Abstract
Every 34 seconds an American experiences a myocardial infarction or cardiac death. Approximately 80% of these coronary artery disease (CAD)-related deaths are attributable to modifiable behaviors, such as a lack of physical exercise training (ET). Regular ET decreases CAD morbidity and mortality through systemic and cardiac-specific adaptations. ET increases myocardial oxygen demand acting as a stimulus to increase coronary blood flow and thus myocardial oxygen supply, which reduces myocardial infarction and angina. ET augments coronary blood flow through direct actions on the vasculature that improve endothelial and coronary smooth muscle function, enhancing coronary vasodilation. Additionally, ET promotes collateralization, thereby, increasing blood flow to ischemic myocardium and also treats macrovascular CAD by attenuating the progression of coronary atherosclerosis and restenosis, potentially through stabilization of atherosclerotic lesions. In summary, ET can be used as a relatively safe and inexpensive way to prevent and treat CAD.
Keywords: exercise, coronary blood flow, coronary artery disease
Introduction
The main purpose of this review in the series is to provide an overview of the effects of chronic exercise training (ET) on the adaptations of the coronary circulation, primarily in the setting of coronary artery disease (CAD), and potential cellular and molecular mechanisms for the adaptations.
Globally, more people die from cardiovascular (CV) diseases (CVD) than any other cause 1, with approximately 40% of these deaths attributable to CAD 2. One of the main pathological processes leading to CAD-related morbidity is atherosclerosis, Which is a lifelong process, which begins in childhood as fatty lesions and can later progress into flow-limiting stenosis of large epicardial coronary arteries, ultimately manifesting as angina and/or myocardial infarction (MI) 2;3. About 80% of CAD and cerebrovascular disease-related morbidities and mortalities are caused by modifiable behaviors such as physical inactivity, unhealthy diets, tobacco use, and alcohol abuse 2. The large number of CAD-related morbidities and mortalities caused by modifiable behaviors suggests that the incidence of CAD can be significantly reduced through a number of interventions. In particular, increasing regular physical activity (PA) which is widely accessible and relatively inexpensive could significantly reduce the number of deaths related to CVD.
Current CAD therapies and recommended PA guidelines
The current goals of the guideline directed medical therapies for patients with CAD are 1) prevention of premature CVD death and complications from CAD, 2) maintaining or restoring a quality of life (QoL) that is acceptable for the patient by reducing angina and improving activities of daily living, and 3) minimizing adverse effects of treatments for CAD 4. In the United States, the current pharmacological therapies for CAD mainly include β-blockers, inhibitors of the renin-angiotensin-aldosterone system, lipid-lowering, antiplatelet agents, nitrates, and calcium channel blockers, which treat hypertension, hypercholesterolemia, and angina. ET has similar therapeutic effects, as will be discussed later in this review. Despite recent advances in these pharmacological agents, there still remains a large incidence of CAD in optimally treated patients, especially those with multiple comorbidities. In addition to pharmacotherapy, lifestyle modifications like increasing chronic PA, eating a healthy diet, maintaining a healthy weight, cessation of smoking, and stress management are part of the guideline directed medical therapy for patients with CAD. If the patient is at a high risk for a CAD event, revascularization procedures, such as percutaneous coronary intervention (PCI) or coronary artery bypass graft surgery, are also highly effective 5.
The current recommendation for PA in stable CAD patients is 30 to 60 minutes of moderate intensity aerobic PA at least 5 days of the week, to elicit a heart rate of 60–85% of maximum 4. The duration, frequency, and intensity of aerobic ET are the key variables in the “dose” of ET that is optimally therapeutic. Highly controlled modes of PA include brisk walking, running, cycling, or swimming that involves large muscle groups which can increase peak aerobic capacity and are inversely related to all-cause mortality in patients with CAD 6. The goal of the current review is to discuss how increasing chronic PA improves coronary blood flow (CBF) and, by implication, CAD morbidity and mortality.
Under-prescription of ET
Compliance and access to the necessary resources are often barriers to implementing healthy lifestyle changes in CAD patients. However, it is likely that lifestyle modifications like exercise are “under-prescribed” to CAD patients by physicians 7{34237}. It has been estimated that 20–30% of eligible patients receive referrals to cardiac rehabilitation (CR)/ET programs 7,8;9{34303}. Of the referred patients, only approximately 40% actually participate in the programs, with women less likely to be referred or participate in the programs, especially if they are elderly or non-white 7;10{34303}. This is alarming because the least fit, inactive individuals comprise the high risk cohort that receives the most benefit from improved cardiorespiratory fitness (CRF) 7{34301}{34302}{34237}.
Systemic benefits of ET
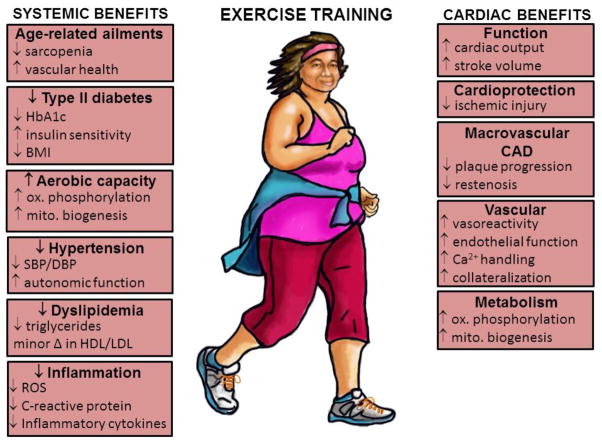
Habitual PA (ET) has been shown to reduce the risk of sudden cardiac death and acute MI 11. Increasing PA is a critically important modifiable behavior that can reduce the relative risk of CAD events because of its systemic benefits (Figure 1), which rival or exceed those achieved with pharmacological interventions and coronary revascularization for secondary treatment of CAD-related events 7. Other systemic benefits of regular endurance ET—including improving CRF, muscle strength, and mood—could enhance even a healthy individual’s QoL. Conversely, being physically inactive is an independent risk factor for CAD 12;13, and it recently has been reported that sitting for 6–7 hours per day negates the benefits of one hour of moderate-intensity ET per day 14. Being physically active also propagates other positive lifestyle modifications such as maintaining a healthy weight and stress management 15. In turn, these positive lifestyle modifications help reduce CAD risk factors such as high blood pressure, high cholesterol, diabetes, and obesity. To date, there has not been any type of pharmacological intervention, i.e. “polypill,” that has provided the benefits to as many organ systems as regular PA.
Figure 1.
Long-term benefits of regular endurance exercise on systemic risk factors and the coronary circulation. HbA1c, glycated hemoglobin; BMI, body mass index; SBP/DBP, systolic/diastolic blood pressure; HDL, high-density lipoprotein; LDL, low-density lipoprotein; ROS, reactive oxygen species; CAD, coronary artery disease.
Cardiac-specific benefits of ET
One of the major CV-specific benefits (Figure 1) is reduction of angina through enhanced oxygen delivery to the myocardium. Macroscopic atherosclerosis is an obstructive CAD that attenuates myocardial oxygen delivery despite increased myocardial demand, in times when heart rate and/or systolic blood pressure are elevated, which often occurs during physical work and/or with disease progression. When myocardial oxygen delivery does not meet the needs of myocardial demand cellular anaerobic pathways are activated, evoking symptoms of angina 7. When CBF to the myocardium is enhanced through ET, the resulting reduction in angina can increase the patient’s ability to perform activities of daily living and thus improve their QoL.
