Abstract
Background:
Power-driven instrumentation of root surfaces during supportive periodontal therapy is an alternative to hand instrumentation. The purpose of this pilot in vitro study was to investigate the efficacy of sub- and supragingival plaque removal with a sonic (AIR: Synea, W and H, Bürmoos, Austria) and two ultrasonic devices (TIG: Tigon+, W and H, Bürmoos, Austria; VEC: Vector, Dürr, Bietigheim-Bissingen, Germany) as well as the health-risk for dental professionals during treatment.
Materials and Methods:
The power-driven devices were utilized to remove plaque from model teeth in dummy heads. The percentage of residual artificial plaque after 2 min of supra- or subgingival instrumentation was calculated by means of image-processing techniques at four sites (n = 576) of each tooth. The Health-Risk-Index (HRI: spatter/residual plaque quotient) with the different power-driven devices was assessed during treatment.
Results:
The smallest amounts of residual plaque were found for the sonic device AIR (8.89% ± 10.92%) and the ultrasonic scaler TIG (8.72% ± 12.02%) (P = 0.707). Significantly more plaque was remained after the use of the ultrasonic scaler VEC (18.76% ± 18.07%) (P < 0.001). Irrespectively of the scaler, efficacy was similar sub- (10.7% ± 11.6%) and supragingivally (13.5% ± 17.2%) (P = 0.901). AIR/TIG demonstrated equal residual amounts of plaque sub- (P = 0.831) as well as supragingivally (P = 0.510). However, AIR/VEC and TIG/VEC were significantly in favor of AIR and TIG (P < 0.001). In contrast, the lowest HRI was found after using VEC (0.0043) and differed considerably for AIR (0.2812) and TIG (0.0287).
Conclusion:
Sonic devices are as effective as ultrasonic devices in the removal of biofilm but bear a higher risk to the dental professional's health concerning the formation of spatter.
Keywords: Aerosol, dental prophylaxis, plaque, scaling
INTRODUCTION
During the last years power-driven instrumentation in periodontal therapy is gaining in significance as an alternative to conventional curette methods.[1] Compared with hand instruments, power-driven devices are relatively blunt and rely on the acceleration of the vibrating tip to disrupt the plaque. Nevertheless, they are easier to handle and less time-consuming than hand instruments.[2] Although, they are indispensable for periodontal treatment, their use is not without perils.[3] They are the major source of spatter and aerosol contamination in dental practices.[4,5,6,7,8] This should be kept in mind regarding the reported resurgence of bacterial diseases such as tuberculosis[9] and the presence of other pathogenic organisms with the potential for airborne transmission.[10,11] For that reason, in the current pilot study we used a newly developed Health-Risk-Index (HRI) as a quotient from spatter and residual plaque to discuss the advantages and disadvantages of different power-driven devices.
MATERIALS AND METHODS
Experimental setup: Operating room and dummy heads
The operating room was darkened and all surfaces 1.5 m2 around the dummy head were colored with matt black lacquer (Plasti Dip® Deutschland GmbH, Aschaffenburg, Germany) [Figure 1]. Dummy heads were equipped with periodontitis models (Frasaco, Tettnang, Germany). The modified models simulated moderate periodontitis with horizontal bone loss and isolated, deep vertical pockets up to 6 mm [Figure 2]. The root and cervical crown surfaces of the eight test teeth (every middle incisor and every first molar in both jaws) had to be cleaned. The molars showed no furcation involvement or deep concavity of the root surface. No calculus was applied locally considering the fact that the primary aim of this study was to ensure effective instrumentation with powered devices over a large area. The tooth surfaces were coated with a thin layer of transparent fluorescent lacquer (Shiny White – Rival de Loop Young, Berlin, Germany) in order to simulate adhering plaque. The coronal border of the coat on the tooth surface was nearly 2 mm above the cementum-enamel-border. Apical border was the alveolar bone [Figure 2a and b]. These borders were the same on each tooth type. To measure the HRI, the spatter was visualized using a 50 mg/l fluorescein water solution (Uranin, Niepötter Labortechnik, Bürstadt, Germany), which would fluoresce with bright orange color when exposed to ultraviolet-A light (350-370 nm).
Figure 1.

The darkened treatment room with the dummy head under normal light conditions and black lacquer coverage of all surfaces
Figure 2.
