Abstract
Blood stasis (BS) is characterized as a disorder of blood circulation. In traditional Korean medicine (TKM), it is viewed as a cause factor of diseases such as multiple sclerosis and stroke. This study investigated differences in the plasma metabolites profiles of subjects displaying BS or non-BS patterns. Thirty-one patients with cerebral infarction diagnosed with BS and an equal number of sex- and age-matched non-BS patients were enrolled. Metabolic profiling was performed using UPLC-MS. The ratio of subjects with a rough pulse and purple coloration of the tongue was higher in patients presenting with BS pattern. Through metabolomics analysis, 82 metabolites that differed significantly between the BS and non-BS pattern were identified, and the two groups were significantly separated using an orthogonal partial least square-discriminant analysis model (P < 0.001). Of these 82 metabolites, acetyl carnitine, leucine, kynurenine, phosphocholine, hexanoyl carnitine, and decanoyl carnitine were present in significantly higher levels in patients with a BS pattern than those with a non-BS pattern. Our results also demonstrated that seven plasma metabolites, including acyl-carnitines and kynurenine, were associated with a BS pattern, suggesting that variant plasma metabolic profiles may serve as a biomarker for diagnosis of BS in patients with cerebral infarction.
1. Introduction
Traditional Korean medicine (TKM) assigns diseases to subtypes according to the combination of symptoms experienced by patients, a process known as pattern identification (PI). PI is a diagnosis system unique to traditional medicines practiced in East Asian countries, including China, Korea, and Japan. In TKM, stroke is assigned to five PI subtypes: Qi-deficiency (QD), dampness-phlegm (DP), blood stasis (BS), Yin-deficiency (YD), and fire and heat (FH) [1].
Blood stasis (BS) is characterized as a disorder of blood circulation with hallmarks including extravagated or sluggish blood circulation and viscous or congested blood, all of which may contribute to various disease pathologies [1]. Similar to its definition in TKM, BS is described in traditional Chinese medicine (TCM) as a slowing or pooling of the blood caused by disruption of heart Qi. BS can be understood in biomedical terms in the context of hematological disorders such as hemorrhage, congestion, thrombosis, and local ischemia (microclots) [2].
The diagnosis of a BS pattern by traditional medical doctors relies on the symptoms exhibited by patients. The subjectivity of this process calls the reliability of a BS diagnosis into question [3], leading many researchers in China, Korea, and Japan to search for serum or plasma biomarkers associated with the diagnosis [4–7].
Metabolomics is a powerful approach to quantitative assessment of endogenous small molecules within biological fluids such as serum and urine [8] and has been utilized to identify candidate biomarkers of several diseases [9–13]. Many studies using this approach have been performed to find metabolites associated with PI subtypes [14–19]. Jian et al. have shown that some free fatty acids and amino acids, including octadecanoic acid, arachidonic acid, and proline, were associated with BS in coronary heart disease [18]. Zhao et al. have reported 37 metabolites associated with BS in patients with unstable angina [19].
In this study, we analyzed the metabolic profiles of plasma from patients with cerebral infarction (CI) by ultraperformance liquid chromatography-quadrupole time of-flight mass spectrometry (UPLC-Q-TOF-MS) to identify metabolites associated with BS.
2. Materials and Methods
2.1. Subjects
Patients with CI were enrolled from 2009 to 2010 at two Korean oriental medical hospitals (Kyung Hee Oriental Medical Center in Seoul and Dae Jeon Oriental Medical Hospital in Daejeon) and one western medical center (Dongguk University Medical Center in Kyunggi-do). Subjects were CI patients enrolled within one month of the onset of symptoms, as characterized by Park et al. [20]. The patients' symptoms were confirmed via diagnostic imaging with computerized tomography (CT) or magnetic resonance imaging (MRI). CI subtypes were determined according to the Trial of ORG 10172 in the Acute Stroke Treatment (TOAST) classification. Subjects with hemorrhaging or with infectious or liver diseases were excluded from the study. Some patient in this study was also included in a previous study [15].
After obtaining written informed consent from all subjects, clinical data and plasma were collected. This study was approved by the Institutional Review Board (IRB) of the KIOM and by both of the oriental medical hospitals.
