Summary
Habituation represents a fundamental form of learning, yet the underlying molecular genetic mechanisms are not well defined. Here we report on a genome-wide genetic screen, coupled with whole genome sequencing, that identified 14 zebrafish startle habituation mutants including mutants of the vertebrate specific gene pregnancy associated plasma protein-aa (pappaa). PAPP-AA encodes an extracellular metalloprotease known to increase IGF bioavailability thereby enhancing IGF receptor signaling. We find that pappaa is expressed by startle circuit neurons, and expression of wildtype, but not a metalloprotease-inactive version of pappaa restores habituation in pappaa mutants. Furthermore, acutely inhibiting IGF1R function in wild-type reduces habituation, while activation of IGF1R downstream effectors in pappaa mutants restores habituation, demonstrating that pappaa promotes learning by acutely and locally increasing IGF bioavailability. In sum, our results define the first functional gene set for habituation learning in a vertebrate, and identify PAPPAA-regulated IGF signaling as a novel mechanism regulating habituation learning.
Keywords: Zebrafish, habituation, learning, IGFR, startle, pyruvate carboxylase, pregnancy associated plasma protein
Introduction
All animals exploit a fundamental mechanism of non-associative learning, called habituation, to filter irrelevant input and prioritize attention (Thompson and Spencer, 1966). Habituation is characterized by progressive response decline to repeatedly experienced, yet inconsequential stimuli (Groves and Thompson, 1970; Rankin et al., 2009). Despite its simplicity, habituation is an attractive form of learning because it provides a quantifiable form of neuroplasticity (Poon and Young, 2006). Deficits in habituation represent hallmark features of human cognitive and behavioral disorders, including schizophrenia, addiction, attention deficit hyperactivity disorder, and other disorders marked by “intellectual disability” (Braff et al., 1992; Jansiewicz et al., 2004; McSweeney et al., 2005). Despite its biological conservation and clinical relevance, our understanding of the genetic mechanisms governing habituation is limited. Identifying the genetic program that governs how neural circuits regulate habituation is therefore instrumental to understanding disorders marked by habituation deficits and for dissecting the genetic basis of higher cognition.
To identify the genetic program that governs vertebrate habituation learning, we took an unbiased, genome wide approach to define a core set of genes critical for habituation of the vertebrate acoustic startle response. Inspired by behavioral screens in Drosophila and C. elegans (Benzer, 1967; Brenner, 1974; Eddison et al., 2012; Ikeda et al., 2008; L'Etoile et al., 2002; Lau et al., 2012; Pierce-Shimomura et al., 2008; Rankin, 2004; Rankin et al., 1990; Swierczek et al., 2011; Wolf et al., 2007), we performed a forward genetic screen using a high throughput behavior testing apparatus that measures zebrafish startle habituation (Wolman et al., 2011), and then applied whole genome sequence (WGS) analysis to molecularly identify the mutated genes. Here, we report on: i) a set of 14 mutants with specific deficits in startle habituation, ii) the molecular identification of two mutants, and iii) the characterization of a novel and vertebrate specific modulator of zebrafish habituation learning.
As predicted, our gene set includes genes with ‘expected’ functions, and genes previously not associated with habituation learning. Specifically, we identified mutations in the pyruvate carboxylase a (pcxa) gene, which encodes a rate limiting enzyme in the production of the glutamate (Hertz et al., 2007), a key neurotransmitter for habituation learning (Bespalov et al., 2007; Bickel et al., 2008; Riedel et al., 2003; Rose and Rankin, 2006). Conversely, we also identified a mutation in the vertebrate specific gene pregnancy associated plasma protein-aa (pappaa). PAPP-AA has not been implicated in any type of learning, and is known to act as an extracellular metalloprotease to enhance local insulin like growth factor (IGF) signaling by cleaving IGF binding protein 4 (IGFBP4), which normally restricts IGF from signaling through cell surface IGF receptors (Conover et al., 2004; Laursen et al., 2007; Laursen et al., 2001; Lawrence et al., 1999). Using a combination of molecular-genetic, pharmacological and behavioral analyses, we uncover a previously unknown role for PAPPAA regulated IGF signaling in mediating habituation learning through an acute and presumptive local mechanism. Overall, our results define the first in vivo function based set of genes regulating vertebrate habituation learning.
Results
Forward genetic screen identifies zebrafish mutants with a startle habituation deficit
By five days of age, zebrafish larvae perform a repertoire of simple sensorimotor behaviors that operate on characterized and accessible neural circuits (Wolman and Granato, 2011). For example, exposure to abrupt acoustic stimuli elicits a startle response, an evolutionary conserved and stereotyped yet modifiable behavior (Burgess and Granato, 2007b; Eaton et al., 1977; Kimmel et al., 1974; Wolman et al., 2011). Repeated acoustic stimulation rapidly prompts habituation by the larvae with identical kinematic and pharmacodynamic parameters observed in adult zebrafish and mammals (Bespalov et al., 2007; Bickel et al., 2008; Riedel et al., 2003; Wolman et al., 2011). To identify genes critical for habituation, we mutagenized adult males using ENU and implemented a three-generation breeding scheme to generate homozygous mutant larvae (Dosch et al., 2004; Mullins et al., 1994). For each F3 clutch, we tested 32 larvae at 5 days post-fertilization (dpf) for short-term habituation to repetitive acoustic stimuli (Wolman et al., 2011). Larvae with morphological defects, hearing loss, or aberration in the highly stereotyped kinematics of the startle response were excluded from subsequent analyses. Heritability of the genetic alteration lesion was verified by observing similarly reduced habituation in subsequent generations.
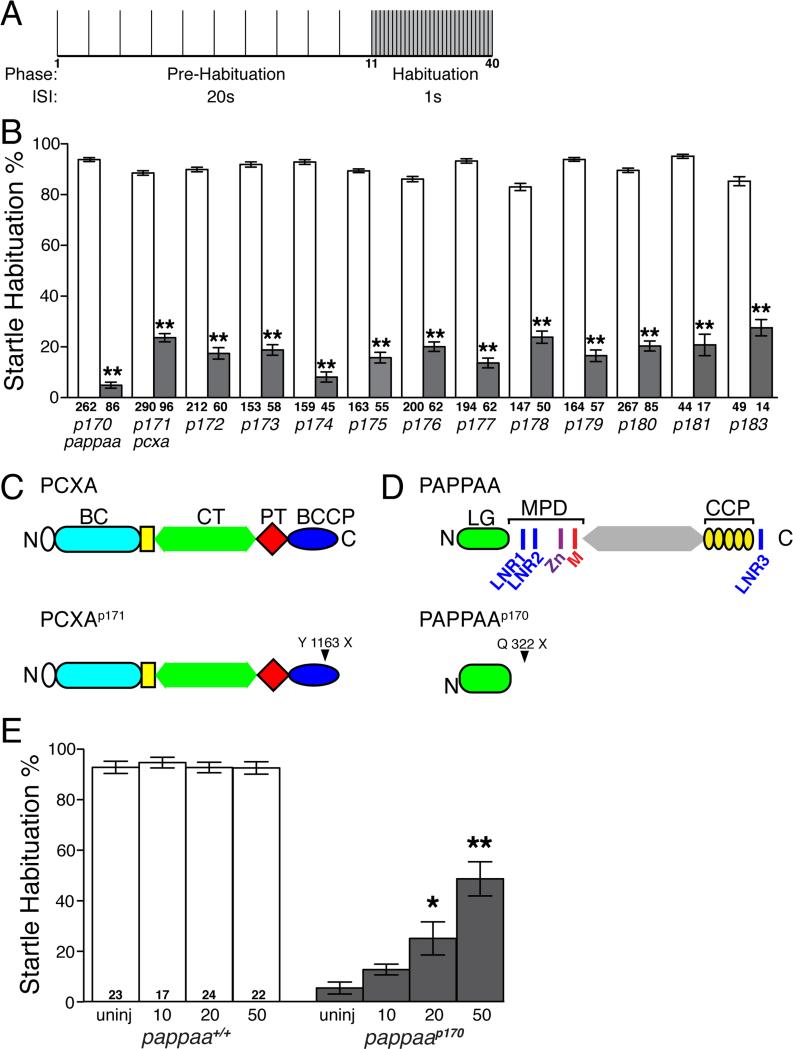
To identify mutants with habituation defects, we used a high-throughput behavioral platform that measures habituation to the acoustic startle response (Wolman et al., 2011). Specifically, 5 dpf larvae were first exposed to 10 acoustic stimuli separated by a 20 second interstimulus interval (ISI) to determine baseline startle responsiveness, and then were given 30 acoustic stimuli with a 1 second ISI to evaluate habituation (Figure 1A). Wild type larvae show a rapid reduction in startle response initiation and stereotypically habituate by more than 80% under these conditions (Wolman et al., 2011). Therefore, clutches with approximately 15-25% of the larvae habituating by less than 50% indicated that the larvae were homozygous for a recessive mutation affecting habituation. Larvae habituating by less than 50% were classified “mutant” (Figure 1B, Movie S1). Using this approach, we screened 405 mutagenized F2 families, corresponding to 614 genomes, and identified 14 habituation mutants (Table 1, Figure 1B).
