A sufficient and balanced supply of mineral nutrients is crucial for plant growth and development. However, excessive amounts of minerals in the soil are harmful to most plants. To reduce the toxic levels of ions in the cytoplasm caused by environmental mineral stress, plants have evolved mechanisms either to promote the exclusion of excessive ions outside of the cells or to sequester ions into the vacuole, an organelle equivalent to the mammalian lysosome. One of the unfavorable ions that plants often encounter in high concentrations is sodium (Na+) coming from soil salinity, which is estimated to affect over 20% of the world’s arable land (1). In the past decades, great efforts have been made to understand the underlying mechanisms of plant salt tolerance. In contrast to Na+, magnesium (Mg2+) is usually considered as an essential macronutrient for plants because of its important roles in chlorophyll synthesis, enzyme activation, and cation balance. However, a high level of Mg2+ in the soil, such as found in the serpentine areas of California (2), is also detrimental to plants. Only serpentine-tolerant plants, such as Microseris douglasii, are able to survive in such high Mg2+ conditions. Numerous studies have focused on the physiological and ecological aspects of plants survived in serpentine soils (2), yet the genetic basis of serpentine adaptation remains elusive. To make better use of serpentine soils for landscape ecology and to potentially improve agricultural production, it is important to understand the molecular mechanism of plant Mg2+ tolerance. In PNAS, Tang et al. (3) have delved into the function of unique tonoplast-localized calcium (Ca2+) sensors, the calcineurin B-like proteins (CBLs) and their downstream components, CBL-interacting protein kinases (CIPKs), in regulating vacuolar sequestration of Mg2+ to detoxify high Mg2+ in plant cells.
The plant vacuole is a membrane-bound organelle that can occupy up to 90% of the total cell volume, and has multiple functions, including storage of nutrients and metabolites, generation of turgor, protein degradation, and plant defense (4, 5). The function of vacuole as a reservoir for ions and metabolites relies on tonoplast-localized transport proteins. Two types of proton pumps, vacuolar H+-ATPase (V-ATPase) and vacuolar H+-pyrophosphatase (V-PPase), are responsible for generating a proton gradient and membrane potential that energize secondary transport processes across the vacuolar membrane (6). On the other hand, Ca2+ functions as an important second messenger to relay various external ionic stress signals, thereby activating transporters that can cope with ionic stress by moving ions out of cells across the plasma membrane (PM) and into the vacuole through the tonoplast. Plant CBL proteins act as Ca2+ sensors and actively participate in environmental stress responses, and particularly in ionic stresses (7, 8). Through interaction with CIPKs, the CBL–CIPK signaling modules activate a number of ion channels or transporters that facilitate uptake, exclusion, or vacuolar sequestration of various ions (7, 8). For example, plant potassium (K+) homeostasis is regulated by the PM-anchored CBL1 and CBL9, as well as their downstream kinase CIPK23. Under K+-deficient conditions, the CBL1/9–CIPK23 complex activates the AKT1 (a shaker-type potassium channel) to enhance K+ uptake and protect plants from low potassium stress (Fig. 1) (8). In plant adaptation to salt stress regulated by the salt overly sensitive (SOS) pathway, CIPK24/SOS2, in complex with CBL4/SOS3, target SOS1 (a PM-localized Na+/H+ antiporter) for activting Na+ extrusion, and by coupling with CBL10 it regulates a yet-unknown transporter for vacuolar Na+ sequestration, thus playing a dual role in conferring plant salt tolerance (Fig. 1) (1, 8).
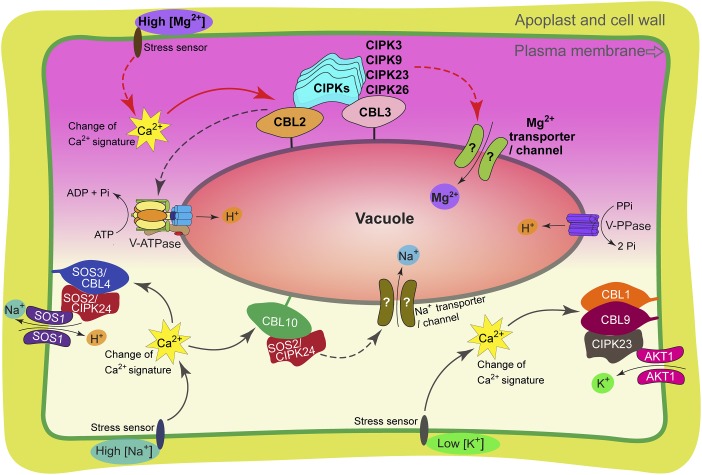
Fig. 1.