2.2. Diagnosis of Blood Stasis Pattern
The symptoms experienced by subjects were collected using the “stroke PI case report form.” Diagnosis of a BS pattern in subjects was determined by two expert TKM doctors based on criteria established in “Korean Standard PIs for Stroke-II” as previously reported by Go et al. [21]. Subjects receiving different BS pattern diagnoses from the two doctors were excluded. There were 31 age- and sex-matched patients in the non-BS and BS groups. The general characteristics of patients in the two groups are shown in Table 1.
Table 1.
Demographic parameters of study subjects.
| Characteristics | Non-BS | BS | P valuea |
|---|---|---|---|
| Anthropometric characteristics | |||
| Sex (M/F) | 14/17 | 15/16 | 1.000* |
| Age (year) | 67.13 ± 10.51 | 66.06 ± 10.78 | 0.659 |
| Smoking (none/stop/active) | 9/7/15 | 5/9/17 | 0.468 |
| Drinking (none/stop/active) | 9/5/17 | 17/3/11 | 0.120 |
| BMI (kg/m2) | 24.03 ± 4.23 | 22.85 ± 2.79 | 0.208 |
| Waist circumference (cm) | 84.10 ± 11.18 | 88.73 ± 9.69 | 0.209 |
| TOAST classification | |||
| LAA/CE/SVO/SUE | 7/3/17/3 | 8/7/11/3 | 0.511 |
| NIHSS | 3.83 ± 3.09 | 3.53 ± 3.47 | 0.732 |
| Medical history | |||
| TIA (yes, %) | 1 (3.23) | 3 (9.68) | 0.612* |
| Hypertension (yes, %) | 19 (61.29) | 20 (64.52) | 1.000* |
| Hyperlipidemia (yes, %) | 2 (6.45) | 7 (22.58) | 0.147* |
| DM (yes, %) | 6 (19.35) | 9 (29.03) | 0.554* |
| IHD (yes, %) | 0 | 3 (9.68) | 0.238* |
| Blood parameter | |||
| WBC (×103) | 7.93 ± 1.98 | 8.12 ± 2.43 | 0.734 |
| RBC (×106) | 4.58 ± 0.48 | 4.44 ± 0.59 | 0.306 |
| Hg (g/dL) | 13.94 ± 1.60 | 13.60 ± 1.77 | 0.441 |
| Hct (%) | 40.10 ± 7.84 | 40.20 ± 4.75 | 0.949 |
| Platelet (×103/ul) | 294.93 ± 98.24 | 284.27 ± 151.11 | 0.750 |
| Fibrinogen (mg/dL) | 371.11 ± 162.05 | 346.78 ± 93.85 | 0.491 |
| Total cholesterol (mg/dL) | 179.10 ± 35.48 | 173.32 ± 35.57 | 0.525 |
| Triglyceride (mg/dL) | 143.13 ± 89.26 | 112.59 ± 48.96 | 0.110 |
| HDL-cholesterol (mg/dL) | 44.07 ± 15.61 | 45.0 ± 12.26 | 0.798 |
| FBS (mg/dL) | 107.92 ± 22.23 | 120.56 ± 62.71 | 0.347 |
a P value using Student's t-test for continuous variables and chi-square test or Fisher's exact test *for categorical variables.
2.3. Plasma Extract Analysis by UPLC-Q-TOF-MS
Plasma protein was precipitated by the addition of cold acetonitrile. After shaking for 30 min at 4°C, the samples were centrifuged at 13,000 rpm for 10 min at 4°C. The supernatant was dissolved in 20% aqueous methanol containing caffeine for ultraperformance liquid chromatography-quadrupole time-of-flight mass spectrometry (UPLC-Q-TOF-MS) analysis.
A UPLC-Q-TOF-MS instrument (Waters, Milford, MA, USA), equipped with a column oven and coupled with a Waters Q-TOF primier, was used. The Q-TOF-MS was operated in positive electrospray ionization (ESI) mode with a scan range of m/z 50–1,000. Cone voltage was 30 V, capillary voltage was 3 kV, and scan time was 0.2 s with an interscan delay of 0.02 s. The source temperature was set at 110°C, and the desolvation flow was set at 700 L/h. The desolvation gas temperature was set at 300°C. The MS was calibrated using sodium format, and leucine enkephalin was used as lock mass. The concentration of leucine enkephalin was 200 ρM, and the flow rate was set at 5 μL/min.