Regulation of CBF
To fully understand the significance of augmented CBF after ET, it is important to understand how CBF is regulated. Regulation of CBF has been reviewed thoroughly in outstanding monographs 16–20, so we only briefly review regulation of left ventricular CBF in the healthy state here. Adequate CBF is essential because it maintains oxygen and substrate delivery to the myocardium, which in turn uses this energy for contraction to produce enough pressure to pump blood and nutrients to the rest of the body. The heart is unique in that it must generate enough pressure to supply its own blood, which increases myocardial metabolism and need for oxygen delivery 20. The heart continuously generates pressure to pump blood by precisely matching myocardial oxygen supply with myocardial demand through efficient oxygen delivery to the myocardium. The efficient delivery of oxygen is enabled by high myocardial capillary density of ~3,000–4,000 capillaries per mm2 of myocardium 16;20. When metabolic demands are lowest in the myocardium (under “resting” conditions with a heart rate about 60 beats per minute), oxygen consumption is about (~50–100·l O2/min/g) 16;21 with about ~70–80% of the delivered oxygen extracted from the blood 17;20;21. The limited myocardial oxygen extraction reserve renders the heart predominantly dependent on CBF for continuous oxygen and nutrient delivery 16;22, highlighting the significance of improved CBF after exercise training in CAD patients.
CBF is directly proportional to the arterial pressure gradient across the coronary vasculature and inversely proportional to coronary vascular resistance (Ohm’s Law). Poiseuille’s equation relates blood flow to vascular resistance, where: , flow (F) is determined by the arterial pressure gradient (ΔP), vessel radius (r4), vessel length (l), and blood viscosity (η). Out of this equation, the coronary artery radius is the main determinant of coronary vascular resistance. Coronary vascular resistance is tightly regulated to myocardial metabolism because the heart has a limited capacity for anaerobic metabolism. CBF is directly related to the underlying level of myocardial oxygen consumption over a wide range of perfusion pressures 5 as long as CBF is not limited, which occurs in CAD patients who have significant arterial stenosis or severe microvascular dysfunction.
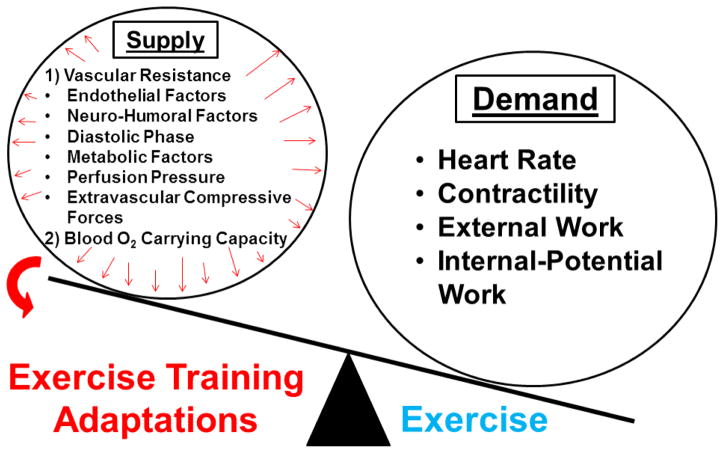
The major determinant of myocardial oxygen supply is vascular resistance, which is influenced by 1) extravascular compressive forces (tissue pressure); 2) diastolic time fraction of the cardiac cycle; 3) coronary perfusion pressure; 4) myocardial metabolism (local metabolic factors); 5) endothelial-derived substances; and 6) neuro-humoral influences. Conversely, the factors driving myocardial demand are 1) heart rate, 2) contractility, 3) ventricular wall tension, and 4) cardiac muscle shortening 17,16. Quantification of the relative contribution of these individual variables to myocardial demand is challenging in vivo because modulation of one of these variables often results in changes in one or more of the other variables 17. In addition, the regulation of CBF in vivo is complicated by mechanical and other factors that modify myocardial oxygen supply and demand. Precise coronary hemodynamic measures described by Hambrecht in this review series have enabled evaluations of the effects of exercise training on CBF and some of the underlying determinants of CBF adaptations.
ET as a stimulus to augment CBF
Certainly, ET is the most important physiological stimulus for increasing myocardial oxygen demand 17, which in turn requires a compensatory increase in myocardial oxygen supply (Figure 2). Regularly participating in ET programs results in adaptations that enhance myocardial oxygen supply, which makes ET a practical therapeutic intervention for primary and secondary treatment of CAD 17.
Figure 2.
Exercise training acts increases the determinants of myocardial oxygen supply due to large increases in myocardial oxygen demand during exercise.
During exercise there are systemic hemodynamic adjustments that increase cardiac output to the exercising skeletal muscles. The magnitude of the hemodynamic adjustments depend on the ET 1) intensity, 2) type (resistance vs. endurance), and 3) amount of muscle mass recruited to perform work (whole body vs. limb ET). The higher the exercise intensity, the more the sympathetic nervous system will be activated. Sympathetic activation of the heart results in higher heart rate, greater contractility, and reduces the diastolic filling time, which increases myocardial oxygen demand while altering myocardial oxygen supply. Further, using more muscle mass during ET requires greater hemodynamic adjustments than ET involving small muscle groups, and whole-body ET may produce the greatest systemic benefits for CAD patients. Resistance and high intensity ET programs have been gaining attention for their beneficial effects on musculoskeletal health and weight loss; however, this review will focus on moderate intensity endurance ET because this has been studied to a greater extent in CAD patients.
Despite the challenges to endocardial oxygen supply during ET due to extravascular compressive forces, the heart is able to adequately perfuse the myocardium because of the ability of the coronary microvasculature to dilate in response to metabolic vasodilators in healthy individuals. During ET aortic pressure only slightly exceeds intramyocardial tissue pressure, which increases effective tissue pressure only by 20–30%. Therefore, the large rise in CBF observed during ET is predominantly due to lowering microvascular resistance 17. This ability to vasodilate the resistance vessels allows for about 40–50% of the total blood flow to occur in systole during strenuous exercise 16. Overall, the extravascular compressive forces do not seem to limit CBF in healthy individuals because coronary vasodilator reserve has been observed even during maximal ET 17. However, during pathological changes from CAD such as severe coronary stenosis of greater than ~70%, this gradient is minimized because of chronic compensatory vasodilation that occurs in the resistance vessels to maintain adequate blood flow to the myocardium. This chronic vasodilation reduces the coronary vasodilator reserve, which could result in extravascular compressive forces causing functional impairments in CBF in CAD 16;17.
Mediators of vascular resistance during ET
Total coronary vascular resistance is the sum of both passive (structural) and active (smooth muscle tone) components 17. As discussed above, the structural and mechanical effects of the beating heart affect myocardial tissue perfusion. At the cellular level, active smooth muscle tone is regulated by a balance of local vasodilator and vasoconstrictor factors 16;17;23, arising from neurohumoral influences 24,18;25, metabolic byproducts from the myocardium 26–31, and the endothelium 31–37. Determining which of the myriad of vasoactive substances are necessary for promoting CBF during exercise has been difficult because of the redundancies in vasomotor control and differences that occurs between animal species and throughout the aging spectrum 17. Hypothetically, any of the numerous molecular signals mediating vascular resistance may undergo adaptation to chronic exercise and provide a mechanistic explanation for the improved CBF in trained animals. We will return to some of these cellular and molecular signals after reviewing the overall coronary functional adaptations in humans and in animal models.
CV benefits of ET and implications for CAD
The “dose” of endurance exercise that will elicit the most benefits in CAD patients is not entirely clear, but it is likely to be specific to each individual based on their CRF level, response to ET, and disease progression. However, several studies suggest that the greatest functional adaptations take longer to occur (>1 year) and require frequent exercise bouts at higher relative intensities 38. This ideal “dose” of endurance ET is debatable—since there is the possibility that too much ET may have ill effects 39. However, the problem in westernized nations is not that CAD patients are over-exercising—rather; they are sedentary and not engaged in any regular PA at all! Therefore, the overall push should be to get the majority of CAD patients to make PA part of their daily routine.
ET reduces mortality in CAD patients
Possibly the most compelling reasons for CAD patients to engage in ET is that exercise-based CR programs have a 27% reduction in total mortality than patients receiving usual care 40;41. In addition, low levels of CRF are associated with an eightfold increase in CVD prevalence compared to their fit counterparts. Simply improving CRF reduces mortality risk by 44% 42 and there is a 52% lower age-adjusted risk of CVD-related mortality 42.
