Abstract
The Lyme disease spirochete, Borrelia burgdorferi, controls protein expression patterns during its tick-mammal infection cycle. Earlier studies demonstrated that B. burgdorferi synthesizes 4,5-dihydroxy-2,3-pentanedione (autoinducer-2 [AI-2]) and responds to AI-2 by measurably changing production of several infection-associated proteins. luxS mutants, which are unable to produce AI-2, exhibit altered production of several proteins. B. burgdorferi cannot utilize the other product of LuxS, homocysteine, indicating that phenotypes of luxS mutants are not due to the absence of that molecule. Although a previous study found that a luxS mutant was capable of infecting mice, a critical caveat to those results is that bacterial loads were not quantified. To more precisely determine whether LuxS serves a role in mammalian infection, mice were simultaneously inoculated with congenic wild-type and luxS strains, and bacterial numbers were assessed using quantitative PCR. The wild-type bacteria substantially outcompeted the mutants, suggesting that LuxS performs a significant function during mammalian infection. These data also provide further evidence that nonquantitative infection studies do not necessarily provide conclusive results and that regulatory factors may not make all-or-none, black-or-white contributions to infectivity.
INTRODUCTION
Borrelia burgdorferi, the spirochetal agent of Lyme disease, encounters numerous environments during the transmission, dissemination, and colonization stages of its tick-mammal infectious cycle. To facilitate those host-pathogen interactions, the bacterium controls production of proteins and other factors throughout the cycle. Investigations into borrelial gene regulation have revealed several overlapping regulons, providing this bacterium with mechanisms to “fine-tune” the expression of genes and proteins to appropriate levels (1–3).
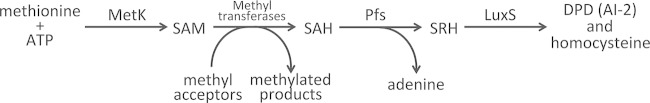
Bacteria use S-adenosylmethionine (SAM) as the methyl donor for methylation reactions (Fig. 1). In many species, including B. burgdorferi, the resulting by-product, S-adenosylhomocysteine (SAH), is detoxified by Pfs to S-ribosylhomocysteine (SRH). That product, in turn, is broken down by LuxS into homocysteine and 4,5-dihydroxy-2,3-pentanedione (DPD) (4–9). Although some bacterial species are able to recycle homocysteine into methionine, genetic and biochemical analyses demonstrated that B. burgdorferi lacks the necessary enzymes and thus cannot use homocysteine (8, 10). Several bacterial species, including the syphilis spirochete, Treponema pallidum, produce a Pfs enzyme but lack LuxS, indicating that SRH is not inhibitory to bacterial growth (8, 11). These observations beg the question of why B. burgdorferi possesses a LuxS enzyme.
FIG 1.
Activated methyl pathway of B. burgdorferi. MetK synthesizes S-adenosylmethionine (SAM) from ATP and methionine. SAM then acts as a methyl donor for many metabolic steps, also producing the by-product S-adenosylhomocysteine (SAH). SAH is toxic, so Pfs cleaves that molecule to produce adenine and S-ribosylhomocysteine (SRH). SRH is nontoxic, and some bacterial species, such as the spirochete Treponema pallidum, end the pathway at this step. B. burgdorferi instead uses LuxS to cleave SRH into homocysteine and 4,5-dihydroxy-2,3-pentanedione (DPD). Biochemical and genetic analyses demonstrated that B. burgdorferi lacks the ability to further metabolize homocysteine (7, 10). DPD, also known as autoinducer-2 (AI-2), is secreted by B. burgdorferi into the environment (7).
The other product of the LuxS-catalyzed reaction, DPD, is also known as autoinducer-2 (AI-2). That molecule is used in an intercellular signaling mechanism by Vibrio harveyi to control bioluminescence (12–15). Salmonella enterica serovar Typhimurium also possesses a mechanism to detect and respond to AI-2 (12, 16–18). Phenotypic changes have been observed in luxS mutants of other bacterial species, although, since many of those bacteria possess a complete activated methyl cycle, it is not always clear whether the effects of luxS mutations are due to an inability to produce AI-2 or homocysteine (19).
As noted above, B. burgdorferi cannot use homocysteine, simplifying interpretations of data obtained from borrelial luxS mutants. Comparative analyses of wild-type and luxS mutant B. burgdorferi strains demonstrated differences in expression levels of numerous proteins (7, 20). Moreover, addition of AI-2 to cultured wild-type and luxS mutant B. burgdorferi strains measurably affected expression levels of several borrelial proteins. These included the VlsE, ErpA, and IpLA7 proteins, which are involved in mammalian infection (4, 7, 20). B. burgdorferi significantly increases luxS transcription during transmission from ticks to mammals (21).
A previous study examined whether a B. burgdorferi luxS mutant is able to infect mice (22). Animals were injected with luxS bacteria, and then tissues from those mice were incubated in borrelial culture medium. Quantification of bacteria in mouse tissues was not attempted. Despite the limitations of this nonquantitative approach, the authors interpreted outgrowth of bacteria as an indication that “a LuxS/AI-2 system is not involved in the overall mammalian infectious process, or, at the very least, in mammalian host adaptation by B. burgdorferi” (22). A subsequent study by the same group, again using nonquantitative methods, demonstrated that luxS mutant B. burgdorferi can also colonize ticks (23).
To reconcile the effects of AI-2 and luxS deletions on borrelial gene and protein expression patterns with the ability of luxS-deficient bacteria to infect mice, we used the same wild-type and luxS strains as in the earlier infection studies, but we performed quantitative PCR (qPCR) to measure bacterial loads in mouse tissues. Detailed analyses indicated that the luxS mutant was significantly less infectious than the wild type. Thus, the data on the previously analyzed luxS mutant actually support the hypothesis that LuxS provides a significant advantage to B. burgdorferi during mammalian infection.
