Abstract
Drugs acting at D3 dopamine receptors have been suggested as medications for cocaine dependence. These experiments examined the effects of intravenously and orally administered buspirone, a D2-like receptor antagonist with high affinity for D3 and D4 receptors, on the relative reinforcing strength of cocaine in group-housed male cynomolgus monkeys. Use of socially housed monkeys permitted the assessment of whether social status, known to influence D2-like receptor availability, modulates the behavioral effects of buspirone. Buspirone was administered acutely to monkeys self-administering cocaine under a food–drug choice procedure in which a cocaine self-administration dose–effect curve was determined daily. When administered by either route, buspirone significantly decreased cocaine choice in dominant-ranked monkeys. In subordinate monkeys, however, i.v. buspirone was ineffective on average, and oral buspirone increased choice of lower cocaine doses. The effects of buspirone only differed according to route of administration in subordinate monkeys. Moreover, it is noteworthy that the effects of buspirone were similar to those of the D3 receptor-selective antagonist PG01037 and qualitatively different than those of less selective drugs that act at D2-like or serotonin (5-HT)1A receptors, suggesting a D3 and possibly D4 receptor mechanism of action for buspirone. Taken together, the data support the utility of drugs targeting D3/D4 receptors as potential treatments for cocaine addiction, particularly in combination with enriching environmental manipulations.
INTRODUCTION
Cocaine addiction persists as a worldwide public health problem for which there is no widely effective pharmacotherapy (Haile et al, 2012). A great deal of preclinical and clinical evidence has implicated D2-like dopamine receptors (composed of D2, D3, and D4 subtypes) in the abuse-related behavioral effects of cocaine (see, eg, Woolverton et al, 1984; Callahan et al, 1991; Caine et al, 2000). Drugs that nonselectively and indirectly stimulate D2-like receptors such as d-amphetamine and methamphetamine can reduce cocaine use (Grabowski et al, 2004; Mooney et al, 2009), but themselves possess high abuse liability. Negative results have been obtained using direct-acting D2-like receptor agonists (cf, Amato et al, 2011). D2-like receptor antagonists similarly have limited utility in treating stimulant abuse because of a general lack of efficacy and the emergence of motor side effects that decrease compliance (see, eg, Gawin, 1986; Farde et al, 1992; Barnes and Edwards, 1993; Ohuoha et al, 1997; Grabowski et al, 2000). In contrast, drugs that selectively interact with receptors of the D3 subtype have generated a great deal of interest in recent years as potential pharmacotherapies (Newman et al, 2005, 2012; Heidbreder, 2013; Paterson et al, 2014). Both post-mortem studies and brain imaging experiments have indicated that D3 receptors are higher in cocaine users than control subjects (Staley and Mash, 1996; Payer et al, 2014). Thus, it is hypothesized that D3 receptor antagonists may block the reinforcing and subjective effects of cocaine without producing extrapyramidal motor side effects. Indeed, D3 receptor antagonists have been shown to decrease the abuse-related effects of cocaine in a number of laboratory animal models of cocaine addiction (reviewed in Heidbreder and Newman, 2010).
In other studies, putative D3 receptor antagonists that decreased cocaine or methamphetamine self-administration did so only at doses that also decreased responding maintained by food (see, eg, Claytor et al, 2006; Martelle et al, 2007; Thomsen et al, 2008; Higley et al, 2011). However, it remains possible that the lack of selective effects on cocaine self-administration in these studies is due to the relatively high D2 receptor affinity and/or lack of sufficient selectivity for D3 versus D2 receptors of the pharmacological tools that were used. It should also be noted that studies in mice with genetically deleted D2 or D3 receptors support the prominence of D2 receptors in mediating the reinforcing effects of cocaine, and suggest that D3 and D4 receptors play a more subtle role (Caine et al, 2002, 2012).
Buspirone (Buspar), used clinically as an anxiolytic, is a serotonin (5-HT)1A receptor partial agonist and antagonist at D2, D3, and D4 receptors (Kula et al, 1994; Tallman et al, 1997; Wong et al, 2007; Bergman et al, 2013). Its relatively high affinity at D3 and D4 receptors (98 and 29 nM, respectively; Bergman et al, 2013) and modest selectivity (5- and 16-fold, respectively; Bergman et al, 2013) versus D2 receptors has focused attention on its use as a potential medication for cocaine abuse (Winhusen et al, 2012, 2014). Supporting its utility as a pharmacotherapy, recent studies in nonhuman primates demonstrated that acute intramuscular administration and chronic intravenous (i.v.) infusion of buspirone decreased cocaine self-administration under a fixed-ratio (FR) 30 (Bergman et al, 2013) and a second-order (specifically, FR2 (variable ratio 16:S)) schedule of reinforcement (Mello et al, 2013). However, a recent clinical trial indicated that buspirone was ineffective in maintaining abstinence in treatment-seeking cocaine abusers (Winhusen et al, 2014). To examine whether the predictive nature of preclinical models could be enhanced, a recent study in monkeys showed that 5 days of treatment with buspirone did not decrease cocaine self-administration when studied under a food–cocaine choice paradigm, consistent with the clinical trials findings (John et al, 2014). In the present experiments, we extended these results by characterizing the effects of buspirone on the reinforcing strength of cocaine relative to food using a choice procedure in socially housed male cynomolgus monkeys and compared these effects with those of a high-affinity (Ki=0.7, 93.3, and 375.0 nM at human hD3, D2, and D4 receptors, respectively) and highly selective (133-fold versus D2 and 540-fold versus D4 receptors) D3 receptor antagonist PG01037 (Grundt et al, 2007).
The monkeys used in this study were housed in groups of four monkeys per pen. We have demonstrated that monkeys that occupy dominant vs. subordinate positions in the social hierarchy differ in D2-like receptor availability as measured with positron emission tomography and in sensitivity to cocaine and other drugs that act at D2-like receptors (Morgan et al, 2002; Czoty et al, 2010; Czoty and Nader, 2013). However, because the radiotracers used in those imaging experiments do not discriminate between D2, D3, and D4 receptors, little is known about the influence of social rank on subtypes within the D2-like family. We hypothesized that the ability of buspirone to decrease cocaine choice may be enhanced when combined with environmental enrichment, in this case being a dominant monkey in a social group. Another noteworthy feature of the present study is that the effects of buspirone were characterized after both oral and i.v. administration. Although atypical in laboratory animal studies, assessment of oral administration of putative pharmacotherapies is critical because it represents the preferred route of medication administration in humans. A comparison of oral versus parenteral administration of buspirone in particular is warranted in light of recent in vivo brain imaging data suggesting that buspirone selectively blocks D3 versus D2 receptors when given orally, but blocks both receptors after intramuscular administration (Kim et al, 2014).
MATERIALS AND METHODS
Subjects
Sixteen adult male cynomolgus monkeys (Macaca fascicularis) served as subjects. Each monkey was fitted with an aluminum collar (Primate Products, Redwood City, CA) and trained to sit calmly in a standard primate chair (Primate Products). Monkeys were weighed weekly and fed enough food daily (Purina LabDiet Chow, St Louis, MO) and fresh fruit and vegetables to maintain healthy body weights without becoming obese. Body weights did not change significantly during these studies and were not different between dominant and subordinate monkeys. Water was available ad libitum in the home cage.
All monkeys lived in stable social groups of four monkeys per pen, in stainless steel cages (0.71 × 1.73 × 1.83 m; Allentown Caging Equipment, Allentown, NJ) with removable wire mesh partitions that separated monkeys into quadrants (0.71 × 0.84 × 0.84 m). Monkeys were separated daily for several hours during operant behavioral sessions and feeding. Social status had previously been determined for each monkey according to the outcomes of agonistic encounters using procedures similar to those described previously (see Kaplan et al, 1982; Czoty et al, 2009). Briefly, two observers separately conducted several 15-min observation sessions per pen. The monkey in each pen aggressing toward all others and submitting to none was ranked no. 1 (most dominant). The no. 2-ranked monkey aggressed toward all but the no. 1-ranked monkey and submitted to him. The no. 3-ranked monkey aggressed towards only the no. 4-ranked monkey and submitted toward the no. 1- and no. 2-ranked pen mates. The monkey designated most subordinate (no. 4) displayed a low frequency of aggressive behaviors and submitted to all other monkeys in the pen. Seven of the subjects were dominant (ie, ranked no. 1 or no. 2), eight were subordinate (ie, ranked no. 3 or no. 4), and one monkey (C-7426) was dominant during determination of effects of i.v. buspirone, but had become subordinate before testing p.o. buspirone. Animal housing and handling and all experimental procedures were performed in accordance with the 2003 National Research Council Guidelines for the Care and Use of Mammals in Neuroscience and Behavioral Research and were approved by the Animal Care and Use Committee of Wake Forest University. Environmental enrichment was provided as outlined in the Animal Care and Use Committee of Wake Forest University Non-Human Primate Environmental Enrichment Plan.