ET reduces multiple CVD risk factors
In addition to improving mortality, ET improves physical work capacity, QoL, and reduces CAD risk factors. Fiuza-Luces et al 43, performed meta-analyses of 1) randomized controlled trials of pharmacotherapies and 2) randomized controlled trials of exercise training alone on reduction of modifiable CAD risk factors. Overall, they found that ET is like a “polypill” in that it reduces more CVD risk factors than any single pharmacological agent alone, albeit probably more modestly. Specifically, ET had benefits on lipid profiles, blood pressure, diabetes, and body weight, making a strong argument that exercise benefits can overcome those of common drugs when one considers “that the ET polypill combines preventive, multi-systemic effects with little adverse consequences and at a lower cost” 43.
Putative mechanisms for these beneficial ET adaptations include increasing cardiac function, regression of coronary atherosclerosis, improving vasoreactivity, and collateralization. When discussing beneficial effects of ET it is important to know that some training adaptations take longer to manifest than others and some benefits may even be transient. The following chronic adaptations discussed below are adaptations that occur after >4 weeks of ET.
ET and coronary plaque burden
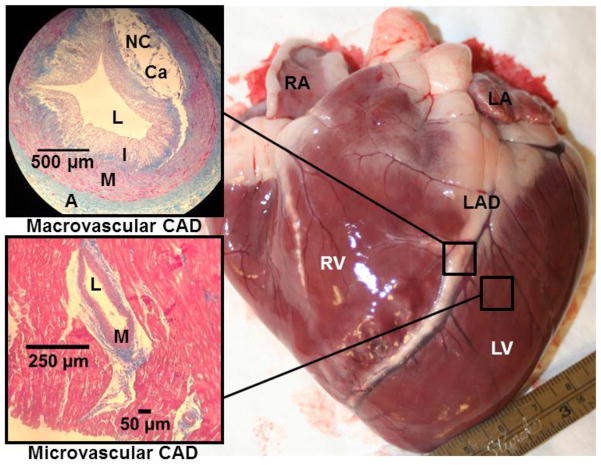
Several studies have examined the influence of ET on regression of atherosclerotic lesions (e.g. Figure 3) in combination with diet and lifestyle modifications. The Lifestyle Heart Trial demonstrated that CAD regression was possible and 5 years of an intensive diet and lifestyle change resulted in a 3.1% regression of relative coronary stenosis versus an 11.8% progression of the lesion in the standard care group, which equated to a 2.5-fold risk reduction in CVD events 44. In another study that examined 7 years of intense ET on patients with CAD, intense ET reduced electrocardiographic ST segment depression and increased rate pressure product during maximal exercise, suggesting improved myocardial oxygenation during ET45. Further, the latter participants had improved total cholesterol/high-density lipoprotein cholesterol (HDL-C) ratio due to significant increases in HDL-C 33. In the Heidelberg Regression Studies, CAD patients either underwent ET and a low-fat diet or were given usual CAD therapy, and had follow up angiographic studies to assess coronary stenosis progression with these interventions. 46,47 ET attenuated coronary stenosis progression observed via angiography versus the usual therapy (pre-statin era) and 47 improved work capacity, myocardial oxygen consumption, and psychological indexes of well-being, while reducing stress-induced myocardial ischemia 47. Hambrecht et al. further examined how leisure time energy expenditure, estimated from questionnaires, affected plaque progression. They found that luminal lesion size increased in the lowest level of leisure time calorie expenditure, while no change in CAD progression occurred in those who expended about 1400 kcal/week 46. Plaque regression only occurred in those participants who spent an average of 2,200 kcal/week (~5 to 6 hrs/week) in leisure time PA, suggesting that some of the macroscopic effects of ET may be dose-dependent. All of the latter long-term ET studies have examined angiographically determined luminal CAD progression. These studies suggest different ET “doses” may explain differences in plaque progression/regression between different training studies. Overall, these studies demonstrated only small changes in coronary lumen diameter over long periods of ET, suggesting that lesion regression may not be the mechanism responsible for improved mortality observed in exercising CAD patients.
Figure 3.
Porcine heart with advanced coronary artery disease (CAD), highlighting the differences in pathological changes that occur in conduit and resistance vessels. The photomicrographs on the left are Masson trichrome staining from a section of the left anterior descending (LAD) artery (top) and resistance arteries embedded within the left ventricle (bottom). Note the large atherosclerotic lesion that is encroaching the lumen (L) of the LAD (top) to form a flow-limiting stenosis. NC, necrotic core; Ca, calcification; I, neointima, M, media; A, adventitia; RA, right atrium; LA, left atrium; LV, left ventricle; RV, right ventricle.
Additional mechanisms that may improve mortality in exercising CAD patients are increased endothelial function and/or plaque stabilization. Long-term ET improved endothelial function in Type 2 diabetics with CAD, but failed to reduce plaque burden assessed by intravascular ultrasound (IVUS) 48. However, most acute coronary syndromes and sudden death have been hypothesized to stem from vulnerable lesions, which are characterized as a thin fibrous cap overlaying a large necrotic core 19. Madssen et al. examined plaque structure and morphology after two different 12 week aerobic ET protocols in CAD patients who were receiving optimal medical therapy (i.e. including statins) after intracoronary stent placement 49. Grayscale IVUS and radiofrequency IVUS (RF-IVUS) were used to assess plaque burden and estimate plaque composition, and ET significantly decreased the necrotic core volume of the plaques distal to stent placement, but only trended towards a reduction in plaque burden (p=0.06). The reduction in the necrotic core resulted in 16 “vulnerable” lesions to be reclassified into a “less vulnerable” category, with only 6 lesions progressing to a “more vulnerable” category 49, suggesting plaque stabilization. The impact of ET on plaque stabilization in vivo is limited by current technology because RF-IVUS can only estimate lesion composition. Devices that enable in vivo chemical resolution of collagen and lipid within the atherosclerotic plaque are required to assess how plaque composition changes from CAD interventions, with longer duration studies needed to relate lesion composition to altered morbidity and mortality 50. In addition, it is unlikely that future studies will be able to isolate the effect of ET because ET- patients must also receive the standard of care (i.e. statins), which can drastically affect plaque burden and composition.
Studies of ET in humans have been supported by controlled, mechanistic studies in animal models that have been able to separate the effects of ET versus that of lifestyle modifications and pharmacological therapies. Animal models of CAD in mice, rabbits, pigs, and nonhuman primates have found that ET reduces the progression or causes regression of atherosclerotic lesions 19. These studies have provided mechanistic information on changes in atherosclerosis lesion morphology and composition that are not possible to perform in humans. Kramsch et al 51 found that ET before the induction of atherosclerosis in primates improved lipid profiles by lowering trigylcerides and low-density lipoprotein cholesterol (LDL-C), while increasing HDL-C. Further, they found that ET suppressed lesion size (intimal thickening), lesion composition, and widened coronary artery luminal diameter 51. Conversely, Williams et al. found that wheel running in monkeys did not slow the progression of CAD lesion size as measured by angiography, but it still improved other markers of CV health and attenuated stress responses from the pituitary adrenocortical axis 52 Link et al. found that in swine the incidence of atherosclerotic regions was reduced with ET 53. In Ossabaw swine with metabolic syndrome, exercise improved LDL-C/HDL-C ratio and reduced the percent of atherosclerotic plaque wall coverage in non-stented regions of coronary arteries assessed by IVUS 54. This was also confirmed in Yucatan swine 55. In Yucatan swine with early stages of CAD development, ET does not seem to halt the progression of CAD 56, which was marked by an appearance of macrophage foam cells in large coronary arteries 56. Despite the lack of CAD regression in some swine models, ET has demonstrated improved vascular/endothelial function and an atheroprotective phenotype in a number of porcine models of CAD 19;57. Stabilization of atherosclerotic plaques may be more important than reducing luminal stenosis in predicting CAD mortality and future CVD events.