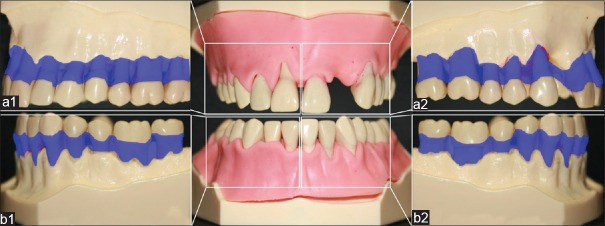
The modified periodontitis models for maxilla and mandible with horizontal bone loss and gingival masks of persistently pliable silicone (Frasaco, Tettnang, Germany); (a1) upper right and (b1) lower right side as well as (a2) upper left and (b2) lower left side. The transparent blue hatching in the right and left picture illustrate the coated tooth surface
Experimental setup: Artificial plaque and spatter distribution
Evaluation of the residual plaque on the tooth surface and the spatter contaminated area was planimetrically assessed [Figure 3]. The dummy head was placed over fixed marks in the treatment room for a reproducible position. After the treatment, the teeth were put in a specially designed apparatus (Lego GmbH, Grasbrunn, Germany) to ensure repeatable positions. These measures were taken to enable standardized evaluation of photos that were taken during and after treatment. To evaluate the residual plaque and the spatter distribution, the program (Excel 2013, Microsoft Corporation, Redmond, USA) was set up to draw standardized data masks with quadrangle arrays, which assisted in including the determined area of interest in the evaluation as illustrated in the enlargement in Figure 3a and b. The percentages of the area of residual plaque as well as the spatter were calculated via selective marking of the contaminated areas. The defined area of interest was set to “2”. In case of residual plaque or spatter remained it was set to “1”. An example is given in Figure 3a.
Figure 3.
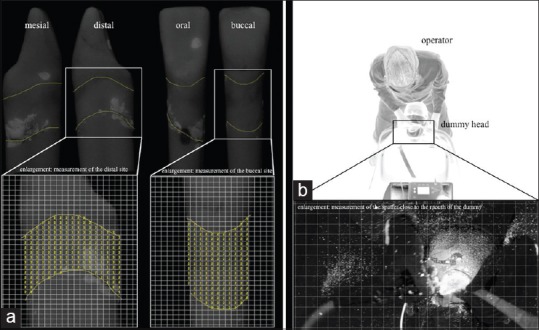
(a) Illustration of the measurement of subgingival residual plaque of tooth 31 after working with a sonic scaler (AIR) for 120 s. The enlargements show the measurement with the standardized data masks with quadrangle arrays. (b) For demonstration purposes; the experimental setup in the darkened treatment room under inverse light conditions with the operator in 12 o’clock treatment position. The enlargement shows part of a photo taken under experimental conditions to measure the spatter distribution
Instrumentation procedure
Badersten et al. and our pretrials have shown that a time span of 2 min for one tooth was sufficient for plaque removal with instruments during supportive periodontal therapy (SPT) and did not restrict the work of a trained operator. Therefore, a maximum treatment time of 2 min for each tooth was defined for our investigation.[12,13] Prior to each trial, the operator adjusted the coolant flow for the sonic and ultrasonic scaler to the clinically ideal level of 25-30 ml/min.[14] During the investigation one sonic scaler AIR (synea, W and H, Bürmoos, Austria) and one newly developed ultrasonic hand-piece TIG (tigon+, W and H, Bürmoos, Austria) as well as an established ultrasonic device VEC (vector, Dürr, Bietigheim-Bissingen, Germany) were used with nondiamond-coated tips (P60 and P1, W and H, Bürmoos, Austria and P1 Dürr, Bietigheim-Bissingen, Germany) [Figure 4]. All of the tips that were used can be categorized as “Slimline”.[15] Furthermore, a polish powder (<10 μm, Dürr, Bietigheim-Bissingen, Germany) was added to the ultrasonic device VEC as recommended by the manufacturer. Only one experienced operator (C.G.) performed the study procedures during all 18 trial series (6 trials, each with AIR, TIG and VEC divided in 3 trials for supra- and subgingival scaling). A high-speed evacuation system (VAS 300 S, Dürr, Bietigheim-Bissingen, Germany) with a standardized suction volume of 300 ml/min and a depression of 180 mbar was used during instrumentation.
Figure 4.
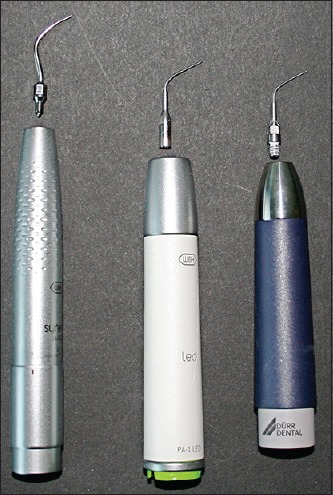
The three tested powered devices with the appending slimline tips. From left to right: Sonic hand-piece (AIR), ultrasonic hand-piece (TIG) and ultrasonic hand-piece (VEC)
Health-Risk-Index
We developed the HRI due to the lack of microorganism measurement in airborne spatter and aerosols in this in vitro study. The HRI is used to discuss the potential infection risk for dental staff and benefit in terms of the percentage of residual deposits for the patient. The HRI is a quotient of spatter and residual plaque after scaling (HRI = spatter/residual plaque). This index is based on the consideration of “effect and side-effect” of each medical therapy. For periodontal therapy it has been concluded that there is no effect of mechanically therapy with powered devices without side-effects.[16] During scaling side effects could be destruction of the root surfaces or hypersensitivity[2,17] and as investigated in this study, the risk of transmission of infectious disease like tuberculosis or hepatitis through generated spatter and aerosol.[9,10] Previous investigations have shown for the used powered devices, that scaling without aerosols and 100% effectiveness was impossible.[16,18]
Therefore, an HRI close to zero represents a high effectiveness of scaling with only a small amount of residual plaque. On the other side, an HRI ≥1 demonstrates a greater risk of spatter/aerosol and little effectiveness of scaling.
