Abstract
Tight control of T follicular helper (Tfh) cells is required for optimal maturation of the germinal centre (GC) response. The molecular mechanisms controlling Tfh-cell differentiation remain incompletely understood. Here we show that microRNA-146a (miR-146a) is highly expressed in Tfh cells and peak miR-146a expression marks the decline of the Tfh response after immunization. Loss of miR-146a causes cell-intrinsic accumulation of Tfh and GC B cells. MiR-146a represses several Tfh-cell-expressed messenger RNAs, and of these, ICOS is the most strongly cell autonomously upregulated target in miR-146a-deficient T cells. In addition, miR-146a deficiency leads to increased ICOSL expression on GC B cells and antigen-presenting cells. Partial blockade of ICOS signalling, either by injections of low dose of ICOSL blocking antibody or by halving the gene dose of Icos in miR-146a-deficient T cells, prevents the Tfh and GC B-cell accumulation. Collectively, miR-146a emerges as a post-transcriptional brake to limit Tfh cells and GC responses.
 Maturation of antibody-producing B cells in germinal centers is orchestrated by T follicular helper cells. Here Pratama et al. show that miR-146a negatively regulates T follicular helper cells by targeting ICOS-ICOS ligand signaling in germinal centers.
Maturation of antibody-producing B cells in germinal centers is orchestrated by T follicular helper cells. Here Pratama et al. show that miR-146a negatively regulates T follicular helper cells by targeting ICOS-ICOS ligand signaling in germinal centers.
T follicular helper (Tfh) cells provide essential survival and selection signals to germinal centre (GC) B cells that are important for long-lived protective antibody responses1,2. Increasing evidence suggests that restricting Tfh-cell numbers in GCs is crucial for optimal GC B-cell selection3,4,5. B cells expressing the highest affinity receptors after somatic hypermutation can capture more antigens and therefore have a competitive advantage in establishing sustained interactions and eliciting survival signals from Tfh cells5. Studies of autoimmune mouse models6,7,8,9 and human patients10,11,12,13,14 suggest that excessive Tfh cells may contribute to the pathogenesis of antibody-mediated autoimmune diseases, potentially by allowing survival and differentiation of self-reactive B cells. While multiple signals are now recognized to be important for Tfh-cell formation and migration3, relatively little is known about the mechanisms that limit Tfh-cell numbers to achieve optimal selection of high affinity B-cell clones. Cell-extrinsic mechanisms such as the actions of T follicular regulatory (Tfr)15,16,17 and follicular CD8+ T cells18 have been reported, but to date, only Roquin is shown to act in a T cell-autonomous manner to prevent spontaneous accumulation of Tfh cells19.
MicroRNA-146a (miR-146a) has recently emerged as a key post-transcriptional regulator in hematopoietic cells. MiR-146a represses TRAF6 and IRAK1 in myeloid cells20 and T cells21 to control their proliferation and NF-κB activation in response to Toll-like receptor and TCR signalling, respectively. Deficiency of miR-146a leads to excessive production of IL-6 and TNF, myeloproliferation, chronic inflammation and a decline in the number and quality of hematopoietic stem cells20,22,23. In the absence of miR-146a, regulatory T (Treg) cells also lose their suppressive ability due to STAT1 overexpression driving increased IFN-γ secretion24. Not surprisingly, dysregulated expression of miR-146a has also been found to correlate with increased incidence of autoimmune diseases, such as lupus25,26,27,28 and rheumatoid arthritis29,30,31,32.
Here we show that miR-146a profoundly represses Tfh-cell numbers: the absence of this miRNA leads to spontaneous Tfh-cell accumulation that precedes myeloid cell dysregulation and is not a consequence of Treg-cell functional deficiency. This is achieved by directly repressing multiple messenger RNAs (mRNAs) targets, most prominently Icos.
Results
MiR-146a is highly expressed in Tfh cells
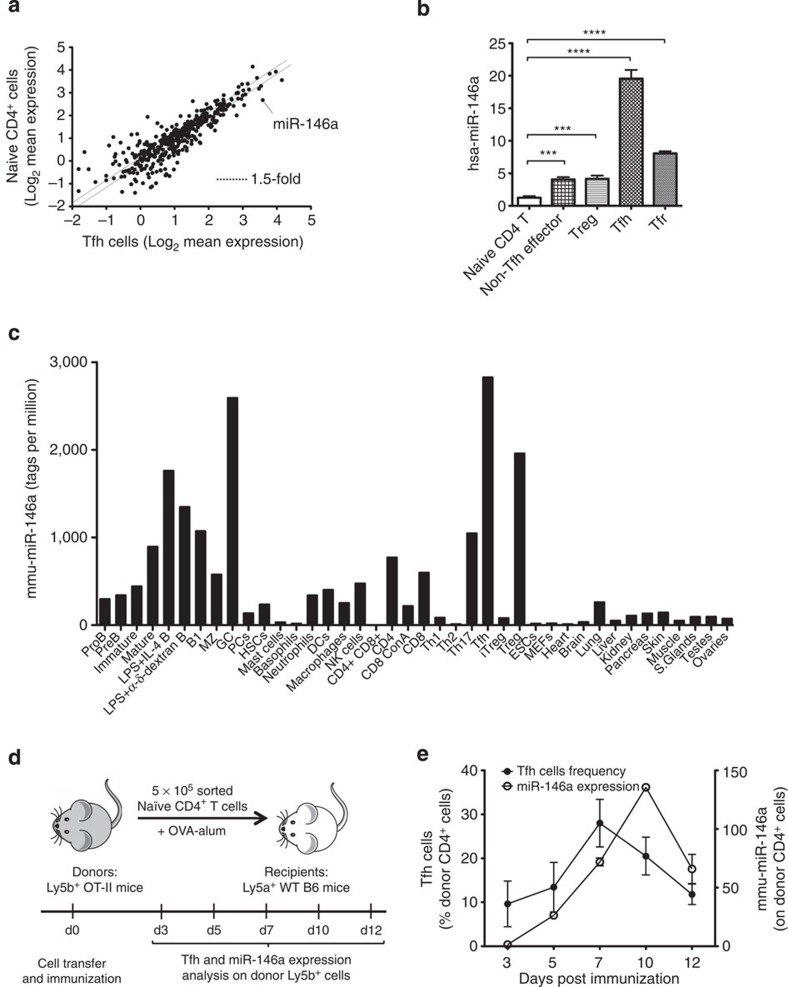
To identify miRNAs that might regulate Tfh-cell development and/or function, we performed a genome-wide expression analysis to identify miRNAs that are highly expressed in human Tfh cells. Compared with naïve (CD44low CD25−) CD4+ T cells, miR-146a was the most differentially expressed miRNA (8.2-fold) in Tfh cells (Fig. 1a) among those that are highly expressed in T cells. Real-time PCRs on T-cell subsets from human tonsils revealed that Tfh cells expressed the highest amounts of miR-146a compared with all other effector (CD44high) subsets (Fig. 1b). Consistent with these results, a deep-sequencing survey of the miRNome in mouse tissues showed the highest levels of miR-146a in Tfh and GC B cells (Fig. 1c and ref. 33). Lower amounts of miR-146a were also present in activated B cells and thymic-derived Tregs, while non-haematopoietic tissues displayed little or no miR-146a expression (Fig. 1c).
Figure 1. MiR-146a is highly expressed in Tfh cells.
(a) Microarray analysis of miRNA expression in naïve CD4+ T and Tfh cells. The scatter plot shows the log2 of averaged (n=3) background-subtracted expression of all Agilent Human miRNA Microarray (V1) Kit 8 × 15K probes by Tfh cells on the x-axis compared with naïve CD4+ T cells on the y-axis. Each dot represents one miRNA probe. The dotted lines are drawn at 1.5-fold difference in expression. (b) Expression of miR-146a, normalized to U6, measured by real-time PCR from human tonsillar T-cell subsets. Cell sorting strategy is described in the Methods. The heights of the bars represent the mean, and the error bars represent the s.d. of three technical replicates. Data are representative of three independent experiments. Statistical significance was determined using unpaired Student’s t-test. ***P<0.001, ****P<0.0001. (c) Bar graphs showing the expression of miR-146a in mouse immune cell subsets and non-hematopoietic tissues as described in Kuchen et al.33. In brief, small RNAs from various mouse tissues were ligated to adapters using Illumina’s protocol. These were then reverse transcribed, amplified by PCR and sequenced on a Genome Analyzer (Illumina). (d,e) Kinetics of the Tfh-cell response (filled circles) and miR-146a expression (open circles) from transferred Ly5b+ OT-II cells analyzed at different time points according to the strategy shown in d. Three recipient mice were killed at each time point. Percentage of Tfh cells among donor Ly5b+ cells was measured using flow cytometry staining of CD4+ CXCR5high PD-1high Foxp3− T cells. MiR-146a expression was measured by real-time PCR of total Ly5b+ CD4+ T cells and was normalized to U6 expression. Filled and open circles in e represent the mean, and error bars represent the s.d. of biological replicates (for Tfh-cell percentage) or technical replicates (for miR-146a expression).
MiR-146a expression peaks at the late stage of GC responses
To study the kinetics of miR-146a expression during a T cell-dependent immune response, we adoptively transferred 5 × 105 naïve CD4+ T cells from Ly5b+ OT-II TCR-transgenic mice into Ly5a+ wild-type C57BL/6 recipient mice, which were then immunized with ovalbumin (OVA) precipitated in alum and killed at different time points (Fig. 1d). The proportion of Tfh cells among donor cells peaked at day 7 post immunization (Fig. 1e) and started to contract by day 10, coinciding with the peak of miR-146a expression in transferred cells (Fig. 1e). This suggests a possible role for miR-146a in downsizing the Tfh-cell population.