Figure 1. Genetic screen identifies mutations affecting acoustic startle habituation.
(A) Schematic of acoustic startle habituation assay. Larvae are exposed to 10 non-habituating acoustic stimuli, delivered at 20s interstimulus intervals (ISI), and then 30 habituating stimuli at a 1s ISI. (B) Mean acoustic startle habituation percentage calculated by comparing the average frequency of startle responsiveness of an individual to stimuli 1-10 and stimuli 31-40 (Wolman et al., 2011). Behaviorally defined wild-type siblings shown in white bars, mutants in grey bars. (C) Estimated truncated PCXAp171 protein in information overloadp171 mutants due to Y1163X mutation. BC: biotin carboxylase; CT: carboxyl transferase; PT: pyruvate carboxylase tetramerization; BCCP: biotincarboxy carrier protein. (D) Estimated truncated PAPP-AAp170 protein product in unfilteredp170 mutants due to Q322X mutation. LG: laminin G-like module; LNR: Lin-12/Notch repeats, MPD: metzincin proteolytic domain containing zinc-binding consensus sequence (Zn) and Met-turn motif (M), CCP: complement control protein modules 1-5. E) pappaap170 larvae injected with increasing doses of wild type pappaa mRNA show improved habituation at 5 dpf. *p<0.01, **p<0.001, ANOVA with Bonferonni correction versus wild-type sibling (B) or uninjected pappaap170 (E) larvae. N= number of larvae shown within or below each bar. Error bars indicate SEM.
Table 1.
Zebrafish Habituation Mutants
| Mutant Allele | Startle Habituation% | Visual Habituation% | Baseline Activity | Startle Kinematics | Startle Sensitivity | Gene Locus |
|---|---|---|---|---|---|---|
| unfilteredp170 | 4.9 +/− 1.2* | No response1 | Normal | Normal | Increased | pappaa |
| information overloadp171 | 23.6 +/− 1.6* | Normal | Normal | Normal | Normal | pcxa |
| ignorance is blissp172 | 17.4 +/− 2.3* | Normal | Reduced | Normal | Increased | Ch. 15 Z4396; z9189 Z13822 |
| irresistiblep173 | 18.8 +/− 2.1* | Normal | Normal | Normal | Increased | Ch. 7 Z7958 |
| slow learnerp174 | 7.9 +/− 2.0* | Reduced | Reduced | Normal | Increased | Ch. 4 Z1366; z1525 Z4951; z7104 |
| uninhibitedp175 | 15.7 +/− 2.1* | Normal | Normal | Normal | Increased | n.d. |
| groundhog dayp176 | 20.1 +/− 1.9* | Normal | Normal | Normal | Increased | n.d. |
| doryp177 | 13.8 +/− 1.9* | Normal | Reduced | Normal | Increased | n.d. |
| divided attentionp178 | 24.0 +/− 2.4* | Normal | Normal | Normal | Increased | n.d. |
| oops I did it againp179 | 16.6 +/− 2.3* | Reduced | Reduced | Normal | Normal | n.d. |
| repeat offenderp180 | 20.5 +/− 2.0* | n.d. | Normal | Normal | Increased | n.d. |
| fool me twicep181 | 21.49 +/− 4.48* | n.d. | n.d. | Normal | Increased | n.d. |
| forgetfulp182; forgetfulp183 | 28.49 +/− 3.1* | n.d. | n.d. | Normal | Normal | n.d. |
Summary of behavioral analyses, including acoustic startle habituation (Figure 1B), visual habituation (Figure S2C), baseline activity (Figure S2B), startle kinematics, and startle sensitivity (Figure S2A) of mutants versus wild type siblings.
No response indicates that pappaap170 mutants do not perform the stereotyped O-bend maneuver to the “dark flash” stimuli and therefore cannot be evaluated for visual habituation. Startle kinematic analysis included latency to initiate C-Bend, turning angle during C-bend, and duration of C-bend (primary data not shown). Genetic locus column shows gene or chromosomal region linked to mutant phenotype. Results from complementation crosses suggest these 14 mutants represent alleles of 13 genes.
p<0.01 mutants versus wild type siblings using ANOVA with Bonferonni correction. n.d. not determined.
The behavioral severity and specificity of each of the 14 mutants was determined by comparing the magnitude of the habituation deficit and performing additional behavioral analyses, including acoustic startle sensitivity, baseline activity level, and habituation to repetitive visual stimuli. A comparative analysis revealed that startle habituation in the 14 mutants was affected to different degrees, ranging from 24% habituation (information overload p171, divided attentionp178) down to the almost complete absence of habituation (unfilteredp170, Table 1, Figure 1B). We further evaluated two of the mutants, unfilteredp170 and information overload p171, for short term habituation to acoustic stimuli delivered at longer ISIs and found that both mutants also show strong habituation deficits to the less frequently delivered stimuli (Figure S1). Startle sensitivity was significantly enhanced in unfilteredp170, ignorance is blissp172, irresistiblep173, slow learnerp174, uninhibitedp175, groundhog dayp176, doryp177, divided attentionp178, repeat offenderp180, and fool me twicep181, but not information overloadp171, oops I did it againp179, or forgetfulp182,p183 mutants (Table 1, Figure S2A). Recording gross movement of unstimulated larvae revealed that none of the mutants displayed spontaneous hyperactivity (Table 1, Figure S2B). In fact, ignorance is blissp172, slow learnerp174, doryp177, and oops I did it againp179 mutants performed less spontaneous, overall movement compared to their siblings (Table 1, Figure S2B). Larvae were also given a series of repetitive visual “dark flashes” (Burgess and Granato, 2007a) to determine whether the acoustic startle habituation mutants were capable of habituating to visual stimuli (see Experimental Procedures). Both slow learnerp174 and oops I did it againp179 mutants showed visual habituation deficits, suggesting circuit and/or molecular overlap between acoustic startle- and visual habituation (Table 1, Figure S2C). Thus, a genome wide genetic screen identified a set of mutants in which acoustic startle habituation is reduced to varying degrees. None of these mutants display obvious deficits in acoustic startle performance (i.e. kinematic parameters) or exhibit increased baseline activity, further underscoring the specificity of our screening assay. Several mutants exhibit deficits in acoustic stimulus sensitivity or in visual habituation, reflecting potential overlap between the neural circuitry and genetic programs underlying these behaviors and startle habituation.
