Abstract
Background
Retroperitoneal lymphoceles (RPLs) caused by injury to the lymphatics are commonly seen after pelvic lymphadenectomy, renal transplantation, and gynecologic tumor resection surgeries. Degenerative disc disease still remains the major cause of low-back pain. Anterior lumbar spinal procedures, such as anterior lumbar interbody fusion and anterior lumbar arthroplasty, have been increasingly performed for treatment of axial back pain. RPLs, as an approach-related complication, though infrequent, have been reported after anterior lumbar spinal surgery. We report a case of RPL after total disc replacement of the lumbar spine. To our knowledge, there has been no prior report of RPL after total disc replacement managed by percutaneous aspiration only.
Methods
A 49-year-old woman who underwent total disc replacement at the L4-5 level presented with a postoperative complication of RPL. The imaging findings, clinical course, and treatment are discussed, and a review of literature is presented.
Results
The patient presented with significant abdominal swelling and discomfort at 4 weeks after surgery without any signs or symptoms of infection. Investigations showed an RPL. She was treated by multiple aspirations under ultrasound guidance. At 12 months’ follow-up, the patient had no further abdominal symptoms and had gone back to her routine activities and work with significant improvement in back pain.
Conclusions
RPL is an uncommon complication after anterior lumbar surgery and can be managed effectively if detected and diagnosed early. Although repeated aspiration is associated with high recurrence and infection, it is a safe and minimally invasive procedure to manage RPL.
Keywords: Retroperitoneal lymphocele, Degenerative disc disease, Anterior lumbar spine surgery, Total disc replacement
Leakage of lymph can occur as a result of traumatic or iatrogenic injury to the lymphatic vessels, leading to formation of a lymphocele, which is cystic in nature with a thick fibrotic wall lacking epithelial lining.1 Postoperative lymphoceles have been reported to occur after genitourinary including kidney transplantation and vascular, gynecologic tumor resection, pelvic lymphadenectomy, and thoracic surgery. 1–3 Approach-related complications after anterior lumbar spine surgery can theoretically cause a lymphocele because similar structures are dissected; however, postoperative retroperitoneal lymphoceles (RPLs) after anterior spinal surgery are infrequent and present difficulties in diagnosis and management. 4 We report a case of RPL after total disc replacement of the L4-5 disc managed by multiple aspirations. A review of the literature regarding the various management options for postoperative RPL is detailed.
Case report
A 49-year-old, female, former elite sports participant presented with worsening low-back pain radiating to the left lower limb of 4 years’ duration. Informed consent was provided by the patient for publication. The back pain was constant, rated at 7 of 10 on a visual analog scale; was aggravated by prolonged sitting and standing; and resulted in moderate restriction of activities of daily living. The leg pain ranged from 2 to 7 out of 10. Examination showed overall excellent balance with some loss of lumbar lordosis and well-preserved spinal column movements with no nerve root stretch signs or neurologic deficit. The patient responded to neither facet injections (bilateral L4-5 and L5-S1) nor pain medications.
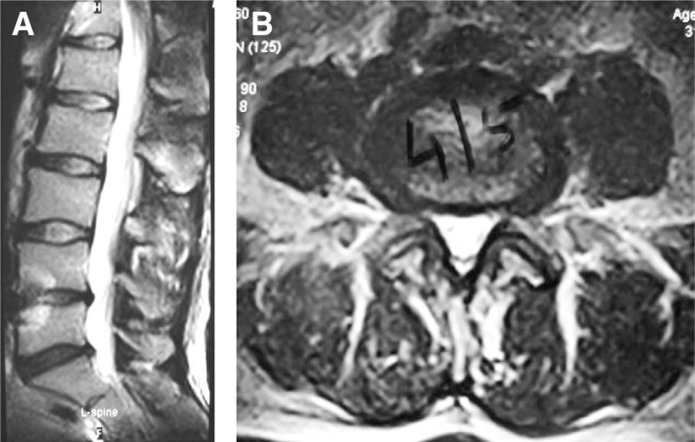
Radiographs of the lumbar spine showed focal kyphosis and reduced disc space at L4-5. Progressive magnetic resonance imaging scans over a period of 4 years showed advancing degenerative disc disease at L4-5 with loss of disc height (Fig. 1). A 3-phase technetium bone scan showed active disco-vertebral arthritis at the L4-5 level without evidence of active facet joint arthritis.