Statistical analysis
Statistical analysis was performed using SPSS (SPSS Statistics 20, IBM, Chicago, IL, USA). The assumption of normal distribution of data was checked by Shapiro–Wilk test. Differences between instruments were analyzed using the Kruskal–Wallis non parametric ANOVA. Post-hoc tests were performed afterwards by the Mann–Whitney U-test, whereby the significance level was adjusted after the Bonferroni method (α devided by the number of performed tests). Deviation within teeth and sites were calculated by the Wilcoxon test. All statistical tests were two-sided and used a significance level of P ≤ 0.05.
RESULTS
Efficacy of plaque removal
With respect to the power-driven instruments the Kruskal–Wallis test showed significant results (P < 0.001) in plaque removal. The highest amount of plaque remained after using the ultrasonic scaler VEC (18.76% ± 18.07%). After post-hoc testing VEC differed significantly (P < 0.001) compared to the sonic scaler AIR (8.89% ± 10.92%) and the ultrasonic scaler TIG (8.72% ± 12.02%) [Table 1]. Irrespectively of the scaler being used, the effectiveness was nearly similar for sub- (10.7% ± 11.6%) and supragingival debridement (13.5% ± 17.2%) (P = 0.901). Comparing residual deposits after sub- and supragingival instrumentation, the effectiveness was similar for AIR (supra: 9.54% ± 11.87%; sub: 8.23% ± 9.9%) and TIG (supra: 8.54% ± 12.72%; sub: 8.91% ± 11.34%) (supra: P = 0.510, sub: P = 0.831). In contrast significant differences for residual plaque were found between AIR and VEC as well as between TIG and VEC (P < 0.001) [Table 1]. Between jaws, maxilla and mandible, were significantly different in favor of the mandible (P < 0.001). On tooth level, more residual plaque was left in molars compared to incisors (at all: P < 0.001, supra: P = 0.011, sub: P = 0.008) [Table 1]. On site level, no differences were found between mesial and distal sites (at all: P = 0.910, supra: P = 0.401, sub: P = 0.330), whereas significantly higher residual plaque was found (P ≤ 0.015) [Table 1] for all other sites compared with the buccal site.
Table 1.
Residual plaque (mean±SD in percent) divided by the different powered devices: Sonic scaler (AIR) and two ultrasonic scalers (TIG and VEC) and localization: Jaws, teeth and sites
Health-Risk-Index
The highest HRI was found when scaling with sonic scaler AIR (0.2812), the lowest HRI was found when scaling with ultrasonic scaler VEC (0.0043) [Table 2 and Figure 5]. VEC/AIR and VEC/TIG were in favor of VEC regarding the HRI [Table 2]. Irrespectively of the power-driven device a lower HRI was found for subgingival scaling (0.0934) versus supragingival scaling (0.1174) as well as for molars (0.0162) compared with incisors (0.0243) irrespectively of the jaw [Table 2].
Table 2.
HRI as the quotient from spatter/residual plaque divided by the different powered devices: Sonic scaler (AIR) and two ultrasonic scalers (TIG and VEC) and localization: jaws and teeth

Figure 5.