Working model of Ca2+-CBL–CIPK signaling network and vacuole functions in plant responses to ionic stresses. The Mg2+ stress may trigger a rapid change of cytosolic Ca2+ concentration. The Ca2+ signal can be perceived by the tonoplast-localized Ca2+ sensors, CBL2/3, which then recruit and activate the downstream kinases CBL3/9/23/26 to activate the elusive tonoplast-localized Mg2+ transporters or channels that are responsible for the translocation of Mg2+, maintaining a nontoxic level of Mg2+ in the cytoplasm. Under salinity stress, the Ca2+ signal, which is perceived by the PM-localized sensor-kinase complex CBL4/SOS3–CIPK24/SOS2, targets the PM-localized Na+/H+ exchanger SOS1 and regulates the exclusion of Na+ outside of the cell. The salinity stress can also activate the tonoplast-localized CBL10–CIPK24/SOS2 complex to target the vacuolar localized transporters for the vacuolar sequestration of Na+. Under low-K+ conditions, the Ca2+ signal activates CBL1/9–CIPK23 that targets the AKT1 K+-channel and enhances K+-uptake. The upper half cell and red arrows highlight the new findings published in PNAS by Tang et al. (3). The dashed lines and question marks indicate the elusive pathways or components to be identified in future studies.
In contrast to the well-defined functions of PM-localized CBL–CIPKs in regulating ion fluxes and stress responses, the function of tonoplast-localized CBL–CIPKs remains quite obscure. The vacuole-localized CBL2 and CBL3 contain the N-terminal tonoplast targeting signals that harbor three cysteine residues required for S-acylation (9, 10). Genetic analysis demonstrated that these two calcium sensors exhibit overlapping functions in the regulation of plant development and ionic homeostasis, which are correlated with V-ATPase activities (10).
The function of CBL2 and CBL3 in regulating vacuole-mediated ion homeostasis has now been further explored in Tang et al.’s study (3). The authors find that the cbl2 cbl3 mutant was hypersensitive to high concentrations of external Mg2+, but analysis of ionic profiles showed a reduced accumulation of Mg2+ in the cbl2 cbl3 double-mutant. Accordingly, by taking advantage of a patch-clamp strategy in isolated intact vacuoles derived from mesophyll cells of wild-type or the cbl2 cbl3 double-mutant, Tang et al. find a significantly reduced Mg2+ influx across the tonoplast in the vacuoles of cbl2 cbl3 plants. These results strongly highlight the important role of vacuolar sequestration of extra Mg2+ in the detoxification of high levels of Mg2+. On the other hand, maintaining Ca2+/Mg2+ homeostasis was thought to be critical for serpentine-tolerant plants (2). Consistent with the previous observation of Ca2+ effects on Mg2+ toxicity, adding more Ca2+ indeed successfully alleviated the oversensitivity of the cbl2 cbl3 mutant to high-Mg2+ (3). Interestingly, CBL2 and CBL3 regulate vacuolar sequestration of Mg2+ in a V-ATPase–independent manner (3), even though CBL2 and CBL3 also manipulate V-ATPase function (10). It will therefore be interesting to further investigate the role of V-PPase, another vacuolar photon pump, in CBL2/3-regulated and vacuole-mediated detoxification of high Mg2+. The work done by Tang et al. (3) identifies a new function for the CBL–CIPK signaling network in vacuole-mediated detoxification of high external Mg2+.
To fulfill the regulatory functions of CBLs in the Ca2+-mediated signaling pathway, these CBL Ca2+ sensors need to bind to and activate their target CIPKs. Tang et al. (3) subsequently perform extensive genetic, gene expression, and biochemical analysis to map the CIPKs downstream of CBL2/3 in the regulation of stress response to high external Mg2+. The authors find that CIPK3/9/23/26 interact with CBL2/3 on the tonoplast, and the quadruple cipk3/9/23/26 mutant could fully phenocopy Mg2+ hypersensitivity of the cbl2/3 double-mutant, and showed a similar ionic profile to what was found in cbl2/3. These results convincingly demonstrate that CIPK3/9/23/26 work together with CBL2/3 at the tonoplast to alleviate the toxic effects of high external Mg2+ concentrations via vacuolar sequestration of extra Mg2+. Some open questions remaining are: (i) Does external Mg2+ stress induce the change of Ca2+ signatures? (ii) Do the vacuoles isolated from the cipk3/9/23/26 mutant also have a similar electrophysiological signature as the cbl2/3 double-mutant when displaying a significantly reduced Mg2+ influx across the tonoplast?