Fig. 1.
(A, B) Midsagittal and axial magnetic resonance images of lumbar spine showing degeneration and reduction in disc height at L4-5.
The patient underwent an L4-5 total disc replacement (ProDisc-L; Synthes, West Chester, Pennsylvania) in November 2008, through a left anterior retroperitoneal approach via a longitudinal median skin incision. The disc was exposed by retracting the inferior vena cava and descending aorta to the right side. The disc was excised after 2 annular flaps were created. An appropriate-sized prosthesis (large × 6° × 10 mm) was inserted under fluoroscopic monitoring. The perioperative period was uneventful, and ambulation commenced the day after surgery. The patient reported significant improvement in symptoms and was discharged home on the fifth postoperative day.
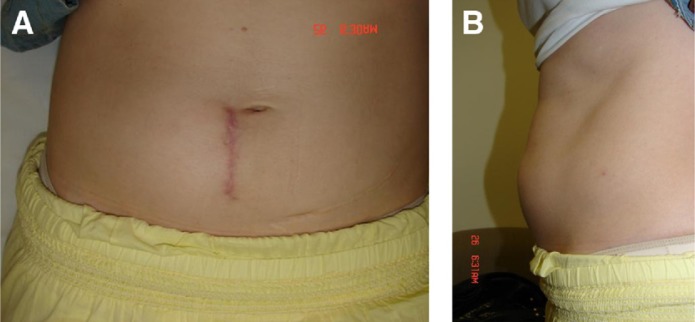
Two weeks after surgery, the patient presented with a soft, nontender left-sided abdominal swelling without evidence of inflammation. By the end of 4 weeks, the swelling had increased in size, become firm, remained nontender, and did not disappear on lying down or produce impulse on coughing. There was no abdominal pain, headache, fever, constitutional symptoms, or aggravation of back or leg pain (Fig. 2).
Fig. 2.
(A, B) Clinical picture of patient at 4 weeks after surgery showing abdominal swelling along with right paramedian scar.
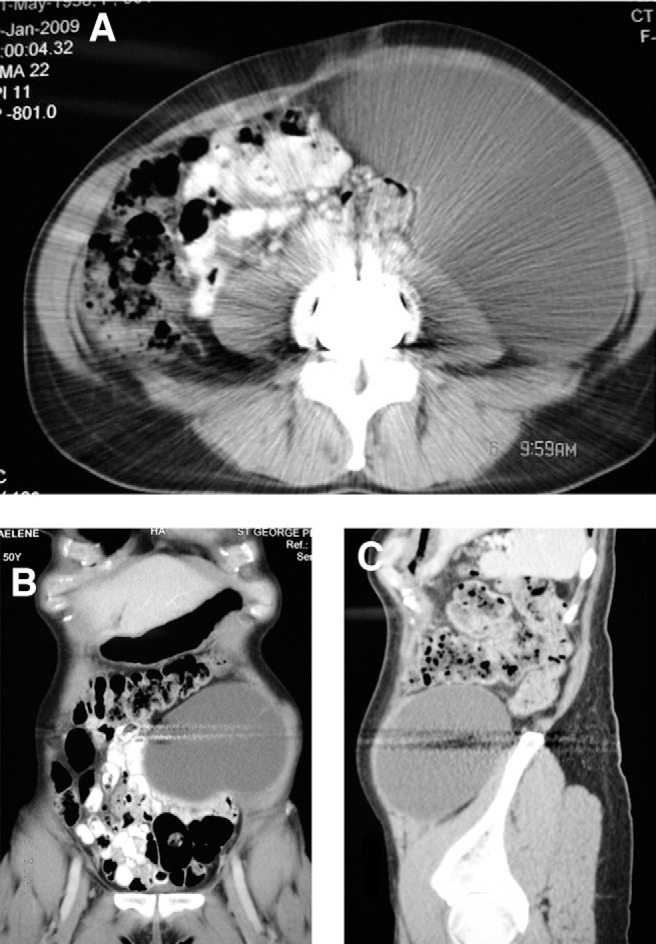
An abdominal computed tomography (CT) scan showed a large retroperitoneal collection measuring 18 × 10 × 13 cm and extending into the left rectus abdominis muscle. Straw-colored fluid (1100 mL), aspirated by use of ultrasound guidance, showed multiple leukocytes but no organisms on Gram stain and culture (Fig. 3).