Catheter Implantation
Each monkey had been prepared with an indwelling venous catheter and subcutaneous vascular access port (VAP; Access Technologies, Skokie, IL) under sterile surgical conditions. An antibiotic (30 mg/kg kefzol, i.m.; Cefazolin sodium, Marsam Pharmaceuticals, Cherry Hill, NJ) was administered 1 h before surgery. Anesthesia was induced with ketamine (15 mg/kg, i.m.) and maintained with ketamine supplements. A catheter was inserted into a major vein (femoral or internal or external jugular) to the level of the vena cava. The distal end of the catheter was passed subcutaneously to a point slightly off the midline of the back, where an incision was made. The end of the catheter was attached to a VAP that was placed in a subcutaneous pocket formed by blunt dissection.
Apparatus and General Behavioral Procedures
Five days per week, monkeys were separated by partitioning the living space into quadrants. Next, each monkey was seated in a primate chair and placed into a ventilated, sound-attenuating chamber (1.5 × 0.74 × 0.76 m; Med Associates, St Albans, VT). The back of the animal was cleaned with chlorhexidine and the VAP was connected to an infusion pump (Cole-Parmer Instrument, Niles, IL) located outside the chamber via a 22-gauge Huber Point Needle (Access Technologies) and tubing. The pump was operated for ∼3 s to fill the port and catheter with the concentration of cocaine available for the session. Two photo-optic switches (Model 117-1007; Stewart Ergonomics, Furlong, PA) were located on one side of the chamber with a horizontal row of three stimulus lights positioned 14 cm above each switch. The switches were positioned to be easily within reach of the monkey seated in the primate chair. A food receptacle, above which was a single white stimulus light, was located between the switches and connected with a Tygon tube to a pellet dispenser (Med Associates) located on the top of the chamber for delivery of 1-g banana-flavored food pellets (Bio-Serv, Frenchtown, NJ). At the conclusion of each behavioral session, monkeys were returned to their home cages. Partitions were left in place for 60–90 min during which time monkeys were fed.
Food–Cocaine Choice
Monkeys had previously been trained to self-administer cocaine under a concurrent FR schedule of food and cocaine availability (Czoty and Nader, 2012). Completion of 30 consecutive responses on one switch (henceforth termed the ‘food switch') always resulted in delivery of a single food pellet; the yellow light above this switch was illuminated during pellet availability. Completion of an FR 30 on the other switch (henceforth termed the ‘drug switch') resulted in activation of the infusion pump and an injection of cocaine (0.003–0.1 mg/kg per injection). Availability of different cocaine doses, administered by varying the duration of pump activation, was associated with illumination of a different set of stimulus lights above the switch (see Czoty and Nader, 2012). If a response was emitted on the alternate switch before an FR was completed, the response requirement on the first switch was reset. Delivery of either reinforcer was accompanied by illumination of the red light above the corresponding switch (for 5 s after a pellet delivery or during an injection) and a subsequent period during which all lights remained off and responding had no scheduled consequences. The total timeout (TO) duration was 30 s.
Each daily session consisted of 5 components in which monkeys chose between food pellets and ascending doses of cocaine (ie, no injection and 0.003, 0.01, 0.03, and 0.1 mg/kg per injection cocaine in components 1–5, respectively). Each component ended when 10 total reinforcers had been earned or 20 min had elapsed, whichever came first; a 2-min TO followed each component. Ratio requirements for food and cocaine were adjusted for each monkey such that allocation of responding to the drug switch increased over the session as the available dose of cocaine increased. As observed in a previous study (see Table 2 in Czoty and Nader, 2012), there was not a significant difference in final ratio values for dominants and subordinates as determined by t-test. Responding was considered stable when ≤20% of reinforcers were earned on the drug switch when the alternative to food was no injection (component 1) or 0.003 mg/kg per injection cocaine (component 2) and ≥80% of reinforcers were earned on the drug switch when the alternative to food was 0.1 mg/kg per injection cocaine (component 5). An additional criterion of stability was observation of a dose-related increase in drug choice. A complete dose–effect curve was determined in each monkey each day, typically 5 days per week.
Once responding was stable, vehicle or a single dose of buspirone was administered i.v. (0.03–0.56 mg/kg, 3 min before session) or orally (3.0–17.8 mg/kg, 45 min before session; Dockens et al, 2011) to dominant (n=6) or subordinate (n=6 for i.v. buspirone; n= 7 for p.o. buspirone) monkeys. Most doses were examined at least twice, except for ineffective low doses and in cases when the highest doses were observed to disrupt behavior markedly in several monkeys. Following completion of studies with buspirone, PG01037 (1.0–5.6 mg/kg, i.v.) was tested in a subset of monkeys in whom buspirone was (n=4; 3 dominant and 1 subordinate) and was not (n=5; 2 dominant and 3 subordinate) effective in reducing cocaine choice. Drugs were typically tested on Tuesdays and Fridays; vehicle was administered on Thursdays.
Data Analysis
Dependent variables of interest were percent cocaine choice (defined as the number of reinforcers earned on the drug-paired switch divided by the total completed choices on both switches, multiplied by 100) and the number of food reinforcers and injections earned per component. Total session cocaine intake was also determined. Monkeys differed in sensitivity to buspirone and PG01037; thus, it was not informative to average data according to dose. However, the effects were qualitatively similar in all monkeys such that two doses were clearly identified that had progressively greater effects on behavior. In all cases, at least one low dose was tested and found to be without effect, and a high dose was identified that produced disruption of responding. For this purpose, a dose was deemed to have ‘disrupted' responding when the total number of reinforcers earned across the session decreased >50% from baseline. The two tested doses below the disruptive dose were termed ‘dose 1' (the lower dose) and ‘dose 2.' Initially, buspirone was administered in half-log increments. On occasions where a lower dose was without any effects on cocaine choice and a higher dose was deemed ‘disruptive,' the intervening quarter-log dose was tested. Doses that represented ‘dose 1' and ‘dose 2' in each monkey are shown in Table 1. For PG01037, data for the ‘best dose' are shown, which was the highest dose tested that was not disruptive. Data were analyzed using repeated-measures two-way analyses of variance (ANOVA), with cocaine dose and treatment drug dose (vehicle, dose 1, and dose 2) as factors, followed by multiple comparisons tests conducted with Prism 6 for Mac OS X software (Graphpad Software). Data for cocaine intake were analyzed using a one-way ANOVA across treatment drug conditions. In all cases, differences were considered significant when p<0.05.
Table 1. Buspirone Doses (mg/kg) That Constituted ‘Dose 1' and ‘Dose 2' by Each Route of Administration and the ‘Best Dose' of PG01037 (mg/kg) in Each Subject.
|
Intravenous |
Oral |
PG01037 | |||
|---|---|---|---|---|---|
| Dose 1 | Dose 2 | Dose 1 | Dose 2 | ||
| Dominant: no. 1 ranked | |||||
| C-7079 | 0.1 | 0.17 | 3.0a | 6.0 | 3.0a |
| C-6628 | 0.1a | 0.3a | 6.0 | 10.0a | 3.0a |
| C-7082 | Not tested | 3.0 | 6.0 | Not tested | |
| Dominant: no. 2 ranked | |||||
| C-6629 | 0.1 | 0.3a | 10.0 | 17.8a | 5.6 |
| C-6625 | 0.1 | 0.3 | 6.0 | 10.0 | 5.6 |
| C-7426b | 0.1 | 0.3a | Not tested | Not tested | |
| C-7081 | 0.1 | 0.3a | Not tested | Not tested | |
| C-7080 | Not tested | 3.0a | 6.0a | Not tested | |
| Subordinate: no. 3 ranked | |||||
| C-7083 | 0.1 | 0.3a | 3.0 | 6.0a | 3.0a |
| C-6527 | 0.1 | 0.17 | 3.0 | 6.0 | 5.6a |
| C-6216 | 0.1 | 0.3 | Not tested | Not tested | |
| C-7425 | 0.1 | 0.3 | 3.0 | 6.0a | 5.6 |
| C-7061 | Not tested | 3.0 | 6.0 | Not tested | |
| Subordinate: no. 4 ranked | |||||
| C-6526 | 0.03 | 0.1 | Not tested | Not tested | |
| C-6955 | 0.1 | 0.3 | 3.0 | 6.0 | 3.0 |
| C-7898 | Not tested | 3.0 | 6.0 | Not tested | |
| C-7426b | Not tested | 6.0 | 10.0 | 3.0a | |
Doses that ‘affected' cocaine choice were operationally defined as an increase in the ED50 of at least 0.25 log units.