Cell-autonomous control of Tfh cells by miR-146a
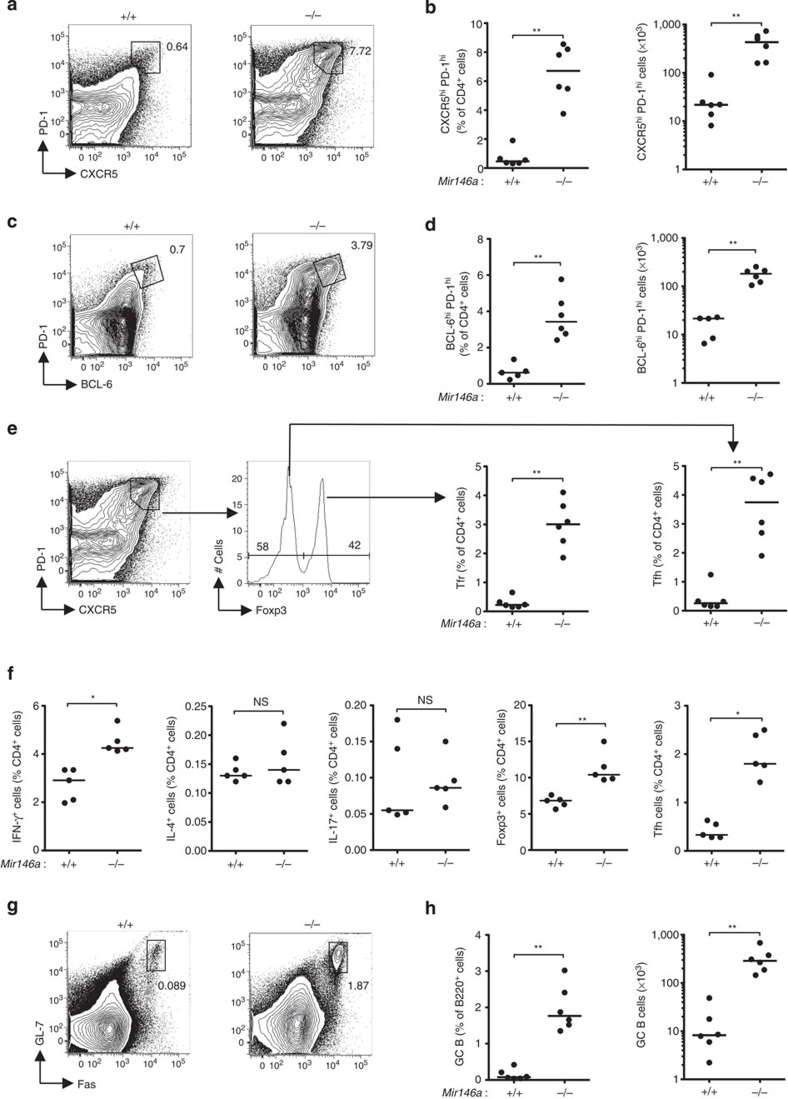
To test whether miR-146a plays a role in the control of Tfh-cell numbers, we studied Tfh-cell development and function in 12- to 16-week-old mice lacking miR-146a. Analysis of total follicular T cells, gated as CXCR5high PD-1high (Fig. 2a,b) or BCL-6high PD-1high (Fig. 2c,d) cells, showed they were expanded in both percentage and total number in unimmunized Mir146a−/− mice compared with wild-type littermates. This expansion was not selective to either Tfh (CXCR5high PD-1high Foxp3−) or Tfr (CXCR5high PD-1high Foxp3+) cells as both subsets increased in parallel (Fig. 2e). This effect of miR-146a deficiency seemed to affect Tfh cells more severely than the other effector CD4+ T cell subsets: compared with the 5-fold increase in Tfh cells, no statistically significant increases were observed in either Th2 (IL-4+) cells or Th17 (IL-17+) cells, while Th1 (IFN-γ+) cells were increased by 1.4-fold and Tregs (Foxp3+), as previously reported24, were expanded by 1.5-fold (Fig. 2f). Total GC B cells were also increased by about 20-fold in Mir146a−/− mice compared with wild-type littermates (Fig. 2g,h). The spontaneous accumulation of Tfh and GC B cells became more severe with age (Supplementary Fig. 1).
Figure 2. Loss of miR-146a causes spontaneous accumulation of Tfh and GC B cells.
(a–d) Flow cytometric contour plots and dot plots showing percentages and absolute cell number of follicular T cells gated as CXCR5high PD-1high cells (a,b) or BCL-6high PD-1high cells (c,d). (e) Gating strategy for Tfh (CXCR5high PD-1high Foxp3−) and Tfr (CXCR5high PD-1high Foxp3+) cells and dot plots showing their percentages. (f) Percentages of Th1 (IFN-γ+), Th2 (IL-4+), Th17 (IL-17+), Treg (Foxp3+) and Tfh (CXCR5high PD-1high Foxp3−) cells among total CD4+ cells. (g,h) Flow cytometric contour plots and dot plots showing percentages and absolute cell numbers of GC B cells. Contour plots shown in a,c,e are gated on CD4+ cells, those in g are gated on B220+ cells. Unimmunized miR-146a-deficient (−/−) and -sufficient (+/+) mice were analyzed. Each symbol represents one mouse and the horizontal bars represent the median values. Data are representative of at least three independent experiments. Statistical significance was determined using Mann–Whitney U-test. NS, not significant, *P<0.05, **P<0.01.
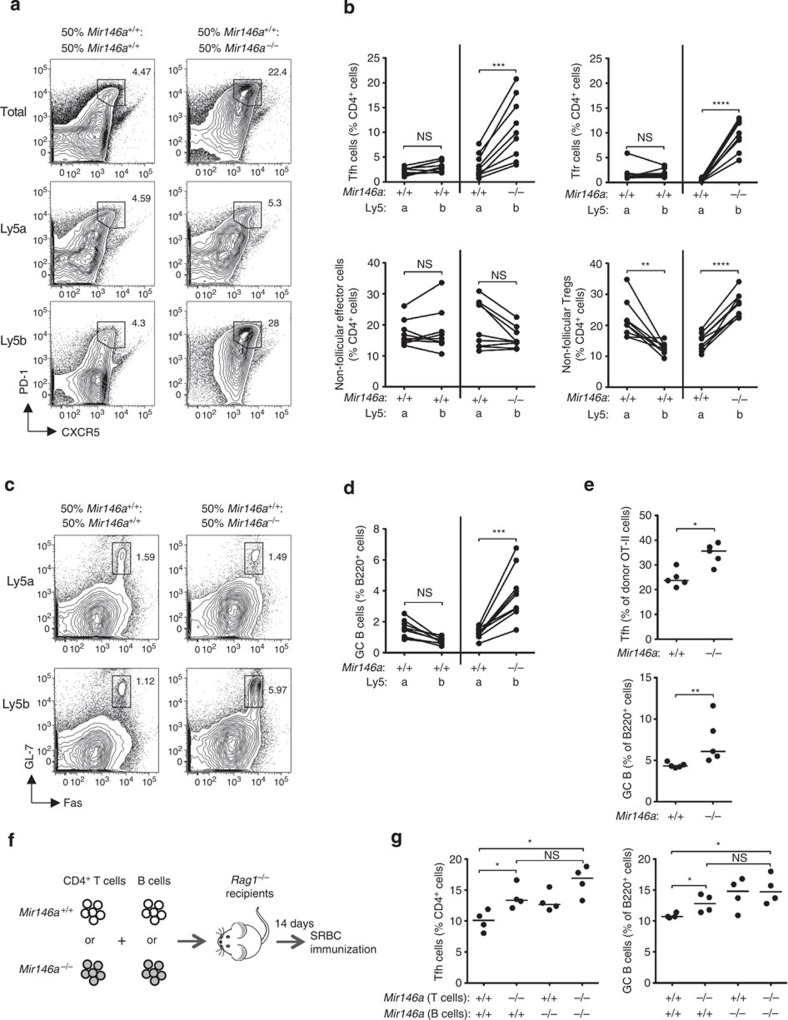
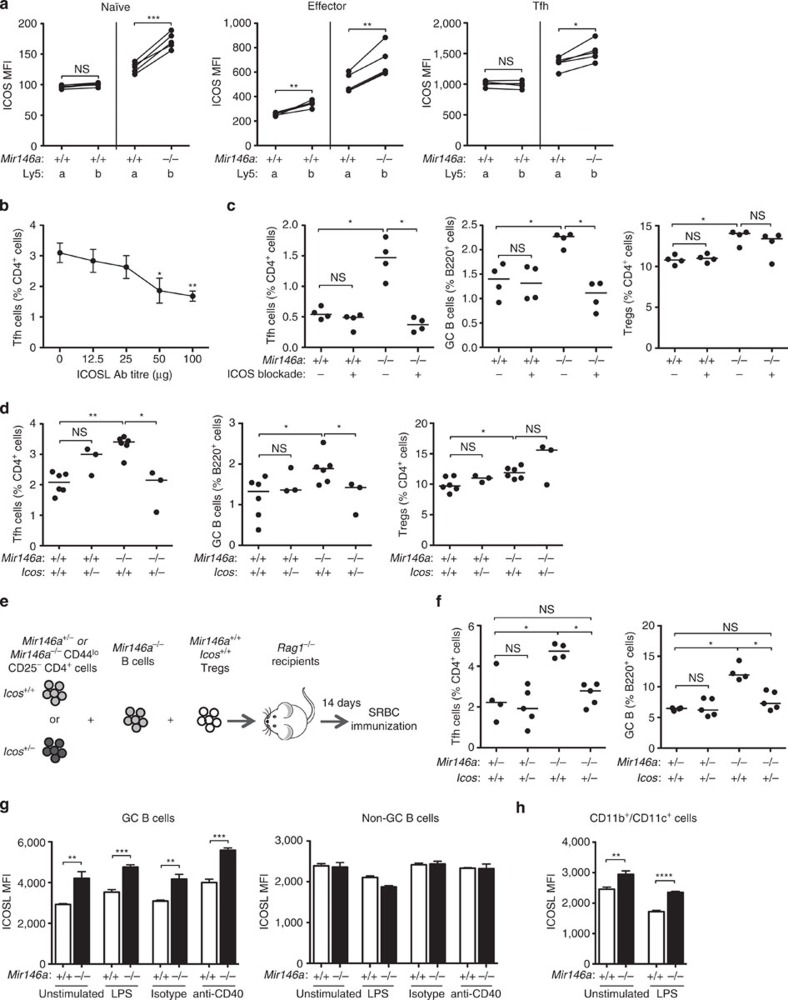
To test whether the accumulation of Tfh cells in Mir146a−/− mice was due to T-cell intrinsic factors, we set up mixed bone marrow chimeras by transferring equal amounts of either Ly5a+.Mir146a+/+: Ly5b+.Mir146a+/+ (WT:WT) or Ly5a+.Mir146a+/+: Ly5b+.Mir146a−/− (WT:KO) bone marrow cells into sub-lethally irradiated Rag1−/− recipients. Four months after reconstitution, the spleens were harvested and the percentage of total CD4+ T cells, effector cells, Tfh cells, total B220+ cells and GC B cells derived from the Ly5a+ or Ly5b+ donor marrow in each mouse was determined (Fig. 3a–d). There was a higher percentage of total follicular T cells in the WT:KO chimeras compared with the WT:WT chimeras (Fig. 3a). This was due to cell-autonomous expansions of Tfh and Tfr cells derived from Ly5b+.Mir146a−/− marrow (Fig. 3b). In each mouse, there was a two to fourfold increase in Ly5b+ Tfh cells compared with Ly5a+ Tfh cells, whereas this was not observed in the control WT:WT chimeras, in which the percentages of Ly5a+ and Ly5b+ Tfh cells were comparable (Fig. 3b). Importantly, the percentages of non-follicular effector CD4+ cells were comparable between Ly5a+ and Ly5b+ cells in both WT:WT and WT:KO mixed chimeras. Consistent with previous reports24, there was a cell-autonomous expansion of non-follicular Tregs derived from Ly5b+.Mir146a−/− marrow in the WT:KO chimeras (Fig. 3b).