WGS identifies novel regulators of habituation learning
To determine the molecular identity of the mutated genes, we selected two mutants with varying degrees of habituation capacity and differing behavioral profiles, and performed WGS. WGS analysis from behaviorally defined unfilteredp170 and information overloadp171 mutant larvae followed by homozygosity analysis (see Experimental Procedures, Figure S3) identified distinct chromosomal intervals for each of these mutants, which we confirmed using bulk segregant analysis (Jain et al., 2011). Within these chromosomal intervals, sequencing data revealed unique nonsense mutations in pyruvate carboxylase a (pcxa) in information overloadp171 mutants, and in pregnancy associated plasma protein-aa (pappaa) in unfilteredp170 mutants, respectively (Figures 1C-D).
PCXA is a biotin-dependent, mitochondrial enzyme that catalyzes the carboxylation of pyruvate to oxaloacetate, a critical step in the synthesis of glucose, fat, amino acids, and neurotransmitters, including glutamate, which is known to influence habituation (Bespalov et al., 2007; Bickel et al., 2008; Jitrapakdee et al., 2008; Rankin and Wicks, 2000; Riedel et al., 2003). Sequencing of pcxa cDNA from behaviorally identified information overloadp171 mutant larvae identified a single nucleotide nonsense mutation in pcxa (nt3489: C to A), which causes a premature stop codon within the biotin-carboxy carrier protein (BCCP) domain (Figure 1C). Mutations to the BCCP domain are associated with the most severe form of pyruvate carboxylase deficiency (type B) due to loss of enzymatic activity (Monnot et al., 2009). Future experiments will determine whether the pcxap171 allele shows reduced enzymatic activity as a result of reduced biotin binding and/or catalytic activity due to improper presentation of biotin to the biotin carboxylase (BC) and/or carboxyl transferase (CT) domains.
Sequencing of pappaa cDNA from behaviorally identified unfilteredp170 larvae confirmed a single nucleotide nonsense mutation in pappaa (nt964: C to T). This mutation causes a premature stop codon in exon 3 at amino acid 322 of 1591, severely truncating PAPP-AA upstream of the metzincin proteolytic domain required for substrate proteolysis, and upstream of the C-terminal domains required for membrane tethering via heparin sulfate proteoglycans (Figure 1D) (Boldt et al., 2001; Laursen et al., 2002; Weyer et al., 2004). To confirm that mutations in the pappaa gene cause the habituation deficit we observe in pappaa/unfilteredp170 mutants, we injected wild type zebrafish pappaa mRNA into one-cell stage pappaap170 embryos. This restored startle habituation in a dose dependent manner, demonstrating that the truncating mutation in zebrafish pappaa causes the habituation deficit (Figure 1E). Thus, WGS analysis reveals pappaa as a novel regulator of habituation learning.
papp-aa is expressed throughout the acoustic startle circuit
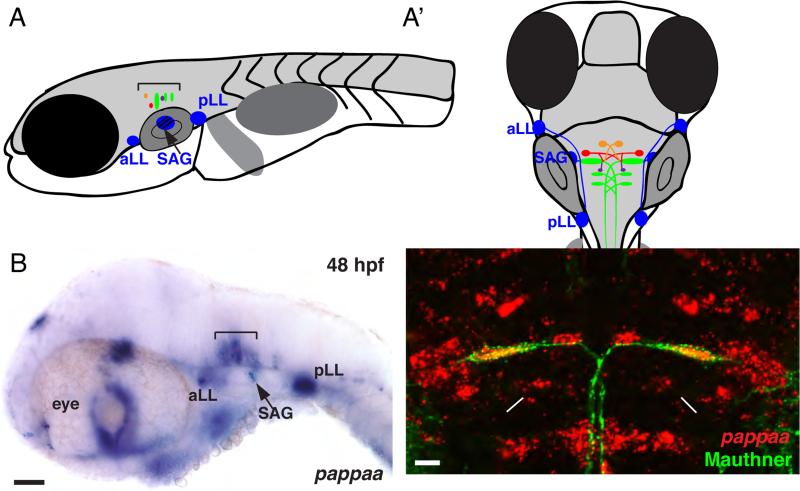
To understand how pappaa modulates startle habituation, we first examined the spatial expression pattern of pappaa mRNA. The acoustic startle response is triggered by activation of one of the bilateral pair of Mauthner neurons in the hindbrain (Figure 2A-A’). Mauthner neurons receive acoustic inputs from the ear (via the statoacoustic ganglia), and the lateral line, and send descending commissural axons down the spinal cord to stimulate contralaterally positioned motor neurons and induce contralateral trunk muscle contraction (Eaton and Emberley, 1991; Eaton et al., 2001; Faber et al., 1989; Faber and Korn, 1978; Liu and Fetcho, 1999). Whole mount in situ hybridization revealed that during embryonic development pappaa mRNA is detectable in several sensory components of the startle circuit: in neurons of the anterior and posterior lateral line ganglia and in neurons of the statoacoustic ganglia, which receive afferent input from sensory hair cells in the lateral line neuromasts and the inner ear, respectively (Figures 2A-B). Importantly, at 5 dpf, when pappaap170 mutants display habituation defects, pappaa mRNA is detectable in the startle command neurons, the Mauthner neurons (Figure 2C). pappaa mRNA is also expressed by several clusters of neighboring hindbrain interneurons known to modulate Mauthner activation (Figure 2C), including passive hyperpolarizing (PHP) neurons, spiral fiber neurons, and the feedback inhibitory neurons (Faber et al., 1989; Hackett and Faber, 1983; Koyama et al., 2011; Lorent et al., 2001; Scott et al., 1994). Molecular markers for these cell types are currently unavailable, yet the location of pappaa mRNA positive hindbrain neurons is consistent with the location of PHP, spiral fiber, and feedback inhibitory neurons (Koyama et al., 2011). Thus, pappaa is expressed by identified hindbrain neurons that are well-characterized components of the acoustic startle circuit.
Figure 2. pappaa expression in neurons of the acoustic startle circuit.