In the MS-MS experiments, argon was used as collision gas, with the collision energy alternating between 10 and 30 eV. A 5 μL aliquot of extracted plasma sample was injected into an Acquity UPLC BEH C18 column (2.1 × 100 mm, 1.7 μm, Waters, Milford, MA, USA). The column oven was set at 40°C, and the sample temperature was 10°C. The mobile phase consisted of 0.1% formic acid in water (A) and 0.1% formic acid in acetonitrile (B), and the flow rate was 0.35 mL/min. MassLynx software version 4.1 (Waters Inc.) was used to control the instrument and calculate accurate masses.
2.4. Data Processing and Pattern Recognition Analysis
UPLC-MS data, including retention time, m/z, and ion intensity, were extracted using MarkerLynx software (Waters Corp., Milford, USA) and assembled into a data matrix. Peaks were collected using a peak width at 5% height, 1 s, a noise elimination of 6, and an intensity threshold of 50. Data were aligned with a mass tolerance of 0.07 Da and a retention time window of 0.2 min. All spectra were aligned and normalized to an external standard.
The resulting data sets were imported into SIMCA-P version 12.0.1 (Umetrics, Umeå, Sweden) for multivariate analysis and were mean-centered scaled. Orthogonal partial least-squares discriminant analysis (OPLS-DA) was conducted, and the quality of each model was determined based on a goodness of fit parameter (R 2) and a goodness of prediction parameter (Q 2). In addition, OPLS-DA models and the reliabilities of models were further validated using a rigorous permutation test (n = 200). To find metabolites that contributed to the discrimination, the S-plot showing a combination of covariance p(1) and correlation p(corr) from the OPLS-DA model was generated to better visualize the metabolites contributing to the discrimination. Assignment of metabolites contributing to the observed variance was performed using the ChemSpider (http://www.chemspider.com/) and the Human Metabolome Database (http://www.hmdb.ca/).
2.5. Statistical Analysis
The statistical analysis of our data was performed with IBM SPSS Statistics 19 (IBM Co., New York, NC, USA). After testing for the normality of continuous variables in clinical data by the Kolmogorov-Smirnov test, significant difference was determined via t-test for parametric variables or Mann-Whitney U test for nonparametric variables. Categorical variables were compared with a chi-square test or Fisher's exact test. The results of LC-MS were tested using independent t-test with Mann-Whitney U test. The statistical significance was set at P < 0.05.
3. Results
3.1. Distribution of Symptoms and Signs in BS and Non-BS Groups
According to Korean Standard PIs for Stroke-II, we investigated the distribution of 11 diagnostic symptoms and signs of BS from patients with stroke. As shown in Table 2, 40.9% of the BS patients displayed purple tongue coloration (P = 0.001), and some patients in the BS group had red or black spots on the surface of the tongue which were not observed in the non-BS group (P = 0.039). In addition, the ratio of subjects with rough pulse in the BS group was significantly higher (64.5%) than that of subjects in the non-BS group (6.5%) (P < 0.001).
Table 2.
Distribution of BS symptoms in BS and non-BS patients.
| Variable | Non-BS | BS | P valuea |
|---|---|---|---|
| Black face with black eyelid | 1 (3.2) | 4 (12.9) | 0.354* |
| Purpura in the sclera | 5 (16.1) | 2 (6.5) | 0.425* |
| Purpura | 6 (19.4) | 9 (29.0) | 0.554* |
| Cyanotic lips | 3 (9.7) | 10 (32.3) | 0.059* |
| Site-fixed headache | 2 (6.5) | 5 (16.1) | 0.425* |
| Headache with a pulling sensation | 1 (3.2) | 3 (9.7) | 0.612* |
| Fishy-smelling mouth odor | 15 (48.4) | 11 (35.5) | 0.440* |
| Tongue purple in color | 2 (6.5) | 13 (41.9) | 0.001 |
| Tongue with red, white, or black spots as well as thornlike protrusions on its surface | 0 (0.0) | 4 (12.9) | 0.039 |
| Bloated feeling in the chest and hypochondriac region | 1 (3.2) | 0 (0.0) | 0.313* |
| Rough pulse | 2 (6.5) | 20 (64.5) | <0.001 |
a P value using a chi-square test or Fisher's exact test (∗) for categorical variables.