Fig. 3.
(A–C) Axial, sagittal, and coronal CT images of abdomen at 4 weeks postoperatively showing significant collection of lymphatic fluid.
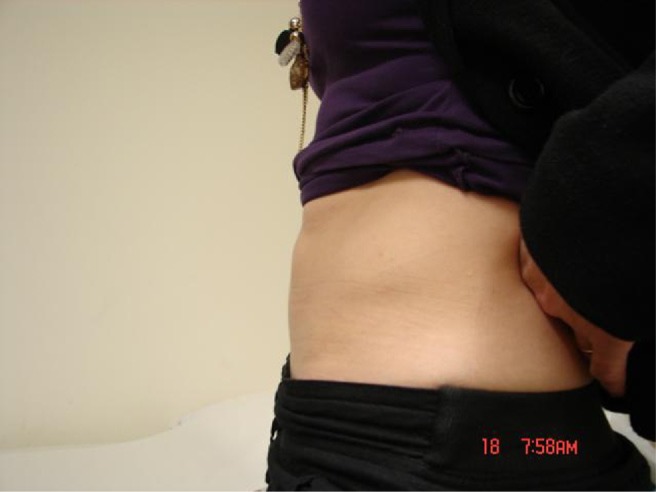
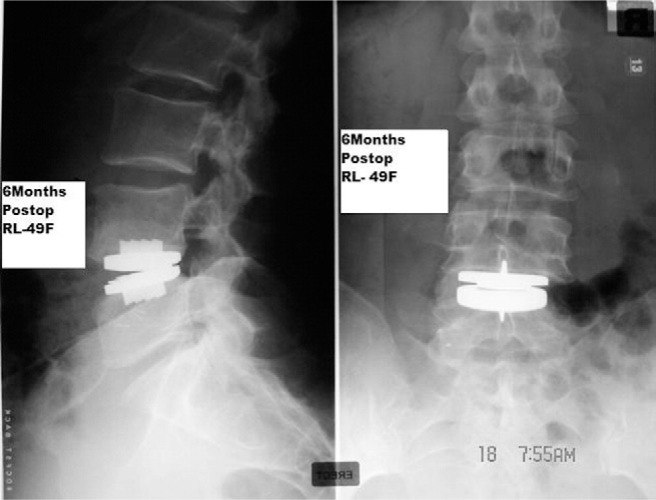
Three weeks later, 1050 mL of fluid was aspirated, which again showed a high leukocyte count with no organisms. Biochemical studies when compared with serum values showed the following: glucose, 2.9 mmol/L (normal range, 3.6–6.0 mmol/L); total protein, 27 g/L (normal range, 60–83 g/L); amylase, 41 U/L (normal range, 10–100 U/L); cholesterol, 0.9 mmol/L (normal range, 3.9 –5.5 mmol/L); and triglycerides, 0.1 mmol/L (normal range, 0.5–1.7 mmol/ L). The biochemistry and microbiology reports were suggestive of a lymphocele and ruled out urinoma and pyogenic abscess. Four weeks later, 675 mL of fluid was aspirated, after which there were no further recurrences. At 6 months’ follow-up, the abdominal swelling had completely resolved and X-rays were satisfactory (Figs. 4 and 5).
Fig. 4.
Clinical picture of patient at 6 months’ follow-up (after aspiration of lymphocele) showing complete resolution of abdominal swelling.
Fig. 5.
Anteroposterior and lateral views of radiograph of lumbosacral spine with prosthesis (ProDisc-L) 6 months postoperatively with maintenance of disc height and lordosis.
As of 1 year after surgery, the patient is working full time at her previous job and the score for back and leg pain on the visual analog scale is 0.