(A-A’) Schematic representation of acoustic startle circuit at larval stage from a lateral (A) and dorsal (A’) perspective. The acoustic startle circuit includes cranial ganglion (blue), Mauthner neuron and homologs (green), spiral fiber neurons (orange), passive hyperpolarizing (PHP) neurons (red), and feedback inhibitory neurons (purple). (B-C) In situ hybridization for pappaa at 48 hpf (B, purple) and 5 dpf (C, red). Brackets (A, B) mark site of hindbrain neurons controlling startle behavior. (C) Dorsal view, anterior to the top. pappaa mRNA in red, Mauthner neuron (M) in green. Arrowheads mark site of spiral fiber neurons, asterisks mark position of PHP neurons, and arrows indicate location of feedback inhibitory neurons. SAG: statoacoustic ganglion; aLL: anterior lateral line ganglion; pLL: posterior lateral line ganglion; M:Mauthner. Scale bars = 50μm (B) and 10μm (C)
Neuronal development of the acoustic startle circuit is intact in pappaap170 mutants
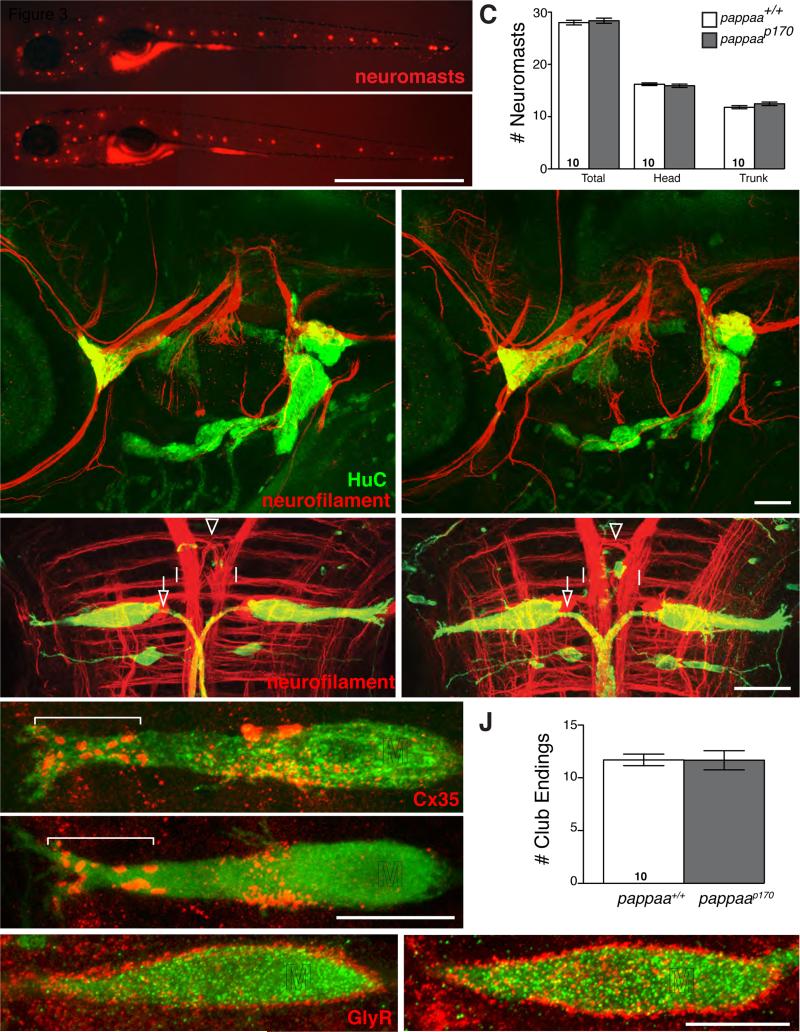
Given the expression of pappaa in several neuronal cell types known to regulate startle modulation and/or execution, we used vital dye and immunolabeling to examine whether pappaa function is required for the development and/or cellular and synaptic integrity of startle circuitry neurons. Analysis of sensory neuromasts that detect acoustic stimuli, sensory cranial ganglia, Mauthner neurons and some of their inputs from other hindbrain modulatory interneurons such as PHP and spiral fibers did not reveal any obvious defects in 5 dpf pappaap170 mutants when compared to wild type animals (Figures 3A-G; n=22 pappaap170, n=24 pappaa+/+). Furthermore, high resolution imaging of the Mauthner neuron showed indistinguishable cellular morphology between pappaap170 mutants and siblings, (Movie S2 (pappaa+/+, n=16) and Movie S3 (pappaap170, n=14); Figure S4). Similarly, large synaptic club endings on the lateral dendrite, the sites of acoustic input to the Mauthner, and glycinergic receptor distribution on the soma of Mauthner neurons revealed no differences when compared to wild type animals (Figures 3H-L; n=12 pappaap170, pappaa+/+ n=10). Taken together, in pappaap170 mutant larvae, sensory afferents to the Mauthner neurons, the Mauthner neurons, and modulatory inputs from neighboring hindbrain neurons appear morphologically indistinguishable from those in wild type.
Figure 3. Acoustic startle circuit appears intact in pappaap170 larvae.
DASPEI labeling (A-B) and mean number (C) of sensory neuromasts. (D-I, K-L) Projections of confocal stacks acquired at 5 dpf . Lateral views, anterior to the left in A-B, D-E. Dorsal views, anterior to the top in F-I, K-L. (D-E) Arrowheads mark projections (anti-neurofilament, red) from cranial ganglia (anti-HuC, green) to hindbrain. (F-I, K-L) Green label marks hspGFF130DMCA:UAS-gap43-citrine expression in Mauthner and Mid2cm Mauthner homologs. (F-G) Open arrows mark Mauthner (M) axon cap. Open arrowhead marks spiral fiber neuron projection and closed arrows mark contralateral passive hyperpolarizing (PHP) projection. (H-I) Brackets mark lateral dendrite of Mauthner with Cx35 positive club endings. Asterisk marks blood cell. (J) Mean number of club endings. (K-L) Glycineric receptors on Mauthner soma. SAG: statoacoustic ganglion; aLL: anterior lateral line ganglion; pLL: posterior lateral line ganglion. N larvae shown within bars. Error bars indicate SEM. Scale bars = 1mm (B), 50μm (E) and 10μm (G, I, L).
PAPP-AA regulates habituation learning through its metalloprotease activity
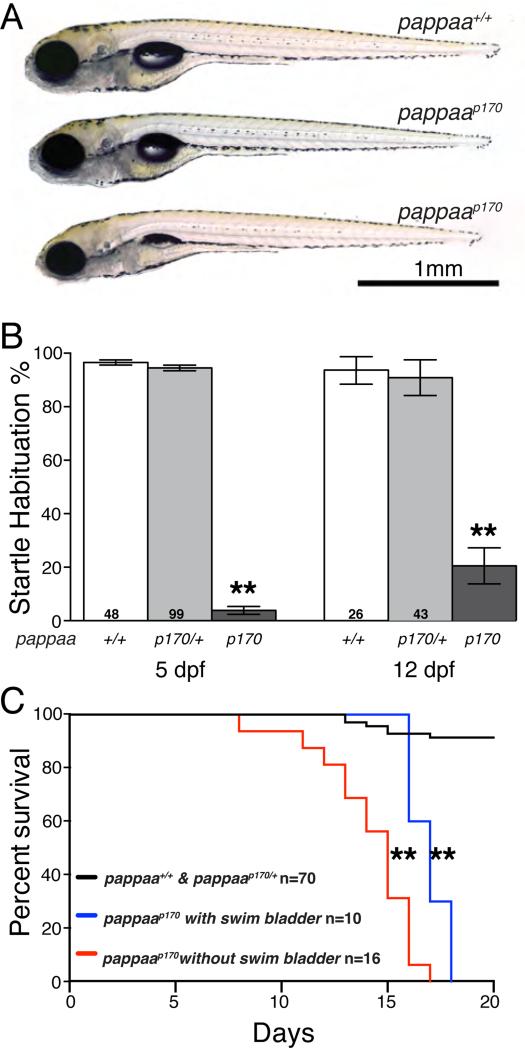
At 5 dpf, when pappaap170 larvae first become behaviorally distinct from their siblings, they appear grossly normal, but often fail to fully inflate their swim bladder (62%, n=26 pappaap170; Figure 4A). By 9 dpf, the pappaap170 mutants are noticeably smaller in size than their wild type siblings (3.93mm +/− 0.07, n=15 pappaap170 vs. 4.65mm +/− 0.03 SEM, n=24 pappaa+/+). Behavioral testing of pappaap170 mutants at 12 dpf revealed a clear deficit in habituation learning, strongly suggesting that PAPP-AA also mediates habituation learning during post-developmental stages (Figure 4B). pappaap170 mutants die at 16.9 dpf (+/− 0.29 days SEM, n=10; Fig. 4C), precluding the analysis of adult mutants. Therefore, we conducted all future experiments between 5 and 12 dpf.
Figure 4. pappaap170 mutants show reduced swim bladder inflation, habituation, and survival.
(A) pappaap170 mutants appear grossly normal at 5 dpf, with exception of some showing an uninflated swim bladder. (B) pappaap170 larvae show startle habituation deficit at 5 and 12 dpf. (C) Kaplan-Meier survival curve shows reduced viability in pappaap170 mutants. *p<0.001, one-way ANOVA (with Bonferonni correction) vs. pappaa+/+, **p< 0.0001 Mantel-Cox test vs. wild type siblings. N= number of larvae shown within bars (B) or in legend (C). Error bars indicate SEM.