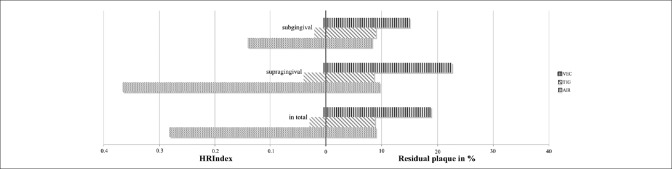
Residual plaque (right side of the graph) and Health-Risk-Index (left side of the graph) divided by the different powered devices: Sonic scaler (AIR) and two ultrasonic scalers (TIG and VEC) as well as the location (supra-, subgingival and in total)
DISCUSSION
Instruments design and efficacy
Ultrasonic and sonic scalers are routinely established in modern prophylaxis and periodontal treatment.[2,15] Hence, the goal of this study was to measure the efficacy of different power-driven instruments in relation to a newly developed HRI and to discuss the advantages and disadvantages of power-driven scaling. In this context, one recently released sonic device (AIR) as well as a newly developed (TIG) and an established ultrasonic device (VEC) were investigated. The tested ultrasonic devices work at a frequency range of 25-35 kHz, whereas the tested sonic device works at a frequency of 6 kHz. The manner of oscillation production for the two classes is different: The sonic device AIR relies on the passage of compressed air over an eccentric rod that is driven to vibrate. Instead the ultrasound generation of the two piezoelectric devices TIG and VEC works through a crystal oscillating in the presence of an electromagnetic field.[15] All investigated devices were highly effective in removing plaque. However, they demonstrated some differences. In terms of efficacy the best results were performed by TIG and AIR with no significant difference (P = 0.707). VEC left nearly 10% more residual deposits (P < 0.001). After subgingival debridement by AIR and TIG a plaque level of 8.23% ± 9.9% and 8.91% ± 11.34% remained. Compared to the data by Kocher et al. with similar instruments and study design the amount of residual plaque was comparatively low.[1] The results came up to 15.35% ± 13.10% for sonic and to 12.33% ± 3.93% for ultrasonic instrumentation. However, this is only a small improvement and the clinical relevance remains unknown.[15,19] The aim of such a treatment is to disrupt the subgingival biofilm and in doing so to reduce the pathogens that are present to allow periodontal health to return.[20,21] Therefore, it isn’t necessary to achieve a plaque reduction of 100%. It is more important to disrupt the plaque at all areas of the root surface, which is a problem in all cases with isolated, deep vertical pockets, concavities of the root surface or furcation involvement.[22,23] Especially for anatomically difficult situations like scaling of approximal sites of the molars[13] our investigation showed high efficacy of 16.8% ± 13.08% (AIR: 16.03% ± 12.74%; TIG: 13.87% ± 12.21%; VEC 20.5% ± 13.89%). On the contrary, less residual plaque after scaling was found especially on buccal sites of all incisors independent from the powered device used (in total 2.77% ± 5.16%). These results were significantly lower (P ≤ 0.001) compared with all other sites. This difference might be explained by accessibility as well as by the differences in tooth anatomy.[15] Furthermore, the buccal sites of the molars showed less residual plaque after scaling (8.37% ± 8.78%, P < 0.001). According to previous studies,[22] the buccal sites of molars were more easily accessed during power-driven instrumentation than proximal (16.8% ± 13.08%) or oral sites (11.09% ± 12.34%). However, the results of the current investigation must be carefully interpreted due to the used artificial tooth design of the first molars without deep concavities or furcation involvement [Figure 2]. According to Walmsley et al.,[15] the instrumentation of buccal or oral sites of molars with furcation involvement takes time and incomplete removal of plaque can furthermore be caused through accessibility problems. Adriaens and Adriaens[24] cited in their review 15-38% residual deposits for probing pocket depth values of 4-6 mm independent of the tooth type and used instruments. In the current investigation there were less residual deposits of 9-19%, more comparable with 4-43% for lower PPD up to 3 mm. Nevertheless, deeper probing depths of >6 mm showed the least effectiveness with 19-66% residual deposits.[24] Therefore, the morphology of the tooth as well as the pocket probing depth seem to influence the effectiveness of scaling and should therefore be mentioned during the discussion.
However, similar to recent investigations our results could not show a real improvement through the use of powered devices compared to previously detected data for hand instrumentation.[1,25,26] Instead the motivation of the trained operator and his ability to realistically assess one's own performance has a strong influence on debridement effectivity.[13] When more residual plaque was detected during the current investigation, it seemed more likely to be an application error [Figure 6] and not a functional failure of the power-driven device. However, all powered devices have the potential to damage the root surface although no definite conclusion has been reached on this topic.[27] Therefore, specially designed tips like Teflon or plastic coated tips were developed e.g. for sonic devices to preserve the root surface during repeated scaling during SPT.[22] Through the use of these Teflon coated tips it is possible to manage plaque removal effectively, whereas the removal of hard deposits like calculus seems insufficient.[28] Diamond-coated scaler tips seem to remove calculus faster,[19] but lead to more damage of the root surface.[29,30] Due to the fact that this aspect was not considered in this current study we will do further investigations. Furthermore a polish powder (<10 μm, Dürr, Bietigheim-Bissingen, Germany) was added to the ultrasonic device VEC in order to increase its efficacy. Similar to the clinical study by Kahl et al.[31] it failed to show a real benefit with an efficacy rate of only 81% compared with the conventional techniques. However, during SPT the use of the ultrasonic device VEC is able to improve periodontal healing similar to other instruments.[31] In addition, it constitutes an advantage over the other tested power-driven devices with a clearly better HRI (0.0043).
Figure 6.