Discussion
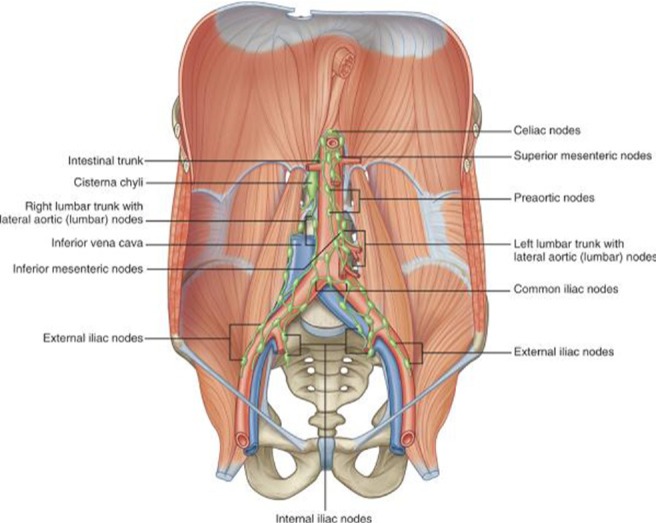
The common iliac lymphatic vessels, draining the lower extremities, pelvis, and genitalia, coalesce to form the ascending vertical lumbar lymphatic trunks, which run cephalad along the aorta and inferior vena cava in the retroperitoneum. The ascending vertical lumbar lymphatic trunks merge with lymphatics draining abdominal and retroperitoneal viscera to form the cisterna chyli, which lies posteromedial to the aorta and anterior to the L1-2 vertebra (Fig. 6). Lymph from the cisterna chyli flows through the thoracic duct to enter the venous circulation at the junction of the subclavian and left jugular veins. The flow of lymph is usually cranial and from right to left.5, 6
Fig. 6.
Anatomy of retroperitoneal lymphatics. (Reprinted with permission from Drake RL, Vogl W, Mitchell AWM. Gray's anatomy for students. Philadelphia: Elsevier; 2005. p. 335).
Lymph drainage below the cisterna chyli is serous, straw colored, and low in triglyceride and high in protein content, in comparison to creamy white and rich in chylomicron lymph (from gastrointestinal lymphatics) cephalad to the cisterna chyli.1, 7, 8 Biochemically, the lymph has a specific gravity greater than 1.012, high triglyceride content, greater than 3% protein, and high leukocyte (lymphocytes) count, and it stains positive with Sudan III reagent.
Low concentrations of clotting factors and absence of platelets prevent lymph from clotting, whereas a lack of smooth muscles in the vessel wall precludes vasoconstriction, allowing lymphatics to remain patent for up to 48 hours after injury. Lymphoceles rarely occur after intraperitoneal surgery, because the peritoneum absorbs the lymph. The reduced lymphatic flow (<1 mL/min), especially in fasting surgical patients, and presence of numerous collateral lymphatic vessels rerouting the fluid from the surgical site obscure intraoperative diagnosis of leakage but allow rapid healing of vessels.
The incidence of approach-related complications (vascular injury, retrograde ejaculation, ureteral injury, and peritoneal and visceral injury) after anterior lumbar surgery is 1% to 3%.9–12 Though uncommon after anterior lumbar surgery, postoperative RPL has been reported after genitourinary and pelvic lymphadenectomy surgeries.13 The incidence of postoperative RPL varies between 0.6% and 18% after renal transplant and between 0.7% and 27% after extraperitoneal lymphadenectomy.2, 3, 14 Lymphoceles typically develop a few weeks after surgery, but late occurrences up to 2 years after surgery have been reported.15 Small asymptomatic lymphoceles disappear spontaneously and do not require treatment. Persistent lymphoceles, with a thick fibrotic wall lacking epithelial lining, may compress the ureter (hydronephrosis), veins (limb edema), rectosigmoid (constipation), bladder (urinary frequency), and pelvic nerves (pain) and may become secondarily infected.2, 16
The differential diagnosis of RPL includes an abscess, ascites, urinoma due to ureteral injury, pancreatic injury with pseudocyst formation, and cerebrospinal fluid leak with pseudo-meningocele. A high index of suspicion, radiologic evaluation, and biochemical analysis of the fluid are necessary to diagnose lymphocele. Compared with urine, lymph is low in potassium and creatinine and high in sodium. Transaminase levels helps to rule out ascites due to chronic liver disease. Biochemical studies on the fluid aspirated from our patient were suggestive of lymph.1, 17
Ultrasound, CT, and magnetic resonance imaging scans are useful in identifying the presence, location, and size of the lymphocele. CT myelogram rules out occult cerebrospinal fluid leak, and intravenous pyelogram can identify ureteral injury leading to urinoma formation. Although contrast lymphangiography provides definitive evidence of lymphatic injury, the procedure is invasive, technically difficult, and fraught with complications such as localized skin necrosis, infection, lymphatic vessel damage, and pulmonary embolus.17, 18