MATERIALS AND METHODS
Bacteria.
The previously described Borrelia burgdorferi strain 297 and an isogenic luxS mutant, AH309, were provided by Michael Norgard (University of Texas Medical Center) (22, 23). B. burgdorferi cells were cultured in Barbour-Stoenner-Kelly II (BSK-II) broth at 35°C (24). Bacterial culture densities were determined by visual enumeration, using dark-field microscopy and a Petroff-Hausser counting chamber. There were no detectable differences in growth rate or other phenotypes between cultured strains 297 and AH309.
Mouse infections.
All animal studies were performed under a protocol approved by the University of Kentucky Institutional Animal Care and Use Committee and in facilities of the University of Kentucky Division of Laboratory Animal Resources. BALB/cJ mice were used for all infection studies because they are as susceptible to B. burgdorferi infection as are other inbred strains, while being less likely to develop stressful arthritis than strains such as C3H/HeN (25, 26).
Mid-exponential-phase cultures (approximately 107 bacteria/ml) were adjusted to a density of 105 spirochetes/ml with sterile phosphate-buffered saline (PBS). For individual infection studies, cohorts of 8 BALB/cJ mice were injected subcutaneously with 100 μl of either B. burgdorferi 297 or AH309, resulting in a dose of 104 spirochetes. For competitive infection studies, 8 BALB/cJ mice were injected subcutaneously with 200 μl of a 1:1 mixture of 105 spirochetes of each strain/ml (i.e., 104 spirochetes of each strain per mouse). After 28 days, mice were euthanized, and hearts, urinary bladders, and ears were collected and snap-frozen at −80°C.
DNA isolation.
For use in PCR specificity studies and as a reference for quantification studies, total bacterial DNAs were purified from mid-exponential-phase cultures (approximately 107 bacteria/ml) of strains 297 and AH309 by use of DNeasy blood and tissue kits (Qiagen, Germantown, MD). For analyses of bacterial loads in mouse tissues, total DNAs were isolated using a Mo Bio Ultraclean tissue and cell DNA isolation kit (Mo Bio Laboratories, Carlsbad, CA).
qPCR.
Bacterial burdens in mouse tissues were assessed using Idaho Technologies/BioFire buffers (BioFire Diagnostics, Salt Lake City, UT) and Platinum Taq polymerase (Life Technologies, Grand Island, NY) with a CFX96 Touch real-time PCR detection platform (Bio-Rad, Hercules, CA). Oligonucleotide primers are listed in Table 1. Cycling was performed as follows: 94°C for 3 min and 40 cycles of 94°C for 10 s followed by 30 s at 60°C. Standard curves for each oligonucleotide pair were generated by diluting a known quantity of genomic DNA in a series of 10-fold serial dilutions. Threshold cycle (CT) values obtained for experimental samples were then plotted against this curve to determine quantities of each target. Melting curve analyses were performed to assess the presence of single products. Results were analyzed using CFX Manager software (Bio-Rad). Data comparisons were analyzed by unpaired two-tailed t tests.
TABLE 1.
Oligonucleotide primers used for PCR
| Purpose | Primer name | Primer sequence (5′ to 3′)a |
|---|---|---|
| Detection of mouse chromosomes | nido-F | CCAGCCACAGAATACCATCC |
| nido-R | GGACATACTCTGCTGCCATC | |
| Detection of B. burgdorferi chromosomes | flaB-F | GGAGCAAACCAAGATGAAGC |
| flaB-R | TCCTGTTGAACACCCTCTTG | |
| Detection of wild-type luxS | luxS-F | GAGCACATAGGAGCTACTTTACTT |
| luxS-R | TGAGACTAAGTCAACAAGATC-TTTAC | |
| Detection of AH309 mutant luxS locus | ermC-F | AAACGCTCATTGGCATTACTTT |
| ermC-R | TGAGCTATTCACTTTAGGTTTAGGA |
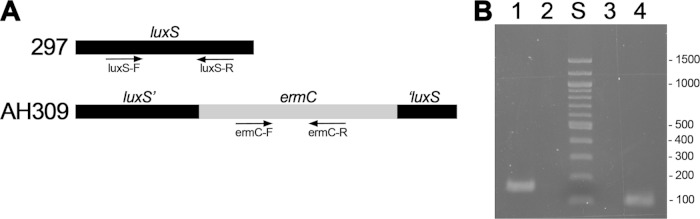
The dash in the luxS-R sequence indicates the point of luxS into which the ermC gene was inserted to create mutant strain AH309. Due to the split of the luxS-R target sequence in AH309, that oligonucleotide cannot serve as a PCR primer for AH309 (see Fig. 2).
For analyses of tissues from mice infected with a single B. burgdorferi strain, quantities of the single-copy bacterial flaB gene were compared with quantities of the single-copy mouse nidogen gene, generating ratios of bacterial chromosomes/mouse chromosomes (27).
For the competition studies, oligonucleotide primer pairs were designed and validated to be specific for either the wild-type luxS gene of strain 297 or the inactivated luxS gene of AH309 (Table 1 and Fig. 2). For detection of the wild-type gene, the 3′ primer consisted of a sequence spanning the site of the insertion in the AH309 locus and therefore could not amplify DNA from the mutant strain AH309. For detection of the mutant locus, primers were used which amplified the inserted ermC gene. Quantities of both amplicons produced from each animal tissue were determined and compared.
FIG 2.