C-7426 was dominant when buspirone was tested i.v., but had become subordinate before it was tested p.o. and was subordinate during PG01037 testing.
Drugs
The (−)-Cocaine HCl was supplied by the National Institute on Drug Abuse (Bethesda, MD) and dissolved in sterile 0.9% saline. Buspirone was obtained from Sigma-Aldrich (St Louis, MO) and was dissolved in sterile water. When administered orally, the appropriate volume of buspirone solution was mixed in mashed banana that had been placed in a small paper cup and given to the monkey. The animal was observed to assure that he had eaten all of the banana. PG01037 was synthesized as described in Grundt et al (2007) at the National Institute on Drug Abuse-Intramural Research Program and dissolved in a vehicle of 40% β-cyclodextrin in sterile water.
RESULTS
Experiment 1: Effects of I.V. Buspirone on Food–Cocaine Choice in Dominant and Subordinate Monkeys
Under vehicle treatment conditions, the frequency of cocaine choice increased in a dose-related manner, with cocaine preference obtained at 0.03 and 0.1 mg/kg per injection cocaine in dominant and subordinate monkeys (Figure 1, open symbols). Under baseline conditions, there were no differences in cocaine choice between dominant and subordinate monkeys. In all the statistical analyses described below except two, a main effect of cocaine dose was observed (all p<0.01). Both dominant and subordinate monkeys typically completed all available trials, except when the highest cocaine dose was studied (Figure 2, top panels). There was a shift in allocation of responding from exclusively food when no injection or a low cocaine dose (0.003 mg/kg per injection) was available to exclusively cocaine when high doses (0.03–0.1 mg/kg per injection) were available (Figure 2, middle and lower panels). There were no differences between social ranks under baseline conditions.
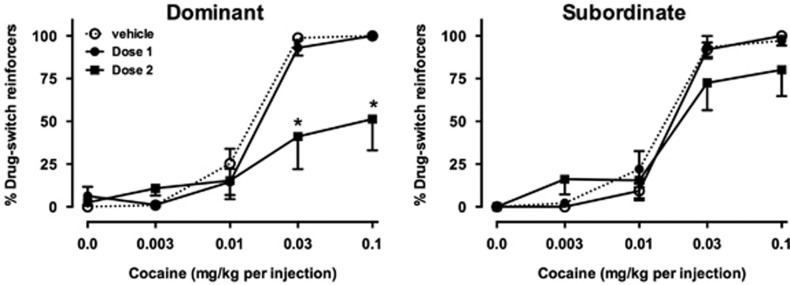
Figure 1.
Effects of intravenous vehicle or buspirone on cocaine choice in dominant (left, n=6) and subordinate (right, n=6) monkeys. Ordinates, percent of reinforcers earned on cocaine-associated switch. Abscissae, dose of cocaine (mg/kg) available as an alternative to a food pellet. Data represent mean±SEM. *P<0.05 compared with data obtained after saline administration at the same cocaine dose.
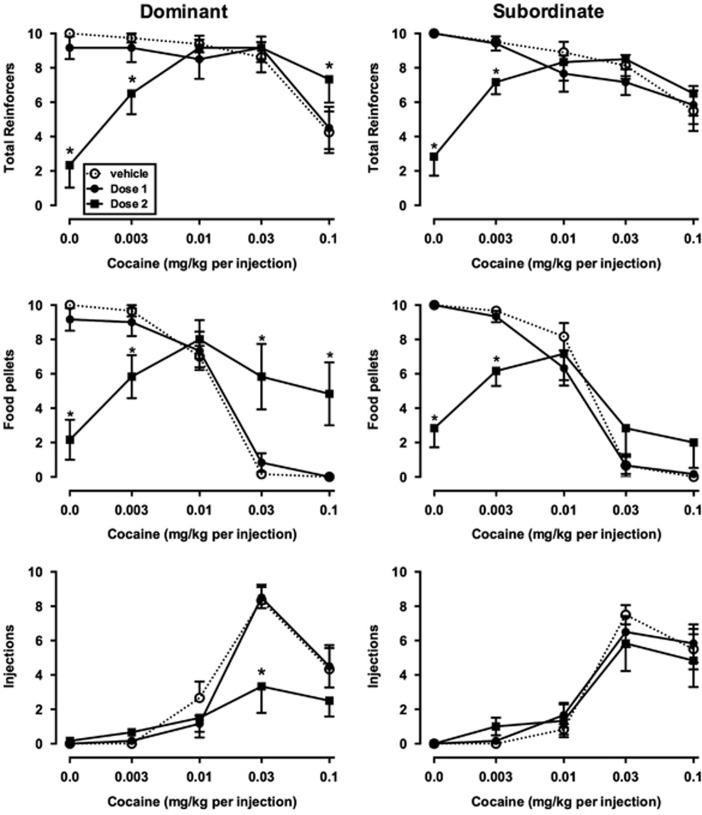
Figure 2.
Effects of intravenous vehicle or buspirone on total reinforcers earned (top row), food pellets earned (middle row), and injections received (bottom row) in dominant (left column, n=6) and subordinate (right column, n=6) monkeys. Ordinates, number of respective reinforcer type earned. Otherwise, as in Figure 1.
In dominant monkeys, i.v. buspirone significantly decreased choice of higher cocaine doses (Figure 1, left panel). There was a significant main effect of buspirone dose (F2, 10=4.20, p<0.05) and a significant interaction (F8, 40=7.13, p<0.0001), with post hoc analyses indicating that choice of the highest two doses of cocaine decreased significantly compared with vehicle treatment (p<0.0001). There was also a main effect of buspirone dose on total reinforcers (F2, 10=8.89, p<0.001; Figure 2, top left), and significant interactions between buspirone dose and cocaine dose for total reinforcers (F8, 40=17.09, p<0.0001), food pellets earned (F8, 40=16.92, p<0.0001), and injections received (F8, 40=5.93, p<0.0001). The higher buspirone dose significantly decreased the number of food pellets earned early in the session (Figure 2, center left panel), but significantly increased the number of food pellets received during the last two components of the session. During these components, monkeys received fewer cocaine injections (Figure 2, bottom left panel), with the effect reaching significance during availability of 0.03 mg/kg cocaine (p<0.0001).
In subordinate monkeys, although choice of higher doses was decreased by i.v. buspirone in some animals, on average this effect did not reach statistical significance (Figure 1, right panel). However, because of decreases in behavior early in the session, there were significant main effects of buspirone on total reinforcers (F2, 10=7.59, p<0.01) and food reinforcers (F2, 10=5.08, p<0.05) earned, but not cocaine injections (Figure 2, right column).