Interestingly, ET has a greater effect on atherosclerotic lesion progression/regression in CAD patients who have undergone PCI. The ETICA trial was a longitudinal study that examined the effects of supervised aerobic ET 3 times per week on functional capacity, QoL, and atherosclerotic plaque progression/regression in CAD patients who underwent PCI; 58 ET improved functional capacity and QoL versus the control group (patients who underwent PCI and only performed activities of daily living). In addition, ET attenuated plaque progression versus control (7.6% and 25% stenosis, respectively) which was measured by coronary angiography 58. In fact, ET was an independent predictor of CVD events and resulted in a lower hospital readmission rate than control, despite similar restenosis rates 58. However, the extent of restenosis was lower in the ET group versus control 58. This study was performed before lipid-lowering drugs like statins were routinely prescribed, making this study unique in that it could independently assess the effects of ET restenosis rate in CAD patients. A recent study that included patients on statin therapy examined the effects of ET on restenosis in patients with drug eluting stents who had a previous MI 59. They found that CR followed by 9 months of community based ET resulted in a reduction of stenosis assessed by angiography. Further, they found that ET resulted in fewer major adverse CVD events and death, but this was not significant which was likely due to the low number of participants enrolled in the study who experienced an event 59. These findings were also recapitulated in swine models of hyperlipidemia 60 and metabolic syndrome 54.
ET improves epicardial (conduit) vessel vasoreactivity
Endothelial function
Some of the most important ET-induced adaptations in the coronary epicardial arteries are increases in vessel diameter, improvements in endothelial function, and stabilization of vascular smooth muscle phenotype. In ET individuals coronary artery tone is greater at rest, so they have a greater conduit artery vasodilator capacity. This is largely due to restoration of endothelial function in the conduit arteries which are responsible for vasodilation in response to shear stress. The endothelium has a major role in regulating vascular smooth muscle tone through release of factors that elicit relaxation and contraction of the underlying vascular smooth muscle cells. Endothelial dysfunction occurs in pathophysiological states such as CAD when the endothelium favors production of vasoconstrictors. In healthy individuals, acetylcholine (Ach) elicits modest vasodilation in coronary conduit arteries by stimulating the endothelium to produce vasodilatory substances, whereas in CAD patients, Ach elicits vasoconstriction due to endothelial dysfunction; 61 ET elicits improvements in flow-mediated and Ach-induced vasodilation of large coronary arteries prior to changes in ventricular remodeling or reduction in heart rate after 7 to 10 days of ET in dogs. The vasodilation was reversed after nitric oxide (NO) synthase (NOS) inhibition suggesting this is largely mediated by NO 62. However, longer periods of ET did not affect the proximal endothelium dependent vasodilation suggesting that progressive outward remodeling of the epicardial arteries occurs so that shear stress levels are normalized, which has a large effect on NO production 19. In CAD patients with endothelial dysfunction, short term ET (4 weeks) on a cycle ergometer 6 times a day for at least 10 minutes attenuated Ach induced vasoconstriction. The landmark study of Hambrecht et al. showed that after 4 weeks of ET coronary vasoconstriction to Ach was attenuated, but was not restored to normal levels. Further, in these patients adenosine-induced vasodilation was restored. Taken together, they found that short term ET could begin to reverse endothelial dysfunction, but longer duration ET interventions may be necessary to fully restore endothelial function 63.
The NO contribution to endothelial function is significant because of its important vasodilatory and vasoprotective properties that help prevent pathological changes that occur during CAD. Specifically, NO protects the vascular wall by inhibiting platelet aggregation, expression of adhesion molecules and penetration of white blood cells in the intimal and medial layers of the artery, and oxidation of lipids. These effects of NO may attenuate atherosclerosis by protecting the vessel from injury and fatty lesion formation. 64 ET increases NO production and bioavailability by 1) increasing endothelial NOS (eNOS) enzyme quantity and activity, 2) increasing availability of eNOS substrate (L-arginine) and essential cofactors (tetrahydrobiopterin (BH4)), and 3) decreasing breakdown of NO by reactive oxygen species (ROS) 65. Shear stress is one of the main physiologic activators of eNOS mRNA and protein 65, providing a key link between NO production and ET that involves repeated bouts of increased shear stress. In coronary artery bypass graft surgery patients, ET resulted in a 2-fold higher eNOS expression and 4-fold higher eNOS phosphorylation, suggesting elevated eNOS protein content and activity 66 Twelve weeks of ET in CAD patients decreases the level of the eNOS substrate antagonist asymmetrical dimethylarginine (ADMA), thereby increasing L-arginine substrate availability for NO production 67. Finally, eNOS activity is dependent on its essential cofactor BH4 and ROS, and when BH4 levels are diminished or there is high ROS, the eNOS dimer is uncoupled and produces superoxide rather than functional NO. Shear stress promotes de novo BH4 synthesis 68 and in hypercholesterolemic adults, infusion of BH4 nearly restored exercise-induced reactive hyperemia 69 and CBF during Ach infusion; 70 ET has also been proposed to be an effective antioxidant therapy 71, resulting in decreases in pro-oxidative enzyme mRNA expression and activity (i.e. NADPH oxidase, angiotensin II receptor type I) and upregulation of antioxidant enzyme proteins (i.e. angiotensin II receptor type II) in internal mammary arteries of CAD patients who underwent ET 72. ET mobilizes endothelial progenitor cells (EPCs) which promote endothelial function. The mechanism for how EPCs improve endothelial function or elicit new vessel development (i.e. vasculogenesis) is not completely understood, but putative mechanisms have been reviewed 73.
Vascular smooth muscle function and phenotype
As previously discussed, ET elicits modest plaque regression, which is unlikely to account for the reduction in CVD events. There is growing evidence that ET may stabilize existing atherosclerotic plaques through phenotypic switching of coronary smooth muscle cells (CSM) 50. Healthy conduit coronary arteries are characterized by medial CSM cells in a differentiated contractile state with a single layer of healthy endothelium comprising the intima. In contrast to many animal species, human intima also contains resident CSM 74. The presence of CAD risk factors, contribute to the development of endothelial dysfunction, macrophage infiltration of the intima and the entrance of lipids and other factors into the artery, forming foam cells within the early lesion. As the atherosclerotic lesion grows, neointimal thickening occurs as de-differentiated, proliferative CSM migrate into the intima, and synthesis of extracellular matrix macromolecules is heightened 50. During CAD progression, phenotypic modulation of CSM involves further differentiation to a more osteogenic phenotype that contributes to vascular calcification 50. As more severe lesions develop, macrophages and CSM become apoptotic and accumulate in the central region of the plaque forming a lipid-laden, necrotic core. In later stages of CAD, eccentric lesions develop into atheromas and form flow-limiting stenosis (Figure 3). The complexity of the atherosclerotic plaque development depends on the magnitude of dyslipidemia, the number of other risk factors present, and the duration of exposure to systemic risk factors 50. Unstable, vulnerable plaques that are prone to rupture and thrombosis are characterized by a large lipid-laden core with a thin, fibrous cap and often contain cholesterol crystals and microvessels.
In CAD patients ET may help promote a healthy, functional contractile phenotype in CSM by altering the regulation of intracellular calcium (Ca2+) handling as reviewed previously 50. Phenotypic switching of CSM to or stabilization in a healthy contractile state may have modest effects on the volume of the atherosclerotic lesion, but may result in more stable, fibrous plaque that has a structured, differentiated CSM and collagen content.
ET and resistance vessels
Following ET, CAD patients exhibited less electrocardiographic ST segment depression at similar myocardial oxygen demands, suggesting improved blood supply to the ischemic subendocardium 38 through improved resistance vessel function. This can occur through improved vasodilator reserve in the resistance vessels or from collateral formation.