3.2. Pattern Recognition Analysis of Plasma Metabolic Profiling
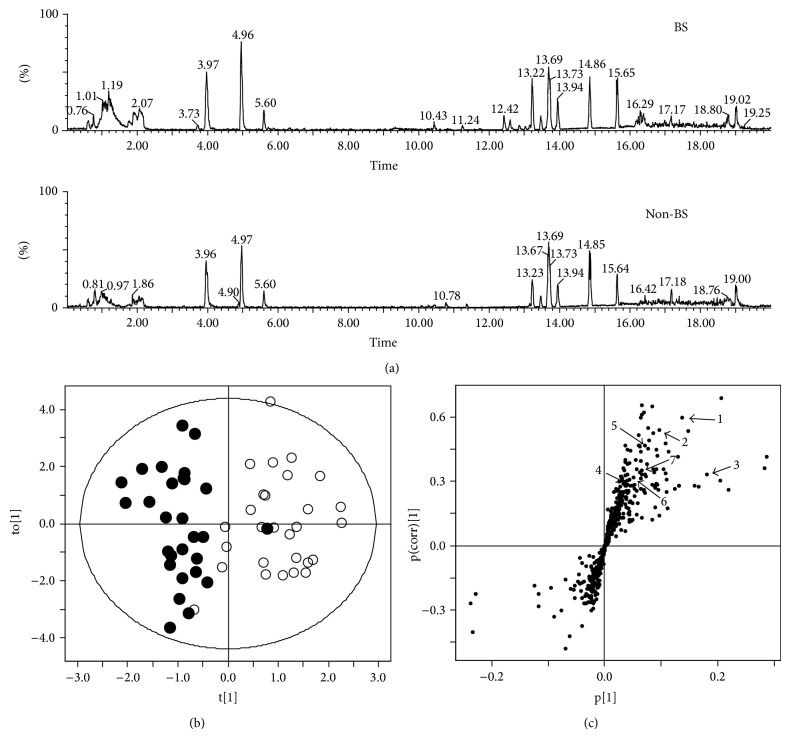
Figure 1(a) shows representative Base peak intensity (BPI) of plasma from the BS (higher) and non-BS patients (lower) obtained via UPLC-Q-TOF MS in positive mode. To identify differences in metabolic pattern between the BS and non-BS patients, OPLS-DA was applied to the UPLC-Q-TOF MS data.
Figure 1.
Ultraperformance liquid chromatography-quadrupole time-of-flight mass spectrometry (UPLC-Q-TOF-MS) profiles of plasma from BS and non-BS subjects in positive mode (a), orthogonal partial least-squares discriminant analysis (OPLS-DA) score (b), and S-plots derived from UPLC-Q-TOF MS data (c) of plasma from patients with stroke. The score plots showed a significant separation between BS and non-BS patients (P < 0.0001) by permutation test.
The OPLS-DA score plot showed different metabolic patterns in the plasma of the BS and non-BS patients, and the R 2 Y and Q 2 Y values of the OPLS-DA model were 0.715 and 0.436, respectively (Figure 1(b)). The model was validated using 200x repeated permutation and with P < 0.0001, indicating that the OPLS-DA model is significant.
3.3. Targeted Metabolic Profiling to Identify a BS Marker
The UPLC-MS data allowed us to find 82 metabolites that differed significantly between the BS and non-BS groups (P < 0.05) in Supplemental Table 1 (see Supplementary Material available online at http://dx.doi.org/10.1155/2015/453423). Furthermore, we identified seven metabolites for which P < 0.05 and the variable importance of projection (VIP) value was higher than 1.0 using public data masses by ion masses determined using positive-ion-modes (Table 3).
Table 3.
Identification of plasma metabolites in patients with cerebral infarction (non-BS and BS) analyzed using UPLC-Q-TOF MS.