A low-fat diet and bed rest decrease lymphatic flow and lead to closure of leaking vessels, especially in injury above the cisterna chyli. A high-protein diet replaces the lost protein, and rarely, total parenteral nutrition may be necessary to manage significant malnutrition.19
Persistent postoperative RPL may require aspiration and/or surgery. Aspiration provides fluid for diagnosis as well as relief of symptoms but is associated with a high incidence of recurrence (up to 80%–90%) and infection (25%–50%).20 Although catheter drainage with transcatheter sclerotherapy by use of ethanol, povidone-iodine, tetracycline and doxycycline, and fibrin glue is reported to be successful in 80% to 95% of cases (comparable to surgery), a recurrence rate of up to 25% has been reported in various series. Our patient underwent 3 percutaneous aspirations, after which there were no recurrences.1, 17, 21–23
Levi24 reported on a 38-year-old woman presenting with urinary incontinence and urgency 1 week after a posterioranterior- posterior L5-S1 spondylolisthesis reduction and fusion. She was diagnosed as having RPL and was managed with repeated drainage and sclerotherapy by use of povidone- iodine and doxycycline. Hanson and Mirkovic13 reported on a patient in whom left leg pain, scrotal pain, and serous wound drainage developed after an anterior L3-4 debridement and reconstruction for osteomyelitis and who underwent percutaneous catheter drainage after a lymphangiogram confirmed a lymphatic injury.
Surgical management of lymphoceles, reserved for recurrent cases, is in the form of open or laparoscopic marsupialization. Marsupialization involves intraperitoneal drainage of lymphocele through a peritoneal window allowing the large surface area and absorptive capacity of the peritoneum to resorb the fluid. Laparoscopic marsupialization, described by McCullough et al,25 is the preferred mode of surgical treatment.1, 3, 5, 17 Abou-Elela et al26 reported 9 symptomatic lymphoceles in 135 consecutive renal transplant patients with no recurrences after marsupialization (10 months’ follow-up), whereas Atray et al27 reported 36 symptomatic lymphoceles in 138 consecutive renal transplant patients with recurrence in 33% after drainage, 25% after sclerotherapy, and 12% after either open or laparoscopic marsupialization.
Pee et al28 reported an RPL after anterior lumbar interbody fusion at L4-5, which recurred after ultrasound-guided aspiration; it was eventually managed by open drainage through the previous retroperitoneal approach and application of lyophilized fibrin adhesive around the iliac vessels and prevertebral soft tissues. Wagner et al,29 in their series of 19 patients describing access strategies for revision or exploration of the Charité lumbar total disc replacement system (DePuy Spine, Raynham, Massachusetts), reported 1 case of RPL with hydronephrosis after 2-level total disc replacement at L4-5 and L5-S1 treated with laparoscopic marsupialization after failure of nonoperative strategies. Patel et al30 reported a case of RPL after combined anterior (L2-5) and posterior (T11-L5) fusion for degenerative scoliosis and stenosis. The patient presented with abdominal pain, nausea, and vomiting 6 weeks after surgery and was managed by laparoscopic marsupialization without further recurrence.
Conclusion
RPL is an uncommon complication after anterior lumbar surgery and can be managed if detected and diagnosed early. Although repeated aspiration is associated with high recurrence, it is a safe and minimally invasive strategy to manage RPL.