Chronic ET and CAD risk factors have heterogeneous effects on coronary conduit and resistance vessel structure and function, which make it important to consider the size of the vessels being examined. For example, Muller-Delp et al. examined the effect of aging and ET on resistance vessels structure and arterial stiffness in rats 75. They found that aging, a CAD risk factor, induced hypertrophic remodeling in the medial wall of coronary resistance arteries while simultaneously reducing arteriole stiffness versus younger controls 75, similar to what has been observed in porcine model of metabolic syndrome 76 and in isolated cutaneous resistance vessels from essential hypertensive humans 77. This change in arteriole stiffness is directionally opposite from reports in conduit vessels 75. Further, Muller-Delp et al. found that exercise training attenuated resistance vessel hypertrophic remodeling, increased vessel stiffness, and restored myogenic responsiveness to levels similar to young sedentary controls 75, suggesting that ET can improve distribution of coronary blood flow and oxygen delivery in the endocardial and epicardial microcirculation. These positive coronary resistance vessel adaptations to ET may induce more positive shear patterns, thus accounting for the positive effects that have been noted in coronary conduit vessel function after ET in humans.
Collateral Formation
A well-developed collateral circulation provides cardioprotective effects on myocardial perfusion, contractile function, reductions in infarct size, and electrocardiographic abnormalities of ischemic myocardium 78. Chronic ET can induce both arteriogenesis (enlargement and remodeling of pre-existing collateral arterioles) and ischemia-induced angiogenesis (growth and proliferation of capillaries), with arteriogenesis being more important for delivery of blood into collateral-dependent regions 78. Physical forces and ischemia are the two principle mechanisms that promote collateral vessel development 38 and may play a role in eliciting collateral development in exercising CAD patients.
Humans have sparse innate collateral vessels, and it may take severe coronary artery stenosis (>90% of normal) to elicit collateral growth and formation 38. Postmortem observations revealed better collateral vessel development in patients with longer duration of ischemic symptoms or more severe coronary arterial narrowing 38. However, both human and animal studies suggest that ET does not enhance native collateral blood flow in healthy hearts 19.
Additionally, ET yields discrepant findings on collateralization development in humans and animals with coronary occlusions; ET did not induce collateralization in angiographic studies in humans 79;80, but this method for assessing collateralization is not sensitive enough to detect the small intramyocardial collateral vessels (20–200 μm) that are recruited by exercise 65. In addition, angiographic studies often use nitrovasodilators in their patients undergoing coronary angiography, which may affect the assessment of collateralization 78. Studies using thallium scintigraphy to assess ischemic myocardial regions in CAD patients after ET have shown improved functional performance of the compromised myocardial regions 38;81–83, which could either be a sign of collateralization or improved coronary resistance vessel function. When ET was combined with heparin in angina patients there was an increase in collateralization that was detectable on angiography 84. These findings suggest that ET in conjunction with pharmacological stimulators may promote collateralization 38. Further human studies are needed to investigate how ET enhances coronary collateral development versus enhanced coronary collateral vasomotor responsiveness in CAD patients 78.
Conclusions
Aerobic ET elicits CV adaptations that can prevent or reverse the pathology of CAD without the use of pharmacological interventions. The enhanced CBF from ET helps prevent myocardial ischemia, reducing the incidence of MI and angina. These improvements in coronary blood flow are due to structural and functional changes in the conduit and resistance vessels of the coronary circulation, which promote vasodilation due to enhanced endothelial and smooth muscle function. Improvements in endothelial function are marked by increased NO-dependent vasodilation, a greater production of vasodilatory factors, and a reduction of ROS and pro-constrictor factors. CSM function is enhanced through improved Ca2+ handling, which may promote a stabilization of coronary atherosclerotic plaques. Similarly, macrovascular atherosclerotic lesion progression is attenuated and/or reversed. There is also evidence that ET promotes collateralization which can improve perfusion of ischemic myocardium. These significant cardiac improvements in CAD highlight the need for increased promotion of PA/ET in CAD patients. Further research is needed to fully understand the benefits of ET and to examine how optimal pharmacological treatment combined with aerobic ET affect CV health. The current evidence supports much more vigorous promotion of PA and ET throughout the health care system {34300}.
Abbreviation List
- Ach
Acetylcholine
- ADMA
Asymmetrical dimethylarginine
- BH4
Tetrahydrobiopterin
- CAD
Coronary artery disease
- CBF
Coronary blood flow
- CR
Cardiac rehabilitation
- CRF
Cardiorespiratory fitness
- CSM
Coronary smooth muscle cells
- CV
Cardiovascular
- CVD
Cardiovascular disease
- EPCs
Endothelial progenitor cells
- eNOS
Endothelial nitric oxide synthase
- ET
Exercise training
- HDL-C
High-density lipoprotein cholesterol
- IVUS
Intravascular ultrasound
- LDL-C
Low-density lipoprotein cholesterol
- MI
Myocardial infarction
- NO
Nitric oxide
- NOS
Nitric oxide synthase
- PCI
Percutaneous coronary intervention
- PA
Physical activity
- QoL
Quality of life
- RF-IVUS
Radiofrequency intravascular ultrasound
- ROS
Reactive oxygen species
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Reference List
- 1.World Health Organization (WHO) [Accessed 8/5/2014];Cardiovascular diseases (CVDs) Fact sheet No. 317. 2013 Mar; at http://www.who.int/mediacentre/factsheets/fs317/en/
- 2.World Health Organization (WHO) Global Atlas on cardiovascular disease prevention and control. Chapter 1. Geneva: 2011. pp. 1–31. [Google Scholar]
- 3.McGill HC, Jr, McMahan CA, Zieske AW, Tracy RE, Malcom GT, Herderick EE, et al. Association of coronary heart disease risk factors with microscopic qualities of coronary atherosclerosis in youth. Circulation. 2000;102(4):374–379. doi: 10.1161/01.cir.102.4.374. [DOI] [PubMed] [Google Scholar]
- 4.Qaseem A, Fihn SD, Dallas P, Williams S, Owens DK, Shekelle P. Management of stable ischemic heart disease: summary of a clinical practice guideline from the American College of Physicians/American College of Cardiology Foundation/American Heart Association/American Association for Thoracic Surgery/Preventive Cardiovascular Nurses Association/Society of Thoracic Surgeons. Ann Intern Med. 2012;157(10):735–743. doi: 10.7326/0003-4819-157-10-201211200-00011. [DOI] [PubMed] [Google Scholar]