Figure 3. Cell-autonomous accumulation of miR-146a-deficient Tfh cells.
(a) Flow cytometric contour plots showing the gating strategy for follicular T cells in 50:50 mixed bone marrow chimeras gated on all CD4+ (top), Ly5a+ CD4+ (middle) and Ly5b+ CD4+ (bottom) cells. (b) Percentages of Tfh, Tfr, non-follicular effector and non-follicular Tregs in the mixed chimeras. (c,d) Flow cytometric contour plots and dot plot showing the percentages of GC B cells in the mixed chimeras. Contour plots in c are gated on Ly5a+ B220+ and Ly5b+ B220+ cells as indicated. Connecting lines between percentages of Ly5a+ and Ly5b+ in b and d indicate that they are from the same chimeric mouse. Chimeras were analyzed 16 weeks post reconstitution. (e) Percentages of OVA-specific (Vβ5+) Tfh cells and total GC B cells in Cd28−/− recipient mice 7 days following adoptive transfer. Data are representative of three independent experiments. (f,g) Adoptive transfer of purified CD4+ T cells and B cells from Mir146a+/+ or Mir146a−/− donor mice into Rag1−/− recipients according to the strategy shown in f. The recipient mice were immunized with sheep red blood cells 14 days after transfer and they were killed 7 days post immunization. (g) Percentages of Tfh and GC B cells in Rag1−/− recipient mice following adoptive transfer and SRBC immunization. Each symbol represents one mouse and the horizontal bars represent the median values. Statistical significance was determined using a paired Student’s t-test (b,d) or Mann–Whitney U-test (e,g). NS, not significant, *P<0.05, **P<0.01, ***P<0.001, ****P<0.0001.
We also observed a statistically significant increase in the percentage of GC B cells derived from Ly5b+.Mir146a−/− marrow compared with those derived from Ly5a+.Mir146a+/+ bone marrow (Fig. 3c,d), suggesting that miR-146a also acts cell autonomously in GC B cells. Intriguingly, despite the significant increase of total follicular T cells in the WT:KO chimeras (Fig. 3a), we only observed expansion of the Ly5b+.Mir146a−/− GC B cells, while the percentage of the neighbouring Ly5a+.Mir146a+/+ GC B cells was comparable to that in the WT:WT chimeras (Fig. 3d). This could indicate that GC expansion requires the concerted actions of miR-146a in T cells and B cells, perhaps through the regulation of a receptor–ligand pair in each cell type. Collectively, these results suggest that miR-146a acts in T cells and B cells to prevent Tfh and GC B-cell accumulation.
MiR-146a deficiency in T cells initiates Tfh-cell expansion
We next investigated whether accumulation of Tfh cells could occur independently of neighbouring Mir146a−/− B cells and myeloid cells in the same mice. To this end, we transferred purified naïve CD4+ T cells from Mir146a+/+ or Mir146a−/− OT-II TCR-transgenic mice into Cd28−/− recipients, which were then immunized with OVA precipitated in alum and killed 7 days later. There was a statistically significant increase in the percentage of OVA-specific Vβ5+ Tfh cells and in the percentage of total GC B cells in mice that received Mir146a−/− OT-II cells, although the magnitude of the effect was smaller than that observed in intact Mir146a−/− mice (Fig. 3e), suggesting that non-T cells, including B cells and myeloid cells, enhance this effect.
In a separate experiment, four different combinations of Mir146a+/+ or Mir146a−/− CD4+ T cells and B cells were co-transferred into Rag1−/− mice, which were immunized 14 days later with sheep red blood cells (SRBCs; Fig. 3f). Similar to the OT-II transfer experiment, we found that in the presence of wild-type B cells and myeloid cells, miR-146a deficiency in T cells was sufficient to cause Tfh-cell accumulation, although the magnitude of the effect was again more modest than that observed in intact Mir146a−/− mice (Fig. 3g). The presence of Mir146a−/− B cells appeared to exacerbate the effect, but this did not reach statistical significance. Similar trends were observed for the increase in GC B cells (Fig. 3g). In summary, our data show that miR-146a deficiency in T cells is sufficient to initiate Tfh and GC B-cell accumulation, but T-cell-extrinsic factors contribute to exacerbate this phenotype.
Contribution of IL-6 to Tfh and GC B-cell accumulation
In view of the reported increased proliferation of myeloid cells in 6- to 8-month-old Mir146a−/− mice20,34, we asked whether expanded myeloid compartment could be one of the T-cell-extrinsic factors contributing to the Tfh and GC B-cell expansion. Assessments of 12- to 16-week-old mice, when the Tfh-cell accumulation is obvious (Supplementary Fig. 1), revealed no significant accumulation of myeloid cell subsets in Mir146a−/− mice (Supplementary Fig. 2a).
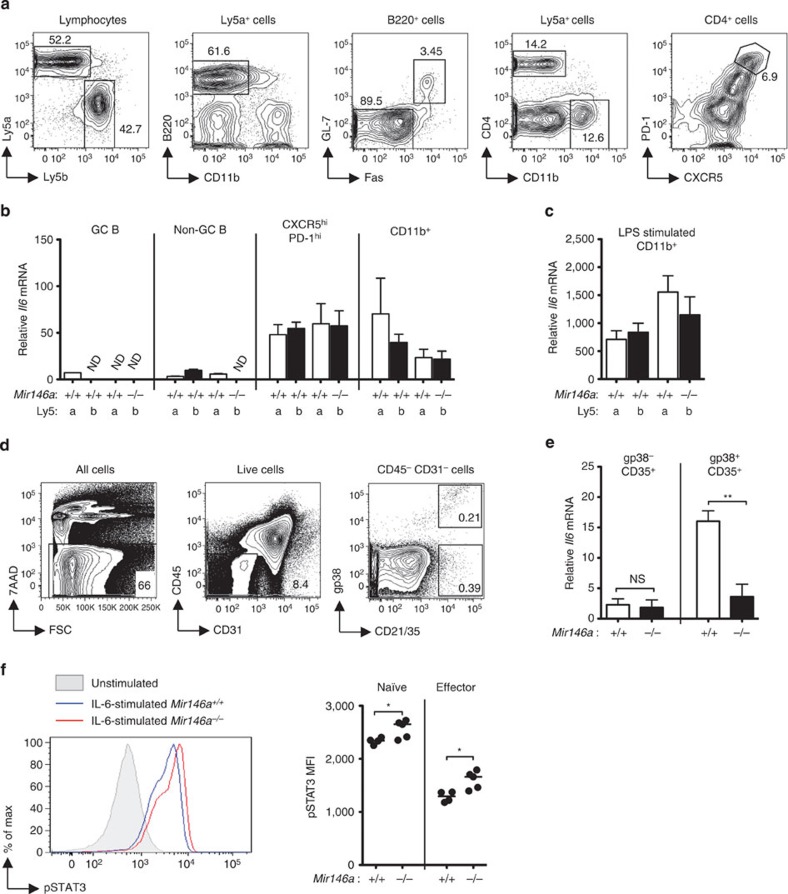
Myeloid cells and T cells have been reported to produce more IL-6 cytokine in 6- to 10-month-old miR-146a-null mice20,23. Since IL-6 is required for Tfh-cell formation35,36, we tested whether IL-6 production was elevated in 12- to 16-week-old Mir146a−/− mice. No significant increase of IL-6 was observed in the serum of these mice (Supplementary Fig. 2b), and we also did not find increased amount of Il6 transcripts in CD11chigh splenic dendritic cells (Supplementary Fig. 2b). Next, we used Ly5a+.Mir146a+/+: Ly5b+.Mir146a−/− mixed bone marrow chimeras to investigate the possibility that during the course of an immune response, IL-6 might be expressed at higher levels in Mir146a−/− GC B cells, Tfh cells or myeloid cells. Despite the expected cell-autonomous accumulation of Mir146a−/− Tfh cells in the mixed chimeric mice (Supplementary Fig. 3), no differences in Il6 mRNA expression were found between miR-146a-sufficient and miR-146a-deficient cells in any of the subsets examined (Fig. 4a–c). Finally, we tested the possibility that follicular dendritic cells (FDCs), which are of non-hematopoietic origin, expressed more IL-6 in the absence of miR-146a; it has been suggested that FDC-derived IL-6 is important for the late stage maintenance of Tfh cells during viral infection35. We isolated FDCs according to published protocols by gating on CD45− CD31− CD21/35+ cells from Mir146a+/+ or Mir146a−/− mice. As both gp38-expressing37 and non-gp38-expressing38 FDCs have been identified, we separated these two cell subsets and measured the expression of IL-6 (Fig. 4d). Again, we did not find evidence of increased Il6 transcripts in either gp38+ or gp38− FDCs from miR-146-deficient mice (Fig. 4e). Nevertheless, a complete blockade of IL-6R using a previously reported dose of monoclonal antibody35 greatly reduced Tfh-cell accumulation in Mir146a−/− mice (Supplementary Fig. 4), indicating that miR-146a-driven Tfh-cell accumulation is still dependent on IL-6-mediated signalling.
Figure 4. Contribution of IL-6 signalling to the Tfh and GC B-cell accumulation in miR-146a-deficient mice.
(a–c) Similar expression of Il6 mRNA between miR-146a-deficient (−/−) and -sufficient (+/+) cells in SRBC-immunized Ly5a+.Mir146a+/+: Ly5b+.Mir146a−/− mixed bone marrow chimeras analyzed 14 weeks post reconstitution. (a) Flow cytometric contour plots showing the gating strategy used to isolate the different cell subsets. (b) Relative amounts of Il6 mRNA in GC B (B220+ GL-7+ Fas+), non-GC B (B220+ GL-7− Fas−), Tfh (CD4+ CXCR5high PD-1high), and CD11b+ cells from Ly5a+ and Ly5b+ cells analyzed directly after isolation. ND=not detectable. The expression of Il6 was normalized to β-actin. (c) Relative amounts of Il6 mRNA in LPS-stimulated CD11b+ cells from the same set of chimeric mice as in b. (d,e) No increased expression of IL-6 in miR-146a-deficient follicular dendritic cells. (d) Flow cytometric contour plots showing the gating strategy used to isolate follicular dendritic cells. (e) Relative amounts of Il6 mRNA in gp38+ and gp38− FDCs (CD45− CD31− CD35+) from Mir146a+/+ or Mir146a−/− mice. (f) Histograms and dot plots showing the amounts of STAT3 phosphorylation in naïve and effector CD4+ T cells following acute stimulation with recombinant mouse IL-6. Data are representative of at least two independent experiments. Heights of bar graph in b,c,e represent mean and error bar represents s.d. Each symbol in f represents one mouse and the horizontal bars represent the median values. Statistical significance was determined using unpaired Student’s t-test (e) or Mann–Whitney U-test (f). NS=not significant, *P<0.05, **P<0.01.