Experiment 2: Effects of Oral Buspirone on Food–Cocaine Choice in Dominant and Subordinate Monkeys
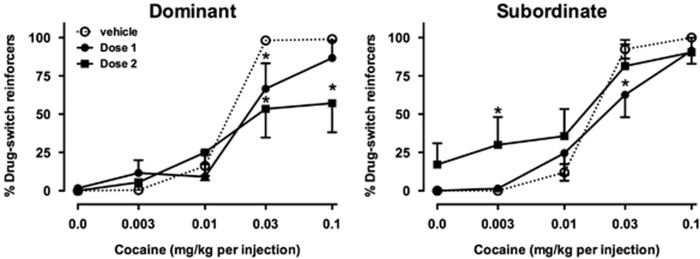
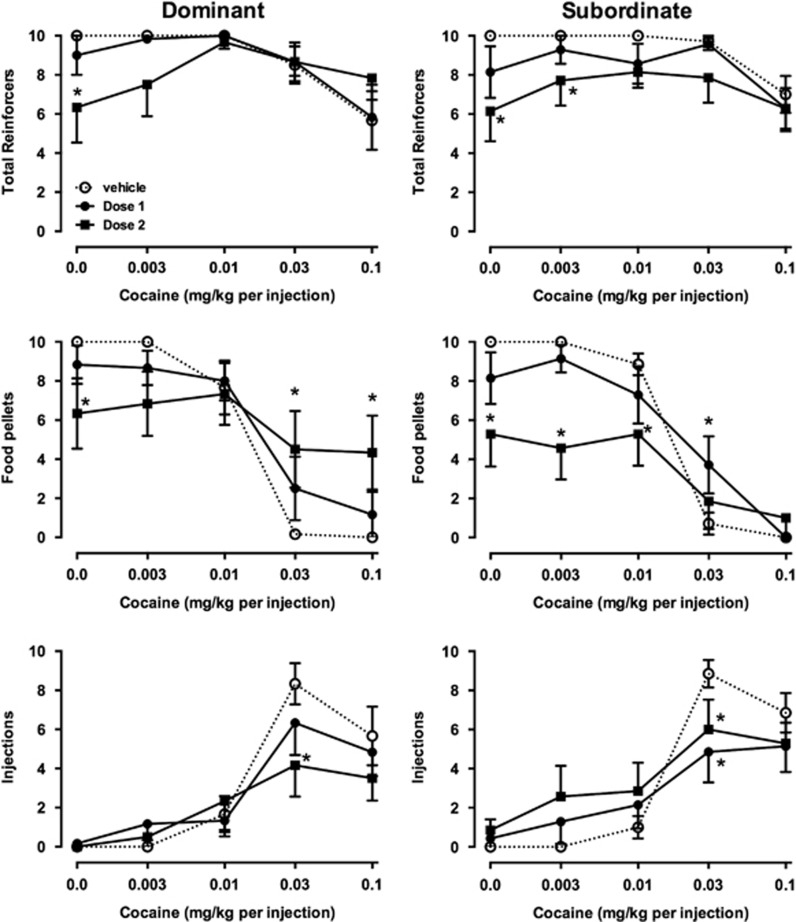
Effects of orally administered buspirone in dominant monkeys were similar to those of i.v. buspirone. As observed after i.v. buspirone, a high buspirone dose given orally significantly decreased choice of higher cocaine doses (Figure 3, left). Although there was no main effect of buspirone on choice, the interaction between buspirone dose and cocaine dose was significant (F8, 40=2.96, p<0.05). The effects of oral buspirone on reinforcers earned (Figure 4, left column) were also qualitatively similar to those of i.v. buspirone. A higher dose of buspirone significantly decreased the number of food pellets earned early in the session but increased pellets earned later in the session, at which time fewer injections were received. A significant interaction was found between buspirone dose and cocaine dose on food pellets (F8, 40=3.94, p<0.01). Qualitatively, the only differences between oral and i.v. buspirone treatment in dominant monkeys was that orally administered buspirone produced less decreases in food pellets earned early in the session, and that the lower dose (dose 1) was effective after oral administration, whereas it was without effect after i.v. administration.
Figure 3.
Effects of orally administered vehicle or buspirone on cocaine choice in dominant (left, n=6) and subordinate (right, n=7) monkeys. Otherwise, as in Figure 1.
Figure 4.
Effects of orally administered vehicle or buspirone on total reinforcers earned (top row), food pellets earned (middle row), and injections received (bottom row) in dominant (left, n=6) and subordinate (right, n=7) monkeys. Otherwise, as in Figure 1.
In subordinates, the effects of oral buspirone differed from the effects of i.v. buspirone. A lower dose, given orally, significantly decreased choice of 0.03 mg/kg per injection cocaine and a higher dose increased choice of lower doses of cocaine. In contrast to i.v. buspirone, a significant interaction was observed between oral buspirone and cocaine (F8, 48=p<0.05). There was a significant main effect of buspirone dose on total reinforcers (F2, 12=10.23, p<0.01), and a significant interaction on food pellets earned (F8, 48=5.08, p<0.0001). Like i.v. buspirone, oral buspirone decreased food pellets earned early in the session, but in contrast to i.v. buspirone, oral buspirone nonsignificantly increased the number of injections earned early in the session and significantly decreased injections of 0.03 mg/kg cocaine.
Effects of Buspirone on Cocaine Intake
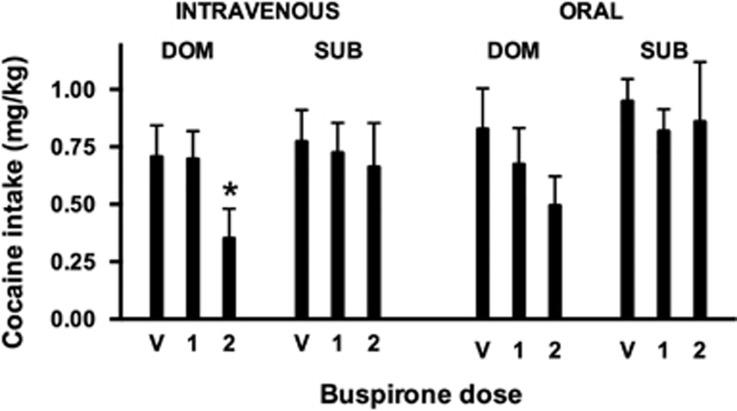
Generally, the interaction between social rank and buspirone on cocaine choice was paralleled by their interaction on cocaine intake (Figure 5). Both i.v. and oral buspirone decreased cocaine intake in dominant monkeys, although the effect reached significance only for i.v. administration (F2, 10=4.57, p<0.05); cocaine intake was unaffected by buspirone in subordinate monkeys. The variability in these data are, in part, a reflection of the noteworthy observation that not all animals of a given social rank responded similarly to buspirone treatment, as we have observed in prior studies with socially housed monkeys (see, eg, Czoty and Nader, 2012, 2013). In this study, buspirone decreased cocaine choice in 4 of 6 dominant monkeys after i.v. administration and 4 of 6 dominant monkeys after oral administration; it was not necessarily the same 4 monkeys that were affected (defined as a shift in the ED50 of 0.25 log units). A much lower proportion of subordinate monkeys were affected by buspirone when given i.v. (1 of 6) or orally (2 of 7).
Figure 5.
Effects of intravenous and oral buspirone on cocaine intake during food–drug choice sessions in dominant and subordinate monkeys. Ordinate, intake (mg/kg per session); abscissa, buspirone dose. V, vehicle; 1, dose 1; 2, dose 2. *P<0.05 compared with respective vehicle treatment.
Experiment 3: Effects of I.V. PG01037 on Food–Cocaine Choice in Dominant and Subordinate Monkeys
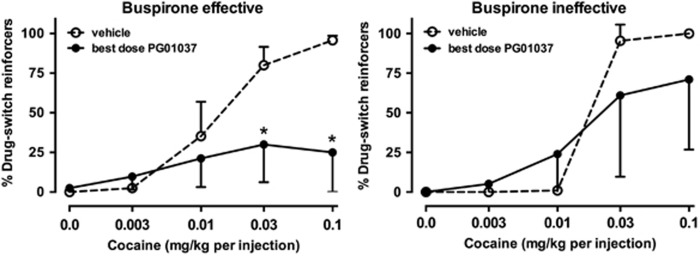
A subset of monkeys was also tested with the DA D3 receptor antagonist PG01037. On average, i.v. administration of PG01037 decreased cocaine choice in monkeys in whom i.v. buspirone had been effective, but did not significantly affect cocaine choice in monkeys in whom buspirone was ineffective (Figure 6). In the monkeys positively affected by buspirone (Figure 6, left), the cocaine choice curve was shifted to the right and down in three of four monkeys. A significant interaction was observed between PG01037 and cocaine dose (F4, 12=6.20, p<0.01) with post hoc analyses indicating that choice of the highest two doses of cocaine decreased significantly compared with vehicle treatment (p<0.05). In monkeys in whom buspirone was ineffective in altering cocaine choice (Figure 6, right), PG01037 did not, on average, alter cocaine choice. In these five monkeys, PG01037 either shifted the curve to the left (C-6625, C-7425, and C-6955) or moderately rightward (C-6527 and C-7079), resulting in a large amount of variability and, on average, no significant effect for the group. The likelihood that PG01037 would decrease cocaine choice was not influenced by social status.