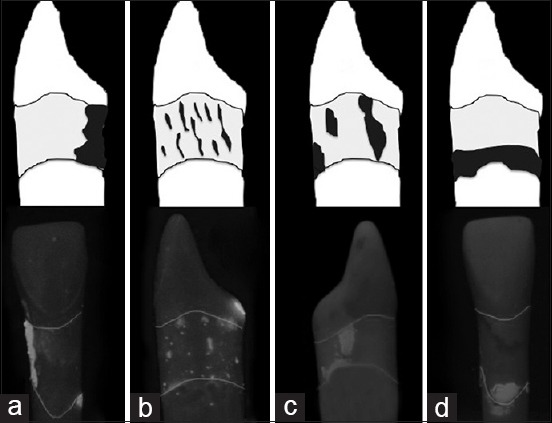
Illustration of residual plaque of tooth 41. Reason for ineffectivity could be: (a) Forgetting to treat a site/area, (b) Too fast and too hectical working strokes, (c) No systematical work, (d) Operator didn’t reach the plaque at the pocket bottom
Health-Risk-Index – benefit and harms of powered devices for scaling
The newly developed HRI was created in the current in vitro investigation to describe the hazards to health[32] in relation to the effectiveness of plaque removal. The study design included only measurement of spatter and aerosol,[11,33] but no air sampling and collecting of microorganism.[34,35] However, this helps to discuss potential spreading[16] of infections between patients and between patient and operator during debridement with powered devices. Regarding the HRI, in the current investigation the VEC seems to reduce the potential health hazards of power-driven devices [Figure 5].[16] Furthermore, it could improve the acceptance of the dental professionals also through the increase of the patient's comfort.[36] One disadvantage of all tested power-driven devices is the formation of spatter and aerosols,[34,37,38,39] which potentially contain microorganisms associated with infectious hepatitis and tuberculosis.[40,41] Nevertheless, there is no evidence that any serious diseases have been contracted in this manner.[16,40] Therefore, recent investigations analyzed the infection risk through spatter in order to create a prediction model of the total number of bacteria. Thus, a positive correlation between the number of decayed teeth and calculus as well as a negative correlation to preoperative rinsing with chlorhexidine was found.[42] Our results, which focus on the used instruments for scaling, showed a clearly higher HRI during sonic instrumentation (AIR: 0.2812). This emphasizes the conclusion from Timmerman et al.,[35] that piezoelectric devices, as a result of their linear tip movement,[43] produce less spatter and aerosol than sonic scalers with nearly round tip movement and a greater amplitude.[44] The HRI of the two piezoelectric devices is considerably lower (VEC: 0.0043 and TIG: 0.0287). Therefore, the tip movement does not only essentially influence the scaling efficacy but also the risk of infection. Conclusively, the instrument should be carefully selected, especially when treating potentially infectious patients with e.g. HIV or tuberculosis and if necessary opt for the use of an ultrasonic scaler. Nevertheless, liquids are essential for cooling during root instrumentation with all powered devices. Our results show that an entire elimination of spatter and aerosols seems impossible and therefore the prevention of infection through hand instrumentation of infectious patients seems to be safer.[16] Further, use of preprocedural rinsing with an antiseptic mouthwash seems to be an effective way to reduce the microbiological hazards of spatter and aerosols.[27,45,46,47] However, a conscientious clinician should therefore inculcate all available steps to overcome the hazards of the powered device scaling.[3] Besides, the root surface being treated seems to be relevant for the health-risk of the operator. In this current investigation, subgingival scaling showed a lower risk of spatter production compared to supragingival scaling (HRI: 0.0934 vs. 0.1174) as well as molars compared to incisors (HRI: 0.0162 vs. 0.0243). This should be taken into consideration especially when only supragingival calculus is removed in the lower jaw.
Finally, the limitations of the study are to be discussed. The advantage of this standardized method, e.g. investigation of defined variables, is mitigated by the fact that the working conditions are not necessarily comparable to a clinical situation. For instance, the material properties of the plastic teeth coated with artificial plaque and the artificial gingiva mask, correspond to the clinical setting to a limited extent only.[1] Another limitation of this study as well as other studies was the two-dimensional visualization of the treatment room and of the root surface divided in four sites.[1,13,48] That means, it is impossible, e.g. to correctly measure the residual plaque at the corners of the root [Figure 3a] or in the furcation area, but especially these areas seem most important because they are more difficult to access.[15,23] When discussing the HRI we have a similar situation, contamination on vertical surfaces or borders such as clothes or the face guard, is only conditionally viewable. Besides that, the size of the area around the dummy head to measure the spatter distribution was limited to 1.35 m × 1.11 m through the dimension of the treatment room. According to a recent investigation, which showed 33% of blood borne aerosols at a distance of 0.5 m from patient undergoing ultrasonic scaling around,[49] the chosen area seems sufficient.