Intriguingly, we observed enhanced IL-6-induced STAT3 phosphorylation in naïve and effector CD4+ T cells from Mir146a−/− mice (Fig. 4f). Lack of increased STAT1 phosphorylation in parallel assays (Supplementary Fig. 5) argued against STAT1 overexpression being the cause of this enhanced sensitivity to IL-6. Collectively, these results suggest that increased IL-6 expression is probably not the main cause of the Tfh-cell accumulation in miR-146a-deficient mice; rather, the increased sensitivity to IL-6 signalling is a more likely contributor to the phenotype.
MiR-146a represses multiple Tfh-cell mRNAs
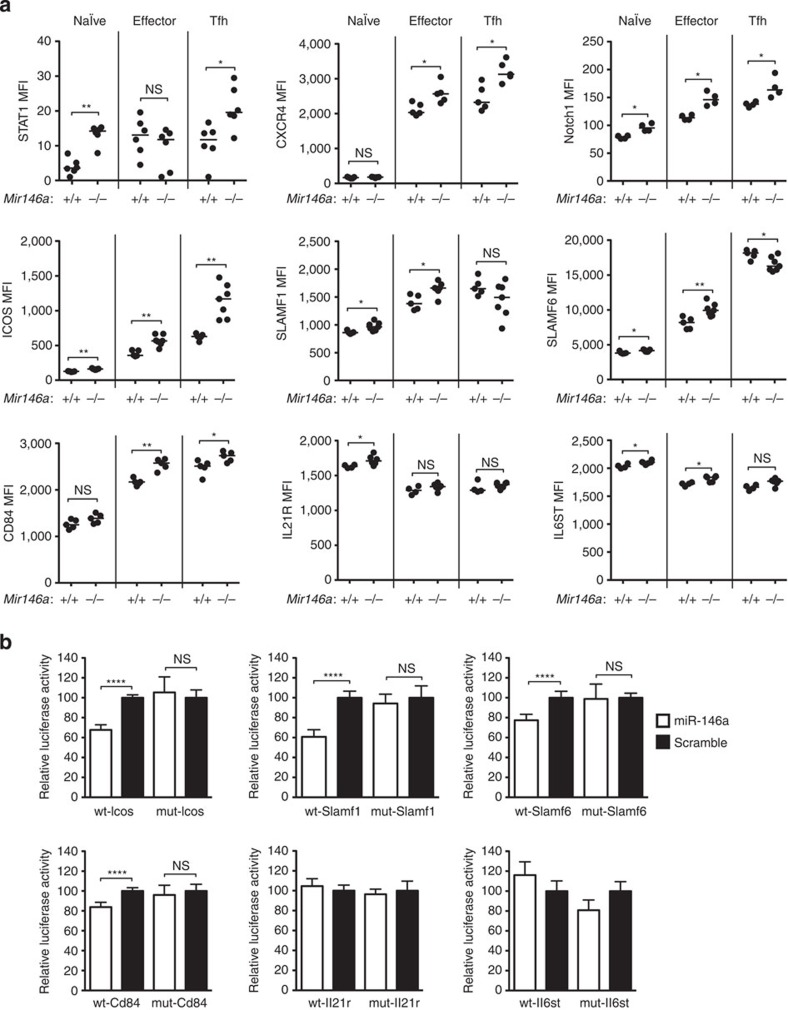
To uncover mRNA targets of miR-146a in T cells, we performed a microarray analysis on sorted naïve CD4+ T cells from Mir146a+/+ and Mir146a−/− mice. A number of differentially expressed genes were identified and for further validation, we selected putative targets based on meeting two of the following three criteria: (1) upregulated mRNA expression in Mir146a−/− T cells assessed by global gene expression profiling, (2) known roles in Tfh and/or GC biology and (3) the presence of target sites for miR-146a seed region within their 3′ untranslated region (UTR). The selected mRNA targets were Stat1, Cxcr4, Notch1, Slamf1, Slamf6, Cd84, Icos, Il6st and Il21r (putative miR-146a binding sites are shown in Supplementary Fig. 6; previously validated target sites in Cxcr4, Notch1 and Stat1 are not shown).
Next we assessed by flow cytometry whether the proteins encoded by these putative RNA targets were upregulated in miR-146a-deficient T cells. STAT1, a validated target of miR-146a in Tregs24 and an important signalling intermediate and transcription factor downstream of IL-6, IL-12, IL-21 and IL-27, was upregulated in Mir146a−/− naïve CD4+, Tfh cells (Fig. 5a) and, as shown previously24, in Tregs (Supplementary Fig. 7). The validated miR-146a targets CXCR4 (ref. 39), known to regulate dark zone versus light zone partitioning in GCs5,40,41, and Notch1 (ref. 42), shown to promote Tfh-cell differentiation43, were both upregulated in Mir146a−/− effector CD4+, Tfh cells (Fig. 5a) and in Tregs (Supplementary Fig. 7). ICOS is required for providing costimulatory signals for optimal Tfh and GC B-cell differentiation44,45,46,47 and for persistent directional motility and follicular recruitment of activated CD4+ T cells48 and failed Icos mRNA repression is associated with accumulation of Tfh cells in lupus-prone Roquin-1/2 mutant mice6,19,49. We observed ICOS overexpression in Mir146a−/− naïve, effector CD4+, Tfh cells (Fig. 5a) and Tregs (Supplementary Fig. 7). Members of the SLAM family of receptors (SLAMF1, SLAMF6 and CD84) mediate prolonged interactions between activated antigen-specific T and B cells and signal via the recruitment of SAP to promote terminal Tfh-cell differentiation50. SLAMF1, SLAMF6 and CD84 were upregulated in Mir146a−/− naïve and effector CD4+ T cells compared with their wild-type counterparts (Fig. 5a). CD84 was upregulated in Mir146a−/− Tfh cells (Fig. 5a), and SLAMF1 and CD84 were also upregulated in Mir146a−/− Tregs (Supplementary Fig. 7). IL-21R and IL6ST are subunits of receptors for the Tfh-cell-inducing cytokines IL-21 and IL-6, respectively. IL-21R was upregulated in Mir146a−/− naïve (Fig. 5a) and Tregs (Supplementary Fig. 7), while IL6ST was upregulated in Mir146a−/− naïve and effector CD4+ T cells (Fig. 5a), but not in Tfh cells (Fig. 5a) or Tregs (Supplementary Fig. 7).
Figure 5. MiR-146a directly represses multiple Tfh-cell mRNAs.
(a) Geometric mean fluorescence intensity (MFI) of STAT1, CXCR4, Notch1, ICOS, SLAMF1, SLAMF6, CD84, IL-21R and IL6ST on naïve (CD4+ CD44low), effector (CD4+ CD44high) and Tfh (CD4+ CXCR5high PD-1high Foxp3−) cells from mice lacking miR-146a (−/−) or wild-type littermates (+/+). Each symbol represents one mouse and the horizontal bars represent the median values. Data are representative of at least two independent experiments. Statistical significance was determined using a Mann–Whitney U-test. (b) Effects of miR-146a (open bars) and scramble (negative control) RNA (filled bars) expression on luciferase reporter constructs containing putative miR-146a target sites as relative luciferase activity (normalized to the Renilla control). Sequence specificity of miRNA–mRNA binding was confirmed by introducing mutations to the predicted target sites at regions complementary to the miR-146a seed region. Relative luciferase activity of cells transfected with miR-146a is expressed as a percentage of that transfected with the scramble RNA. Data are representative of at least two independent experiments. The heights of the bars represent the mean, and the error bars represent the s.d. of six technical replicates. Statistical significance was determined using unpaired Student’s t-test. NS, not significant, *P<0.05, **P<0.01, ****P<0.0001.
The putative targets that had not been previously validated (Slamf1, Slamf6, Cd84, Icos, Il6st and Il21r) were further tested for direct repression by miR-146a using luciferase reporter assays. The predicted target sequence from Stat1 revealed a 25% repression (Supplementary Fig. 8), comparable to that reported by Rudensky and co-workers24. Similar repression was observed using luciferase reporters containing the minimal target sequences from Icos, Slamf1, Slamf6 or Cd84. All of these targets were specifically repressed by miR-146a, but not by a ‘scramble’ miRNA control (a commercially available, random sequence miRNA mimic; Fig. 5b). Luciferase reporter constructs containing mutations in the sequences recognized by the miR-146a seed region were no longer repressed by miR-146a. This not only validated the sequence specificity of the miRNA–mRNA binding but also mapped the miRNA recognition sequence in the 3′UTR (Fig. 5b). By contrast, luciferase reporter constructs containing the predicted target sequences from Il21r and Il6st were not repressed by miR-146a (Fig. 5b). Hence, Icos, Slamf1, Slamf6 and Cd84, but not Il21r and Il6st, are likely to be novel direct targets of miR-146a.
Luciferase assays reveal targets of miRNAs when the miRNA is overexpressed in HEK293T cells. To assess which of the above targets are physiologically relevant and a direct consequence of miR-146a regulation in CD4+ T cells (naïve, effectors, Treg and Tfh cells), we assessed their protein expression in Ly5a+.Mir146a+/+: Ly5b+.Mir146a−/− mixed bone marrow chimeras. For this, the geometric mean fluorescence intensity was compared between Ly5a+ and Ly5b+ cells for each target. Of all targets, the only one that appeared to be controlled cell autonomously by miR-146a in all CD4+ subsets examined (including Tfh cells) was ICOS (Fig. 6a). None of the other targets was cell autonomously upregulated in Tfh cells, but some cell-autonomous upregulation was observed in naïve CD4+ T cells (Supplementary Fig. 9: CXCR4, SLAMF1, SLAMF6 and CD84), effector CD4+ T cells (Supplementary Fig. 9: CXCR4) and Tregs (Supplementary Fig. 10: CXCR4, SLAMF1, CD84 and STAT1). As expected from the luciferase assay results, neither IL6ST nor IL-21R were upregulated in a cell-intrinsic manner in Mir146a−/− T cells (Supplementary Figs 9 and 10). Collectively, our data suggest that miR-146a acts in T cells to directly repress the expression of Icos, Slamf1, Slamf6, Cd84 and Cxcr4, and of these, the only cell-autonomous effect in Tfh cells is exerted on Icos.
Figure 6. Increased ICOS and ICOSL expression is likely to be the major driver of Tfh-cell accumulation in the miR-146a-deficient mice.
