Abstract
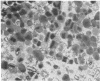
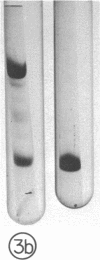
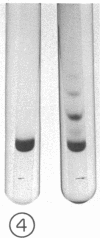
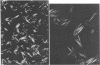
Guinea pig eosinophil granules contain a protein, the major basic protein (MBP), which accounts for more than half of the total granule protein, has a high content of arginine, and displays a remarkable tendency to form disulfide-linked aggregates. In this study we have purified a similar protein from human eosinophil granules and have compared the human MBP to the protein comprising the Charcot-Leyden crystal (CLC). Eosinophils from patients with various diseases were purified and disrupted, and the granule fraction was obtained. Examination of the granule fraction by transmission electron microscopy showed numerous typical eosinophil granules. Analyses of granule lysates by gel filtration and by polyacrylamide gel electrophoresis revealed the presence of peroxidase and MBP with properties similar to that previously found in guinea pig eosinophil granules. The human MBP had a molecular weight of 9,200, contained less than 1% carbohydrate, was rich in arginine, and readily formed disulfide-bonded aggregates. CLC were prepared from eosinophil-rich cell suspensions by homogenization in hypotonic saline. The supernates following centrifugation of cell debris spontaneously formed CLC. Analysis of CLC revealed the presence of a protein with a molecular weight of 13,000 containing 1.2% carbohydrate. The protein displayed a remarkable tendency to aggregate even in the presence of 0.2 M acetic acid. Human MBP and CLC protein differed in their molecular weights, carbohydrate compositions, and amino acid analyses. Mixtures of the MBP and the CLC protein yielded two bands in polyacrylamide gel electrophoresis. Neither eosinophil protein increased vascular permeability in the guinea pig skin or contracted the guinea pig ileum. The results indicate that the human MBP and the CLC are distinct substances with properties such that one cannot be derived from the other.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- ARCHER G. T., BLACKWOOD A. FORMATION OF CHARCOT-LEYDEN CRYSTALS IN HUMAN EOSINOPHILS AND BASOPHILS AND STUDY OF THE COMPOSITION OF ISOLATED CRYSTALS. J Exp Med. 1965 Jul 1;122:173–180. doi: 10.1084/jem.122.1.173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- BEAVER P. C. Biological and physiopathological ideas on eosinophilia. Observations on the nature of Charcot-Leyden crystals. Bull Soc Pathol Exot Filiales. 1962 Jul-Aug;55:471–476. [PubMed] [Google Scholar]
- BUDDECKE E., ESSELLIER A. F., MARTI H. R. Uber die chemische Natur der Charcot-Leydenschen Kristalle. Hoppe Seylers Z Physiol Chem. 1956 Sep 25;305(4-6):203–206. [PubMed] [Google Scholar]
- Chodirker W. B., Bock G. N., Vaughan J. H. Isolation of human PMN leukocytes and granules: observations on early blood diluion and on heparin. J Lab Clin Med. 1968 Jan;71(1):9–19. [PubMed] [Google Scholar]
- Day R. P. Eosinophil cell separation from human peripheral blood. Immunology. 1970 Jun;18(6):955–959. [PMC free article] [PubMed] [Google Scholar]
- Fairbanks G., Steck T. L., Wallach D. F. Electrophoretic analysis of the major polypeptides of the human erythrocyte membrane. Biochemistry. 1971 Jun 22;10(13):2606–2617. doi: 10.1021/bi00789a030. [DOI] [PubMed] [Google Scholar]
- Freireich E. J., Judson G., Levin R. H. Separation and collection of leukocytes. Cancer Res. 1965 Oct;25(9):1516–1520. [PubMed] [Google Scholar]
- Gleich G. J., Loegering D. A., Kueppers F., Bajaj S. P., Mann K. G. Physiochemical and biological properties of the major basic protein from guinea pig eosinophil granules. J Exp Med. 1974 Aug 1;140(2):313–332. doi: 10.1084/jem.140.2.313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gleich G. J., Loegering D. A., Maldonado J. E. Identification of a major basic protein in guinea pig eosinophil granules. J Exp Med. 1973 Jun 1;137(6):1459–1471. doi: 10.1084/jem.137.6.1459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gleich G. J., Loegering D. Selective stimulation and purification of eosinophils and neutrophils from guinea pig peritoneal fluids. J Lab Clin Med. 1973 Sep;82(3):522–528. [PubMed] [Google Scholar]
- HORNUNG M. Preservation recrystallization and preliminary biochemical characterization of Charcot-Leyden crystals. Proc Soc Exp Biol Med. 1962 May;110:119–124. doi: 10.3181/00379727-110-27442. [DOI] [PubMed] [Google Scholar]
- Liu T. Y., Chang Y. H. Hydrolysis of proteins with p-toluenesulfonic acid. Determination of tryptophan. J Biol Chem. 1971 May 10;246(9):2842–2848. [PubMed] [Google Scholar]
- MILES A. A., MILES E. M. Vascular reactions to histamine, histamine-liberator and leukotaxine in the skin of guinea-pigs. J Physiol. 1952 Oct;118(2):228–257. doi: 10.1113/jphysiol.1952.sp004789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mann K. G., Fish W. W. Protein polypeptide chain molecular weights by gel chromatography in guanidinium chloride. Methods Enzymol. 1972;26:28–42. doi: 10.1016/s0076-6879(72)26004-9. [DOI] [PubMed] [Google Scholar]
- el-Hashimi W. Charcot-Leyden crystals. Formation from primate and lack of formation from nonprimate eosinophils. Am J Pathol. 1971 Nov;65(2):311–324. [PMC free article] [PubMed] [Google Scholar]