- 5.Mosher P, Ross J, Jr, MCFATE PA, SHAW RF. Control of Coronary Blood Flow by an Autoregulatory Mechanism. Circ Res. 1964;14:250–259. doi: 10.1161/01.res.14.3.250. [DOI] [PubMed] [Google Scholar]
- 6.Keteyian SJ, Brawner CA, Savage PD, Ehrman JK, Schairer J, Divine G, et al. Peak aerobic capacity predicts prognosis in patients with coronary heart disease. Am Heart J. 2008;156(2):292–300. doi: 10.1016/j.ahj.2008.03.017. [DOI] [PubMed] [Google Scholar]
- 7.Boden WE, Franklin B, Berra K, Haskell WL, Calfas KJ, Zimmerman FH, et al. Exercise as a Therapeutic Intervention in Patients With Stable Ischemic Heart Disease: An Underfilled Prescription (narrative review) Am J Med. 2014 doi: 10.1016/j.amjmed.2014.05.007. [DOI] [PubMed] [Google Scholar]
- 8.Blackburn GG, Foody JM, Sprecher DL, Park E, Apperson-Hansen C, Pashkow FJ. Cardiac rehabilitation participation patterns in a large, tertiary care center: evidence for selection bias. J Cardiopulm Rehabil. 2000;20(3):189–195. doi: 10.1097/00008483-200005000-00007. [DOI] [PubMed] [Google Scholar]
- 9.Mazzini MJ, Stevens GR, Whalen D, Ozonoff A, Balady GJ. Effect of an American Heart Association Get With the Guidelines program-based clinical pathway on referral and enrollment into cardiac rehabilitation after acute myocardial infarction. Am J Cardiol. 2008;101(8):1084–1087. doi: 10.1016/j.amjcard.2007.11.063. [DOI] [PubMed] [Google Scholar]
- 10.Alpert JS. Exercise is just as important as your medication. Am J Med. 2014 doi: 10.1016/j.amjmed.2014.05.029. [DOI] [PubMed] [Google Scholar]
- 11.Petersen CB, Gronbaek M, Helge JW, Thygesen LC, Schnohr P, Tolstrup JS. Changes in physical activity in leisure time and the risk of myocardial infarction, ischemic heart disease, and all-cause mortality. Eur J Epidemiol. 2012;27(2):91–99. doi: 10.1007/s10654-012-9656-z. [DOI] [PubMed] [Google Scholar]
- 12.Morris JN, Heady JA, Raffle P, Roberts CG, Parks JW. Coronary heart disease and physical activity of work. Lancet. 1953;2:1053–1057. doi: 10.1016/s0140-6736(53)90665-5. [DOI] [PubMed] [Google Scholar]
- 13.Fletcher GF, Blair SN, Blumenthal J, Caspersen C, Chaitman B, Epstein S, et al. Statement on exercise: Benefits and recommendations for physical activity programs for all Americans: A statement for health professionals by the Committee on Exercise and Cardiac Rehabilitation of the Council on Clinical Cardiology, American Heart Association. Circulation. 1992;86:340–344. doi: 10.1161/01.cir.86.1.340. [DOI] [PubMed] [Google Scholar]
- 14.Kulinski JP, Khera A, Ayers CR, Das SR, De Lemos JA, Blair SN, et al. Association Between Cardiorespiratory Fitness and Accelerometer-Derived Physical Activity and Sedentary Time in the General Population. Mayo Clin Proc. 2014 doi: 10.1016/j.mayocp.2014.04.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.American College of Sports Medicine: American College of Sports Medicine Position Stand. Exercise for patients with coronary artery disease. Med Sci Sports Exerc. 1994;26(3):i–v. [PubMed] [Google Scholar]
- 16.Tune JD. Morgan & Claypool. 2014. Coronary Circulation. [Google Scholar]
- 17.Duncker DJ, Bache RJ. Regulation of coronary blood flow during exercise. Physiol Rev. 2008;88(3):1009–1086. doi: 10.1152/physrev.00045.2006. [DOI] [PubMed] [Google Scholar]
- 18.Duncker DJ, Bache RJ, Merkus D. Regulation of coronary resistance vessel tone in response to exercise. J Mol Cell Cardiol. 2012;52(4):802–813. doi: 10.1016/j.yjmcc.2011.10.007. [DOI] [PubMed] [Google Scholar]
- 19.Laughlin MH, Bowles DK, Duncker DJ. The coronary circulation in exercise training. American Journal of Physiology - Heart and Circulatory Physiology. 2012;302(1):H10–H23. doi: 10.1152/ajpheart.00574.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Rowell LB. Human Cardiovascular Control. New York: Oxford University Press; 1993. [Google Scholar]
- 21.Tune JD, Gorman MW, Feigl EO. Matching coronary blood flow to myocardial oxygen consumption. J Appl Physiol. 2004;97(1):404–415. doi: 10.1152/japplphysiol.01345.2003. [DOI] [PubMed] [Google Scholar]
- 22.Feigl EO. Coronary physiology. Physiol Rev. 1983;63:1–205. doi: 10.1152/physrev.1983.63.1.1. [DOI] [PubMed] [Google Scholar]
- 23.Merkus D, Haitsma DB, Fung TY, Assen YJ, Verdouw PD, Duncker DJ. Coronary blood flow regulation in exercising swine involves parallel rather than redundant vasodilator pathways. Am J Physiol Heart Circ Physiol. 2003;285(1):H424–H433. doi: 10.1152/ajpheart.00916.2002. [DOI] [PubMed] [Google Scholar]
- 24.Rush JW, Aultman CD. Vascular biology of angiotensin and the impact of physical activity. Appl Physiol Nutr Metab. 2008;33(1):162–172. doi: 10.1139/H07-147. [DOI] [PubMed] [Google Scholar]
- 25.Jorgensen CR, Wang K, Wang Y, Gobel FL, Nelson RR, Taylor H. Effect of propranolol on myocardial oxygen consumption and its hemodynamic correlates during upright exercise. Circulation. 1973;48(6):1173–1182. doi: 10.1161/01.cir.48.6.1173. [DOI] [PubMed] [Google Scholar]
- 26.Taverne YJ, de Beer VJ, Hoogteijling BA, Juni RP, Moens AL, Duncker DJ, et al. Nitroso-redox balance in control of coronary vasomotor tone. J Appl Physiol (1985) 2012;112(10):1644–1652. doi: 10.1152/japplphysiol.00479.2011. [DOI] [PubMed] [Google Scholar]
- 27.Berne RM. Cardiac nucleotides in hypoxia: possible role in regulation of coronary blood flow. Am J Physiol. 1963;204(2):317–322. doi: 10.1152/ajplegacy.1963.204.2.317. [DOI] [PubMed] [Google Scholar]
- 28.Rossen JD, Oskarsson H, Minor RL, Jr, Talman CL, Winniford MD. Effect of adenosine antagonism on metabolically mediated coronary vasodilation in humans. J Am Coll Cardiol. 1994;23(6):1421–1426. doi: 10.1016/0735-1097(94)90386-7. [DOI] [PubMed] [Google Scholar]
- 29.Edlund A, Conradsson T, Sollevi A. A role for adenosine in coronary vasoregulation in man. Effects of theophylline and enprofylline. Clin Physiol. 1995;15:623–636. doi: 10.1111/j.1475-097x.1995.tb00549.x. [DOI] [PubMed] [Google Scholar]
- 30.Edlund A, Sollevi A. Theophylline increases coronary vascular tone in humans: Evidence for a role of endogenous adenosine in flow regulation. Acta Physiol Scand. 1995;155:303–311. doi: 10.1111/j.1748-1716.1995.tb09978.x. [DOI] [PubMed] [Google Scholar]
- 31.Tune JD, Richmond KN, Gorman MW, Feigl EO. Role of nitric oxide and adenosine in control of coronary blood flow in exercising dogs. Circulation. 2000;101(25):2942–2948. doi: 10.1161/01.cir.101.25.2942. [DOI] [PubMed] [Google Scholar]
- 32.Quyyumi AA, Dakak N, Mulcahy D, Andrews NP, Husain S, Panza JA, et al. Nitric oxide activity in the atherosclerotic human coronary circulation. J Am Coll Cardiol. 1997;29:308–317. doi: 10.1016/s0735-1097(96)00472-x. [DOI] [PubMed] [Google Scholar]
- 33.Lefroy DC, Crake T, Uren NG, Davies GJ, Maseri A. Effect of inhibition of nitric oxide synthesis on epicardial coronary artery caliber and coronary blood flow in humans. Circulation. 1993;88(1):43–54. doi: 10.1161/01.cir.88.1.43. [DOI] [PubMed] [Google Scholar]