Figure 6.
Effects of intravenous PG01037 on cocaine choice in monkeys in whom cocaine choice was decreased (left) or was unaffected (right) by intravenous buspirone. Otherwise, as in Figure 1.
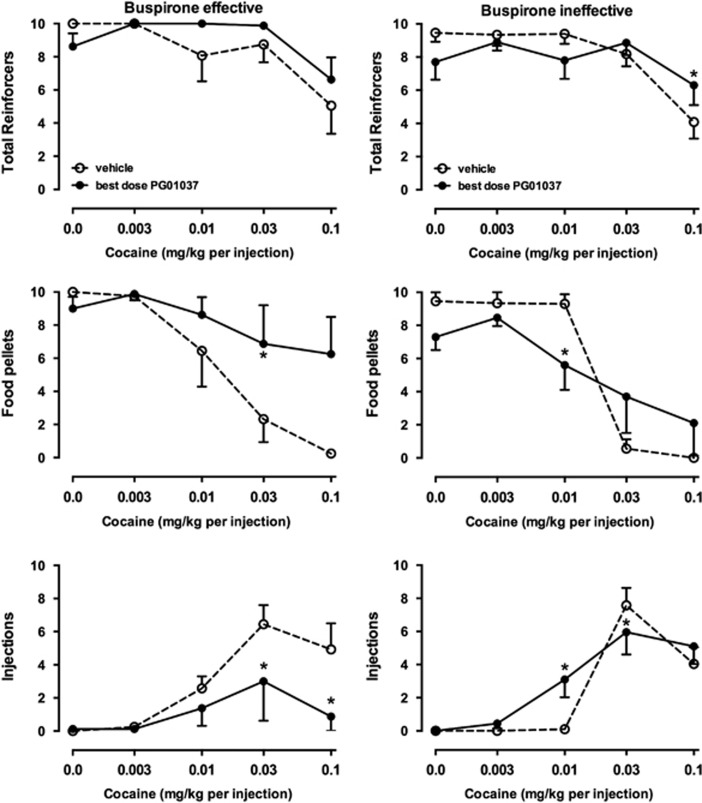
The pattern of effects of PG01037 on the numbers of reinforcers earned reflects this differential effect on cocaine choice. In monkeys that were affected by buspirone (Figure 7, left panels), there was a statistically significant effect of PG01037 pretreatment on injections (F4, 12=3.52, p<0.05) and an effect on food reinforcers that approached significance (F4, 12=3.16, p=0.054). Consistent with decreases in cocaine choice (Figure 6, left), PG01037 increased food reinforcers and decreased injections delivered when higher doses of cocaine were available. In contrast, effects of PG01037 were more variable in monkeys in whom buspirone was ineffective (Figure 7, right panels). Biphasic effects were observed for both food pellets and injections delivered, resulting in significant interactions (F4, 16=4.40 and F4, 16=26.19, respectively, both p<0.05.
Figure 7.
Effects of intravenous vehicle or PG01037 on total reinforcers earned (top row), food pellets earned (middle row), and injections received (bottom row) in monkeys in which buspirone was effective (left column, n=4) and ineffective (right column, n=5) in decreasing cocaine choice. Ordinates, number of respective reinforcer type earned. Otherwise, as in Figure 1.
DISCUSSION
The primary goal of these studies was to determine whether the effects of buspirone on food–cocaine choice could be influenced by the social environment. A second goal was to examine whether the behavioral effects of buspirone were influenced by route of drug administration. In dominant-ranked monkeys, buspirone administered i.v. or orally shifted allocation of responding from higher doses of cocaine to food. In contrast, i.v. buspirone did not affect cocaine choice in subordinate monkeys up to doses that decreased total trials per session, whereas biphasic effects were observed following oral buspirone. In the context of a recent clinical trial showing that buspirone was ineffective in maintaining abstinence in treatment-seeking cocaine abusers (Winhusen et al, 2014), the present findings suggest that improvement in social context may enhance the clinical utility of buspirone. A third goal was to determine whether D3 receptor-mediated mechanisms contributed to the effects of buspirone by comparing its effect with that of a highly selective D3 receptor antagonist, PG01037. Taken together with previous studies of cocaine choice that examined effects of drugs acting nonselectively at DA D2-like receptors and others at 5-HT1A receptors, the data support a D3 receptor mechanism for the effects of buspirone, with possible involvement of D4 receptors.
Earlier research in individually housed monkeys examined effects of acute and chronic parenteral administration of buspirone on cocaine self-administration under a simple FR and a second-order schedule of reinforcement (Bergman et al, 2013; Mello et al, 2013) and reported positive outcomes, which were not borne out in a recent clinical trial (Winhusen et al, 2014). It has been hypothesized that choice paradigms are more predictive of clinical efficacy (Banks and Negus, 2012), and hence our recent findings in individually housed monkeys showing negative effects of buspirone supported this hypothesis (John et al, 2014). Data from subordinate monkeys treated acutely with i.v. buspirone also reported no decreases in cocaine choice. The major finding from this study was that monkeys living in what has been hypothesized to be an enriched environment (Nader et al, 2012a) showed decreases in cocaine choice following acute buspirone administration. As in previous studies (Bergman et al, 2013; Mello et al, 2013), buspirone exhibited behavioral effects that interfered with responding early in the experimental session. Such effects on schedule-controlled behavior are not uncommon for DA receptor antagonists and likely reflect motor-disrupting effects (which were more pronounced at higher doses) rather than a decrease in the reinforcing effects of food pellets. Once these effects dissipated later in the session, a clear reduction of cocaine choice was revealed. This shift in allocation of behavior away from obtaining drug and toward obtaining another reinforcer supports further research into the potential utility of buspirone and other D3/D4 receptor antagonists as medications for cocaine dependence.
As mentioned above, the food–cocaine choice studies in individually housed rhesus monkeys and socially housed subordinate monkeys support the negative findings observed in a recent clinical study (Winhusen et al, 2014), whereas socially housed dominant monkeys showed reductions in cocaine choice following buspirone administration. There are some important methodological considerations to be made when comparing preclinical results with those of clinical studies. The goal of the Winhusen et al (2014) study was relapse prevention, whereas this study examined reductions in cocaine self-administration. In addition, the treatment group in the study of Winhusen et al (2014) only received one dose of buspirone (60 mg per day), whereas dose was adjusted on an individual-subject basis in this study. Perhaps most relevant was that the people receiving buspirone were in an inpatient/residential area at the beginning of the trial, but were outpatient for most of the 15-week study. The present findings suggest that enriched environments may be necessary to show efficacy of buspirone.
Pharmacological Mechanism of Buspirone's Effects
Characterizing the specific receptor mechanisms responsible for the encouraging effects of buspirone will aid the development of buspirone and other drugs as medications. Buspirone is perhaps best known as a 5-HT1A receptor partial agonist, as this mechanism is likely responsible for its well-known anxiolytic effects (Peroutka, 1985). In addition, although the binding profile of buspirone demonstrates a modest selectivity for D3 and D4 receptors versus D2 receptors, the affinity at D2 receptors is not negligible (see, eg, Kula et al, 1994; Tallman et al, 1997). Comparison of the present data with previous studies examining the effects of D2-like receptor antagonists and a 5-HT1A partial agonist provides support for a D3/D4-based mechanism. Unlike the effects of buspirone in the present studies, D2-like receptor antagonists and low-efficacy agonists, including aripiprazole, chlorpromazine, haloperidol, flupenthixol, and eticlopride, have been shown to increase choice of low doses of cocaine without decreasing choice of higher doses (Woolverton and Balster, 1981; Negus, 2003; Czoty and Nader, 2013). The effects of buspirone are also unlike those of the 5-HT1A receptor partial agonist (±)-8-hydroxy-2-(di-N-propylamino)tetralin (8-OH-DPAT) that also increased choice of low cocaine doses in cynomolgus monkeys (Czoty et al, 2005b). Moreover, another 5-HT1A receptor partial agonist, gepirone, was ineffective in decreasing cocaine self-administration under an FR 10 schedule of reinforcement (Gold and Balster, 1992). Finally, we found that the D3 receptor subtype-selective antagonist PG01037 decreased cocaine choice in the same monkeys in whom buspirone was effective, whereas PG01037 did not consistently alter cocaine choice in monkeys that were unaffected by buspirone. It is also noteworthy that the early-session decreases in food reinforcers earned following buspirone treatment were not observed after PG01037 treatment. This difference could suggest that D2 receptor stimulation is responsible for the decreases in responding observed after buspirone. Although the present experiments cannot completely rule out a contribution of 5-HT1A stimulation or D2/D4 antagonism, the similarity in effects of buspirone and PG01037 and the dissimilar effects of buspirone versus 5-HT1A agonists and less selective D2-like receptor antagonists suggest that the effects of buspirone on cocaine choice are mediated by D3 receptors. One caveat inherent to food–drug choice procedures is the possibility that a shift in responding from drug to food reflects increased reinforcing effects of food rather than decreased reinforcing effects of the drug alternative. However, whereas 5-HT1A receptor agonists can increase feeding (reviewed in Curzon, 1991), clinical data have reported either a decrease or lack of effect of buspirone on appetite (Enkelmann, 1991; Mohammadi et al, 2012).