(a) Geometric mean fluorescence intensity (MFI) of ICOS on naïve, effector CD4+ and Tfh cells from Mir146a+/+: Mir146a−/− and Mir146a+/+: Mir146a+/+ mixed bone marrow chimeras 16 weeks post reconstitution. (b) Percentages of Tfh cells in wild-type mice immunized with sheep red blood cells (SRBC) and treated with different amounts of anti-ICOSL blocking antibody on days 0, 3 and 6 post immunization. Each symbol represents the mean and error bars represent the s.d. of three biological replicates. (c) Percentages of Tfh, GC B and Treg cells in 9-week-old wild-type (+/+) and miR-146a-deficient mice (−/−) treated with 25 μg anti-ICOSL blocking antibody or PBS control on day 0, 3 and 6 following SRBC immunization. (d) Percentages of Tfh, GC B and Treg cells in 9-week-old Mir146a+/+ Icos+/+, Mir146a+/+ Icos+/−, Mir146a−/− Icos+/+, Mir146a−/− Icos+/− mice following SRBC immunization. (e,f) Adoptive transfer of purified naïve (CD44low CD25−) CD4+ T cells from either Mir146a+/− Icos+/+, Mir146a+/− Icos+/−, Mir146a−/− Icos+/+ or Mir146a−/− Icos+/−, together with Mir146a−/− Icos+/+ B cells and Mir146a+/+ Icos+/+ Tregs into Rag1−/− recipients according to the strategy shown in e. The recipient mice were immunized with SRBC 14 days after transfer and killed 7 days post immunization. (f) Percentages of Tfh and GC B cells in Rag1−/− recipient mice following adoptive transfer and SRBC immunization. (g,h) ICOSL MFI on GC B (g, left), non-GC B (g, right) and a mixture of CD11b+ and CD11c+ cells (h) from SRBC-immunized wild-type (+/+) or miR-146a-deficient (−/−) mice following stimulation with LPS, anti-CD40 antibody or isotype control. Heights of bars represent the mean and error bars represent s.d. of three technical replicates. Each symbol represents one mouse and the horizontal bars represent the median values. Data are representative of at least two independent experiments. Statistical significance was determined using paired Student’s t-test (a), unpaired Student’s t-test (b,g,h) or Mann–Whitney U-test (c,d,f). NS, not significant, *P<0.05, **P<0.01, ***P<0.001, ****P<0.0001.
Increased ICOS signalling in T cells causes Tfh-cell accumulation
In light of the profound cell-autonomous control of ICOS by miR-146a and the known roles of ICOS in promoting Tfh-cell differentiation19,48,51, we set out to formally test the contribution of excessive ICOS signalling to the expansion of Tfh cells in the absence of miR-146a. To this end, we first titrated anti-ICOSL antibody treatment in mice in search of a dose sufficiently low so as to partially suppress ICOS signalling without abolishing Tfh-cell formation in wild-type mice. Injections of 25 μg of anti-ICOSL antibody given every 3 days to wild-type mice after immunization with SRBCs had no significant effect on wild-type Tfh cells (Fig. 6b). We then used this anti-ICOSL treatment regime on 9-week-old Mir146a+/+ and Mir146a−/− mice immunized with SRBC. This treatment completely and consistently corrected the accumulation of Tfh and GC B cells in Mir146a−/− mice and still had no effect on wild-type mice (Fig. 6c). Of note, the increase in Tregs seen in Mir146a−/− mice was not consistently corrected by the anti-ICOSL treatment (Fig. 6c and Supplementary Fig. 11).
We also used a genetic approach to reduce but not eliminate ICOS expression in Mir146a−/− mice. Icos hemizygosity was achieved by crossing Mir146a−/− to Icos−/− mice to generate Mir146a−/− Icos+/− mice. As seen with the antibody blockade, lowering Icos gene dose reduced the percentage of Tfh and GC B cells in Mir146a−/− Icos+/− mice to the amount seen in wild-type Mir146a+/+ Icos+/+ mice (Fig. 6d). Importantly, Icos hemizygosity did not rescue Treg-cell accumulation in mice lacking miR-146a (Fig. 6d), suggesting that dysregulation of Treg number is not the cause of Tfh-cell accumulation. Thus, our data indicate that overexpression of ICOS is likely to be a prominent contributor to the Tfh-cell accumulation in Mir146a−/− mice.
To exclude the possibility that the observed effect of Icos gene dose reduction was a consequence of modulating ICOS expression on Tregs, we co-transferred sorted naïve (CD44low CD25−) CD4+ T cells from Mir146a+/− or Mir146a−/− mice, which were either Icos+/+ or Icos+/−, with Mir146a−/− Icos+/+ B cells and Mir146a+/+ Icos+/+ Tregs into Rag1−/− recipient mice (Fig. 6e). Mir146a+/− mice behaved identically to Mir146a+/+ with no evidence of spontaneous Tfh or GC B-cell accumulation (Supplementary Fig. 12a) or ICOS overexpression (Supplementary Fig. 12b). Fourteen days after adoptive transfer, T cells in all experimental groups showed comparable activation status as measured by the proportion of cells upregulating CD44 or CD25 (Supplementary Fig. 13). Following SRBC immunization, recipients of Mir146a−/− Icos+/− T cells had a ~50% reduction in the percentage of Tfh and GC B cells compared with those that received Mir146a−/− Icos+/+ T cells (Fig. 6f). Furthermore, halving the gene dose of Icos on Mir146a−/− T cells virtually corrected the Tfh-cell accumulation caused by the lack of miR-146a in T cells: there was no statistically significant difference between the Mir146a+/− Icos+/+ and Mir146a−/− Icos+/− groups. These results suggest that Icos overexpression in Tfh-cell precursors is indeed responsible, at least in part, for the Tfh-cell accumulation in miR-146a-deficient mice.
MiR-146a limits ICOSL in GC B cells and dendritic cells
The mixed bone marrow chimera results showed that despite the increase in total follicular T cells in the Ly5a+.Mir146a+/+: Ly5b+.Mir146a−/− chimeras, only the Ly5b+.Mir146a−/− GC B cells, and not the Ly5a+.Mir146a+/+ B cells, expanded (Fig. 3c,d). One of the plausible explanations is that miR-146a co-ordinately regulates a receptor–ligand pair in Tfh and GC B cells, so overexpression of both T-cell-expressed receptor and GC B-cell ligand (or vice versa) are required for Mir146a−/− GC B cells to accumulate. Since our data revealed ICOS as the most prominent target of miR-146a in Tfh cells, and T-cell-expressed ICOS interacts with B-cell-expressed ICOS ligand (ICOSL) at different stages of Tfh-cell and GC formation48,51, we assessed whether miR-146a also regulates ICOSL in B cells. Although we did not find any predicted miR-146a binding site in Icosl 3′UTR, previous studies have shown that Icosl transcription is regulated by NF-κB signalling52, which is partly controlled by miR-146a (refs 20, 21, 23).
To test whether miR-146a controls ICOSL expression in B cells, we sorted total B cells from Mir146a+/+ and Mir146a−/− mice, stimulated them in culture for 8 h using lipopolysaccharides (LPS) or anti-CD40 antibody, and compared ICOSL expression on either GC or non-GC B cells. No differences in ICOSL expression were observed in non-GC B cells under any conditions (Fig. 6g). On the other hand, the expression of ICOSL was consistently higher in Mir146a−/− GC B cells—known to be dependent on NF-κB signalling53,54,55—across all conditions (Fig. 6g). An increase in ICOSL expression was also observed in miR-146a-deficient CD11b+ and CD11c+ cells following in vitro culture in the absence or presence of LPS (Fig. 6h). These results suggest that overexpression of ICOSL on GC B cells and myeloid/dendritic cells are likely to enhance the effects of increased ICOS expression on Tfh-cell accumulation. They also suggest that ICOSL overexpression may be required for Mir146a−/− GC B-cell expansion when ICOS is concomitantly overexpressed in T cells.
Finally, we explored the role of other miR-146a targets that may contribute to the Tfh-cell accumulation. To this end, we investigated the consequences of genetic manipulation of STAT1 that has known Tfh-cell-promoting effects51 and SLAMF1. Mir146a−/− mice were crossed with mice carrying a loss-of-function mutation in the Stat1 gene (Stat1fae; MGI:3611892) to generate Mir146a−/− Stat1fae/+ mice (Supplementary Fig. 14). The percentages of Tfh and GC B cells in Mir146a−/− Stat1fae/+ mice were significantly lower than those in the Mir146a−/− Stat1+/+ mice, but remained about eightfold higher than those seen in wild-type mice (Supplementary Fig. 14). This result suggests that STAT1 contributes to the Tfh-cell accumulation in Mir146a−/− mice, perhaps by crippling Treg control of Tfh responses or by actions within Tfh cells and/or other cells. We also studied the effect of increased Slamf1 copy number in Tfh-cell formation using NOD.Nkrp1b.Tg(Slamf1)1 (SLAM A—6 copies of Slamf1) and NOD.Nkrp1b.Tg (Slamf1)2 (SLAM B—44 copies56) mice (Supplementary Fig. 15). No differences were identified in either Tfh-cell or GC formation. Thus, in addition to the regulation of ICOS, miR-146a also represses ICOSL and STAT1 expression, which collectively may contribute to the accumulation of Tfh cells in mice lacking miR-146a.
Discussion
We have shown that miR-146a, a microRNA highly expressed in Tfh cells, limits Tfh-cell number. Indeed, in the absence of miR-146a, there was spontaneous expansion of Tfh and GC B cells. MiR-146a emerges as the first miRNA to negatively regulate Tfh-cell accumulation. It therefore opposes the action of the miR-17~92 cluster, recently shown to promote Tfh-cell differentiation by repressing Pten, Phlpp2 and Rora mRNAs57,58.
Our luciferase assays, together with published reports, have shown that ectopic expression of miR-146a can lead to repression of multiple Tfh-cell-expressed mRNAs, including Icos, Slamf1, Slamf6, Cd84, Stat1, Cxcr4 and Notch1. Although all of the above targets were upregulated in T cells from miR-146a-deficient mice, ICOS was the only target upregulated cell autonomously in Tfh cells and the most robustly upregulated across all T-cell subsets examined. Strikingly, dampening ICOS-mediated signalling in Mir146a−/− mice, either by generating Mir146a−/− Icos+/− mice or by injecting a suboptimal dose of anti-ICOSL antibody, completely prevented miR-146a-driven Tfh-cell accumulation. Furthermore, adoptive transfer experiments in which only non-Tregs lacked one allele of Icos demonstrated that repressing ICOS signalling in Tfh-cell precursors themselves, rather than in Tregs, is required to control Tfh-cell number. This is consistent with previous reports showing that cell-autonomous Icos mRNA overexpression due to defective Roquin function contributes to excessive Tfh-cell number19.
We have also found that miR-146a acts cell autonomously in GC B cells to limit their number. Increased expression of ICOSL on GC B cells is a plausible contributor to their accumulation in the absence of miR-146a, and may explain why despite the striking increase of Mir146a−/− Tfh cells in Mir146a+/+: Mir146a−/− chimeras only Mir146a−/− GC B cells expanded. Limiting ICOSL signalling is likely to be important to prevent excessive B-cell activation. Indeed, mice overexpressing soluble ICOSL-Fc developed hypergammaglobulinemia and plasmacytosis59. Our results suggest that concomitant control of ICOS on Tfh cells and ICOSL on GC B may be necessary to control the number of GC B cells.