References
- 1.Karcaaltincaba M, Akhan O. Radiological imaging and percutaneous treatment of pelvic lymphocele. Eur J Radiol. 2005;55:340–54. doi: 10.1016/j.ejrad.2005.03.007. [DOI] [PubMed] [Google Scholar]
- 2.Dodd GD, Rutledge F, Wallace S. Postoperative pelvic lymphocysts. AJR Am J Roentgenol. 1970;108:312–23. doi: 10.2214/ajr.108.2.312. [DOI] [PubMed] [Google Scholar]
- 3.Glass LL, Cockett AT. Lymphoceles: diagnosis and management in urologic patients. Urology. 1998;51(Suppl 5A):135–40. doi: 10.1016/s0090-4295(98)00090-9. [DOI] [PubMed] [Google Scholar]
- 4.Gumbs AA, Shah RV, Yue JJ, et al. The open anterior paramedian retroperitoneal approach for spine procedures. Arch Surg. 2005;140:339–43. doi: 10.1001/archsurg.140.4.339. [DOI] [PubMed] [Google Scholar]
- 5.Leibovitch I, Mor Y, Golomb J, et al. The diagnosis and management of postoperative chylous ascites. J Urol. 2002;167:449–57. doi: 10.1016/S0022-5347(01)69064-5. [DOI] [PubMed] [Google Scholar]
- 6.Anderson JK, Kabalin JN, Cadeddu JA. Surgical anatomy of retroperitoneum. In: Wein AJ, Kavoussi LR, Novick AC, Partin AW, Peters CA, editors. Campbell-Walsh urology. 9th ed. Philadelphia: Saunders; 2007. pp. 3–19. [Google Scholar]
- 7.Miller T, Stewart L. The lymphatic system. In: Wilson SE, Veith FJ, editors. Vascular surgery—principles and practice. New York: McGraw-Hill; 1987. pp. 801–10. [Google Scholar]
- 8.Prost-Proctor SL, Rinsky LA, Bleck EE. The cisterna chyli in orthopaedic surgery. Spine (Phila Pa 1976) 1983;8:787–92. doi: 10.1097/00007632-198310000-00017. [DOI] [PubMed] [Google Scholar]
- 9.Brau SA, Delamarter RB, Schiffman ML, et al. Vascular injury during anterior lumbar surgery. Spine (Phila Pa 1976) 2004;4:409–12. doi: 10.1016/j.spinee.2003.12.003. [DOI] [PubMed] [Google Scholar]
- 10.Escobar E, Transfeldt E, Garvey T, et al. Video-assisted versus open anterior lumbar spine fusion surgery: a comparison of four techniques and complications in 135 patients. Spine (Phila Pa 1976) 2003;28:729–32. doi: 10.1097/01.BRS.0000051912.04345.96. [DOI] [PubMed] [Google Scholar]
- 11.Sasso RC, Best NM, Mummaneni PV, et al. Analysis of operative complications in a series of 471 anterior lumbar interbody fusion procedures. Spine (Phila Pa 1976) 2005;30:670–4. doi: 10.1097/01.brs.0000155423.18218.75. [DOI] [PubMed] [Google Scholar]
- 12.Tiusanen H, Seitsalo S, Osterman K, et al. Retrograde ejaculation after anterior interbody lumbar fusion. Eur Spine J. 1995;4:339–42. doi: 10.1007/BF00300293. [DOI] [PubMed] [Google Scholar]
- 13.Hanson D, Mirkovic S. Lymphatic drainage after lumbar surgery. Spine (Phila Pa 1976) 1998;23:956–8. doi: 10.1097/00007632-199804150-00023. [DOI] [PubMed] [Google Scholar]
- 14.Conte M, Panici PB, Guariglia L, et al. Pelvic lymphocele following radical paraaortic and pelvic lymphadenectomy for cervical carcinoma: incidence rate and percutaneous management. Obstet Gynecol. 1990;76:268–71. [PubMed] [Google Scholar]
- 15.Greenberg BM, Perloff LJ, Grossman RA, Naji A, Barker CF. Treatment of lymphocele in renal allograft recipients. Arch Surg. 1985;120:501–4. doi: 10.1001/archsurg.1985.01390280087019. [DOI] [PubMed] [Google Scholar]