| Numbera | Identity | Exact mass (M + H) |
Actual mass (M + H) |
Mass error (mDa) |
MS fragments (ESI) |
Fold change (BS versus non-BS) |
P valueb | VIP |
|---|---|---|---|---|---|---|---|---|
| 1 | Creatinine | 114.0667 | 114.0670 | −0.3 | 99, 86, 72, 71 | 1.46 | 0.010 | 2.88 |
| 2 | Acetyl carnitine | 204.1236 | 204.1262 | −2.6 | 145, 144, 85, 60 | 1.44 | 0.037 | 1.75 |
| 3 | Leucine | 132.1025 | 132.1001 | 2.4 | 86 | 1.31 | 0.042 | 4.12 |
| 4 | Kynurenine | 209.0926 | 209.0998 | −7.2 | 174, 146, 136, 94 | 2.22 | 0.010 | 1.05 |
| 5 | Phosphocholine | 185.0817 | 185.1326 | −50.9 | 125, 98, 86 | 1.61 | 0.000 | 1.73 |
| 6 | Hexanoyl carnitine | 260.1862 | 260.1897 | −3.5 | 201, 183, 144, 85 | 5.18 | 0.000 | 1.28 |
| 7 | Decanoyl carnitine | 316.2488 | 316.2512 | −2.4 | 137, 129, 144, 85 | 1.98 | 0.012 | 1.42 |
aNumber is the number of metabolites marked in Figure 1(c).
b P value was calculated using Mann-Whitney U test.
Levels of carnitine-related metabolites, including acetyl carnitine, decanoyl carnitine, and hexanoyl carnitine, were significantly higher in the BS pattern than the non-BS pattern patients (P < 0.05). Kynurenine, which is produced by oxidation of tryptophan, was increased 2.2-fold in the BS pattern patients (P = 0.010). Creatinine, leucine, and phosphocholine were also increased 1.3–1.6-fold in the BS pattern patients (P = 0.010, P = 0.042, and P < 0.001, resp.).
4. Discussion
BS is defined as a state of stagnated or extravagated blood in a blood vessel that causes hematological disorders such as hemorrhage, congestion, thrombosis, and local ischemia (microclots) as well as changes in tissue [22]. In TCM, the major symptoms of BS pattern include purple coloration or the emergence of purple spots on the tongue. Additionally, BS often presents with a choppy or rough pulse [23]. The description of BS symptoms is similar in TKM [24], although the subjects in our study showed more exaggerated signs of purple color on tongue and rough pulse (Table 2). Inspection of the tongue holds a prominent place in TKM diagnostics, as various pathological conditions rooted in disruption of circulation manifest there [25, 26]. In particular, when the tongue is dark purple, stasis or hypoxia is present, rendering the microvenules dark blue [27]. Previous study reported that fibrinogen and platelet in plasma, which are parameters involved in blood coagulation, were increased in patients with BS [28], but those levels were not different in BS pattern (Table 1) and the syndrome of blood stasis in this study. However, it is unknown how these physiological changes occur and how they affect BS pattern.
Many studies have been performed in China, Korea, and Japan to find biological molecules affecting the BS patterns of various diseases [5, 17, 29]. Metabolomics is a powerful method for identifying small molecules present at different levels in the biofluids of patients affected by disease. Studies have been performed to relate BS patterns, as defined by TCM, with physiologically defined coronary heart disease, diabetes, and psoriasis [18, 19, 30]. In this study, we investigated plasma metabolites in BS and non-BS groups of patients with cerebral infarction using UPLC-Q-TOF-MS methods and found that the two groups were significantly separated in an OPLS-DA model obtained by plasma metabolic profiling (Figure 1(b)).
Several of metabolites, acetyl carnitine, hexanoyl carnitine, and decanoyl carnitine were significantly increased in the patients with a BS pattern (Table 3). Particularly, decanoyl carnitine and hexanoyl carnitine were related with tongue purple in color (data not shown). Carnitine also showed a tendency to increase in the patients with a BS pattern, in agreement with a previous study that elucidated BS of myocardial ischemia using TCM methods [31]. In addition, some studies also showed that the hypoxia-induced accumulation of long-chain acylcarnitine and repeated episodes of ischemia may cause chronic accumulation of short-chain acylcarnitine in plasma in patients with peripheral vascular disease [32].
Another metabolite, kynurenine, which is an intermediate in the tryptophan metabolic pathway producing niacin, was increased over twofold in the patients with a BS pattern (Table 3). Kynurenine is synthesized by tryptophan dioxygenase and indoleamine 2,3-dioxygenase cascades [33] and correlated with cardiovascular disease and stroke [34, 35]. The role of kynurenine in the BS pattern is unknown, but the relationship between kynurenine and BS pattern has been suggested by previous studies. One cause of BS pattern from a pathophysiological perspective is stagnant blood in a blood vessel, a phenotypic result of multiple sclerosis. Plasma kynurenine and indoleamine 2,3-dioxygenase activity have been known to increase in patients with atherosclerosis and coronary heart disease [36, 37]. Recently, Sulo et al. showed that the kynurenine-tryptophan ratio was an important predictor of coronary events, including multiple sclerosis [38]. We also observed that the kynurenine/tryptophan ratio in the BS subjects was increased. This means that the BS pattern caused by coagulation may activate the kynurenine pathway.