Specificity of PCR oligonucleotide primer pairs to detect wild-type (297) and luxS mutant (AH309) B. burgdorferi strains. (A) Schematic representation of locations of sequences complementary to PCR primers. Primers ermC-F and ermC-R both correspond to sequences within the ermC gene that is inserted into luxS of AH309 (22). Oligonucleotide luxS-R overlaps the ermC insertion site of the AH309 locus and thus cannot serve to prime PCR for that strain. (B) Purified genomic DNAs from strains 297 and AH309 were subjected to PCRs using each primer pair and then subjected to agarose gel electrophoresis and ethidium bromide staining. Lane 1, 297 with primers luxS-F and luxS-R; lane 2, 297 with primers ermC-F and ermC-R; lane S, molecular size markers; lane 3, AH309 with primers luxS-F and luxS-R; lane 4, AH309 with primers ermC-F and ermC-R. Sizes of markers are indicated to the right of the gel.
Fold differences were calculated for competition studies by comparing the CT values of the wild-type and mutant bacteria for each tissue specimen. A ΔCT value of 1 is equivalent to a doubling of starting nucleic acid material. Therefore, 2ΔCT converts the difference in observed CT to the fold difference for queried DNAs.
RESULTS
Infections with individual bacterial strains.
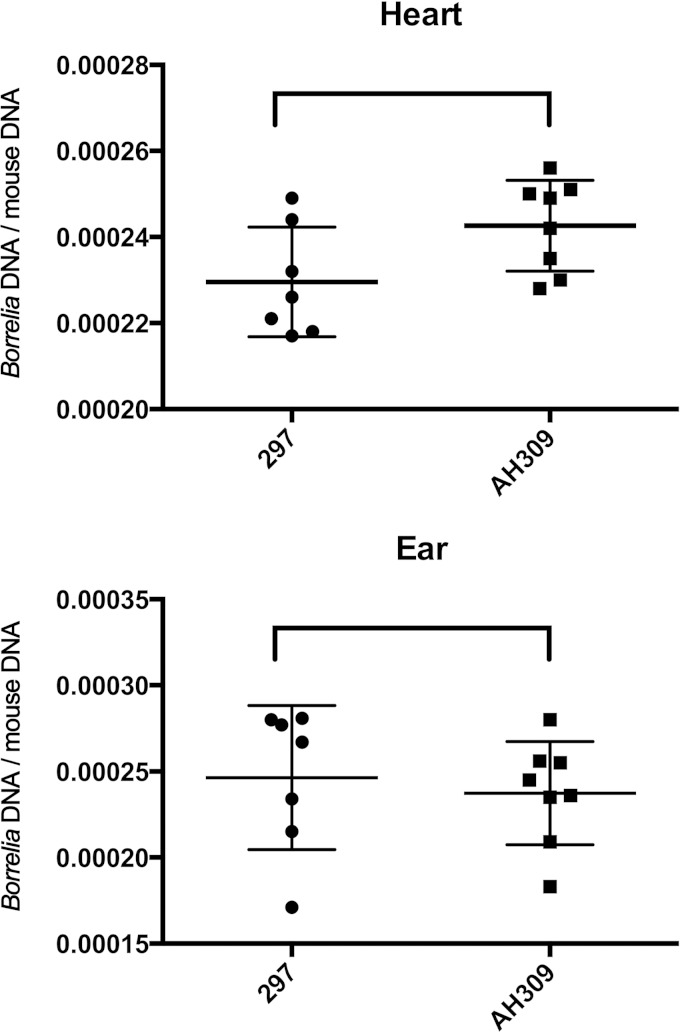
Prior comparisons of wild-type and luxS B. burgdorferi infectivities examined whether or not mice became infected but did not quantify bacterial loads of the infected animals (22). To address that deficiency, cohorts of mice were inoculated with 104 bacteria of either the wild-type (297) or luxS mutant (AH309) strain. After 28 days, mice were euthanized and total DNA (bacteria and mouse) was purified from heart and ear tissues. Bacterial loads in each tissue were determined by ratios of bacterial genomes to the mouse genome, assessed by qPCR. Numbers of copies of the B. burgdorferi flaB locus served as proxies for numbers of bacterial chromosomes, and the mouse nidogen locus served as the target to determined numbers of mouse genomes. Mice infected with AH309 contained slightly larger numbers of bacteria in their hearts than did mice inoculated with 297, with the difference bordering on statistical significance (P = 0.049), but there were no significant differences in the bacterial loads of ears from mice infected with 297 or AH309 (Fig. 3). These data demonstrate that the luxS mutant does not have any metabolic deficiencies that inhibit mammalian infection.
FIG 3.
qPCR analyses of tissues from mice that were singly infected with either strain 297 or AH309. For each tissue, chromosomal loci of B. burgdorferi and mice (flaB and nidogen, respectively) were quantified, and data were plotted as numbers of B. burgdorferi genome equivalents per mouse genome equivalent. Differences in bacterial loads of heart tissues were equivocal (P = 0.049), while there were no significant differences in bacterial loads of ear tissues (P > 0.05).
Wild-type versus mutant competition infections.
For a more sensitive analysis of the role of LuxS during mammalian infection, head-to-head competitions were undertaken. Mice were simultaneously inoculated with equal numbers of both wild-type and luxS mutant bacteria. The rationale for such studies is that if two strains are equally virulent, then mice will become infected with equal numbers of both strains, while differences in infectivity will be reflected by differences in relative bacterial loads.
To discriminate between the wild-type and luxS mutant strains, PCR primers were designed which specifically amplify only the wild-type or mutant luxS locus (Fig. 2). Control PCRs with purified DNA from each strain confirmed that the oligonucleotide pairs were equally efficient at priming PCRs from their respective templates.
The strain-specific primer pairs were then used for qPCR analyses of heart, ear, and urinary bladder tissues of eight doubly inoculated mice. The analyzed tissues were all distant from the site of inoculation and therefore measured the bacteria's ability to disseminate through mice and colonize three different types of tissues. Examination of distal tissues also ensured that detected bacteria had survived the processes of injection and dissemination. Comparisons of resultant data indicated that all tissues of all mice contained significantly larger numbers of wild-type than mutant bacteria (Table 2 and Fig. 4). Variations in wild-type/mutant ratios occurred between mice and between tissues of the same animals. The greatest degree of variation was found for the ears, with 3-fold to 18-fold more wild-type borreliae. Less variability was seen in the internal organs, i.e., the hearts and urinary bladders.