- 16.Braun WE, Banowsky LH, Straffon RA. Lymphoceles associated with renal transplantation: report of 15 cases and review of the literature. Am J Med. 1974;57:714–29. doi: 10.1016/0002-9343(74)90845-6. [DOI] [PubMed] [Google Scholar]
- 17.Hamza A, Fischer K, Koch E, et al. Diagnostics and therapy of lymphoceles after kidney transplantation. Transplant Proc. 2006;38:701–6. doi: 10.1016/j.transproceed.2006.01.065. [DOI] [PubMed] [Google Scholar]
- 18.Kinmonth JB. Lymphangiography in man: a method of outlining lymphatic trunks at operation. Clin Sci. 1952;11:13–20. [PubMed] [Google Scholar]
- 19.Hart AK, Greinwald JH, Jr, Shaffrey CI, et al. Thoracic duct injury during anterior cervical discectomy: a rare complication. Case report. J Neurosurg. 1998;88:151–4. doi: 10.3171/jns.1998.88.1.0151. [DOI] [PubMed] [Google Scholar]
- 20.White M, Mueller PR, Ferrucci JT, et al. Percutaneous drainage of postoperative abdominal and pelvic lymphoceles. AJR Am J Roentgenol. 1985;145:1065–9. doi: 10.2214/ajr.145.5.1065. [DOI] [PubMed] [Google Scholar]
- 21.Kim JK, Jeong YY, Kim YH, et al. Postoperative pelvic lymphocele: treatment with simple percutaneous catheter drainage. Radiology. 1999;212:390–4. doi: 10.1148/radiology.212.2.r99au12390. [DOI] [PubMed] [Google Scholar]
- 22.Chin A, Ragavendra N, Hilborne L, et al. Fibrin sealant sclerotherapy for treatment of lymphoceles following renal transplantation. J Urol. 2003;170(Pt 1):380–3. doi: 10.1097/01.ju.0000074940.12565.80. [DOI] [PubMed] [Google Scholar]
- 23.Gilliland JD, Spies JB, Brown SB, et al. Lymphoceles: percutaneous treatment with povidone-iodine sclerosis. Radiology. 1989;171:227–9. doi: 10.1148/radiology.171.1.2648473. [DOI] [PubMed] [Google Scholar]
- 24.Levi AD. Treatment of a retroperitoneal lymphocele after lumbar fusion surgery with intralesional povidone iodine: technical case report. Neurosurgery. 1999;45:658–60. doi: 10.1097/00006123-199909000-00047. [DOI] [PubMed] [Google Scholar]
- 25.McCullough CS, Soper NJ, Clayman RV, et al. Laparoscopic drainage of a post transplant lymphocele. Transplantation. 1991;51:725–7. doi: 10.1097/00007890-199103000-00034. [DOI] [PubMed] [Google Scholar]
- 26.Abou-Elela A, Reyad I, Torky M, et al. Laparoscopic marsupialization of postrenal transplantation lymphoceles. J Endourol. 2006;20:904–9. doi: 10.1089/end.2006.20.904. [DOI] [PubMed] [Google Scholar]
- 27.Atray NK, Moore F, Zaman F, et al. Post transplant lymphocele: a single centre experience. Clin Transplant. 2004;18(Suppl 12):46–9. doi: 10.1111/j.1399-0012.2004.00217.x. [DOI] [PubMed] [Google Scholar]
- 28.Pee YH, Kim KJ, Choi YG, et al. Lymphocele formation after anterior lumbar interbody fusion at L4-5. J Neurosurg Spine. 2007;7:566–70. doi: 10.3171/SPI-07/11/566. [DOI] [PubMed] [Google Scholar]
- 29.Wagner WH, Regan JJ, Leary SP, et al. Access strategies for revision or explanation of the Charite lumber disc replacement. J Vasc Surg. 2006;44:1266–72. doi: 10.1016/j.jvs.2006.07.046. [DOI] [PubMed] [Google Scholar]
- 30.Patel AA, Spiker WR, Daubs MD, et al. Retroperitoneal lymphocele after anterior spinal surgery. Spine (Phila Pa 1976) 2008;33:E648–52. doi: 10.1097/BRS.0b013e31817c6ced. [DOI] [PubMed] [Google Scholar]