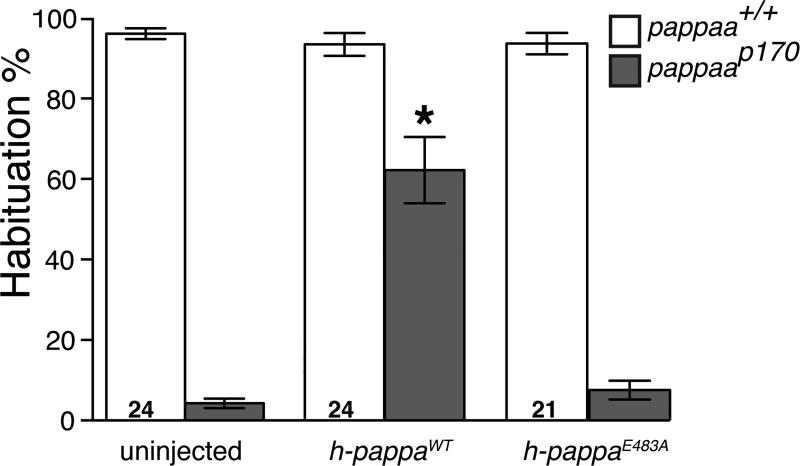
PAPP-A encodes an extracellular metalloprotease know to cleave IGF binding protein 4 (IGFBP4), and hereby increasing insulin like growth factor (IGF) locally available to bind its receptor (Conover et al., 2004; Laursen et al., 2007; Laursen et al., 2001; Lawrence et al., 1999). To ask whether PAPP-AA functions as a protease for habituation learning, we tested the ability of a previously characterized version of human PAPP-A lacking protease activity (h-pappaE483A), to restore habituation learning in zebrafish pappaap170 mutant larvae (Boldt et al., 2001). While control injections of h-pappa mRNA into 1-cell stage embryos significantly improved habituation in pappaap170 mutants, injection of equimolar amounts of h-pappaE483A mRNA did not improve habituation deficits in pappaap170 mutants (Figure 5). Importantly, RT-PCR analysis revealed that wild type h-pappa and h-pappaE483A mRNA persisted through 5 dpf (Figure S5), demonstrating that PAPP-AA metalloprotease activity is required for acoustic habituation learning.
Figure 5. PAPP-AA metalloprotease activity is required for startle habituation.
Mean habituation percentage after injection of human wild type pappa mRNA or proteolytically inactive pappaE483A mRNA. *p<0.001, ANOVA with Bonferonni correction versus DMSO treated larvae of the same genotype. N larvae shown within bars. Error bars indicate SEM.
PAPP-AA regulates habituation through acute control of IGF1R signaling
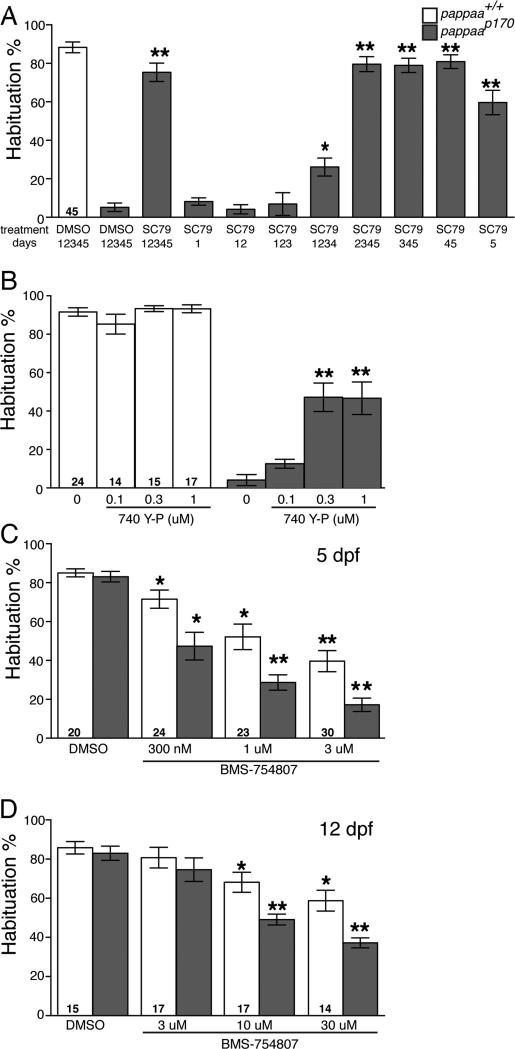
Given that PAPP-AA's metalloprotease activity is required for habituation learning, we next asked whether PAPP-AA regulates this process through canonical IGFR signaling. For this we exposed pappaap170 mutants to SC79, a pharmacological activator of Akt, a canonical downstream effector of IGF1R signaling (Anlar et al., 1999; Jo et al., 2012; Laviola et al., 2007). SC79 treatment from 1-5 dpf improved habituation in pappaap170 mutants to near wild type levels (Figure 6A), suggesting that PAPP-AA regulates acoustic startle habituation through IGFR1-Akt signaling. We next asked whether PAPP-AA acts throughout the period of startle circuit development, or whether PAPP-AA regulates IGF1R-Akt signaling acutely during the habituation process. We hypothesized that periods of SC79 exposure sufficient to improve habituation in pappaap170 mutants would indicate critical periods of PAPP-AA/ IGF1R/Akt signaling. SC79 treatment restricted prior to 3 dpf failed to improve habituation in pappaap170 mutants. In contrast, SC79 treatment between 3-5 dpf, even beginning as late as 5 dpf, restored habituation in pappaap170 mutants. Consistent with these results, acute exposure to 740 Y-P, a cell permeable phosphopeptide activator of PI 3-kinase (PI3K) (Williams and Doherty, 1999), also improved habituation in pappaap170 mutants (Figure 6B). Finally, exposure to BMS-754807, a pharmacological inhibitor of IGF1R (Carboni et al., 2009; Kamei et al., 2011) for 30 minutes prior to and during habituation testing, also significantly reduced startle habituation in a dose dependent manner in wild type zebrafish at both 5 and 12 dpf (Figures 6C-D). Together, these results reveal a novel, acute role for PAPP-AA/IGF1R/PI3K/Akt signaling in promoting startle habituation.
Figure 6. PAPP-AA mediates habituation through acute regulation of IGF signaling.
(A) Mean acoustic startle habituation percentage at 5 dpf of larvae treated with DMSO or 1μM SC79. Treatment period (dpf) indicated below bars. (B) Mean acoustic startle habituation percentage of 5 dpf larvae treated with 740 Y-P for 30 min prior to and during habituation assay. (C-D) Mean acoustic startle habituation percentage after 30 min treatment with BMS-754807 or DMSO at 5 dpf (C) or 12 dpf (D). *p<0.01, **p<0.001, ANOVA with Bonferonni correction versus DMSO treated larvae of the same genotype. N= number of larvae shown within bars. Error bars indicate SEM.