Influence of Social Rank on the Effects of Buspirone
The present experiments were performed in monkeys that occupied both dominant and subordinate ranks in the social hierarchy. We have previously demonstrated that social rank can influence brain D2-like receptor availability; dominance was associated with an increase in the availability of D2-like receptors (Morgan et al, 2002; Czoty et al, 2010; Nader et al, 2012b). Dominant-ranked male monkeys were subsequently found to be less sensitive to the reinforcing effects of cocaine (Morgan et al, 2002; Czoty et al, 2005a), and to the effects of drugs acting directly at D2-like receptors that varied in intrinsic efficacy (Czoty and Nader, 2013). In this study, i.v. buspirone decreased cocaine choice in dominant monkeys, but was ineffective in subordinates. The results are reminiscent of the effects of aripiprazole, an extremely low-efficacy D2-like receptor agonist with ∼18- and ∼500- fold higher affinity for receptors of the D2 subtype versus D3 or D4 subtype, respectively (Lawler et al, 1999). When administered chronically, i.v. aripiprazole decreased cocaine choice in dominant monkeys but not subordinates (Czoty and Nader, 2013). Taken together, the results support the hypothesis that social dominance produces an increase in D2-like receptor densities (and/or a decrease in extracellular dopamine levels) that decreases sensitivity to cocaine and increases the ability of drugs acting at D2-like receptors to attenuate cocaine choice. Little is known about the effects of environmental variables on specific D2-like receptor subtypes; it may be that the environmental variables associates with the social hierarchy differentially affect D2, D3, and D4 receptors. Considering the well-documented ability of environmentally derived stress and enrichment to modulate cocaine self-administration (see, eg, Nader et al, 2012a; Bardo et al, 2013) the effects of environmental variables on D3 and D4 receptors warrant further study.
Influence of Route of Administration
One impetus for examining oral as well as i.v. buspirone was to better reflect the route by which it is given clinically. A second purpose was to examine a conclusion raised by a recent brain imaging study in baboons that indicated that buspirone blocked D2 and D3 receptors nonselectively when administered parenterally, but was a selective D3 receptor antagonist via the oral route (Kim et al, 2014). These differential effects were hypothesized to result from the generation of active metabolites after oral administration (ie, 5- and 6-hydroxybuspirone) that possess higher selectivity for the D3 versus D2 subtype than the parent compound. For example, Bergman et al (2013) demonstrated that compared with buspirone these metabolites had 2- to 8-fold lower affinity at D3 and D4 receptors but >10-fold lower affinity at D2 receptors. In the present studies, buspirone was equally effective orally and i.v. in dominant monkeys, although there were predictable differences in potency via the different routes. These data do not support a role for differential metabolism of p.o. versus i.v. buspirone in the effects of buspirone. In subordinates, however, oral buspirone displayed a poor profile of effects and i.v. buspirone was without effects. Although this result may indicate that buspirone is metabolized differently in subordinates compared with dominant monkeys, the similarity of oral buspirone in the present study to those of nonselective D2-like antagonists also does not support the hypothesis that oral buspirone is metabolized into D3-selective metabolites. The difficulty in reconciling the present behavioral results with the brain imaging data of Kim et al (2014) may arise from the differences in doses used and times after drug administration at which the behavioral or brain effects were measured. Alternatively, differences in the pattern of effects of i.v. and p.o. buspirone in dominant vs. subordinate monkeys may occur because exposure to chronic environmental stress vs. enrichment leads to differential regulation of D2-like receptor subtypes as discussed above. Further pharmacokinetic experiments and brain imaging studies with selective radiotracers (eg, [11C]PHNO, Wilson et al, 2005; Payer et al, 2014) will help to illuminate the mechanisms underlying the observed differences in the behavioral effects of buspirone and other D2-like drugs in dominant and subordinate monkeys.
Limitations and Clinical Implications
As described above, the finding of primary importance in this study was that buspirone decreased cocaine choice in dominant monkeys and this was consistent with the effects observed following administration of the D3 receptor antagonist PG01037. One limitation to the study was that buspirone and PG01037 were administered acutely, and hence it is possible that tolerance could have developed to the positive effects observed in dominant monkeys. A particular advantage of the experimental design was that the use of a choice procedure enabled us to demonstrate that buspirone not only decreased cocaine intake, but also shifted allocation of behavior away from cocaine and toward an alternative reinforcer. This outcome more clearly represents the end point of clinical treatment, making the results of food–drug choice procedures highly relevant and translational (cf, Banks and Negus, 2012), and supporting further research into buspirone and similar drugs as putative pharmacotherapies for cocaine addiction. Some more subtle aspects of the results are worthy of further comment. First, buspirone was clearly much more effective in decreasing cocaine choice in dominant versus subordinate monkeys. The fact that not all subjects were affected by the treatment reflects the clinical reality; in this case, the variability can be linked in part to social rank. Considering our conceptualization of the nonhuman primate social hierarchy as a continuum of chronic social stress in subordinate monkeys to conditions of environmental enrichment in dominants (see, eg, Nader and Czoty, 2005), these results suggest that buspirone may be most effective in conjunction with positive changes to the patient's environment. Second, the transient decrease in food-maintained responding early in the session, at the same buspirone doses that decreased responding later in the session, may manifest as sedation or other unpleasant effects in humans that would need to be managed to ensure compliance. Moreover, future studies should examine chronic treatment with buspirone to verify that the effects seen here in dominant monkeys would be sustained under those conditions.
FUNDING AND DISCLOSURE
The authors declare no conflict of interest.
Acknowledgments
We thank Dr Amy Newman, NIDA-IRP, for the supply of PG01037 and Michael Coller for excellent technical assistance. The research described in this study was supported by grants from the National Institute on Drug Abuse (DA 10584 and DA 12460).