This study has several limitations. First, it is a cross-sectional study and, as such, does not demonstrate a change in plasma metabolite composition after treatment. Second, the sample size was small and may prevent generalizing the association of metabolites such as acylcarnitine and kynurenine with a BS pattern. Third, the degree of BS pattern in each of the patients was not quantified in this study; so, we could not determine whether a correlation between degree of BS and level of plasma metabolite biomarkers was present. Fourth, PI diagnosis was dependent on two expert TKM doctors, and the subjectivity of this diagnosis forced us to perform an observational study. Despite these limitations, this study provided metabolic information about BS patterns of stroke, and further studies should be performed in subjects of other large populations to generalize its conclusions.
Supplementary Material
Suppl. Table 1: Identification of plasma metabolites in stroke patients (Non-BS and BS) analyzed using ultra-performance liquid chromatography-quadrupole time-of-flight mass spectrometry.
Acknowledgments
The work is supported by the Korean Institute of Oriental Medicine (K14280) and the Korean Food Research Institute (K14281).
Conflict of Interests
The authors declare that there is no conflict of interests regarding the publication of this paper.
Authors' Contribution
Min Ho Cha and Min Jung Kim contributed equally to this work.
References
- 1.Lee J. A., Park T.-Y., Moon T.-W., et al. Developing indicators of pattern identification in patients with stroke using traditional Korean medicine. BMC Research Notes. 2012;5, article 136 doi: 10.1186/1756-0500-5-136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Bensky D., Gamble A. Chinese Herbal Medicine Materia Medica. Eastland Press; 1993. [Google Scholar]
- 3.Wu R., Xie J.-X., Zhao F.-D. Modernized study on eye's signs of blood-stasis syndrome. Zhongguo Zhong Xi Yi Jie He Za Zhi. 2011;31(3):319–322. [PubMed] [Google Scholar]
- 4.Liu Z., Yu Z., Ouyang X., Du J., Lan X., Zhao M. Applied research on serum protein fingerprints for prediction of Qi deficiency syndrome and phlegm and blood stasis in patients with non-small cell lung cancer. Journal of Traditional Chinese Medicine. 2012;32(3):350–354. doi: 10.1016/s0254-6272(13)60036-9. [DOI] [PubMed] [Google Scholar]
- 5.Matsumoto C., Kojima T., Ogawa K., et al. A proteomic approach for the diagnosis of ‘Oketsu’ (blood stasis), a pathophysiologic concept of Japanese traditional (Kampo) medicine. Evidence-Based Complementary and Alternative Medicine. 2008;5(4):463–474. doi: 10.1093/ecam/nem049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Chen Y., Zhu L., Qian Y.-M. Diagnostic value of assay of brain natriuretic peptide in patients with sepsis of blood-stasis syndrome. Zhong Xi Yi Jie He Xue Bao. 2008;6(3):266–269. doi: 10.3736/jcim20080309. [DOI] [PubMed] [Google Scholar]
- 7.Liu Y., Liu P., Dai R., et al. Analysis of plasma proteome from cases of the different traditional Chinese medicine syndromes in patients with chronic hepatitis B. Journal of Pharmaceutical and Biomedical Analysis. 2012;59(1):173–178. doi: 10.1016/j.jpba.2011.10.002. [DOI] [PubMed] [Google Scholar]
- 8.Nicholson J. K., Lindon J. C. Systems biology: metabonomics. Nature. 2008;455(7216):1054–1066. doi: 10.1038/4551054a. [DOI] [PubMed] [Google Scholar]
- 9.Spratlin J. L., Serkova N. J., Eckhardt S. G. Clinical applications of metabolomics in oncology: a review. Clinical Cancer Research. 2009;15(2):431–440. doi: 10.1158/1078-0432.ccr-08-1059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Claudino W. M., Quattrone A., Biganzoli L., Pestrin M., Bertini I., Di Leo A. Metabolomics: available results, current research projects in breast cancer, and future applications. Journal of Clinical Oncology. 2007;25(19):2840–2846. doi: 10.1200/jco.2006.09.7550. [DOI] [PubMed] [Google Scholar]