Discussion
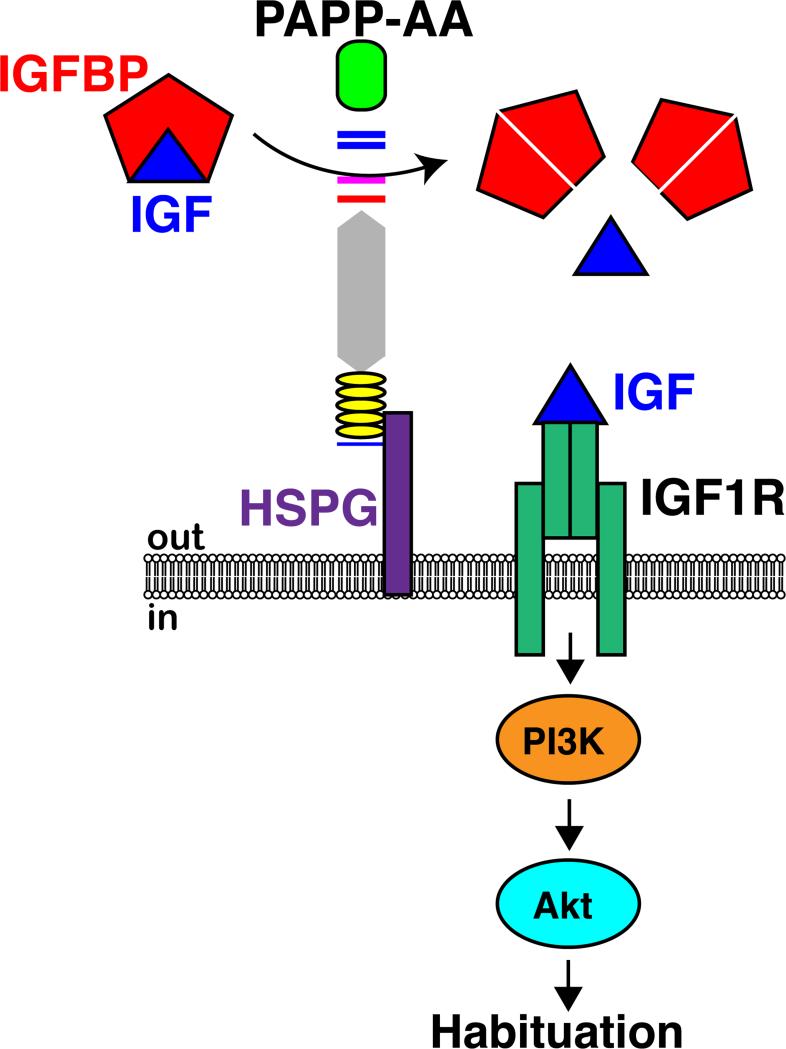
Habituation is a fundamental form of learning during which an animal's response to repetitive, identical stimuli gradually declines (Groves and Thompson, 1970; Thompson and Spencer, 1966). This decline is not due to sensory adaptation or motor fatigue (Rankin et al., 2009; Thompson and Spencer, 1966; Wolman et al., 2011) and is conserved across species. In addition to its conservation, habituation is a particularly interesting form of learning because it provides a measurable form of neuroplasticity that enables animals to ignore irrelevant stimuli in favor of higher priority stimuli (Poon and Young, 2006), and in humans disruption of habituation is strongly correlated with cognitive impairments (Braff et al., 1992; Jansiewicz et al., 2004; McSweeney et al., 2005). Despite extensive characterization of various forms of habituation learning, including olfactory, mechanosensory, and startle habituation (Engel and Wu, 2009; Giles and Rankin, 2009; Glanzman, 2009; Halberstadt and Geyer, 2009; Koch and Schnitzler, 1997; Schmid et al., 2003; Wilson, 2009), there remains a clear need for functional gene sets underlying habituation learning, particularly in vertebrates. Here, we report on results from a genome wide genetic screen to identify a gene set selected solely for a functional role in startle habituation. Importantly, this work identifies pappaa as a novel, vertebrate specific regulator of startle habituation. Based on a series of in vivo experiments we propose a model by which PAPP-AA acts as a metalloprotease to promote startle habituation by increasing IGF receptor signaling during the process of habituation (Figure 7).
Figure 7. Model of PAPP-AA-IGF regulated habituation.
PAPP-AA cleaves IGFBP to increase bioavailable IGF. “Free” IGF then binds and signals through the IGF1R, which triggers activation of PI3K and Akt to regulate habituation.
A forward genetic screen identifies startle habituation mutants
With the exception of an unbiased, forward genetic screen for olfactory habituation mutants in Drosophila (Eddison et al., 2012; Wolf et al., 2007), only candidate gene approaches have been used to identify and dissect genetic mechanisms underlying habituation (Castro-Alamancos and Torres-Aleman, 1994; Das et al., 2011; Engel and Wu, 1998; Lau et al., 2012; Morrison and van der Kooy, 2001; Ohta et al., 2014; Rankin and Wicks, 2000; Rose et al., 2003; Sanyal et al., 2004; Swierczek et al., 2011; Typlt et al., 2013). To complement previous work and identify a broad set of core genes critical for vertebrate startle habituation, we conducted a classical three generation forward genetic screen. This approach yielded a set of 14 mutants with unaltered startle performance, but impaired startle habituation (Figure 1). Because these mutants do not exhibit any morphological or motility defects, they likely represent mutants previous not identified in the major zebrafish screens from Tubingen and Boston in the mid-1990s (see Development, 1996, Issue 123). We noticed that several of the mutants displayed enhanced sensitivity to acoustic stimuli (Figure S2A), consistent with the idea that the mechanisms controlling startle sensitivity are located ‘upstream’ within the neural circuitry regulating startle habituation (Hoffman and Searle, 1968; Pilz and Schnitzler, 1996). Importantly, not all habituation mutants exhibit increased startle sensitivity (e.g. information overloadp171, oops I did it againp179, and forgetfulp182,p183). Conversely, in a companion screen, we identified mutants with much greater enhanced startle sensitivity than the mutants described here, yet these mutants habituate normally (data not shown). We find that several habituation mutants (e.g. information overloadp171, ignorance is blissp172, irresistiblep173, slow learnerp174, uninhibitedp175, groundhog dayp176, and divided attentionp177) are homozygous viable, and display startle habituation deficits when tested as young adults (~3 month), confirming that the molecular-genetic mechanisms driving habituation learning in 5 dpf larvae persists into adulthood. Finally, we find that two of mutants with reduced habituation to acoustic stimuli also displayed visual habituation deficits (Figure S2C; slow learnerp174 and oops I did it againp179), providing evidence that these two affected genes play a role in habituation independent of sensory modality. Thus, we have identified the first set of startle habituation genes characterized solely by their functional requirement for habituation, and given that we previously demonstrated that acoustic startle habituation in zebrafish larvae can be reversed, i.e. dis-habituated by a novel stimulus (Wolman et al, 2011), we expect to find already well characterized genes as well as genes previously not known to affect the central process of habituation learning.
A role for pyruvate carboxylase a (pcxa) in habituation learning
Whole genome sequence analysis and high resolution linkage analyses reveal that the information overloadp171 habituation phenotype co-segregates with a premature stop codon in the biotin-carboxy carrier protein (BCCP) domain of the pyruvate carboxylase a (pcxa) gene (Figure 1C). PCXA catalyzes the carboxylation of pyruvate to oxaloacetate, a necessary step towards the production of glucose, fat, and amino acids, including glutamate (Jitrapakdee et al., 2008). Importantly, pyruvate carboxylase activity is undetectable in neurons but is active in astrocytes where it plays a pivotal role in the maintenance of the glutamate neurotransmitter pool (Hertz, 2004; Schousboe et al., 2013; Whitfield et al., 1996). In humans, pyruvate carboxylase deficiency results in severe psychomotor retardation, and affected individuals die within months to a few years after birth (Monnot et al., 2009). It might therefore appear surprising that pcxap171 mutant zebrafish larvae appear grossly normal and that they are at least partially homozygous viable (data not shown). One likely explanation for the viability of pcxa mutants is that a second paralog gene, pyruvate carboxylase b (pcxb) located on chromosome 7, attenuates the impact of pcxa deficiency. How then does pcxa affect habituation learning? It is well documented that glutamate neurotransmission regulates habituation learning in both mammals and zebrafish (Bespalov et al., 2007; Bickel et al., 2008; Roberts et al., 2013; Wolman et al., 2011). It is therefore tempting to speculate that glia associated with neurons of the startle circuit require pcxa to maintain a constant pool of glutamate. Further analyses are necessary to test this hypothesis, and to understand how pyruvate carboxylase dependent glutamate synthesis promotes neuronal plasticity, including habituation learning.
