, Fiorotto R, et al. Effects of angiogenic factor overexpression by human and rodent cholangiocytes in polycystic liver diseases. Hepatology. 2006;43:1001–12. doi: 10.1002/hep.21143. [DOI] [PubMed] [Google Scholar]
- 7.Nichols MT, Gidey E, Matzakos T, et al. Secretion of cytokines and growth factors into autosomal dominant polycystic kidney disease liver cyst fluid. Hepatology. 2004;40:836–46. doi: 10.1002/hep.20401. [DOI] [PubMed] [Google Scholar]
- 8.Catania JM, Chen G, Parrish AR. Role of matrix metalloproteinases in renal pathophysiologies. Am J Physiol Renal Physiol. 2007;292:F905–11. doi: 10.1152/ajprenal.00421.2006. [DOI] [PubMed] [Google Scholar]
- 9.Murray SL, Grubman SA, Perrone RD, et al. Matrix metalloproteinase activity in human intrahepatic biliary epithelial cell lines from patients with autosomal dominant polycystic kidney disease. Connect Tissue Res. 1996;33:249–56. doi: 10.3109/03008209609028882. [DOI] [PubMed] [Google Scholar]
- 10.Waanders E, Van Krieken JH, Lameris AL, et al. Disrupted cell adhesion but not proliferation mediates cyst formation in polycystic liver disease. Mod Pathol. 2008;21:1293–302. doi: 10.1038/modpathol.2008.115. [DOI] [PubMed] [Google Scholar]
- 11.Page-McCaw A, Ewald AJ, Werb Z. Matrix metalloproteinases and the regulation of tissue remodelling. Nat Rev Mol Cell Biol. 2007;8:221–33. doi: 10.1038/nrm2125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Sbardella D, Fasciglione GF, Gioia M, et al. Human matrix metalloproteinases: an ubiquitarian class of enzymes involved in several pathological processes. Mol Aspects Med. 2012;33:119–208. doi: 10.1016/j.mam.2011.10.015. [DOI] [PubMed] [Google Scholar]
- 13.Yasoshima M, Sato Y, Furubo S, et al. Matrix proteins of basement membrane of intrahepatic bile ducts are degraded in congenital hepatic fibrosis and Caroli’s disease. J Pathol. 2009;217:442–51. doi: 10.1002/path.2472. [DOI] [PubMed] [Google Scholar]
- 14.Klein T, Bischoff R. Physiology and pathophysiology of matrix metalloproteases. Amino Acids. 2011;41:271–90. doi: 10.1007/s00726-010-0689-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Banales JM, Saez E, Uriz M, et al. Up-regulation of microRNA 506 leads to decreased anion exchanger 2 expression in biliary epithelium of patients with primary biliary cirrhosis. Hepatology. 2012;56:687–97. doi: 10.1002/hep.25691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Banales JM, Masyuk TV, Gradilone SA, et al. The cAMP effectors Epac and protein kinase a (PKA) are involved in the hepatic cystogenesis of an animal model of autosomal recessive polycystic kidney disease (ARPKD) Hepatology. 2009;49:160–74. doi: 10.1002/hep.22636. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Muff MA, Masyuk TV, Stroope AJ, et al. Development and characterization of a cholangiocyte cell line from the PCK rat, an animal model of autosomal recessive polycystic kidney disease. Lab Invest. 2006;86:940–50. doi: 10.1038/labinvest.3700448. [DOI] [PubMed] [Google Scholar]
- 18.Van Hove I, Lemmens K, Van de Velde S, et al. Matrix metalloproteinase-3 in the central nervous system: a look on the bright side. J Neurochem. 2012;123:203–16. doi: 10.1111/j.1471-4159.2012.07900.x. [DOI] [PubMed] [Google Scholar]
- 19.Groves MD, Puduvalli VK, Hess KR, et al. Phase II trial of temozolomide plus the matrix metalloproteinase inhibitor, marimastat, in recurrent and progressive glioblastoma multiforme. J Clin Oncol. 2002;20:1383–8. doi: 10.1200/JCO.2002.20.5.1383. [DOI] [PubMed] [Google Scholar]
- 20.Nemunaitis J, Poole C, Primrose J, et al. Combined analysis of studies of the effects of the matrix metalloproteinase inhibitor marimastat on serum tumor markers in advanced cancer: selection of a biologically active and tolerable dose for longer-term studies. Clin Cancer Res. 1998;4:1101–9. [PubMed] [Google Scholar]
- 21.Novak KB, Le HD, Christison-Lagay ER, et al. Effects of metalloproteinase inhibition in a murine model of renal ischemia–reperfusion injury. Pediatr Res. 2010;67:257–62. doi: 10.1203/PDR.0b013e3181ca0aa2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Rosenbaum E, Zahurak M, Sinibaldi V, et al. Marimastat in the treatment of patients with biochemically relapsed prostate cancer: a prospective randomized, double-blind, phase I/II trial. Clin Cancer Res. 2005;11:4437–43. doi: 10.1158/1078-0432.CCR-04-2252. [DOI] [PubMed] [Google Scholar]
- 23.Banales JM, Masyuk TV, Bogert PS, et al. Hepatic cystogenesis is associated with abnormal expression and location of ion transporters and water channels in an animal model of autosomal recessive polycystic kidney disease. Am J Pathol. 2008;173:1637–46. doi: 10.2353/ajpath.2008.080125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Lee SO, Masyuk T, Splinter P, et al. MicroRNA15a modulates expression of the cell-cycle regulator Cdc25A and affects hepatic cystogenesis in a rat model of polycystic kidney disease. J Clin Invest. 2008;118:3714–24. doi: 10.1172/JCI34922. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Murphy G. Tissue inhibitors of metalloproteinases. Genome Biol. 2011;12:233. doi: 10.1186/gb-2011-12-11-233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Akira S, Nishio Y, Inoue M, et al. Molecular cloning of APRF, a novel IFN-stimulated gene factor 3 p91-related transcription factor involved in the gp130-mediated signaling pathway. Cell. 1994;77:63–71. doi: 10.1016/0092-8674(94)90235-6. [DOI] [PubMed] [Google Scholar]
- 27.Yang Q, Zhang XF, Pollard TD, et al. Arp2/3 complex-dependent actin networks constrain myosin II function in driving retrograde actin flow. J Cell Biol. 2012;197:939–56. doi: 10.1083/jcb.201111052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Rothenberg ML, Nelson AR, Hande KR. New drugs on the horizon: matrix metalloproteinase inhibitors. Stem Cells. 1999;17:237–40. doi: 10.1002/stem.170237. [DOI] [PubMed] [Google Scholar]
- 29.Obermuller N, Morente N, Kranzlin B, et al. A possible role for metalloproteinases in renal cyst development. Am J Physiol Renal Physiol. 2001;280:F540–50. doi: 10.1152/ajprenal.2001.280.3.F540. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