- 34.Quyyumi AA, Mulcahy D, Andrews NP, Husain S, Panza JA, Cannon RO. Coronary vascular nitric oxide activity in hypertension and hypercholesterolemia - Comparison of acetylcholine and substance P. Circulation. 1997;95:104–110. doi: 10.1161/01.cir.95.1.104. [DOI] [PubMed] [Google Scholar]
- 35.Edlund A, Sollevi A, Wennmalm Å. The role of adenosine and prostacyclin in coronary flow regulation in healthy man. Acta Physiol Scand. 1989;135:39–46. doi: 10.1111/j.1748-1716.1989.tb08548.x. [DOI] [PubMed] [Google Scholar]
- 36.Friedman PL, Brown EJ, Jr, Gunther S, Alexander RW, Barry WH, Mudge GH, Jr, et al. Coronary vasoconstrictor effect of indomethacin in patients with coronary-artery disease. N Engl J Med. 1981;305(20):1171–1175. doi: 10.1056/NEJM198111123052002. [DOI] [PubMed] [Google Scholar]
- 37.Puybasset L, Béa ML, Ghaleh B, Giudicelli JF, Berdeaux A. Coronary and systemic hemodynamic effects of sustained inhibition of nitric oxide synthesis in conscious dogs - Evidence for cross talk between nitric oxide and cyclooxygenase in coronary vessels. Circ Res. 1996;79:343–357. doi: 10.1161/01.res.79.2.343. [DOI] [PubMed] [Google Scholar]
- 38.McKirnan MD, Bloor CM. Clinical significance of coronary vascular adaptations to exercise training. Med Sci Sports Exerc. 1994;26:1262–1268. [PubMed] [Google Scholar]
- 39.Williams PT, Thompson PD. Increased Cardiovascular Disease Mortality Associated With Excessive Exercise in Heart Attack Survivors. Mayo Clin Proc. 2014 doi: 10.1016/j.mayocp.2014.05.006. [DOI] [PubMed] [Google Scholar]
- 40.Taylor RS, Brown A, Ebrahim S, Jolliffe J, Noorani H, Rees K, et al. Exercise-based rehabilitation for patients with coronary heart disease: systematic review and meta-analysis of randomized controlled trials. Am J Med. 2004;116(10):682–692. doi: 10.1016/j.amjmed.2004.01.009. [DOI] [PubMed] [Google Scholar]
- 41.Jolliffe JA, Rees K, Taylor RS, Thompson D, Oldridge N, Ebrahim S. Exercise-based rehabilitation for coronary heart disease. Cochrane Database Syst Rev. 2001;(1):CD001800. doi: 10.1002/14651858.CD001800. [DOI] [PubMed] [Google Scholar]
- 42.Blair SN, Kohl HW, III, Barlow CE, Paffenbarger RS, Jr, Gibbons LW, Macera CA. Changes in physical fitness and all-cause mortality. A prospective study of healthy and unhealthy men. JAMA. 1995;273(14):1093–1098. [PubMed] [Google Scholar]
- 43.Fiuza-Luces C, Garatachea N, Berger NA, Lucia A. Exercise is the real polypill. Physiology (Bethesda) 2013;28(5):330–358. doi: 10.1152/physiol.00019.2013. [DOI] [PubMed] [Google Scholar]
- 44.Ornish D, Scherwitz LW, Billings JH, Gould KL, Merritt TA, Sparler S, et al. Intensive lifestyle changes for reversal of coronary heart disease. JAMA. 1998;280:2001–2007. doi: 10.1001/jama.280.23.2001. [DOI] [PubMed] [Google Scholar]
- 45.Rogers MA, Yamamoto C, Hagberg JM, Holloszy JO, Ehsani AA. The effect of 7 years of intense exercise training on patients with coronary artery disease. J Am Coll Cardiol. 1987;10(2):321–326. doi: 10.1016/s0735-1097(87)80014-1. [DOI] [PubMed] [Google Scholar]
- 46.Hambrecht R, Niebauer J, Marburger C, Grunze M, Kalberer B, Hauer K, et al. Various intensities of leisure time physical activity in patients with coronary artery disease: effects on cardiorespiratory fitness and progression of coronary atherosclerotic lesions. J Am Coll Cardiol. 1993;22(2):468–477. doi: 10.1016/0735-1097(93)90051-2. [DOI] [PubMed] [Google Scholar]
- 47.Schuler G, Hambrecht R, Schlierf G, Niebauer J, Hauer K, Neumann J, et al. Regular physical exercise and low-fat diet: Effects on progression of coronary artery disease. Circulation. 1992;86:1–11. doi: 10.1161/01.cir.86.1.1. [DOI] [PubMed] [Google Scholar]
- 48.Sixt S, Beer S, Bluher M, Korff N, Peschel T, Sonnabend M, et al. Long- but not short-term multifactorial intervention with focus on exercise training improves coronary endothelial dysfunction in diabetes mellitus type 2 and coronary artery disease. Eur Heart J. 2010;31(1):112–119. doi: 10.1093/eurheartj/ehp398. [DOI] [PubMed] [Google Scholar]
- 49.Madssen E, Moholdt T, Videm V, Wisloff U, Hegbom K, Wiseth R. Coronary Atheroma Regression and Plaque Characteristics Assessed by Grayscale and Radiofrequency Intravascular Ultrasound After Aerobic Exercise. Am J Cardiol. 2014 doi: 10.1016/j.amjcard.2014.08.012. [DOI] [PubMed] [Google Scholar]
- 50.Sturek M. Ca2+ regulatory mechanisms of exercise protection against coronary artery disease in metabolic syndrome and diabetes. J Appl Physiol. 2011;111:573–586. doi: 10.1152/japplphysiol.00373.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Kramsch DM, Aspen AJ, Abramowitz BM, Kreimendahl T, Wood WB., Jr Reduction of coronary atherosclerosis by moderate conditioning exercise in monkeys on an atherogenic diet. N Engl J Med. 1981;305(25):1483–1489. doi: 10.1056/NEJM198112173052501. [DOI] [PubMed] [Google Scholar]
- 52.Williams JK, Kaplan JR, Suparto IH, Fox JL, Manuck SB. Effects of exercise on cardiovascular outcomes in monkeys with risk factors for coronary heart disease. Arterioscler Thromb Vasc Biol. 2003;23:864–871. doi: 10.1161/01.ATV.0000067934.12783.6A. [DOI] [PubMed] [Google Scholar]
- 53.Link RP, Pedersoli WM, Safanie AH. Effect of exercise on development of atherosclerosis in swine. Atherosclerosis. 1972;15:107–122. doi: 10.1016/0021-9150(72)90044-5. [DOI] [PubMed] [Google Scholar]
- 54.Edwards JM, Neeb ZP, Alloosh MA, Long X, Bratz IN, Peller CR, et al. Exercise training decreases store-operated Ca2+ entry associated with metabolic syndrome and coronary atherosclerosis. Cardiovasc Res. 2010;85:631–640. doi: 10.1093/cvr/cvp308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Long X, Bratz IN, Alloosh M, Edwards JM, Sturek M. Short-term exercise training prevents micro- and macrovascular disease following coronary stenting. J Appl Physiol. 2010;108:1766–1774. doi: 10.1152/japplphysiol.01014.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Arce-Esquivel AA, Kreutzer KV, Rush JW, Turk JR, Laughlin MH. Exercise does not attenuate early CAD progression in a pig model. Med Sci Sports Exerc. 2012;44(1):27–38. doi: 10.1249/MSS.0b013e318228879b. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Wamhoff BR, Bowles DK, Dietz NJ, Hu Q, Sturek M. Exercise training attenuates coronary smooth muscle phenotypic modulation and nuclear Ca2+ signaling. Am J Physiol Heart Circ Physiol. 2002;283:H2397–H2410. doi: 10.1152/ajpheart.00371.2001. [DOI] [PubMed] [Google Scholar]
- 58.Belardinelli R, Paolini I, Cianci G, Piva R, Georgiou D, Purcaro A. Exercise training intervention after coronary angioplasty: the ETICA trial. J Am Coll Cardiol. 2001;37(7):1891–1900. doi: 10.1016/s0735-1097(01)01236-0. [DOI] [PubMed] [Google Scholar]
- 59.Lee HY, Kim JH, Kim BO, Byun YS, Cho S, Goh CW, et al. Regular exercise training reduces coronary restenosis after percutaneous coronary intervention in patients with acute myocardial infarction. Int J Cardiol. 2013;167(6):2617–2622. doi: 10.1016/j.ijcard.2012.06.122. [DOI] [PubMed] [Google Scholar]