References
- Amato L, Minozzi S, Pani PP, Solimini R, Vecchi S, Zuccaro P, et al. Dopamine agonists for the treatment of cocaine dependence. Cochrane Database Syst Rev. 2011;12:CD003352. doi: 10.1002/14651858.CD003352.pub3. [DOI] [PubMed] [Google Scholar]
- Banks ML, Negus SS.2012Preclinical determinants of drug choice under concurrent schedules of drug self-administration Adv Pharmacol Sci 2012281768. http://dx.doi.org/10.1155/20212/281768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bardo MT, Neisewander JL, Kelly TH. Individual differences and social influences on the neurobehavioral pharmacology of abused drugs. Pharmacol Rev. 2013;65:255–290. doi: 10.1124/pr.111.005124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barnes TR, Edwards JG.1993The side-effects of antipsychotic drugs. I. CNS and neuromuscular effectsIn: Barnes TR (ed). Antipsychotic Drugs and Their Side-Effects. Neuroscience Perspectives Academic Press: San Diego, CA; 213–247. [Google Scholar]
- Bergman J, Roof RA, Furman CA, Conroy JL, Mello NK, Sibley DR, et al. Modification of cocaine self-administration by buspirone (Buspar®): potential involvement of D3 and D4 receptors. Int J Neuropsychopharmacol. 2013;16:445–458. doi: 10.1017/S1461145712000661. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Caine SB, Negus SS, Mello NK, Bergman J. Effects of dopamine D1-like and D2-like agonists in rats trained to discriminate cocaine from saline: influence of experimental history. Exp Clin Psychopharmacol. 2000;8:404–414. doi: 10.1037//1064-1297.8.3.404. [DOI] [PubMed] [Google Scholar]
- Caine SB, Negus SS, Mello NK, Patel S, Bristow L, Kulagowski J, et al. Role of dopamine D2-like receptors in cocaine self-administration: studies with D2 receptor mutant mice and novel D2 receptor antagonists. J Neurosci. 2002;22:2977–2988. doi: 10.1523/JNEUROSCI.22-07-02977.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Caine SB, Thomsen M, Barrett AC, Collins GT, Grundt P, Newman AH, et al. Cocaine self-administration in dopamine D3 receptor knockout mice. Exp Clin Psychopharmacol. 2012;20:352–363. doi: 10.1037/a0029135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Callahan PM, Appel JB, Cunningham KA. Dopamine D1 and D2 mediation of the discriminative stimulus properties of d-amphetamine and cocaine. Psychopharmacology. 1991;103:50–55. doi: 10.1007/BF02244073. [DOI] [PubMed] [Google Scholar]
- Claytor R, Lile JA, Nader MA. The effects of eticlopride and the selective D3-antagonist PNU 99194-A on food- and cocaine-maintained responding in rhesus monkeys. Pharmacol Biochem Behav. 2006;83:456–464. doi: 10.1016/j.pbb.2006.03.007. [DOI] [PubMed] [Google Scholar]
- Curzon G. Effects of tryptophan and of 5-hydroxytryptamine receptor subtype agonists on feeding. Adv Exp Med Biol. 1991;294:377–388. doi: 10.1007/978-1-4684-5952-4_35. [DOI] [PubMed] [Google Scholar]
- Czoty PW, Gage HD, Nader MA. Differences in D2 dopamine receptor availability and reaction to novelty in socially housed male monkeys during abstinence from cocaine. Psychopharmacology. 2010;208:585–592. doi: 10.1007/s00213-009-1756-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Czoty PW, Gould RW, Nader MA. Relationship between social rank and cortisol and testosterone concentrations in male cynomolgus monkeys (Macaca fascicularis) J Neuroendocrinol. 2009;21:68–76. doi: 10.1111/j.1365-2826.2008.01800.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Czoty PW, McCabe C, Nader MA. Assessment of the reinforcing strength of cocaine in socially housed monkeys using a choice procedure. J Pharmacol Exp Ther. 2005;312:96–102. doi: 10.1124/jpet.104.073411. [DOI] [PubMed] [Google Scholar]
- Czoty PW, McCabe C, Nader MA. Effects of the 5-HT(1A) agonist (+/-)-8-hydroxy-(di-n-propylamino)tetralin (8-OH-DPAT) on cocaine choice in cynomolgus monkeys. Behav Pharmacol. 2005;16:187–191. doi: 10.1097/00008877-200505000-00008. [DOI] [PubMed] [Google Scholar]
- Czoty PW, Nader MA. Individual differences in the effects of environmental stimuli on cocaine choice in socially housed male cynomolgus monkeys. Psychopharmacology. 2012;224:69–79. doi: 10.1007/s00213-011-2562-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Czoty PW, Nader MA. Effects of dopamine D2/D3 receptor ligands on food-cocaine choice in socially housed male cynomolgus monkeys. J Pharmacol Exp Ther. 2013;344:329–338. doi: 10.1124/jpet.112.201012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dockens RC, Salazar DE, Fulmor E, Wehling M, Arnold ME, Croop R. Pharmacokinetics of a newly identified active metabolite of buspirone after administration of buspirone over its therapeutic dose range. J Clin Pharmacol. 2011;46:1308–1312. doi: 10.1177/0091270006292250. [DOI] [PubMed] [Google Scholar]
- Enkelmann R. Alprazolam versus buspirone in the treatment of outpatients with generalized anxiety disorder. Psychopharmacology. 1991;105:428–432. doi: 10.1007/BF02244440. [DOI] [PubMed] [Google Scholar]
- Farde L, Nordstrom AL, Wiesel FA, Pauli S, Halldin C, Sedvall G. Positron emission tomographic analysis of central D1 and D2 dopamine receptor occupancy in patients treated with classical neuroleptics and clozapine. Relation to extrapyramidal side effects. Arch Gen Psychiatry. 1992;49:538–544. doi: 10.1001/archpsyc.1992.01820070032005. [DOI] [PubMed] [Google Scholar]
- Gawin FH. Neuroleptic reduction of cocaine-induced paranoia but not euphoria. Psychopharmacology. 1986;90:142–143. doi: 10.1007/BF00172886. [DOI] [PubMed] [Google Scholar]
- Gold LH, Balster RL. Effects of buspirone and gepirone on i.v. cocaine self-administration in rhesus monkeys. Psychopharmacology. 1992;108:289–294. doi: 10.1007/BF02245114. [DOI] [PubMed] [Google Scholar]
- Grabowski J, Rhoades H, Silverman P, Schmitz JM, Stotts A, Creson D, et al. Risperidone for the treatment of cocaine dependence: randomized, double-blind trial. J Clin Psychopharmacol. 2000;20:305–310. doi: 10.1097/00004714-200006000-00003. [DOI] [PubMed] [Google Scholar]
- Grabowski J, Shearer J, Merrill J, Negus SS. Agonist-like, replacement pharmacotherapy for stimulant abuse and dependence. Addict Behav. 2004;29:1439–1464. doi: 10.1016/j.addbeh.2004.06.018. [DOI] [PubMed] [Google Scholar]
- Grundt P, Prevatt KM, Cao J, Taylor J, Floresca CZ, Choi J-K, et al. Heterocyclic analogues of N-(4-(4-(2,3-dichlorophenyl)piperazin-1-yl)-butyl)-aryl-carboxamides with functionalized linking chains as novel dopamine D3 receptor ligands: potential substance abuse therapeutic agents. J Med Chem. 2007;50:4135–4146. doi: 10.1021/jm0704200. [DOI] [PubMed] [Google Scholar]
- Haile CN, Mahoney JJ, 3rd, Newton TF, De la Garza R., 2nd Pharmacotherapeutics directed at deficiencies associated with cocaine dependence: focus on dopamine, norepinephrine and glutamate. Pharmacol Ther. 2012;134:260–277. doi: 10.1016/j.pharmthera.2012.01.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heidbreder C. Rationale in support of the use of selective dopamine D3 receptor antagonists for the pharmacotherapeutic management of substance use disorders. Naunyn Schmiedebergs Arch Pharmacol. 2013;386:167–176. doi: 10.1007/s00210-012-0803-6. [DOI] [PubMed] [Google Scholar]
- Heidbreder CA, Newman AH. Current perspectives on selective dopamine D(3) receptor antagonists and pharmacotherapeutics for addictions and related disorders. Ann NY Acad Sci. 2010;1187:4–34. doi: 10.1111/j.1749-6632.2009.05149.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Higley AE, Spiller K, Grundt P, Newman AH, Kiefer SW, Xi ZX, et al. PC01037, a novel dopamine D3 receptor antagonist, inhibits the effects of methamphetamine in rats. J Psychopharmacol. 2011;25:263–273. doi: 10.1177/0269881109358201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- John WS, Banala AK, Newman AH, Nader MA.2014Effects of buspirone and the dopamine D3 receptor compound PG 619 on cocaine and methamphetamine self-administration in rhesus monkeys using a food-drug choice paradigm Psychopharmacology(in press). [DOI] [PMC free article] [PubMed]