- 11.Qiu Y., Cai G., Su M., et al. Serum metabolite profiling of human colorectal cancer using GC-TOFMS and UPLC-QTOFMS. Journal of Proteome Research. 2009;8(10):4844–4850. doi: 10.1021/pr9004162. [DOI] [PubMed] [Google Scholar]
- 12.Shah S. H., Sun J.-L., Stevens R. D., et al. Baseline metabolomic profiles predict cardiovascular events in patients at risk for coronary artery disease. The American Heart Journal. 2012;163(5):844–850. doi: 10.1016/j.ahj.2012.02.005. [DOI] [PubMed] [Google Scholar]
- 13.Ciborowski M., Teul J., Martin-Ventura J. L., Egido J., Barbas C. Metabolomics with LC-QTOF-MS permits the prediction of disease stage in aortic abdominal aneurysm based on plasma metabolic fingerprint. PLoS ONE. 2012;7(2) doi: 10.1371/journal.pone.0031982.e31982 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Yan B., JiYe A., Hao H., et al. Metabonomic phenotype and identification of ‘heart blood stasis obstruction pattern’ and ‘qi and yin deficiency pattern’ of myocardial ischemia rat models. Science in China Series C: Life Sciences. 2009;52:1081–1090. doi: 10.1007/s11427-009-0136-y. [DOI] [PubMed] [Google Scholar]
- 15.Cha M. H., Jones A. D., Ko M. M., Zhang C., Lee M. S. Metabolic profiles distinguish non-dampness-phlegm and dampness-phlegm patterns among korean patients with acute cerebral infarction. Evidence-based Complementary and Alternative Medicine. 2013;2013:9. doi: 10.1155/2013/517018.517018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Bai D., Song J. Plasma metabolic biomarkers for syndrome of phlegm and blood stasis in hyperlipidemia and atherosclerosis. Journal of Traditional Chinese Medicine. 2012;32(4):578–583. doi: 10.1016/S0254-6272(13)60074-6. [DOI] [PubMed] [Google Scholar]
- 17.Lu C., Deng J., Li L., et al. Application of metabolomics on diagnosis and treatment of patients with psoriasis in traditional Chinese medicine. Magnetic Resonance in Chemistry. 2013;51:549–556. doi: 10.1016/j.bbapap.2013.05.019. [DOI] [PubMed] [Google Scholar]
- 18.Jian W.-X., Yuan Z.-K., Huang X.-P. Detection and analysis on plasma metabolomics in patient with coronary heart disease of Xin-blood stasis syndrome pattern. Zhongguo Zhong Xi Yi Jie He Za Zhi. 2010;30(6):579–584. [PubMed] [Google Scholar]
- 19.Zhao H., Chen J., Shi Q., et al. Metabolomics-based study of clinical and animal plasma samples in coronary heart disease with blood stasis syndrome. Evidence-based Complementary and Alternative Medicine. 2012;2012:6. doi: 10.1155/2012/638723.638723 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Park T.-Y., Lee J. A., Cha M. H., et al. The fundamental study for the standardization and objectification of pattern identification in traditional Korean medicine for stroke (SOPI-Stroke): an overview of phase I. European Journal of Integrative Medicine. 2012;4(2):e125–e131. doi: 10.1016/j.eujim.2012.01.003. [DOI] [Google Scholar]
- 21.Go H. Y., Kim Y. K., Kang B. K., et al. Report on the Korean standard differentiation of the symptoms and signs for the stroke-2. Korean Journal of Oriental Physiology Pathology. 2006;20:1789–1791. [Google Scholar]
- 22.Bensky D., Gamble A. Chinese Herbal Medicine Materia Medica. 2004. [Google Scholar]
- 23.Walsh S., King E. Pulse Diagnosis: A Clinical Guide. Shanghai, China: Elsevier; 2008. [Google Scholar]
- 24.Lee J. S., Kim S. Y., Kang B. K., et al. A review of static blood pattern in stroke pattern diagnosis. Korean Journal of Integrative Medicine. 2009;30:813–820. [Google Scholar]