TABLE 2.
Results of dual-infection studies
| Animal no. | Raw CT valuea |
|||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Heart |
Bladder |
Ear |
||||||||||
| 297 |
AH309 |
297 |
AH309 |
297 |
AH309 |
|||||||
| Mean | SD | Mean | SD | Mean | SD | Mean | SD | Mean | SD | Mean | SD | |
| 1 | 31.068 | 0.729 | 32.860 | 0.281 | 30.863 | 1.422 | 33.555 | 0.208 | 30.550 | 0.811 | 32.775 | 0.661 |
| 2 | 30.265 | 0.296 | 32.763 | 0.272 | 30.673 | 0.534 | 33.893 | 0.419 | 31.620 | 0.440 | 32.798 | 0.511 |
| 3 | 31.173 | 1.667 | 33.180 | 0.913 | 30.293 | 0.571 | 32.170 | 0.612 | 30.493 | 0.456 | 33.683 | 1.429 |
| 4 | 30.370 | 0.921 | 33.143 | 0.452 | 30.728 | 2.049 | 31.733 | 1.112 | 30.865 | 0.389 | 33.325 | 0.106 |
| 5 | 30.703 | 1.517 | 33.130 | 1.300 | 30.138 | 0.676 | 32.763 | 1.055 | 30.810 | 0.819 | 34.763 | 2.401 |
| 6 | 31.395 | 1.759 | 33.015 | 0.843 | 30.680 | 1.953 | 30.738 | 0.787 | 30.170 | 0.561 | 32.355 | 0.324 |
| 7 | 29.795 | 1.186 | 33.035 | 1.138 | 30.638 | 0.296 | 31.880 | 1.612 | 30.578 | 0.096 | 32.883 | 1.000 |
| 8 | 30.295 | 1.193 | 31.790 | 0.707 | 30.375 | 0.423 | 33.003 | 0.604 | 33.083 | 2.005 | 34.080 | 0.042 |
Values represent the means for two separate trials, with two measurements per trial. Student's t test was performed, and CT values obtained for target loci of strains 297 and AH309 were found to differ significantly across all tissues in all animals, with P values of <0.0001, 0.0015, and 0.0003 for the heart, bladder, and ear, respectively.
FIG 4.
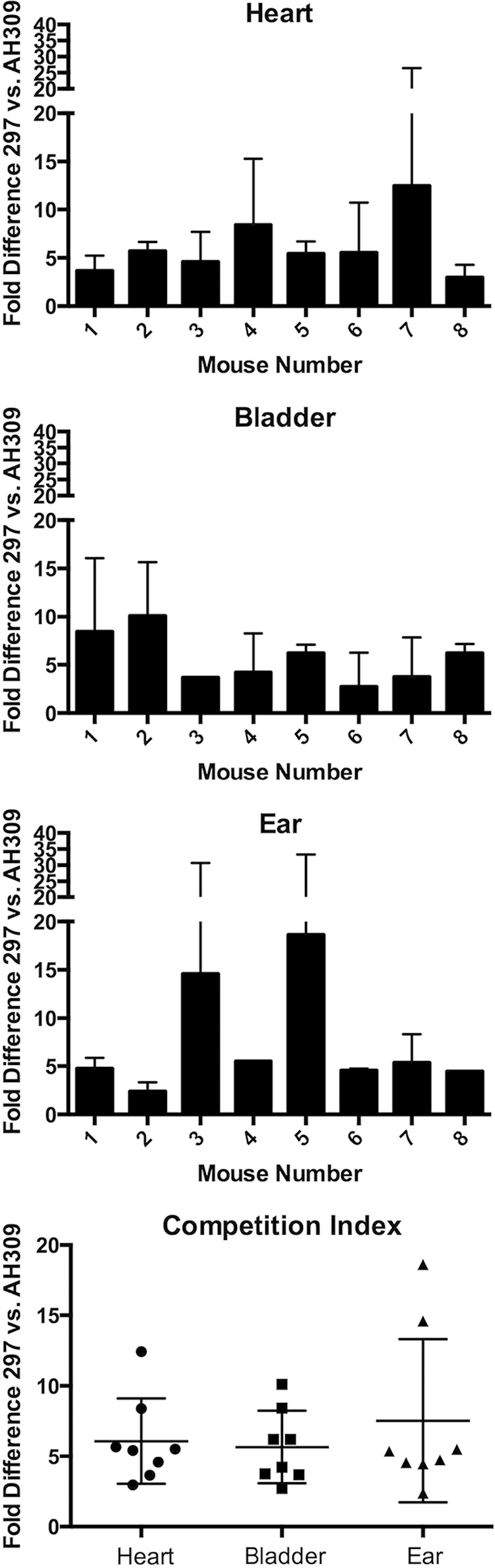
qPCR analyses of tissues from mice that were simultaneously injected with 104 bacteria (each) of both strains 297 and AH309. Total DNAs were extracted from the hearts, urinary bladders, and ear pinnae of eight mice. Ratios of wild-type (strain 297) to luxS mutant (strain AH309) bacteria were calculated from the ΔCT values for each strain in each tissue. Competition indexes were calculated for every mouse tissue.
DISCUSSION
The two strains utilized in the current studies were previously used for nonquantitative analysis of infectivity, the results of which were published in this journal with statements such as “AH309 appeared to be fully infectious at wild-type levels” (22). The main purpose of the present studies was to determine whether or not the previous conclusions were accurate. Quantification of the bacterial loads in dually infected mice revealed that all three examined tissue types of all eight infected mice carried significantly greater loads of wild-type than luxS mutant B. burgdorferi. All examined tissues—ears, hearts, and urinary bladders—are distal from the midscapular injection site, which required bacteria to survive, disseminate, and colonize three different tissues. Although qPCR cannot discriminate live from dead bacteria, these analyses indicated that substantially larger numbers of wild-type bacteria migrated to all tissues, which in itself is a significant difference. Thus, it can be concluded that mouse infection studies with wild-type strain 297 and the congenic luxS mutant AH309 do not eliminate a role for LuxS during mammalian infection but instead provide support for the hypothesis that LuxS makes a significant contribution.