- Kaplan JR, Manuck SB, Clarkson TB, Lusso FM, Taub DM. Social status, environment, and atherosclerosis in cynomolgus monkeys. Arteriosclerosis. 1982;2:359–368. doi: 10.1161/01.atv.2.5.359. [DOI] [PubMed] [Google Scholar]
- Kim SW, Fowler J, Skolnik P, Muench L, Kang Y, Shea C, et al. Therapeutic doses of buspirone block D3 receptors in the living primate brain. Int J Neuropsychopharmacol. 2014;28:1–11. doi: 10.1017/S1461145714000194. [DOI] [PubMed] [Google Scholar]
- Kula NS, Baldesarini RJ, Kebabian JW, Neumeyer JL. S-(+)-aporphines are not selective for human D3 dopamine receptors. Cell Mol Neurobiol. 1994;14:185–191. doi: 10.1007/BF02090784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lawler CP, Prioleau C, Lewis MM, Mak C, Jiang D, Schetz JA, et al. Interaction of the novel antipsychotic aripiprazole (OPC-14597) with dopamine and serotonin receptor subtypes. Neuropsychopharmacology. 1999;20:612–627. doi: 10.1016/S0893-133X(98)00099-2. [DOI] [PubMed] [Google Scholar]
- Martelle JL, Claytor R, Ross JT, Reboussin BA, Newman AH, Nader MA. Effects of two novel D3-selective compounds, NGB 2904 [N-(4-(4-(2,3-Dichlorophenyl)piperazin-1-yl)butyl)-9H-fluorene-2-carboxamide] and CJB 090 [N-(4-(4-(2,3-Dichlorophenyl)piperazin-1-yl)butyl)-4-(pyridin-2-yl)benzamide], on the reinforcing and discriminative stimulus effects of cocaine in rhesus monkeys. J Pharmacol Exp Ther. 2007;321:573–582. doi: 10.1124/jpet.106.113571. [DOI] [PubMed] [Google Scholar]
- Mello NK, Fivel PA, Kohut SJ, Bergman J. Effects of chronic buspirone on cocaine self-administration. Neuropsychopharmacology. 2013;38:1264–1275. doi: 10.1038/npp.2013.25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mohammadi MR, Hafezi P, Galeiha A, Hajiaghaee R, Akhondzadeh S. Buspirone versus methylphenidate in the treatment of children with attention-deficit/hyperactivity disorder: randomized, double-blind study. Acta Med Iran. 2012;50:723–728. [PubMed] [Google Scholar]
- Mooney ME, Herin DV, Schmitz JM, Moukaddam N, Green CE, Grabowski J. Effects of oral methamphetamine on cocaine use: a randomized, double-blind, placebo-controlled trial. Drug Alcohol Depend. 2009;101:34–41. doi: 10.1016/j.drugalcdep.2008.10.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morgan D, Grant KA, Gage HD, Mach RH, Kaplan JR, Prioleau O, et al. Social dominance in monkeys: dopamine D2 receptors and cocaine addiction. Nat Neurosci. 2002;5:169–174. doi: 10.1038/nn798. [DOI] [PubMed] [Google Scholar]
- Nader MA, Czoty PW. PET imaging of dopamine D2 receptors in monkeys: genetic predisposition vs. environmental modulation. Am J Psychiatry. 2005;162:1473–1482. doi: 10.1176/appi.ajp.162.8.1473. [DOI] [PubMed] [Google Scholar]
- Nader MA, Czoty PW, Nader SH, Morgan D. Nonhuman primate models of social behavior and cocaine abuse. Psychopharmacology. 2012;224:57–67. doi: 10.1007/s00213-012-2843-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nader MA, Nader SH, Czoty PW, Riddick NV, Gage HD, Gould RW, et al. Social dominance in female monkeys: dopamine receptor function and cocaine reinforcement. Biol Psychiatry. 2012;72:414–421. doi: 10.1016/j.biopsych.2012.03.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Negus SS. Rapid assessment of choice between cocaine and food in rhesus monkeys: effects of environmental manipulations and treatment with d-amphetamine and flupenthixol. Neuropsychopharmacology. 2003;28:919–931. doi: 10.1038/sj.npp.1300096. [DOI] [PubMed] [Google Scholar]
- Newman AH, Blaylock BL, Nader MA, Bergman J, Sibley DR, Skolnik P. Medication discovery for addiction: translating the dopamine D3 receptor hypothesis. Biochem Pharmacol. 2012;84:882–890. doi: 10.1016/j.bcp.2012.06.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Newman AH, Grundt P, Nader MA. Dopamine D3 receptor partial agonists and antagonists as potential drug abuse therapeutics. J Med Chem. 2005;48:1–17. doi: 10.1021/jm040190e. [DOI] [PubMed] [Google Scholar]
- Ohuoha DC, Maxwell JA, Thomson LE, 3rd, Cadet JL, Rohman RB. Effect of dopamine receptor antagonists on cocaine subjective effects: a naturalistic case study. J Subst Abuse Treat. 1997;14:24–258. doi: 10.1016/s0740-5472(96)00161-4. [DOI] [PubMed] [Google Scholar]
- Paterson NE, Vocci F, Sevak RJ, Wagreich E, London ED. Dopamine D3 receptors as a therapeutic target for methamphetamine dependence. Am J Alcohol Drug Abuse. 2014;40:1–9. doi: 10.3109/00952990.2013.858723. [DOI] [PubMed] [Google Scholar]
- Payer DE, Behzadi A, Kish SJ, Houle S, Wilson AA, Rusjan PM, et al. Heightened D(3) dopamine receptor levels in cocaine dependence and contributions to the addiction behavioral phenotype: a positron emission tomography study with [11C]-(+)-PHNO. Neuropsychopharmacology. 2014;39:311–318. doi: 10.1038/npp.2013.192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peroutka SJ. Selective interaction of novel anxiolytics with 5-hydroxytryptamine1A receptors. Biol Psychiatry. 1985;20:971–979. doi: 10.1016/0006-3223(85)90194-5. [DOI] [PubMed] [Google Scholar]
- Staley JK, Mash DC. Adaptive increase in D3 dopamine receptors in the brain reward circuits of human cocaine fatalities. J Neurosci. 1996;16:6100–6106. doi: 10.1523/JNEUROSCI.16-19-06100.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tallman JF, Primus RJ, Brodbeck R, Cornfield L, Meade R, Woodruff K, et al. I. NGD 94-1L identification of a novel, high-affinity antagonist at the human dopamine D4 receptor. J Pharmacol Exp Ther. 1997;282:1011–1019. [PubMed] [Google Scholar]
- Thomsen M, Fink-Jensen A, Woldbye DP, Wortwein G, Sager TN, Holm R, et al. Effects of acute and chronic aripiprazole treatment on choice between cocaine self-administration and food under a concurrent schedule of reinforcement in rats. Psychopharmacology. 2008;201:43–53. doi: 10.1007/s00213-008-1245-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wilson AA, McCormick P, Kapur S, Willeit M, Garcia A, Hussey D, et al. Radiosynthesis and evaluation of [11C]-(+)-4-propyl-3,4,4a,5,6,10b-hexahydro-2H-naphtho[1,2-b][1,4]oxazin-9-ol as a potential radiotracer for in vivo imaging of the dopamine D2 high-affinity state with positron emission tomography. J Med Chem. 2005;48:4153–4160. doi: 10.1021/jm050155n. [DOI] [PubMed] [Google Scholar]
- Winhusen T, Brady KT, Stitzer M, Woody G, Lindblab R, Kropp F, et al. Evaluation of buspirone for relapse-prevention in adults with cocaine dependence: An efficacy trial conducted in the real world. Contemp Clin Trial. 2012;33:992–1002. doi: 10.1016/j.cct.2012.05.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Winhusen TM, Kropp F, Lindblad R, Douaihy A, Haynes L, Hodgkins C, et al. Multisite, randomized, double-blind, placebo-controlled pilot clinical trial to evaluate the efficacy of buspirone as a relapse-prevention treatment for cocaine dependence. J Clin Psychiatry. 2014;75:757–764. doi: 10.4088/JCP.13m08862. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wong H, Dockens RC, Pajor L, Yeola S, Grace JE, Jr, Stark AD, et al. 6-hydroxybuspirone is a major active metabolite of buspirone: assessment of pharmacokinetics and 5-hydroxytryptamine1A receptor occupancy in rats. Drug Metab Dispos. 2007;35:1387–1392. doi: 10.1124/dmd.107.015768. [DOI] [PubMed] [Google Scholar]
- Woolverton WL, Balster RL. Effects of antipsychotic compounds in rhesus monkeys given a choice between cocaine and food. Drug Alcohol Depend. 1981;8:69–78. doi: 10.1016/0376-8716(81)90088-0. [DOI] [PubMed] [Google Scholar]
- Woolverton WL, Goldberg LI, Ginos JZ. Intravenous self-administration of dopamine receptor agonists by rhesus monkeys. J Pharmacol Exp Ther. 1984;230:678–683. [PubMed] [Google Scholar]