CONCLUSIONS
Despite the limitations of the current pilot in vitro investigation, the tested power-driven devices show a high efficacy of >81% up to 91% in plaque removal, but it failed to show results which improve the discussion of their clinical relevance. It should be mentioned the importance of cleaning an area with efficacy of 80% or 90% is unknown. Nevertheless, it seems to be evident, that there is a significant difference in the HRI between sonic and ultrasonic root instrumentation. Therefore, the lower risk to the dental professional's health concerning the formation of spatter, recommends the use of ultrasonic devices. Further investigations under clinical conditions with patients have to follow, as the influence of airborne microorganism transmission is still unclear.
Footnotes
Source of Support: This study was supported by the Dürr Company (Dürr, Bietigheim-Bissingen, Germany)
Conflict of Interest: None declared.
REFERENCES
- 1.Kocher T, Rühling A, Momsen H, Plagmann HC. Effectiveness of subgingival instrumentation with power-driven instruments in the hands of experienced and inexperienced operators. A study on manikins. J Clin Periodontol. 1997;24:498–504. doi: 10.1111/j.1600-051x.1997.tb00218.x. [DOI] [PubMed] [Google Scholar]
- 2.Tunkel J, Heinecke A, Flemmig TF. A systematic review of efficacy of machine-driven and manual subgingival debridement in the treatment of chronic periodontitis. J Clin Periodontol. 2002;29(Suppl 3):72–81. doi: 10.1034/j.1600-051x.29.s3.4.x. [DOI] [PubMed] [Google Scholar]
- 3.Paramashivaiah R, Prabhuji ML. Mechanized scaling with ultrasonics: Perils and proactive measures. J Indian Soc Periodontol. 2013;17:423–8. doi: 10.4103/0972-124X.118310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Legnani P, Checchi L, Pelliccioni GA, D’Achille C. Atmospheric contamination during dental procedures. Quintessence Int. 1994;25:435–9. [PubMed] [Google Scholar]
- 5.Bentley CD, Burkhart NW, Crawford JJ. Evaluating spatter and aerosol contamination during dental procedures. J Am Dent Assoc. 1994;125:579–84. doi: 10.14219/jada.archive.1994.0093. [DOI] [PubMed] [Google Scholar]
- 6.Lu DP, Zambito RF. Aerosols and cross infection in dental practice - A historic view. Gen Dent. 1981;29:136–42. [PubMed] [Google Scholar]
- 7.Leggat PA, Kedjarune U. Bacterial aerosols in the dental clinic: A review. Int Dent J. 2001;51:39–44. doi: 10.1002/j.1875-595x.2001.tb00816.x. [DOI] [PubMed] [Google Scholar]
- 8.Szymañska J. Dental bioaerosol as an occupational hazard in a dentist's workplace. Ann Agric Environ Med. 2007;14:203–7. [PubMed] [Google Scholar]
- 9.Palakuru SK, Lakshman VK, Bhat KG. Microbiological analysis of oral samples for detection of Mycobacterium tuberculosis by nested polymerase chain reaction in tuberculosis patients with periodontitis. Dent Res J (Isfahan) 2012;9:688–93. [PMC free article] [PubMed] [Google Scholar]
- 10.Faecher RS, Thomas JE, Bender BS. Tuberculosis: A growing concern for dentistry? J Am Dent Assoc. 1993;124:94–104. doi: 10.14219/jada.archive.1993.0003. [DOI] [PubMed] [Google Scholar]
- 11.Harrel SK, Barnes JB, Rivera-Hidalgo F. Reduction of aerosols produced by ultrasonic scalers. J Periodontol. 1996;67:28–32. doi: 10.1902/jop.1996.67.1.28. [DOI] [PubMed] [Google Scholar]
- 12.Badersten A, Nilveus R, Egelberg J. Effect of nonsurgical periodontal therapy. II. Severely advanced periodontitis. J Clin Periodontol. 1984;11:63–76. doi: 10.1111/j.1600-051x.1984.tb01309.x. [DOI] [PubMed] [Google Scholar]
- 13.Rühling A, Schlemme H, König J, Kocher T, Schwahn C, Plagmann HC. Learning root debridement with curettes and power-driven instruments. Part I: A training program to increase effectivity. J Clin Periodontol. 2002;29:622–9. doi: 10.1034/j.1600-051x.2002.290706.x. [DOI] [PubMed] [Google Scholar]
- 14.Kocher T, Plagmann HC. Heat propagation in dentin during instrumentation with different sonic scaler tips. Quintessence Int. 1996;27:259–64. [PubMed] [Google Scholar]