These results indicate that further studies of the function(s) of LuxS during mammalian infection are warranted. At present, there are no published methods to produce clean genetic complementation of B. burgdorferi mutants. Small differences in expression of regulatory factors can have large effect on targets, so complementation of the luxS mutation with the wild-type gene on a plasmid would not be useful. Likewise, restoration of wild-type luxS to its chromosomal locus by use of an adjacent selectable marker would also be questionable, since the marker's insertion would alter the structure of the chromosome at that locus. Until tools are available to examine the final molecular Koch's postulate on B. burgdorferi LuxS, the question of the enzyme's contribution to mammalian infection must remain unanswered, with the understanding that all available data indicate that LuxS and AI-2 affect the expression of numerous borrelial proteins and have a positive effect on mammalian infection.
Competition studies between mutant and wild-type bacteria can effectively identify bacterial factors that contribute to mammalian infection, because the question being asked is not whether the mutant is merely capable of infection but whether the infectivity of the mutant is equivalent to that of the wild-type pathogen. Previous studies demonstrated that competitive index studies magnify the negative impacts of mutations, making them more readily detectable (e.g., see references 28 and 29). Such analyses are particularly relevant to B. burgdorferi, since transmitting ticks are generally colonized with mixtures of borrelial variants (3). Thus, there is intense selective pressure on each Lyme disease spirochete to maximize its efficiency of infection and to avoid being outcompeted.
Although regulatory factors are often perceived as on/off switches, many function more like rheostats, incrementally adjusting transcription to meet specific needs of the organism. Prior analyses of the effects of AI-2 on B. burgdorferi indicated that this molecule affects borrelial protein levels in a rheostat manner (4, 7, 20). The previous (22, 23) and current studies indicate that the inability to produce LuxS and AI-2 does not render B. burgdorferi noninfectious, yet the impacts of LuxS/AI-2 on borrelial protein expression suggest an appreciable benefit of these factors (4, 7, 20). Combining results of these infection studies with previous studies of cultured borreliae, we hypothesize that AI-2 enables precise control of important host-interactive proteins.
Pathogens produce numerous substances that can be described as “virulence factors,” or proteins and other molecules that contribute to infectivity. In addition to LuxS, the significance of at least three other B. burgdorferi factors became evident after quantitative infection assays. B. burgdorferi organisms that are unable to produce BBA03, a surface protein of unknown function, are capable of infecting mice yet were outcompeted in dual-infection studies with wild-type bacteria (30). Similar analyses indicated that the naturally occurring replicon lp28-3 encodes at least one factor that makes a significant contribution to mammalian infection (31). Mutant B. burgdorferi organisms that are unable to produce the surface-exposed fibronectin-binding protein BBK32 are capable of infecting mice (32). However, the 50% infective dose (ID50) of a bbk32 mutant was determined to be approximately 10-fold greater than that of the wild type (33). Subsequent studies found that BBK32 plays a role in B. burgdorferi escape from the bloodstream (34).
B. burgdorferi secretes AI-2 into the culture medium and can respond to externally supplied AI-2, as do V. harveyi and S. Typhimurium (4, 7). The mechanism by which AI-2 influences B. burgdorferi protein expression is not yet known. V. harveyi and S. Typhimurium each use very different mechanisms to detect and respond to AI-2 (12, 14, 15). V. harveyi utilizes a two-component phosphorelay system that senses external AI-2 and sends an internal signal (15). B. burgdorferi possesses two two-component regulatory systems, the triggers for neither of which have been determined (3, 10). However, mutations in either of the two borrelial two-component systems have more drastic effects than does disruption of LuxS function, suggesting that they are probably not key to AI-2 signaling (35, 36). In contrast, S. Typhimurium uses an ABC transport system to internalize AI-2 (17, 18). DPD/AI-2 structurally resembles ribose, and the S. Typhimurium transporter is related to ribose transporters (13, 16–18). Along that line, B. burgdorferi cannot use ribose as a carbon source, yet the spirochete encodes a putative ABC transport system that resembles known ribose transporters (10, 37). B. burgdorferi also encodes two proteins that resemble pentose-sensing DNA-binding proteins, one of which has been named BadR and demonstrated to be a transcriptional regulator (10, 38). Alternatively, noting that two proteobacteria use radically different methods to respond to AI-2, it is possible that a distantly related spirochete may have evolved yet another mechanism.
As do other studied bacterial species, B. burgdorferi maximally produces DPD/AI-2 during periods of high metabolic activity (7, 39, 40). Thus, concentrations of AI-2 are reflective of the bacterial growth rate (40). In this way, AI-2 differs from “typical” quorum sensing molecules, which are thought to be indicative of bacterial density (41, 42). The rate at which B. burgdorferi grows varies during its tick-mammal infection cycle, with essentially no bacterial growth during colonization of the nutrient-poor environment in the unfed tick midgut but rapid growth during transmission, when the tick ingests nutritious blood, and during infection of tissues throughout the vertebrate host (1, 3, 43–49). Corresponding with the changes in growth rate, expression of luxS increases significantly during transmission of B. burgdorferi from ticks to mammals (21). The concomitant increase in AI-2 production may benefit B. burgdorferi at stages in which multiple bacteria are found in close proximity, such as during periods of cell division. Molecules that are secreted and have their concentrations sensed, such as AI-2, can also provide individual bacteria with information on the nature of their local environment: in an open environment, such as the bloodstream, AI-2 will rapidly diffuse away, whereas it will accumulate in a closed environment, such as the solid tissues that are preferred by B. burgdorferi for vertebrate colonization (3, 50, 51). This hypothesis can explain the observed variations in bacterial loads and differences in wild-type/mutant ratios possibly being affected by animal-to-animal differences in microvasculature structures or extracellular matrix distribution.