PAPP-AA is a novel regulator of habituation learning and acutely promotes IGFR signaling
Mammalian pregnancy associated plasma protein a (PAPP-A) was originally purified from late pregnancy plasma, and later shown to encode a membrane associated metalloprotease that specifically cleaves insulin-like growth factor binding proteins IGFBP-4 and -5, thereby releasing IGF from their binding partners and hence promoting IGF receptor activation (Laursen et al., 2007; Laursen et al., 2001; Lawrence et al., 1999; Lin et al., 1974). In mice, knock-out of pappa results in viable offspring about 60% the size of wild type at birth, identical to the phenotype observed in IGF-II null animals (Conover et al., 2004; DeChiara et al., 1990). Although pappa is expressed in the mouse brain, a requirement for pappa in neural development, function or behavior has not been reported (Conover et al., 2004). The zebrafish genome contains two pappa orthologs, pappaa and pappab, and both have been shown to cleave IGFBP-4 (Kjaer-Sorensen et al., 2013). Furthermore, morpholino knockdown of pappab affects developmental growth independent of its proteolytic activity (Kjaer-Sorensen et al., 2013). In contrast, the role of zebrafish pappaa has not been examined.
We find that the startle habituation phenotype in pappaa/unfilteredp170 mutants is caused by a mutation in pappaa, and that pappaa promotes startle habituation through its metalloprotease activity (Figures 1B, 5). Our results support a mechanism in which PAPP-AA regulates habituation by targeting IGFBPs for cleavage and thereby enhancing IGF1R signaling. IGF1R signaling serves extensive roles during all stages of circuit development, and also regulates circuit plasticity (Fernandez and Torres-Aleman, 2012). Although we cannot exclude the possibility that pappaa might contribute to some aspects of neural development, the ability to restore habituation in pappaa p170 mutants by acute activation of Akt and PI3K combined with the observation that acute inhibition of IGF1R reduces startle habituation strongly argues for an acute, post-developmental role for PAPP-AA during habituation learning.
Post-developmental roles for IGF1-IGF1R in learning behavior have been documented from C. elegans to rodent models of IGF signaling deficiency (Castro-Alamancos and Torres-Aleman, 1994; Tomioka et al., 2006). For example, insulin receptor signaling in C. elegans has been shown to play a critical role in experience-dependent temperature habituation, and in taste avoidance learning (Ohno et al., 2014; Ohta et al., 2014). Furthermore, in rats administration of insulin-like growth factor II enhances memory retention and prevents forgetting (Chen et al., 2011), and in humans insulin treatment has been reported to improve memory function (Benedict et al 2004), while IGF-1 mediated signaling is thought to contribute to age-related cognitive decline (Deak and Sonntag, 2012). Reduced IGF availability and signaling has also been linked to schizophrenia (Venkatasubramanian et al., 2007), a disorder with a pronounced habituation deficit (Braff et al., 1992). This association warrants further examination of PAPP-A as a potential therapeutic target to stimulate IGF signaling in patients suffering from disorders like schizophrenia.
pappaa is expressed by neurons known to modulate startle behaviors, and as outlined above is likely to promote habituation learning through an acute, IGFR dependent process. IGFR signaling is known to regulate synaptic strength through ion channel modulation and neurotransmitter release, trafficking, and receptor activity (Blair and Marshall, 1997; Chen and Roche, 2009; Liou et al., 2003; Wang and Linden, 2000; Xing et al., 2007). In the future, resolving the precise mechanism by which pappaa modulates IGFR signaling and how IGFR signaling in turn promotes plasticity will be critical to understand the process of habituation leaning.
Experimental Procedures
Fish Maintenance and Mutagenesis
ENU mutagenesis was performed in TLF and WIK Danio rerio strains as previously described (Dosch et al., 2004; Mullins et al., 1994). Embryos/larvae were maintained on a 14/10 hour light/dark cycle at 29°C and raised as previously described (Gyda et al., 2012; Kimmel et al., 1995). Behavioral experiments were conducted on 5-12 dpf larvae.
Behavioral assays and behavioral analysis
Behavioral experiments were performed and analyzed with the FLOTE software package as previously described (Burgess and Granato, 2007a, b; Hao le et al., 2013; Wolman et al., 2011). The acoustic startle habituation assay was performed as described in Figure 1A (Wolman et al., 2011). Short latency C-bend startle response is measured by defined kinematic parameters including C-turn initiation latency, C-turning angle, C-turn duration, and C-turn maximum angular velocity (Burgess and Granato, 2007b). For secondary behavioral analyses of startle sensitivity, baseline activity, and visual habituation, habituation mutant larvae and wild type siblings were identified based on acoustic startle habituation percentage (mutants < 50%, siblings >50% habituation). Startle sensitivity assay was built into the habituation assay (Wolman et al., 2011). Baseline activity was determined by measuring total distance traveled over 160 sec. For visual habituation testing, larvae were grouped by startle habituation phenotype in a 6 cm petri dish at a density of 12-15 larvae per dish. The visual habituation assay consisted of exposing larvae to 10 1s long dark flashes at 30s ISI to establish baseline dark-flash responsiveness and then 30 1s long dark flashes at 3s ISI to test visual habituation.
Recombination mapping, whole genome sequencing, and molecular cloning of pappaa and pcxa
A three-generation mapping cross was built into our breeding scheme by crossing F1 heterozygotes from mutagenized TLF and WIK backgrounds. Pools of 25 behaviorally identified F3 mutant and sibling larvae were collected at 5 dpf and used for bulk segregant mapping with simple sequence length polymorphic markers (Supplemental Table 1) (Jain et al., 2011) and/or whole genome sequencing.
For whole genome sequencing, we used 100 base-pair paired-end sequencing on the Illumina HiSeq 2000 platform and compared gDNA from our pooled F3 mutants to gDNA prepped from the ENU-mutagenized males of both the TLF and WIK-L11 strains. Each sample was given its own lane in the flow cell, allowing us to achieve an average ~20x coverage of the genome for each sample. Sequence data for each sample was mapped to the zebrafish Zv9 assembly (Ensembl) using Burrows-Wheeler Aligner software to map reads (Li and Durbin, 2009). Duplicate reads were eliminated with Picard tools (http://picard.sourceforge.net.). We then adapted a fast homozygosity mapping strategy from Voz et al (Voz et al., 2012) to our screen. First we isolated all of the 100% homozygous bases unique to our TLF and WIK-L11 reference using the GATK software (McKenna et al., 2010). This generated 463,379 single nucleotide polymorphisms (SNP) markers. Using this set of SNPs, we calculated homozygosity scores by assigning TLF alleles a value of 1 and WIK alleles a value of 0 in a rolling 100-SNP window incrementing one SNP at a time. We analyzed the homozyogosity of our mutant samples across the genome and identified regions with scores > 0.9 or < 0.1 to indicate strong linkage to TLF or WIK-L11 alleles, respectively. To identify potentially causative mutations, we isolated all SNPs in the linked region that were unique to the mutant sample by comparing each mutant sequence to a combined reference sequence comprised of our TLF and WIK sequences, the Ensembl reference sequence, and our other mutant sequences. We restricted our list of candidate mutations to SNPs with <1% allele frequency in this reference sequence that also causes a change in amino acid sequence (nonsense, missense, or splice site mutations). For both unfilteredp170 and information overloadp171, these criteria produced a single SNP candidate.