- 15.Walmsley AD, Lea SC, Landini G, Moses AJ. Advances in power driven pocket/root instrumentation. J Clin Periodontol. 2008;35:22–8. doi: 10.1111/j.1600-051X.2008.01258.x. [DOI] [PubMed] [Google Scholar]
- 16.Trenter SC, Walmsley AD. Ultrasonic dental scaler: Associated hazards. J Clin Periodontol. 2003;30:95–101. doi: 10.1034/j.1600-051x.2003.00276.x. [DOI] [PubMed] [Google Scholar]
- 17.Gunsolley JC. The need for pain control during scaling and root planing. Compend Contin Educ Dent. 2005;26:3–5. [PubMed] [Google Scholar]
- 18.Kepic TJ, O’Leary TJ, Kafrawy AH. Total calculus removal: An attainable objective? J Periodontol. 1990;61:16–20. doi: 10.1902/jop.1990.61.1.16. [DOI] [PubMed] [Google Scholar]
- 19.Yukna RA, Vastardis S, Mayer ET. Calculus removal with diamond-coated ultrasonic inserts in vitro. J Periodontol. 2007;78:122–6. doi: 10.1902/jop.2007.060207. [DOI] [PubMed] [Google Scholar]
- 20.Slots J. Subgingival microflora and periodontal disease. J Clin Periodontol. 1979;6:351–82. doi: 10.1111/j.1600-051x.1979.tb01935.x. [DOI] [PubMed] [Google Scholar]
- 21.Haffajee AD, Teles RP, Socransky SS. The effect of periodontal therapy on the composition of the subgingival microbiota. Periodontol 2000. 2006;42:219–58. doi: 10.1111/j.1600-0757.2006.00191.x. [DOI] [PubMed] [Google Scholar]
- 22.Kocher T, Rühling A, Herweg M, Plagman HC. Proof of efficacy of different modified sonic scaler inserts used for debridement in furcations – A dummy head trial. J Clin Periodontol. 1996;23:662–9. doi: 10.1111/j.1600-051x.1996.tb00591.x. [DOI] [PubMed] [Google Scholar]
- 23.Zappa UE. Factors determining the outcome of scaling and root planing. Probe. 1992;26:152–9. [PubMed] [Google Scholar]
- 24.Adriaens PA, Adriaens LM. Effects of nonsurgical periodontal therapy on hard and soft tissues. Periodontol 2000. 2004;36:121–45. doi: 10.1111/j.1600-0757.2004.03676.x. [DOI] [PubMed] [Google Scholar]
- 25.Leonhardt A, Bergström C, Krok L, Cardaropoli G. Microbiological effect of the use of an ultrasonic device and iodine irrigation in patients with severe chronic periodontal disease: A randomized controlled clinical study. Acta Odontol Scand. 2007;65:52–9. doi: 10.1080/00016350600973078. [DOI] [PubMed] [Google Scholar]
- 26.Del Peloso Ribeiro E, Bittencourt S, Nociti FH, Jr, Sallum EA, Sallum AW, Casati MZ. Comparative study of ultrasonic instrumentation for the non-surgical treatment of interproximal and non-interproximal furcation involvements. J Periodontol. 2007;78:224–30. doi: 10.1902/jop.2007.060312. [DOI] [PubMed] [Google Scholar]
- 27.Drisko CL, Cochran DL, Blieden T, Bouwsma OJ, Cohen RE, Damoulis P, et al. Position paper: Sonic and ultrasonic scalers in periodontics. Research, Science and Therapy Committee of the American Academy of Periodontology. J Periodontol. 2000;71:1792–801. doi: 10.1902/jop.2000.71.11.1792. [DOI] [PubMed] [Google Scholar]
- 28.Rühling A, Bernhardt O, Kocher T. Subgingival debridement with a teflon-coated sonic scaler insert in comparison to conventional instruments and assessment of substance removal on extracted teeth. Quintessence Int. 2005;36:446–52. [PubMed] [Google Scholar]
- 29.Kocher T, König J, Hansen P, Rühling A. Subgingival polishing compared to scaling with steel curettes: A clinical pilot study. J Clin Periodontol. 2001;28:194–9. doi: 10.1034/j.1600-051x.2001.028002194.x. [DOI] [PubMed] [Google Scholar]
- 30.Vastardis S, Yukna RA, Rice DA, Mercante D. Root surface removal and resultant surface texture with diamond-coated ultrasonic inserts: An in vitro and SEM study. J Clin Periodontol. 2005;32:467–73. doi: 10.1111/j.1600-051X.2005.00705.x. [DOI] [PubMed] [Google Scholar]
- 31.Kahl M, Haase E, Kocher T, Rühling A. Clinical effects after subgingival polishing with a non-aggressive ultrasonic device in initial therapy. J Clin Periodontol. 2007;34:318–24. doi: 10.1111/j.1600-051X.2007.01056.x. [DOI] [PubMed] [Google Scholar]