- 25.Maciocia G. Tongue Diagnosis in Chinese Medicine. Seattle, Wash, USA: Eastland; 1995. [Google Scholar]
- 26.Kirschbaum B. Atlas of Chinese Tongue Diagnosis. Seattle, DC, USA: Eastland; 2000. [Google Scholar]
- 27.Rui-Juan X. Microcirculation and traditional chinese medicine. The Journal of the American Medical Association. 1988;260(12):1755–1757. doi: 10.1001/jama.1988.03410120101035. [DOI] [PubMed] [Google Scholar]
- 28.Liu Y., Yin H.-J., Shi D.-Z., Chen K.-J. Chinese herb and formulas for promoting blood circulation and removing blood stasis and antiplatelet therapies. Evidence-based Complementary and Alternative Medicine. 2012;2012:8. doi: 10.1155/2012/184503.184503 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Park S. C., Lee S. C., Park Y. B. Correlation model between questionnaires of oriental medicine and bio-signal. The Journal of the Korea Contents Association. 2013;13(3):299–306. doi: 10.5392/JKCA.2013.13.03.299. [DOI] [Google Scholar]
- 30.Gao Z.-Y., Xu H., Shi D.-Z., Wen C., Liu B.-Y. Analysis on outcome of 5284 patients with coronary artery disease: the role of integrative medicine. Journal of Ethnopharmacology. 2012;141(2):578–583. doi: 10.1016/j.jep.2011.08.071. [DOI] [PubMed] [Google Scholar]
- 31.Wang Y., Li C., Guo S., et al. Serum metabolomics of blood-stasis syndrome of chronic myocardial ischemia based on CPMG pulse sequence. Journal of Beijing University of Traditional Chinese Medicine. 2012;20122011-12 [Google Scholar]
- 32.Yamada K. A., McHowat J., Yan G. X., et al. Cellular uncoupling induced by accumulation of long-chain acylcarnitine during ischemia. Circulation Research. 1994;74(1):83–95. doi: 10.1161/01.res.74.1.83. [DOI] [PubMed] [Google Scholar]
- 33.Opitz C. A., Litzenburger U. M., Sahm F., et al. An endogenous tumour-promoting ligand of the human aryl hydrocarbon receptor. Nature. 2011;478(7368):197–203. doi: 10.1038/nature10491. [DOI] [PubMed] [Google Scholar]
- 34.Wirleitner B., Rudzite V., Neurauter G., et al. Immune activation and degradation of tryptophan in coronary heart disease. European Journal of Clinical Investigation. 2003;33(7):550–554. doi: 10.1046/j.1365-2362.2003.01186.x. [DOI] [PubMed] [Google Scholar]
- 35.Ormstad H., Verkerk R., Amthor K.-F., Sandvik L. Activation of the kynurenine pathway in the acute phase of stroke and its role in fatigue and depression following stroke. Journal of Molecular Neuroscience. 2014;54:181–187. doi: 10.1007/s12031-014-0272-0. [DOI] [PubMed] [Google Scholar]
- 36.Pawlak K., Myśliwiec M., Pawlak D. Kynurenine pathway—a new link between endothelial dysfunction and carotid atherosclerosis in chronic kidney disease patients. Advances in Medical Sciences. 2010;55(2):196–203. doi: 10.2478/v10039-010-0015-6. [DOI] [PubMed] [Google Scholar]
- 37.Eleftheriadis T., Antoniadi G., Liakopoulos V., Stefanidis I., Galaktidou G. Plasma indoleamine 2,3-dioxygenase concentration is increased in hemodialysis patients and may contribute to the pathogenesis of coronary heart disease. Renal Failure. 2012;34(1):68–72. doi: 10.3109/0886022x.2011.623562. [DOI] [PubMed] [Google Scholar]
- 38.Sulo G., Vollset S. E., Nygård O., et al. Neopterin and kynurenine-tryptophan ratio as predictors of coronary events in older adults, the Hordaland Health Study. International Journal of Cardiology. 2013;168(2):1435–1440. doi: 10.1016/j.ijcard.2012.12.090. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Suppl. Table 1: Identification of plasma metabolites in stroke patients (Non-BS and BS) analyzed using ultra-performance liquid chromatography-quadrupole time-of-flight mass spectrometry.