In conclusion, quantitative analyses of a previously examined B. burgdorferi luxS mutant demonstrated that the mutant is significantly impaired in the ability to infect mammals. These data disprove prior conclusions and suggest that LuxS does indeed perform a significant function(s) during mammalian infection. Previous studies indicated that AI-2 affects the expression levels of several proteins known to be involved in mammalian infection (4, 7, 20), suggesting that the defects of the luxS mutant are probably pleiotropic. Further studies should be undertaken to discover the mechanism by which the Lyme disease spirochete responds to AI-2. These results also serve as reminders that determination of whether or not a bacterial component is involved with infection cannot always be achieved through nonquantitative methods.
ACKNOWLEDGMENTS
These studies were funded by NIH grant R01 AI53101 to B.S.
We thank Jennifer Taylor for technical support.
REFERENCES
- 1.Stevenson B, von Lackum K, Riley SP, Cooley AE, Woodman ME, Bykowski T. 2006. Evolving models of Lyme disease spirochete gene regulation. Wien Klin Wochenschr 118:643–652. doi: 10.1007/s00508-006-0690-2. [DOI] [PubMed] [Google Scholar]
- 2.Samuels DS. 2011. Gene regulation in Borrelia burgdorferi. Annu Rev Microbiol 65:479–499. doi: 10.1146/annurev.micro.112408.134040. [DOI] [PubMed] [Google Scholar]
- 3.Radolf JD, Caimano MJ, Stevenson B, Hu LT. 2012. Of ticks, mice, and men: understanding the dual-host lifestyle of Lyme disease spirochaetes. Nat Rev Microbiol 10:87–99. doi: 10.1038/nrmicro2714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Stevenson B, Babb K. 2002. LuxS-mediated quorum sensing in Borrelia burgdorferi, the Lyme disease spirochete. Infect Immun 70:4099–4105. doi: 10.1128/IAI.70.8.4099-4105.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Stevenson B, von Lackum K, Wattier RL, McAlister JD, Miller JC, Babb K. 2003. Quorum sensing by the Lyme disease spirochete. Microbes Infect 5:991–997. doi: 10.1016/S1286-4579(03)00184-9. [DOI] [PubMed] [Google Scholar]
- 6.Sun J, Daniel R, Wagner-Dobler I, Zeng AP. 2004. Is autoinducer-2 a universal signal for interspecies communication: a comparative genomic and phylogenetic analysis of the synthesis and signal transduction pathways. BMC Evol Biol 4:36. doi: 10.1186/1471-2148-4-36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Babb K, von Lackum K, Wattier RL, Riley SP, Stevenson B. 2005. Synthesis of autoinducer 2 by the Lyme disease spirochete, Borrelia burgdorferi. J Bacteriol 187:3079–3087. doi: 10.1128/JB.187.9.3079-3087.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.von Lackum K, Babb K, Riley SP, Wattier RL, Bykowski T, Stevenson B. 2006. Functionality of Borrelia burgdorferi LuxS: the Lyme disease spirochete produces and responds to the pheromone autoinducer-2, and lacks a complete activated-methyl cycle. Int J Med Microbiol 296(Suppl 1):92–102. doi: 10.1016/j.ijmm.2005.12.011. [DOI] [PubMed] [Google Scholar]
- 9.Riley SP, Bykowski T, Babb K, von Lackum K, Stevenson B. 2007. Genetic and physiological characterization of the Borrelia burgdorferi ORF BB0374-pfs-metK-luxS operon. Microbiology 153:2304–2311. doi: 10.1099/mic.0.2006/004424-0. [DOI] [PubMed] [Google Scholar]
- 10.Fraser CM, Casjens S, Huang WM, Sutton GG, Clayton R, Lathigra R, White O, Ketchum KA, Dodson R, Hickey EK, Gwinn M, Dougherty B, Tomb J-F, Fleischmann RD, Richardson D, Peterson J, Kerlavage AR, Quackenbush J, Salzberg S, Hanson M, van Vugt R, Palmer N, Adams MD, Gocayne J, Weidmann J, Utterback T, Watthey L, McDonald L, Artiach P, Bowman C, Garland S, Fujii C, Cotton MD, Horst K, Roberts K, Hatch B, Smith HO, Venter JC. 1997. Genomic sequence of a Lyme disease spirochaete, Borrelia burgdorferi. Nature 390:580–586. doi: 10.1038/37551. [DOI] [PubMed] [Google Scholar]
- 11.Fraser CM, Norris SJ, Weinstock GM, White O, Sutton GG, Dodson R, Gwinn M, Hickey EK, Clayton R, Ketchum KA, Sodergren E, Hardham JM, McLeod MP, Salzberg S, Peterson J, Khalak H, Richardson D, Howell JK, Chidambaram M, Utterback T, McDonald L, Artiach P, Bowman C, Cotton MD, Fujii C, Garland S, Hatch B, Horst K, Roberts K, Watthey L, Weidman J, Smith HO, Venter JC. 1998. Complete genome sequence of Treponema pallidum, the syphilis spirochete. Science 281:375–388. doi: 10.1126/science.281.5375.375. [DOI] [PubMed] [Google Scholar]
- 12.Surette MG, Miller MB, Bassler BL. 1999. Quorum sensing in Escherichia coli, Salmonella typhimurium, and Vibrio harveyi: a new family of genes responsible for autoinducer production. Proc Natl Acad Sci U S A 96:1639–1644. doi: 10.1073/pnas.96.4.1639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Chen X, Schauder S, Potier N, Van Dorsselaer A, Pelczer I, Bassler BL, Hughson FM. 2002. Structural identification of a bacterial quorum-sensing signal containing boron. Nature 415:545–549. doi: 10.1038/415545a. [DOI] [PubMed] [Google Scholar]