Tonoplast-localized transporters are likely to be involved in vacuolar Mg2+ transport. AtMHX was the first identified plant Mg2+/H+ antiporter localized in the vacuolar membrane, which presumably contributes to vacuolar Mg uptake (11). However, atmhx mutant plants show a wild-type–like response toward high Mg2+ stress (3), excluding the possible involvement of AtMHX as a transporter coupling with CBL2/3 and CIPK3/9/23/26 in the detoxification of high Mg2+ in the cytoplasm. Both MGT2 and MGT3 are also known Mg2+ transporters localized to the tonoplast (12), but their mutants showed no significant phenotypic changes under high Mg2+ conditions (3). Therefore, identification of the transporters that are active under Mg2+ toxicity conditions is pivotal to understand the underlying mechanism of magnesium detoxification in plants.
In summary, plant vacuoles serve as the most important reservoir of ions and metabolites for plants to buffer the fluctuating concentrations of external nutrients and thereby alleviate the stressful effects of toxic compounds. The work by Tang et al. (3) uncovers a novel function of the CBL–CIPK signaling network in the vacuolar sequestration of excessive Mg2+, thereby helping plants to survive under Mg2+ stress (Fig. 1). This Mg2+ partitioning process in the vacuole controlled by the CBL–CIPK pathway may represent a general mechanism underlying the detoxification of other ions, including Na+ (Fig. 1). Thus, this work not only sheds new light on our understanding about plant adaptation to serpentine soils, but also provides an example how plants might adapt to changes in other minerals at the molecular level and potentially assists in efforts to improve crop productivity under adverse conditions.
Footnotes
The authors declare no conflict of interest.
See companion article on page 3134.
References
- 1.Zhu JK. Salt and drought stress signal transduction in plants. Annu Rev Plant Biol. 2002;53:247–273. doi: 10.1146/annurev.arplant.53.091401.143329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Brady KU, Kruckeberg AR, Bradshaw HD. Evolutionary ecology of plant adaptation to serpentine soils. Annu Rev Ecol Evol Syst. 2005;36:243–266. [Google Scholar]
- 3.Tang R-J, et al. Tonoplast CBL–CIPK calcium signaling network regulates magnesium homeostasis in Arabidopsis. Proc Natl Acad Sci USA. 2015;112:3134–3139. doi: 10.1073/pnas.1420944112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Gao C, et al. Dual roles of an Arabidopsis ESCRT component FREE1 in regulating vacuolar protein transport and autophagic degradation. Proc Natl Acad Sci USA. 2015;112(6):1886–1891. doi: 10.1073/pnas.1421271112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Müntz K. Protein dynamics and proteolysis in plant vacuoles. J Exp Bot. 2007;58(10):2391–2407. doi: 10.1093/jxb/erm089. [DOI] [PubMed] [Google Scholar]
- 6.Krebs M, et al. Arabidopsis V-ATPase activity at the tonoplast is required for efficient nutrient storage but not for sodium accumulation. Proc Natl Acad Sci USA. 2010;107(7):3251–3256. doi: 10.1073/pnas.0913035107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Luan S. The CBL-CIPK network in plant calcium signaling. Trends Plant Sci. 2009;14(1):37–42. doi: 10.1016/j.tplants.2008.10.005. [DOI] [PubMed] [Google Scholar]
- 8.Luan S, Lan W, Chul Lee S. Potassium nutrition, sodium toxicity, and calcium signaling: Connections through the CBL-CIPK network. Curr Opin Plant Biol. 2009;12(3):339–346. doi: 10.1016/j.pbi.2009.05.003. [DOI] [PubMed] [Google Scholar]
- 9.Batistič O, et al. S-acylation-dependent association of the calcium sensor CBL2 with the vacuolar membrane is essential for proper abscisic acid responses. Cell Res. 2012;22(7):1155–1168. doi: 10.1038/cr.2012.71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Tang RJ, et al. Tonoplast calcium sensors CBL2 and CBL3 control plant growth and ion homeostasis through regulating V-ATPase activity in Arabidopsis. Cell Res. 2012;22(12):1650–1665. doi: 10.1038/cr.2012.161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Shaul O, et al. Cloning and characterization of a novel Mg(2+)/H(+) exchanger. EMBO J. 1999;18(14):3973–3980. doi: 10.1093/emboj/18.14.3973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Conn SJ, et al. Magnesium transporters, MGT2/MRS2-1 and MGT3/MRS2-5, are important for magnesium partitioning within Arabidopsis thaliana mesophyll vacuoles. New Phytol. 2011;190(3):583–594. doi: 10.1111/j.1469-8137.2010.03619.x. [DOI] [PubMed] [Google Scholar]