- 32.Suppipat N. Ultrasonics in periodontics. J Clin Periodontol. 1974;1:206–13. doi: 10.1111/j.1600-051x.1974.tb01259.x. [DOI] [PubMed] [Google Scholar]
- 33.Harrel SK. Clinical use of an aerosol-reduction device with an ultrasonic scaler. Compend Contin Educ Dent. 1996;17:1185–93. [PubMed] [Google Scholar]
- 34.Gross KB, Overman PR, Cobb C, Brockmann S. Aerosol generation by two ultrasonic scalers and one sonic scaler. A comparative study. J Dent Hyg. 1992;66:314–8. [PubMed] [Google Scholar]
- 35.Timmerman MF, Menso L, Steinfort J, van Winkelhoff AJ, van der Weijden GA. Atmospheric contamination during ultrasonic scaling. J Clin Periodontol. 2004;31:458–62. doi: 10.1111/j.1600-051X.2004.00511.x. [DOI] [PubMed] [Google Scholar]
- 36.Braun A, Krause F, Nolden R, Frentzen M. Subjective intensity of pain during the treatment of periodontal lesions with the vector-system. J Periodontal Res. 2003;38:135–40. doi: 10.1034/j.1600-0765.2003.00356.x. [DOI] [PubMed] [Google Scholar]
- 37.Larato DC, Ruskin PF, Martin A. Effect of an ultrasonic scaler on bacterial counts in air. J Periodontol. 1967;38:550–4. doi: 10.1902/jop.1967.38.6_part1.550. [DOI] [PubMed] [Google Scholar]
- 38.Walmsley AD. Potential hazards of the dental ultrasonic descaler. Ultrasound Med Biol. 1988;14:15–20. doi: 10.1016/0301-5629(88)90159-7. [DOI] [PubMed] [Google Scholar]
- 39.Dutil S, Meriaux A, de Latremoille MC, Lazure L, Barbeau J, Duchaine C. Measurement of airborne bacteria and endotoxin generated during dental cleaning. J Occup Environ Hyg. 2009;6:121–30. doi: 10.1080/15459620802633957. [DOI] [PubMed] [Google Scholar]
- 40.Holbrook WP, Muir KF, Macphee IT, Ross PW. Bacteriological investigation of the aerosol from ultrasonic scalers. Br Dent J. 1978;144:245–7. doi: 10.1038/sj.bdj.4804072. [DOI] [PubMed] [Google Scholar]
- 41.Goldman HS, Hartman KS. Infectious diseases. Their disease, our unease: Infectious diseases and dental practice. Va Dent J. 1986;63:10–9. [PubMed] [Google Scholar]
- 42.Serban D, Banu A, Serban C, Tuþã-Sas I, Vlaicu B. Predictors of quantitative microbiological analysis of spatter and aerosolization during scaling. Rev Med Chir Soc Med Nat Iasi. 2013;117:503–8. [PubMed] [Google Scholar]
- 43.Flemmig TF, Petersilka GJ, Mehl A, Hickel R, Klaiber B. The effect of working parameters on root substance removal using a piezoelectric ultrasonic scaler in vitro. J Clin Periodontol. 1998;25:158–63. doi: 10.1111/j.1600-051x.1998.tb02422.x. [DOI] [PubMed] [Google Scholar]
- 44.Flemmig TF, Petersilka GJ, Mehl A, Rüdiger S, Hickel R, Klaiber B. Working parameters of a sonic scaler influencing root substance removal in vitro. Clin Oral Investig. 1997;1:55–60. doi: 10.1007/s007840050011. [DOI] [PubMed] [Google Scholar]
- 45.Veksler AE, Kayrouz GA, Newman MG. Reduction of salivary bacteria by pre-procedural rinses with chlorhexidine 0.12% J Periodontol. 1991;62:649–51. doi: 10.1902/jop.1991.62.11.649. [DOI] [PubMed] [Google Scholar]
- 46.Fine DH, Mendieta C, Barnett ML, Furgang D, Meyers R, Olshan A, et al. Efficacy of preprocedural rinsing with an antiseptic in reducing viable bacteria in dental aerosols. J Periodontol. 1992;63:821–4. doi: 10.1902/jop.1992.63.10.821. [DOI] [PubMed] [Google Scholar]
- 47.Fine DH, Furgang D, Korik I, Olshan A, Barnett ML, Vincent JW. Reduction of viable bacteria in dental aerosols by preprocedural rinsing with an antiseptic mouthrinse. Am J Dent. 1993;6:219–21. [PubMed] [Google Scholar]
- 48.Reitemeier B, Jatzwauk L, Jesinghaus S, Reitemeier C, Neumann K. Effective reduction of aerosol-possibilities and limitations. Zahnheilkunde-Management-Kultur. 2010;26:662–73. [Google Scholar]
- 49.Yamada H, Ishihama K, Yasuda K, Hasumi-Nakayama Y, Shimoji S, Furusawa K. Aerial dispersal of blood-contaminated aerosols during dental procedures. Quintessence Int. 2011;42:399–405. [PubMed] [Google Scholar]