In addition to ICOS–ICOSL regulation, miR-146a-mediated repression of other T-cell-expressed targets might also contribute to limit Tfh cells. Our data suggest STAT1 is one such target: lowering the dose of STAT1 had a significant corrective effect on Tfh-cell accumulation. This effect is likely to, at least in part, be a consequence of STAT1 overexpression in Treg cells, including Tfr cells15, leading to impaired repressive ability24. In addition, STAT1 is known to function downstream of several Tfh-inducing cytokines, including IL-6, IL-12, IL-21 and IL-27 (refs 3, 51, 60), thus STAT1 overexpression may enhance the sensitivity of miR-146a-deficient T cells to stimulation by these cytokines and increase their propensity to differentiate along the Tfh lineage. Nevertheless, we did not observe an increase in IL-6-induced STAT1 phosphorylation in Mir146a−/− T cells. Another candidate that may contribute to the Tfh-cell accumulation in miR-146a-deficient mice is IL6ST. We found a subtle increase in IL6ST expression on Mir146a−/− T cells, although our luciferase assay data indicate that it is unlikely to be a direct target of miR-146a. We speculate that increased IL6ST expression in Mir146a−/− T cells might cause the observed heightened sensitivity of these T cells to IL-6 signalling as measured by STAT3 phosphorylation.
Although this work has focused on identifying Tfh cell-autonomous targets of miR-146a, the milder phenotype observed when only T cells lack miR-146a suggests that T-cell extrinsic factors also contribute to the accumulation of Tfh cells in animals lacking miR-146a. IL-6, which was increased in aged miR-146a-deficient mice20,23, did not appear to be upregulated in any of the cell types examined in 12- to 16-week-old mice, although a role of IL-6 derived from other haematopoietic cells and/or non-haematopoietic cells cannot be completely excluded. Furthermore, given the significant effect of IL-6R blockade treatment, it is possible that Mir146a−/− T cells or myeloid cells do produce increased IL-6 protein in vivo that would not have been detected by our transcriptional studies. On the other hand, antigen-presenting cells that are typically CD11c+ and/or CD11b+ did express increased amounts of ICOSL, which may enhance T-cell priming and exacerbate Tfh-cell accumulation in Mir146a−/− mice61. Tfr cells are required to control Tfh cells and GC B cells15,16,17 and have been shown to derive from Foxp3+ thymic Tregs, which are less functional in Mir146a−/− mice due to STAT1 overexpression24. Thus, despite their accumulation, it is likely that Tfr cells are also defective in Mir146a−/− mice and this may also exacerbate the spontaneous Tfh-cell accumulation. In addition, increased expression of the costimulatory molecules CD80 (ref. 62) and CD86 (ref. 63) and the reported enhanced sensitivity of T cells to antigen stimulation21 due to dysregulated NF-κB activation may also contribute to the Tfh-cell expansion. It has been reported previously that strong TCR signalling can skew the differentiation pathway of naïve CD4+ T cells towards the Tfh lineage64,65. Collectively, all of these miR-146a-mediated effects in various cell types are likely to act cooperatively to cause the accumulation of Tfh cells.
In conclusion, this report identifies miR-146a as an important negative regulator of Tfh and GC B-cell accumulation. It is likely that this contributes to maintaining immunological tolerance and preventing the autoimmune phenotypes observed in mice lacking this microRNA. These findings on miR-146a-mediated control of Tfh and GC B-cell number may therefore improve our understanding of human autoimmunity and inflammatory syndromes.
Note added in proof: A role for miR-146a in repressing miR-155-induced Tfh-cell accumulation was reported while this work was under consideration66.
Methods
Mice and immunizations
The mice used were C57BL/6 (B6), B6.Ly5a, B6.Rag1−/−, B6.Cd28−/−, B6.OT-II, B6.Mir146a−/− (ref. 20), B6.Stat1fae/+, B6.Icos+/− (Australian Phenomics Facility, ANU), NOD.Nkrp1b.Tg(Slamf1)1 (ref. 56) and NOD.Nkrp1b.Tg(Slamf1)2 (ref. 56; Immunogenetics Research Facility, James Cook University). Male and female mice were used in experiments at approximately equal ratio between the wild-type/untreated and mutant/treated groups. Mir146a−/− mice were provided by David Baltimore (Caltech, USA). Stat1-face (Stat1fae) mice were derived from an N-ethyl N-nitrosourea mutagenesis screen67 and kindly donated by Christopher C. Goodnow (ANU, Australia). Mir146a−/−, Mir146a−/− Stat1fae/+, NOD.Nkrp1b.Tg(Slamf1)1 (ref. 56) and NOD.Nkrp1b.Tg (Slamf1)2 (ref. 56) mice were taken down at 12 to 16 weeks old unless otherwise stated. All mice were housed under a specific pathogen-free environment. All experiments were approved by the Australian National University Animal Experimentation Ethics Committee and the James Cook University Animal Ethics Committee. To generate thymus-dependent GC responses, where mentioned, mice were immunized i.v. with 2 × 108 SRBCs (Applied Biological Products Management, Australia) and were taken down at day 7 post immunization.
Human tonsils
Human tonsils were obtained from consenting patients undergoing routine tonsillectomy at the Canberra Hospital (Canberra, Australia) with the approval from the institutional human research ethics committee. Tonsillar cells were isolated by mechanical disruption and Ficoll-Paque density gradient centrifugation.
Adoptive transfers
To study the kinetics of miR-146a expression in a thymus-dependent GC response, 5 × 105 sorted naïve OT-II CD4+ T cells were adoptively transferred into Ly5a+ wild-type B6 recipient mice via i.v. injection. To limit miR-146a deficiency to T cells, 1 × 105 sorted naïve Mir146a−/− OT-II or Mir146a+/+ OT-II T cells were adoptively transferred into Cd28−/− recipient mice. The recipients were immediately immunized i.p. with 100 μg OVA (Sigma) precipitated in alum. To study the contributions of miR-146a deficiency in T and B cells, 4 × 106 CD4+ T cells and 6 × 106 B cells from Mir146a+/+ or Mir146a−/− mice were adoptively transferred into Rag1−/− mice. The recipients were immunized with SRBC 14 days after transfer and they were killed 7 days post immunization. In addition, to examine the contribution of ICOS overexpression specifically in Tfh-cell precursors to the Tfh-cell accumulation, 7.5 × 105 CD44low CD25− CD4+ T cells from either Mir146a+/− Icos+/+, Mir146a+/− Icos+/−, Mir146a−/− Icos+/+ or Mir146a−/− Icos+/− donor mice were co-transferred with 3 × 106 Mir146a−/− Icos+/+ B cells and 7.5 × 104 Mir146a+/+ Icos+/+ Tregs into Rag1−/− recipients. The recipients were bled (to assess the activation status of T cells) at day 14 following adoptive transfer and immediately immunized with SRBC. The mice were then killed 7 days post immunization.
Antibodies
Antibodies and streptavidin conjugates used for flow cytometry were (respective vendors, catalogue numbers and dilutions at which they were used are shown in parentheses): anti-mouse Ly5a-Pacific Blue (Biolegend, 110722, 1:200), Ly5a-Alexa Fluor 700 (BD, 561235, 1:200), Ly5b-Alexa Fluor 700 (BD, 560693, 1:200), Ly5b-PerCP Cy5.5 (BD, 552950, 1:200), CD4-Alexa Fluor 700 (BD, 557956, 1:800), CD4-APC Cy7 (BD, 552051, 1:800), B220-PerCP (BD, 553093, 1:800), CXCR5-biotin (BD, 551960, 1:50), PD-1-PE (BD, 551892, 1:100), PD-1-Brilliant Violet 421 (BD, 562584, 1:400), CD25-Alexa Fluor 647 (BD, 563598, 1:100), CD44-APC (BD, 559250, 1:400), CD44-Alexa Fluor 700 (BD, 560567, 1:400), CD44-PE CF594 (BD, 562464, 1:400), Foxp3-FITC (eBioscience, 11-5773-82, 1:400), Foxp3-eFluor 450 (eBioscience, 48-5773-82, 1:400), GL-7-FITC (BD, 553666, 1:400), Fas-PE Cy7 (BD, 557653, 1:400), Vβ5.1/5.2-FITC (BD, 562087, 1:400), ICOSL-PE (Biolegend, 107405, 1:400), CD11b-APC Cy7 (BD, 557657, 1:200), CD11c-PerCP Cy5.5 (BD, 560584, 1:200), IFN-γ-FITC (BD, 554411, 1:400), IL-4-PE Cy7 (BD, 560699, 1:100), IL-17A-Alexa Fluor 700 (BD, 560820, 1:100), STAT1 (pY701)-Pacific Blue (BD, 560310, 1:5), STAT3 (pY705)-PE (BD, 562072, 1:20), STAT1-Alexa Fluor 647 (BD, 558560, 1:40), CXCR4-biotin (BD, 551968, 1:200), Notch1-PE (BD, 552768, 1:200), ICOS-PE (BD, 552146, 1:200), SLAMF1-PE (BD, 562651, 1:200), SLAMF6-Alexa Fluor 647 (BD, 561547, 1:100), CD84-PE (Biolegend, 122806, 1:200), IL-21R-APC (Biolegend, 131910, 1:100), gp130/IL6ST-APC (R&D Systems, FAB4681A-100, 1:100), CD31-PE (BD, 561073, 1:200), gp38-Alexa Fluor 488 (eBioscience, 53-5381-82, 1:200), CD21/35-biotin (BD, 562796, 1:200), anti-human CD4-APC Cy7 (BD, 557871, 1:800), CXCR5-Alexa Fluor 647 (BD, 558113, 1:100), PD-1-PE (eBioscience, 12-9969-42, 1:200), CD25-PE Cy7 (Biolegend, 356108, 1:100), CD44-Alexa Fluor 700 (BD, 560567, 1:400), and streptavidin-PE Cy7 (BD, 557598, 1:800). Monoclonal blocking antibody against ICOSL (clone MIL-5733)68 was provided by A.H. (DRFZ, Germany) and blocking antibody against IL-6R (catalogue number BE0047, 150 μg per mouse) was purchased from BioXCell. Mouse anti-CD40 (catalogue number BE0016-2, 10 μg ml−1) antibody was also purchased from BioXCell.
Bone marrow chimeras
Chimeras were generated by sub-lethally irradiating Rag1−/− recipient mice (500 cGy, X-rad) and reconstituting their immune system by transferring 4 × 106 bone marrow cells from donor mice. Chimeric mice were taken down 16 weeks post reconstitution unless otherwise stated.