- 60.Fleenor BS, Bowles DK. Exercise training decreases the size and alters the composition of the neointima in a porcine model of percutaneous transluminal coronary angioplasty (PTCA) J Appl Physiol. 2009;107:937–945. doi: 10.1152/japplphysiol.91444.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Ludmer PL, Selwyn AP, Shook TL, Wayne RR, Mudge GH, Alexander RW, et al. Paradoxical vasoconstriction induced by acetylcholine in atherosclerotic coronary arteries. N Engl J Med. 1986;315(17):1046–1051. doi: 10.1056/NEJM198610233151702. [DOI] [PubMed] [Google Scholar]
- 62.Wang J, Wolin MS, Hintze TH. Chronic exercise enhances endothelium-mediated dilation of epicardial coronary artery in conscious dogs. Circ Res. 1993;73:829–838. doi: 10.1161/01.res.73.5.829. [DOI] [PubMed] [Google Scholar]
- 63.Hambrecht R, Wolf A, Gielen S, Linke A, Hofer J, Erbs S, et al. Effect of exercise on coronary endothelial function in patients with coronary artery disease. N Engl J Med. 2000;342(7):454–460. doi: 10.1056/NEJM200002173420702. [DOI] [PubMed] [Google Scholar]
- 64.Feletou M, Kohler R, Vanhoutte PM. Nitric oxide: orchestrator of endothelium-dependent responses. Ann Med. 2012;44(7):694–716. doi: 10.3109/07853890.2011.585658. [DOI] [PubMed] [Google Scholar]
- 65.Wienbergen H, Hambrecht R. Physical exercise and its effects on coronary artery disease. Current Opinion in Pharmacology. 2013;13(2):218–225. doi: 10.1016/j.coph.2012.12.003. [DOI] [PubMed] [Google Scholar]
- 66.Hambrecht R, Adams V, Erbs S, Linke A, Krankel N, Shu Y, et al. Regular physical activity improves endothelial function in patients with coronary artery disease by increasing phosphorylation of endothelial nitric oxide synthase. Circulation. 2003;107(25):3152–3158. doi: 10.1161/01.CIR.0000074229.93804.5C. [DOI] [PubMed] [Google Scholar]
- 67.Richter B, Niessner A, Penka M, Grdic M, Steiner S, Strasser B, et al. Endurance training reduces circulating asymmetric dimethylarginine and myeloperoxidase levels in persons at risk of coronary events. Thromb Haemost. 2005;94(6):1306–1311. doi: 10.1160/TH05-03-0158. [DOI] [PubMed] [Google Scholar]
- 68.Widder JD, Chen W, Li L, Dikalov S, Thony B, Hatakeyama K, et al. Regulation of tetrahydrobiopterin biosynthesis by shear stress. Circ Res. 2007;101(8):830–838. doi: 10.1161/CIRCRESAHA.107.153809. [DOI] [PubMed] [Google Scholar]
- 69.Wyss CA, Koepfli P, Namdar M, Siegrist PT, Luscher TF, Camici PG, et al. Tetrahydrobiopterin restores impaired coronary microvascular dysfunction in hypercholesterolaemia. Eur J Nucl Med Mol Imaging. 2005;32(1):84–91. doi: 10.1007/s00259-004-1621-y. [DOI] [PubMed] [Google Scholar]
- 70.Fukuda Y, Teragawa H, Matsuda K, Yamagata T, Matsuura H, Chayama K. Tetrahydrobiopterin restores endothelial function of coronary arteries in patients with hypercholesterolaemia. Heart. 2002;87(3):264–269. doi: 10.1136/heart.87.3.264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Kojda G, Hambrecht R. Molecular mechanisms of vascular adaptations to exercise. Physical activity as an effective antioxidant therapy? Cardiovasc Res. 2005;67(2):187–197. doi: 10.1016/j.cardiores.2005.04.032. [DOI] [PubMed] [Google Scholar]
- 72.Adams V, Linke A, Krankel N, Erbs S, Gielen S, Mobius-Winkler S, et al. Impact of Regular Physical Activity on the NAD(P)H Oxidase and Angiotensin Receptor System in Patients With Coronary Artery Disease. Circulation. 2005;111(5):555–562. doi: 10.1161/01.CIR.0000154560.88933.7E. [DOI] [PubMed] [Google Scholar]
- 73.Lenk K, Uhlemann M, Schuler G, Adams V. Role of endothelial progenitor cells in the beneficial effects of physical exercise on atherosclerosis and coronary artery disease. J Appl Physiol (1985) 2011;111(1):321–328. doi: 10.1152/japplphysiol.01464.2010. [DOI] [PubMed] [Google Scholar]
- 74.Libby P, Ridker PM, Hansson GK. Progress and challenges in translating the biology of atherosclerosis. Nature. 2011;473(7347):317–325. doi: 10.1038/nature10146. [DOI] [PubMed] [Google Scholar]
- 75.Hanna MA, Taylor CR, Chen B, La HS, Maraj JJ, Kilar CR, et al. Structural remodeling of coronary resistance arteries: effects of age and exercise training. J Appl Physiol (1985) 2014;117(6):616–623. doi: 10.1152/japplphysiol.01296.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Trask AJ, Katz PS, Kelly AP, Galantowicz ML, Cismowski MJ, West TA, et al. Dynamic micro- and macro-vascular remodeling in coronary circulation of obese Ossabaw pigs with metabolic syndrome. J Appl Physiol. 2012;113(7):1128–1140. doi: 10.1152/japplphysiol.00604.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Aalkjaer C, Heagerty AM, Petersen KK, Swales JD, Mulvany MJ. Evidence for increased media thickness, increased neuronal amine uptake, and depressed excitation--contraction coupling in isolated resistance vessels from essential hypertensives. Circ Res. 1987;61(2):181–186. doi: 10.1161/01.res.61.2.181. [DOI] [PubMed] [Google Scholar]
- 78.Heaps CL, Parker JL. Effects of exercise training on coronary collateralization and control of collateral resistance. J Appl Physiol. 2011;111(2):587–598. doi: 10.1152/japplphysiol.00338.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Franklin BA. Exercise training and coronary collateral circulation. Med Sci Sports Exerc. 1991;23(6):648–653. [PubMed] [Google Scholar]
- 80.Niebauer J, Hambrecht R, Marburger C, Hauer K, Velich T, von HE, et al. Impact of intensive physical exercise and low-fat diet on collateral vessel formation in stable angina pectoris and angiographically confirmed coronary artery disease. Am J Cardiol. 1995;76(11):771–775. doi: 10.1016/s0002-9149(99)80224-0. [DOI] [PubMed] [Google Scholar]
- 81.Belardinelli R, Georgiou D, Ginzton L, Cianci G, Purcaro A. Effects of moderate exercise training on thallium uptake and contractile response to low-dose dobutamine of dysfunctional myocardium in patients with ischemic cardiomyopathy. Circulation. 1998;97(6):553–561. doi: 10.1161/01.cir.97.6.553. [DOI] [PubMed] [Google Scholar]
- 82.Todd IC, Bradnam MS, Cooke MB, Ballantyne D. Effects of daily high-intensity exercise on myocardial perfusion in angina pectoris. Am J Cardiol. 1991;68(17):1593–1599. doi: 10.1016/0002-9149(91)90315-c. [DOI] [PubMed] [Google Scholar]
- 83.Schuler G, Schlierf G, Wirth A, Mautner HP, Scheurlen H, Thumm M, et al. Low-fat diet and regular, supervised physical exercise in patients with symptomatic coronary artery disease: reduction of stress-induced myocardial ischemia. Circulation. 1988;77(1):172–181. doi: 10.1161/01.cir.77.1.172. [DOI] [PubMed] [Google Scholar]
- 84.Fujita M, Sasayama S, Asanoi H, Nakajima H, Sakai O, Ohno A. Improvement of treadmill capacity and collateral circulation as a result of exercise with heparin pretreatment in patients with effort angina. Circulation. 1988;77(5):1022–1029. doi: 10.1161/01.cir.77.5.1022. [DOI] [PubMed] [Google Scholar]