- 14.Schauder S, Shokat S, Surette MG, Bassler BL. 2001. The LuxS family of bacterial autoinducers: biosynthesis of a novel quorum-sensing signal molecule. Mol Microbiol 41:463–476. doi: 10.1046/j.1365-2958.2001.02532.x. [DOI] [PubMed] [Google Scholar]
- 15.Bassler BL, Wright M, Silverman MR. 1994. Multiple signalling systems controlling expression of luminescence in Vibrio harveyi: sequence and function of genes encoding a second sensory pathway. Mol Microbiol 13:273–286. doi: 10.1111/j.1365-2958.1994.tb00422.x. [DOI] [PubMed] [Google Scholar]
- 16.Miller ST, Xavier KB, Campagna SR, Taga ME, Semmelhack MF, Bassler BL, Hughson FM. 2004. Salmonella typhimurium recognizes a chemically distinct form of the bacterial quorum-sensing signal AI-2. Mol Cell 15:677–687. doi: 10.1016/j.molcel.2004.07.020. [DOI] [PubMed] [Google Scholar]
- 17.Taga ME, Miller ST, Bassler BL. 2003. Lsr-mediated transport and processing of AI-2 in Salmonella typhimurium. Mol Microbiol 50:1411–1427. doi: 10.1046/j.1365-2958.2003.03781.x. [DOI] [PubMed] [Google Scholar]
- 18.Taga ME, Semmelhack JL, Bassler BL. 2001. The LuxS-dependent autoinducer AI-2 controls the expression of an ABC transporter that functions in AI-2 uptake in Salmonella typhimurium. Mol Microbiol 42:777–793. doi: 10.1046/j.1365-2958.2001.02669.x. [DOI] [PubMed] [Google Scholar]
- 19.Vendeville A, Winzer K, Heurlier K, Tang CM, Hardie KR. 2005. Making ‘sense’ of metabolism: autoinducer-2, LuxS and pathogenic bacteria. Nat Rev Microbiol 3:383–396. doi: 10.1038/nrmicro1146. [DOI] [PubMed] [Google Scholar]
- 20.von Lackum K, Ollison KM, Bykowski T, Nowalk AJ, Hughes JL, Carroll JA, Zückert WR, Stevenson B. 2007. Regulated synthesis of the Borrelia burgdorferi inner-membrane lipoprotein IpLA7 (P22, P22-A) during the Lyme disease spirochaete's mammal-tick infectious cycle. Microbiology 153:1361–1371. doi: 10.1099/mic.0.2006/003350-0. [DOI] [PubMed] [Google Scholar]
- 21.Narasimhan S, Santiago F, Koski RA, Brei B, Anderson JF, Fish D, Fikrig E. 2002. Examination of the Borrelia burgdorferi transcriptome in Ixodes scapularis during feeding. J Bacteriol 184:3122–3125. doi: 10.1128/JB.184.11.3122-3125.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Hübner A, Revel AT, Nolen DM, Hagman KE, Norgard MV. 2003. Expression of a luxS gene is not required for Borrelia burgdorferi infection of mice via needle inoculation. Infect Immun 71:2892–2896. doi: 10.1128/IAI.71.5.2892-2896.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Blevins JS, Revel AT, Caimano MJ, Yang XF, Richardson JA, Hagman KE, Norgard MV. 2004. The luxS gene is not required for Borrelia burgdorferi tick colonization, transmission to a mammalian host, or induction of disease. Infect Immun 72:4864–4867. doi: 10.1128/IAI.72.8.4864-4867.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Zückert WR. 2007. Laboratory maintenance of Borrelia burgdorferi. Curr Protocols Microbiol 12C:1–10. doi: 10.1002/9780471729259.mc12c01s4. [DOI] [PubMed] [Google Scholar]
- 25.Barthold SW, de Souza M, Fikrig E, Persing DH. 1992. Lyme borreliosis in the laboratory mouse, p 223–242. In Schutzer SE. (ed), Lyme disease: molecular and immunologic approaches. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY. [Google Scholar]
- 26.Wooten RM, Weis JJ. 2001. Host-pathogen interactions promoting inflammatory Lyme arthritis: use of mouse models for dissection of disease processes. Curr Opin Microbiol 4:274–279. doi: 10.1016/S1369-5274(00)00202-2. [DOI] [PubMed] [Google Scholar]
- 27.Morrison TB, Ma Y, Weis JH, Weis JJ. 1999. Rapid and sensitive quantification of Borrelia burgdorferi-infected mouse tissues by continuous fluorescent monitoring of PCR. J Clin Microbiol 37:987–992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Auerbuch V, Lenz LL, Portnoy DA. 2001. Development of a competitive index assay to evaluate the virulence of Listeria monocytogenes actA mutants during primary and secondary infection of mice. Infect Immun 69:5953–5957. doi: 10.1128/IAI.69.9.5953-5957.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Beuzón CR, Holden DW. 2001. Use of mixed infections with Salmonella strains to study virulence genes and their interactions in vivo. Microbes Infect 3:1345–1352. doi: 10.1016/S1286-4579(01)01496-4. [DOI] [PubMed] [Google Scholar]
- 30.Bestor A, Rego RO, Tilly K, Rosa PA. 2012. Competitive advantage of Borrelia burgdorferi with outer surface protein BBA03 during tick-mediated infection of the mammalian host. Infect Immun 80:3501–3511. doi: 10.1128/IAI.00521-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Dulebohn DP, Bestor A, Rosa PA. 2013. Borrelia burgdorferi linear plasmid 28-3 confers a selective advantage in an experimental mouse-tick infection model. Infect Immun 81:2986–2996. doi: 10.1128/IAI.00219-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Li X, Liu X, Beck DS, Kantor FS, Fikrig E. 2006. Borrelia burgdorferi lacking BBK32, a fibronectin-binding protein, retains full pathogenicity. Infect Immun 74:3305–3313. doi: 10.1128/IAI.02035-05. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Seshu J, Esteve-Gassent MD, Labandiera-Rey M, Kim JH, Trzeciakowski JP, Höök M, Skare JT. 2006. Inactivation of the fibronectin-binding adhesin gene bbk32 significantly attenuates the infectivity potential of Borrelia burgdorferi. Mol Microbiol 59:1591–1601. doi: 10.1111/j.1365-2958.2005.05042.x. [DOI] [PubMed] [Google Scholar]