Cell isolation and stimulation
Single-cell suspensions were prepared from spleens of unimmunized and/or immunized mice. Suspensions were prepared in RPMI 1640 media (Gibco) supplemented with 10% heat-inactivated foetal calf serum (FCS), 100 U penicillin–streptomycin (Gibco), 2 mM L-Glutamine (Gibco), 10 mM HEPES (Sigma), 0.1 mM non-essential amino acids (Gibco), 1 mM sodium pyruvate (Sigma), 55 μM 2-mercaptoethanol (Gibco) by mechanical disruption and gentle pipetting through 70 μm nylon mesh filters (BD Biosciences). For cytokine staining (IFN-γ, IL-4 and IL-17), cells were cultured for 4 h in a 37 °C/5% CO2 incubator with 50 ng ml−1 PMA (Sigma) and 1 μM ionomycin (Sigma) in the presence of Golgi Stop (BD Biosciences). For ICOSL induction, B220+ cells and combined CD11b+/CD11c+ cells were isolated from SRBC-immunized Mir146a+/+ or Mir146a−/− mice by negative and positive selection, respectively, using MACS columns (Miltenyi Biotec) according to the manufacturer’s instructions. About 5 × 105 B cells or CD11b+/CD11c+ cells were cultured per well in a 96-well plate (Corning) with different conditions: complete RPMI only (unstimulated), 1 μg ml−1 LPS from Escherichia coli (Sigma), 10 μg ml−1 rat IgG2a isotype control antibody (BioXCell) or 10 μg ml−1 rat anti-mouse CD40 antibody (BioXCell) for 8 h in a 37 °C/5% CO2 incubator. To induce IL-6 expression, purified CD11b+ cells from mixed bone marrow chimeras were stimulated overnight with 100 ng ml−1 LPS (Sigma) in a 37 °C/5% CO2 incubator. To study the responsiveness of T cells to IL-6 signalling, total splenocytes were stimulated with recombinant mouse IL-6 (20 ng ml−1) for 15 min in a 37 °C/5% CO2 incubator and levels of STAT3 phosphorylation were analyzed according to published protocols69.
Flow cytometry
For lymphocyte analysis, single-cell suspensions were prepared as described in the previous section. For myeloid cell-population analysis, spleens were treated with Collagenase P (Roche) and DNase I (Sigma) before preparing single-cell suspensions for staining. FDCs were isolated as described previously35. Briefly, spleens were digested in 1 ml dissociation media (complete RPMI, 1 mg ml−1 collagenase D, 200 μg ml−1 dispase and 100 μg ml−1 DNase I) for 30 min in a 37 °C, 5% CO2 incubator. Cell suspensions were then gently triturated through P1000 pipette tips and incubated for a further 30 min. Dissociated cells were washed with complete RPMI to remove excess digestive enzymes. To stain for surface markers, cells were incubated with each antibody layer for 30 min in the dark at 4 °C and washed thoroughly with ice-cold FACS buffer (2% FCS and 0.1% NaN3 in PBS) between each layer. Intracellular staining was performed using the Intracellular Staining Kit (eBioscience) according to the manufacturer’s instructions. Serum IL-6 was measured using CBA bead assays (BD Biosciences) following the manufacturer’s instructions. A BD LSRII Flow Cytometer with FACSDiva software were used for flow cytometry acquisition and FlowJo (Tree Star) was used for analysis. FACS Aria (BD Biosciences) was used for cell sorting, cells were collected in FCS. Tonsillar T cells were sorted using the following strategy: CD4+ CD25− CD44low CXCR5int/low PD-1int/low naïve T cells, CD4+ CD25− CD44high CXCR5int/low PD-1int/low non-Tfh effector cells, CD4+ CD25+ CD44int CXCR5int/low PD-1int/low Tregs, CD4+ CD25− CXCR5high PD-1high Tfh cells, CD4+ CD25+ CXCR5high PD-1high Tfr cells.
Luciferase reporter assay
HEK293T cells cultured in DMEM media (Gibco) supplemented with 10% heat-inactivated FCS, 100 U penicillin–streptomycin (Gibco) and 2 mM L-Glutamine (Gibco) in 24-well plates were co-transfected with pmirGLO (Promega) luciferase reporter plasmids containing either wild-type or mutated putative miR-146a minimal target sequences (Supplementary Table 1; designed according to pmirGLO’s protocol) and mirVana miR-146a mimic (Life Technologies) or mirVana negative control #1 (scramble; Life Technologies) using Lipofectamine 2000 (Life Technologies) according to the manufacturer’s instructions. The luciferase construct containing Stat1 3′UTR was purchased from Genecopoeia. After 24 h, 3 × 105 transfected cells were moved into opaque flat-bottom 96-well plates and incubated for another 24 h. Cells were then harvested and luciferase activity was assessed using Dual-Glo Luciferase Assay System (Promega) according to manufacturer’s protocol.
Microarray
Total RNA extraction from sorted tonsillar cells was done using the RNeasy Kit (QIAGEN) according to the manufacturer’s protocol. RNA quantity and quality was determined using a Nanodrop 1000 and an Agilent 2100 Bioanalyzer, respectively. Hybridization on Agilent Human miRNA Microarray (V1) Kit was performed at The Ramaciotti Center for Genomics (University of New South Wales, Australia). Triplicates (for human miRNA array) were collected and averaged in GeneSpring. The microarray data has been deposited in NCBI's Gene Expression Omnibus and is accessible through GEO Series accession number GSE64833.
Real-time PCR analysis
RNA was extracted using Trizol reagent (Life Technologies) according to the manufacturer’s protocol, and complementary DNA was synthesized using the miScript II reverse transcription Kit (QIAGEN) following the manufacturer’s instructions. Quantitative PCR to detect for miR-146a expression was performed using the miScript SYBR Green PCR Kit (QIAGEN) and miScript Primer Assays (QIAGEN) to amplify mature Mir146a and the U6 small nuclear RNA. Il6 and β-actin mRNAs were amplified using specific primer pairs purchased from Origene Technologies(Il6F: 3′- TACCACTTCACAAGTCGGAGGC -5′; Il6R: 3′- CTGCAAGTGCATCATCGTTGTTC -5′; ActinF: 3′- CATTGCTGACAGGATGCAGAAGG -5′; ActinR:3′- TGCTGGAAGGTGGACAGTGAGG -5′). Quantitative PCR was performed on an Applied Biosystems 7900 machine. The expression of Mir146a was normalized to U6, while the expression of Il6 was normalized to β-actin. The gene specific expression change was calculated using the 2−ΔΔCt method.
Statistical analysis
Statistical significance was determined using the non-parametric Mann–Whitney U-test unless specified otherwise. The sample size was chosen so that it would be possible to conduct Mann–Whitney test with at least 95% confidence. Consequently in most experiments, at least five mice per group were used. All statistical analyses were performed using the GraphPad Prism software. NS, not significant, *P<0.05, **P<0.01, ***P<0.001, ****P<0.0001.
Author contributions
A.P. designed and performed experiments, analyzed the data and wrote the manuscript. M.S. performed the human miRNA array and miR-146a-deficient mouse microarray experiments and analyzed the data. N.J.W., I.P., S.K.L. performed experiments. X.T.D. and M.A.J. performed experiments on Slamf1 transgenic mice, analyzed the data and reviewed the manuscript. A.H. contributed the anti-ICOSL antibody and reviewed the manuscript. J.L.Z. reviewed the manuscript. R.C. contributed data on the transcription factor analysis and reviewed the manuscript. V.A. performed experiments and reviewed the manuscript. C.G.V. designed experiments, wrote the manuscript and supervised the study.
Additional information
How to cite this article: Pratama, A. et al. MicroRNA-146a regulates ICOS–ICOSL signalling to limit accumulation of T follicular helper cells and germinal centers. Nat. Commun. 6:6436 doi: 10.1038/ncomms7436 (2015).
Supplementary Material
Supplementary Figures 1-15 and Supplementary Table 1
Acknowledgments
We thank M. Devoy and H. Vohra (ANU) for technical assistance on fluorescence-activated cell sorting, R.A. Sweet, R.J. Rigby, P. Gonzalez and I.A. Parish (ANU) for technical assistance in several experiments, H. Qi and D. Liu (Tsinghua University, China) for helpful discussions and for providing the stimulation protocols to induce ICOSL expression, and C. C. Goodnow and A. Yates (ANU) for kind provision and initial characterization of the Stat1fae mice. This work was funded by the National Health and Medical Research Council (NHMRC) program and project grants and Elizabeth Blackburn Fellowship to C.G.V., International Postgraduate Research Scholarship to A.P., NHMRC/MSRA Betty Cuthbert Fellowship to M.A.J., National Research Service Award F30HL110691 and UCLA/Caltech Medical Scientist Training Program to J.L.Z.
References
- Crotty S. Follicular helper CD4 T cells (TFH). Annu. Rev. Immunol. 29, 621–663 (2011) . [DOI] [PubMed] [Google Scholar]
- Craft J. E. Follicular helper T cells in immunity and systemic autoimmunity. Nat. Rev. Rheumatol. 8, 337–347 (2012) . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pratama A. & Vinuesa C. G. Control of TFH cell numbers: why and how? Immunol. Cell Biol. 92, 40–48 (2014) . [DOI] [PubMed] [Google Scholar]
- Schwickert T. A. et al. A dynamic T cell-limited checkpoint regulates affinity-dependent B cell entry into the germinal center. J. Exp. Med. 208, 1243–1252 (2011) . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Victora G. D. et al. Germinal center dynamics revealed by multiphoton microscopy with a photoactivatable fluorescent reporter. Cell 143, 592–605 (2010) . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vinuesa C. G. et al. A RING-type ubiquitin ligase family member required to repress follicular helper T cells and autoimmunity. Nature 435, 452–458 (2005) . [DOI] [PubMed] [Google Scholar]
- Linterman M. A. et al. Follicular helper T cells are required for systemic autoimmunity. J. Exp. Med. 206, 561–576 (2009) . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ozaki K. et al. Regulation of B cell differentiation and plasma cell generation by IL-21, a novel inducer of Blimp-1 and Bcl-6. J. Immunol. 173, 5361–5371 (2004) . [DOI] [PubMed] [Google Scholar]
- Odegard J. M. et al. ICOS-dependent extrafollicular helper T cells elicit IgG production via IL-21 in systemic autoimmunity. J. Exp. Med. 205, 2873–2886 (2008) . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ma J. et al. Increased frequency of circulating follicular helper T cells in patients with rheumatoid arthritis. Clin. Dev. Immunol. 2012, 827480 (2012) . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morita R. et al. Human blood CXCR5(+)CD4(+) T cells are counterparts of T follicular cells and contain specific subsets that differentially support antibody secretion. Immunity 34, 108–121 (2011) . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simpson N. et al. Expansion of circulating T cells resembling follicular helper T cells is a fixed phenotype that identifies a subset of severe systemic lupus erythematosus. Arthritis Rheum. 62, 234–244 (2010) . [DOI] [PubMed] [Google Scholar]
- He J. et al. Circulating precursor CCR7(lo)PD-1(hi) CXCR5(+) CD4(+) T cells indicate Tfh cell activity and promote antibody responses upon antigen reexposure. Immunity 39, 770–781 (2013) . [DOI] [PubMed] [Google Scholar]
- Szabo K. et al. Follicular helper T cells may play an important role in the severity of primary Sjogren's syndrome. Clin. Immunol. 147, 95–104 (2013) . [DOI] [PubMed] [Google Scholar]
- Linterman M. A. et al. Foxp3+ follicular regulatory T cells control the germinal center response. Nat. Med. 17, 975–982 (2011) . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chung Y. et al. Follicular regulatory T cells expressing Foxp3 and Bcl-6 suppress germinal center reactions. Nat. Med. 17, 983–988 (2011) . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wollenberg I. et al. Regulation of the germinal center reaction by Foxp3+ follicular regulatory T cells. J. Immunol. 187, 4553–4560 (2011) . [DOI] [PubMed] [Google Scholar]
- Kim H. J., Verbinnen B., Tang X., Lu L. & Cantor H. Inhibition of follicular T-helper cells by CD8(+) regulatory T cells is essential for self tolerance. Nature 467, 328–332 (2010) . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu D. et al. Roquin represses autoimmunity by limiting inducible T-cell co-stimulator messenger RNA. Nature 450, 299–303 (2007) . [DOI] [PubMed] [Google Scholar]
- Boldin M. P. et al. miR-146a is a significant brake on autoimmunity, myeloproliferation, and cancer in mice. J. Exp. Med. 208, 1189–1201 (2011) . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang L. et al. miR-146a controls the resolution of T cell responses in mice. J. Exp. Med. 209, 1655–1670 (2012) . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Etzrodt M. et al. Regulation of monocyte functional heterogeneity by miR-146a and Relb. Cell Rep. 1, 317–324 (2012) . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao J. L., Rao D. S., O'Connell R. M., Garcia-Flores Y. & Baltimore D. MicroRNA-146a acts as a guardian of the quality and longevity of hematopoietic stem cells in mice. Elife 2, e00537 (2013) . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lu L. F. et al. Function of miR-146a in controlling Treg cell-mediated regulation of Th1 responses. Cell 142, 914–929 (2010) . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luo X. et al. A functional variant in microRNA-146a promoter modulates its expression and confers disease risk for systemic lupus erythematosus. PLoS Genet. 7, e1002128 (2011) . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tang Y. et al. MicroRNA-146A contributes to abnormal activation of the type I interferon pathway in human lupus by targeting the key signaling proteins. Arthritis Rheum. 60, 1065–1075 (2009) . [DOI] [PubMed] [Google Scholar]
- Lofgren S. E. et al. Genetic association of miRNA-146a with systemic lupus erythematosus in Europeans through decreased expression of the gene. Genes. Immun. 13, 268–274 (2012) . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang G. et al. Serum and urinary cell-free MiR-146a and MiR-155 in patients with systemic lupus erythematosus. J. Rheumatol. 37, 2516–2522 (2010) . [DOI] [PubMed] [Google Scholar]
- Li J. et al. Altered microRNA expression profile with miR-146a upregulation in CD4+ T cells from patients with rheumatoid arthritis. Arthritis Res. Ther. 12, R81 (2010) . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakasa T. et al. Expression of microRNA-146 in rheumatoid arthritis synovial tissue. Arthritis Rheum. 58, 1284–1292 (2008) . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pauley K. M. et al. Upregulated miR-146a expression in peripheral blood mononuclear cells from rheumatoid arthritis patients. Arthritis Res. Ther. 10, R101 (2008) . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stanczyk J. et al. Altered expression of MicroRNA in synovial fibroblasts and synovial tissue in rheumatoid arthritis. Arthritis Rheum. 58, 1001–1009 (2008) . [DOI] [PubMed] [Google Scholar]
- Kuchen S. et al. Regulation of microRNA expression and abundance during lymphopoiesis. Immunity 32, 828–839 (2010) . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao J. L. et al. NF-kappaB dysregulation in microRNA-146a-deficient mice drives the development of myeloid malignancies. Proc. Natl Acad. Sci. USA 108, 9184–9189 (2011) . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harker J. A., Lewis G. M., Mack L. & Zuniga E. I. Late interleukin-6 escalates T follicular helper cell responses and controls a chronic viral infection. Science 334, 825–829 (2011) . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nurieva R. I. et al. Bcl6 mediates the development of T follicular helper cells. Science 325, 1001–1005 (2009) . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jarjour M. et al. Fate mapping reveals origin and dynamics of lymph node follicular dendritic cells. J. Exp. Med. 211, 1109–1122 (2014) . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Link A. et al. Fibroblastic reticular cells in lymph nodes regulate the homeostasis of naive T cells. Nat. Immunol. 8, 1255–1265 (2007) . [DOI] [PubMed] [Google Scholar]
- Labbaye C. et al. A three-step pathway comprising PLZF/miR-146a/CXCR4 controls megakaryopoiesis. Nat. Cell Biol. 10, 788–801 (2008) . [DOI] [PubMed] [Google Scholar]
- Victora G. D. et al. Identification of human germinal center light and dark zone cells and their relationship to human B-cell lymphomas. Blood 120, 2240–2248 (2012) . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Allen C. D. et al. Germinal center dark and light zone organization is mediated by CXCR4 and CXCR5. Nat. Immunol. 5, 943–952 (2004) . [DOI] [PubMed] [Google Scholar]
- Mei J., Bachoo R. & Zhang C. L. MicroRNA-146a inhibits glioma development by targeting Notch1. Mol. Cell Biol. 31, 3584–3592 (2011) . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Auderset F. et al. Notch signaling regulates follicular helper T cell differentiation. J. Immunol. 191, 2344–2350 (2013) . [DOI] [PubMed] [Google Scholar]
- Bauquet A. T. et al. The costimulatory molecule ICOS regulates the expression of c-Maf and IL-21 in the development of follicular T helper cells and TH-17 cells. Nat. Immunol. 10, 167–175 (2009) . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bossaller L. et al. ICOS deficiency is associated with a severe reduction of CXCR5+CD4 germinal center Th cells. J. Immunol. 177, 4927–4932 (2006) . [DOI] [PubMed] [Google Scholar]
- Warnatz K. et al. Human ICOS deficiency abrogates the germinal center reaction and provides a monogenic model for common variable immunodeficiency. Blood 107, 3045–3052 (2006) . [DOI] [PubMed] [Google Scholar]
- Dong C. et al. ICOS co-stimulatory receptor is essential for T-cell activation and function. Nature 409, 97–101 (2001) . [DOI] [PubMed] [Google Scholar]
- Xu H. et al. Follicular T-helper cell recruitment governed by bystander B cells and ICOS-driven motility. Nature 496, 523–527 (2013) . [DOI] [PubMed] [Google Scholar]
- Vogel K. U. et al. Roquin paralogs 1 and 2 redundantly repress the Icos and Ox40 costimulator mRNAs and control follicular helper T cell differentiation. Immunity 38, 655–668 (2013) . [DOI] [PubMed] [Google Scholar]
- Cannons J. L., Tangye S. G. & Schwartzberg P. L. SLAM family receptors and SAP adaptors in immunity. Annu. Rev. Immunol. 29, 665–705 (2011) . [DOI] [PubMed] [Google Scholar]
- Choi Y. S., Eto D., Yang J. A., Lao C. & Crotty S. Cutting edge: STAT1 is required for IL-6-mediated Bcl6 induction for early follicular helper cell differentiation. J. Immunol. 190, 3049–3053 (2013) . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hu H. et al. Noncanonical NF-kappaB regulates inducible costimulator (ICOS) ligand expression and T follicular helper cell development. Proc. Natl Acad. Sci. USA 108, 12827–12832 (2011) . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen G. et al. Regulation of the IL-21 gene by the NF-kappaB transcription factor c-Rel. J. Immunol. 185, 2350–2359 (2010) . [DOI] [PubMed] [Google Scholar]
- Pohl T. et al. The combined absence of NF-kappa B1 and c-Rel reveals that overlapping roles for these transcription factors in the B cell lineage are restricted to the activation and function of mature cells. Proc. Natl Acad. Sci. USA 99, 4514–4519 (2002) . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tumang J. R. et al. c-Rel is essential for B lymphocyte survival and cell cycle progression. Eur. J. Immunol. 28, 4299–4312 (1998) . [DOI] [PubMed] [Google Scholar]
- Jordan M. A. et al. Role of SLAM in NKT cell development revealed by transgenic complementation in NOD mice. J. Immunol. 186, 3953–3965 (2011) . [DOI] [PubMed] [Google Scholar]
- Kang S. G. et al. MicroRNAs of the miR-17 approximately 92 family are critical regulators of TFH differentiation. Nat. Immunol. 14, 849–857 (2013) . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baumjohann D. et al. The microRNA cluster miR-17 approximately 92 promotes TFH cell differentiation and represses subset-inappropriate gene expression. Nat. Immunol. 14, 840–848 (2013) . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoshinaga S. K. et al. T-cell co-stimulation through B7RP-1 and ICOS. Nature 402, 827–832 (1999) . [DOI] [PubMed] [Google Scholar]
- Ma C. S. et al. Functional STAT3 deficiency compromises the generation of human T follicular helper cells. Blood 119, 3997–4008 (2012) . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Choi Y. S. et al. ICOS receptor instructs T follicular helper cell versus effector cell differentiation via induction of the transcriptional repressor Bcl6. Immunity 34, 932–946 (2011) . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao J., Freeman G. J., Gray G. S., Nadler L. M. & Glimcher L. H. A cell type-specific enhancer in the human B7.1 gene regulated by NF-kappaB. J. Exp. Med. 183, 777–789 (1996) . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li J. et al. T suppressor lymphocytes inhibit NF-kappa B-mediated transcription of CD86 gene in APC. J. Immunol. 163, 6386–6392 (1999) . [PubMed] [Google Scholar]
- Fazilleau N., McHeyzer-Williams L. J., Rosen H. & McHeyzer-Williams M. G. The function of follicular helper T cells is regulated by the strength of T cell antigen receptor binding. Nat. Immunol. 10, 375–384 (2009) . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tubo N. J. et al. Single naive CD4+ T cells from a diverse repertoire produce different effector cell types during infection. Cell 153, 785–796 (2013) . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hu et al. miR-155 promotes T follicular helper cell accumulation during chronic, low-grade inflammation. Immunity 41, 605–619 (2014) . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Andrews T. D. et al. Massively parallel sequencing of the mouse exome to accurately identify rare, induced mutations: an immediate source for thousands of new mouse models. Open Biol. 2, 120061 (2012) . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frey O. et al. Inducible costimulator (ICOS) blockade inhibits accumulation of polyfunctional T helper 1/T helper 17 cells and mitigates autoimmune arthritis. Ann. Rheum. Dis. 69, 1495–1501 (2010) . [DOI] [PubMed] [Google Scholar]
- Schulz K. R., Danna E. A., Krutzik P. O. & Nolan G. P. Single-cell phospho-protein analysis by flow cytometry. Curr. Protoc. Immunol. Chapter 8, Unit 8.17–20 (2012) . [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary Figures 1-15 and Supplementary Table 1