- 34.Norman MU, Moriarty TJ, Dresser AR, Millen B, Kubes P, Chaconas G. 2008. Molecular mechanisms involved in vascular interactions of the Lyme disease pathogen in a living host. PLoS Pathog 4:e1000169. doi: 10.1371/journal.ppat.1000169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Yang XF, Alani SM, Norgard MV. 2003. The response regulator Rrp2 is essential for the expression of major membrane lipoproteins in Borrelia burgdorferi. Proc Natl Acad Sci U S A 100:11001–11006. doi: 10.1073/pnas.1834315100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Rogers EA, Terekhova D, Zhang HM, Hovis KM, Schwartz I, Marconi RT. 2009. Rrp1, a cyclic-di-GMP-producing response regulator, is an important regulator of Borrelia burgdorferi core cellular functions. Mol Microbiol 71:1551–1573. doi: 10.1111/j.1365-2958.2009.06621.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.von Lackum K, Stevenson B. 2005. Carbohydrate utilization by the Lyme borreliosis spirochete, Borrelia burgdorferi. FEMS Microbiol Lett 243:173–179. doi: 10.1016/j.femsle.2004.12.002. [DOI] [PubMed] [Google Scholar]
- 38.Miller CL, Karna SL, Seshu J. 2013. Borrelia host adaptation regulator (BadR) regulates rpoS to modulate host adaptation and virulence factors in Borrelia burgdorferi. Mol Microbiol 88:105–124. doi: 10.1111/mmi.12171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Surette MG, Bassler BL. 1999. Regulation of autoinducer production in Salmonella typhimurium. Mol Microbiol 31:585–595. doi: 10.1046/j.1365-2958.1999.01199.x. [DOI] [PubMed] [Google Scholar]
- 40.Xavier KB, Bassler BL. 2003. LuxS quorum sensing: more than just a numbers game. Curr Opin Microbiol 6:191–197. doi: 10.1016/S1369-5274(03)00028-6. [DOI] [PubMed] [Google Scholar]
- 41.Fuqua C, Winans SC, Greenberg EP. 1996. Census and consensus in bacterial ecosystems: the LuxR-LuxI family of quorum-sensing transcriptional regulators. Annu Rev Microbiol 50:727–751. doi: 10.1146/annurev.micro.50.1.727. [DOI] [PubMed] [Google Scholar]
- 42.de Kievit TR, Iglewski BH. 2000. Bacterial quorum sensing in pathogenic relationships. Infect Immun 68:4839–4849. doi: 10.1128/IAI.68.9.4839-4849.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Piesman J, Oliver JR, Sinsky RJ. 1990. Growth kinetics of the Lyme disease spirochete (Borrelia burgdorferi) in vector ticks (Ixodes dammini). Am J Trop Med Hyg 42:352–357. [DOI] [PubMed] [Google Scholar]
- 44.Burkot TR, Piesman J, Wirtz RA. 1994. Quantitation of the Borrelia burgdorferi outer surface protein A in Ixodes scapularis: fluctuations during the tick life cycle, doubling times and loss while feeding. J Infect Dis 170:883–889. doi: 10.1093/infdis/170.4.883. [DOI] [PubMed] [Google Scholar]
- 45.de Silva AM, Fikrig E. 1995. Growth and migration of Borrelia burgdorferi in Ixodes ticks during blood feeding. Am J Trop Med Hyg 53:397–404. [DOI] [PubMed] [Google Scholar]
- 46.Piesman J, Schneider BS, Zeidner NS. 2001. Use of quantitative PCR to measure density of Borrelia burgdorferi in the midgut and salivary glands of feeding tick vectors. J Clin Microbiol 39:4145–4148. doi: 10.1128/JCM.39.11.4145-4148.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Piesman J, Schneider BS. 2002. Dynamic changes in Lyme disease spirochetes during transmission by nymphal ticks. Exp Appl Acarol 28:141–145. doi: 10.1023/A:1025351727785. [DOI] [PubMed] [Google Scholar]
- 48.Jutras BL, Chenail AM, Stevenson B. 2013. Changes in bacterial growth rate govern expression of the Borrelia burgdorferi OspC and Erp infection-associated surface proteins. J Bacteriol 195:757–764. doi: 10.1128/JB.01956-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Dunham-Ems SM, Caimano MJ, Pal U, Wolgemuth CW, Eggers CH, Balic A, Radolf JD. 2009. Live imaging reveals a biphasic mode of dissemination of Borrelia burgdorferi within ticks. J Clin Invest 119:3652–3665. doi: 10.1172/JCI39401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Redfield RJ. 2002. Is quorum sensing a side effect of diffusion sensing? Trends Microbiol 10:365–370. doi: 10.1016/S0966-842X(02)02400-9. [DOI] [PubMed] [Google Scholar]
- 51.Koerber AJ, King JR, Williams P. 2005. Deterministic and stochastic modelling of endosome escape by Staphylococcus aureus: “quorum” sensing by a single bacterium. J Math Biol 50:440–488. doi: 10.1007/s00285-004-0296-0. [DOI] [PubMed] [Google Scholar]