To confirm candidate nonsense mutations in pappaa and pcxa, cDNA was prepared from total mRNA extraction from 5 dpf larvae as previously described (Peterson and Freeman, 2009). pappaa and pcxa cDNA were amplified with primers (Supplemental Table 1) designed against pappaa and pcxa reference sequence (Ensembl) with the following RT-PCR conditions: 94°C for 3 min and then 35 cycles of 94°C for 30 sec, 57°C for 1 min, and 70°C for 1 min. Products were gel purified and cloned into the pCR2.1-TOPO-TA vector for sequencing.
For zebrafish pappaa RNA injection, cDNA was prepared from wild type TLF larvae and amplified with the z-pappaa:FL primers using similar PCR conditions to those above, but with extension time increased to 3 min. Full-length pappaa was cloned into pCS2+ vector, transcribed using the mMessage mMachine kit (Ambion), and injected at the 1-cell stage at doses ranging from 1-200 picograms. Full length human pappa and pappaE483A constructs (Boldt et al., 2001) similarly prepped and injected. Embryos injected with greater than 50pg of zebrafish pappaa or 100pg h-pappa or h-pappaaE483A mRNA showed gross morphological abnormalities and necrosis, whereas embryos injected with 50pg pappaa or 100pg h-pappa (or less) appeared morphologically normal.
Genotyping, confocal imaging, morphological analyses, immunolabeling and in situ hybridization, pharmacological applications, and statistical analyses are described in Supplemental Experimental Procedures.
Supplementary Material
Acknowledgements
The authors would like to thank Dr. Oxvig for the human PAPP-A constructs, Drs. Mullins and Fisher, Mr. Vought and Ms. Roy for help with the genetic screen, and Drs. Kawakami and Raper for the hspGFF130DMCA and UAS-GAP43-citrine fish lines, respectively. This work is supported by grants to M. G. (NIH MH092257, MH102680 and MH103545). JBH is supported by the National Institute of Neurological Disorders and Stroke (1R01NS054794-06 to JBH), the Defense Advanced Research Projects Agency (DARPA-D12AP00025, to John Harer, Duke University), and by the Penn Genome Frontiers Institute under a HRFF grant with the Pennsylvania Department of Health.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Author Contributions
M.A.W., R.A.J., K.M., J.H., and M.G. designed research;
M.A.W., R.A.J., K.M., H.B., J.S. and K.H performed research;
M.A.W., R.A.J., K.M., K.H., J.H. and M.G. analyzed data;
M.A.W. and M.G wrote the paper.
References
- Anlar B, Sullivan KA, Feldman EL. Insulin-like growth factor-I and central nervous system development. Horm Metab Res. 1999;31:120–125. doi: 10.1055/s-2007-978708. [DOI] [PubMed] [Google Scholar]
- Benzer S. BEHAVIORAL MUTANTS OF Drosophila ISOLATED BY COUNTERCURRENT DISTRIBUTION. Proc Natl Acad Sci U S A. 1967;58:1112–1119. doi: 10.1073/pnas.58.3.1112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bespalov A, Jongen-Relo AL, van Gaalen M, Harich S, Schoemaker H, Gross G. Habituation deficits induced by metabotropic glutamate receptors 2/3 receptor blockade in mice: reversal by antipsychotic drugs. The Journal of pharmacology and experimental therapeutics. 2007;320:944–950. doi: 10.1124/jpet.106.110684. [DOI] [PubMed] [Google Scholar]
- Bickel S, Lipp HP, Umbricht D. Early auditory sensory processing deficits in mouse mutants with reduced NMDA receptor function. Neuropsychopharmacology. 2008;33:1680–1689. doi: 10.1038/sj.npp.1301536. [DOI] [PubMed] [Google Scholar]
- Blair LA, Marshall J. IGF-1 modulates N and L calcium channels in a PI 3-kinase-dependent manner. Neuron. 1997;19:421–429. doi: 10.1016/s0896-6273(00)80950-2. [DOI] [PubMed] [Google Scholar]
- Boldt HB, Overgaard MT, Laursen LS, Weyer K, Sottrup-Jensen L, Oxvig C. Mutational analysis of the proteolytic domain of pregnancy-associated plasma protein-A (PAPP-A): classification as a metzincin. Biochem J. 2001;358:359–367. doi: 10.1042/0264-6021:3580359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Braff DL, Grillon C, Geyer MA. Gating and habituation of the startle reflex in schizophrenic patients. Archives of general psychiatry. 1992;49:206–215. doi: 10.1001/archpsyc.1992.01820030038005. [DOI] [PubMed] [Google Scholar]
- Brenner S. The genetics of Caenorhabditis elegans. Genetics. 1974;77:71–94. doi: 10.1093/genetics/77.1.71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burgess HA, Granato M. Modulation of locomotor activity in larval zebrafish during light adaptation. The Journal of experimental biology. 2007a;210:2526–2539. doi: 10.1242/jeb.003939. [DOI] [PubMed] [Google Scholar]
- Burgess HA, Granato M. Sensorimotor gating in larval zebrafish. J Neurosci. 2007b;27:4984–4994. doi: 10.1523/JNEUROSCI.0615-07.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carboni JM, Wittman M, Yang Z, Lee F, Greer A, Hurlburt W, Hillerman S, Cao C, Cantor GH, Dell-John J, et al. BMS-754807, a small molecule inhibitor of insulin-like growth factor-1R/IR. Molecular cancer therapeutics. 2009;8:3341–3349. doi: 10.1158/1535-7163.MCT-09-0499. [DOI] [PubMed] [Google Scholar]
- Castro-Alamancos MA, Torres-Aleman I. Learning of the conditioned eye-blink response is impaired by an antisense insulin-like growth factor I oligonucleotide. Proc Natl Acad Sci U S A. 1994;91:10203–10207. doi: 10.1073/pnas.91.21.10203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen BS, Roche KW. Growth factor-dependent trafficking of cerebellar NMDA receptors via protein kinase B/Akt phosphorylation of NR2C. Neuron. 2009;62:471–478. doi: 10.1016/j.neuron.2009.04.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen DY, Stern SA, Garcia-Osta A, Saunier-Rebori B, Pollonini G, Bambah-Mukku D, Blitzer RD, Alberini CM. A critical role for IGF-II in memory consolidation and enhancement. Nature. 2011;469:491–497. doi: 10.1038/nature09667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Conover CA, Bale LK, Overgaard MT, Johnstone EW, Laursen UH, Fuchtbauer EM, Oxvig C, van Deursen J. Metalloproteinase pregnancy-associated plasma protein A is a critical growth regulatory factor during fetal development. Development (Cambridge, England) 2004;131:1187–1194. doi: 10.1242/dev.00997. [DOI] [PubMed] [Google Scholar]
- Das S, Sadanandappa MK, Dervan A, Larkin A, Lee JA, Sudhakaran IP, Priya R, Heidari R, Holohan EE, Pimentel A, et al. Plasticity of local GABAergic interneurons drives olfactory habituation. Proc Natl Acad Sci U S A. 2011;108:E646–654. doi: 10.1073/pnas.1106411108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deak F, Sonntag WE. Aging, synaptic dysfunction, and insulin-like growth factor (IGF)-1. The journals of gerontology Series A, Biological sciences and medical sciences. 2012;67:611–625. doi: 10.1093/gerona/gls118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DeChiara TM, Efstratiadis A, Robertson EJ. A growth-deficiency phenotype in heterozygous mice carrying an insulin-like growth factor II gene disrupted by targeting. Nature. 1990;345:78–80. doi: 10.1038/345078a0. [DOI] [PubMed] [Google Scholar]
- Dosch R, Wagner DS, Mintzer KA, Runke G, Wiemelt AP, Mullins MC. Maternal control of vertebrate development before the midblastula transition: mutants from the zebrafish I. Developmental cell. 2004;6:771–780. doi: 10.1016/j.devcel.2004.05.002. [DOI] [PubMed] [Google Scholar]
- Eaton RC, Bombardieri RA, Meyer DL. The Mauthner-initiated startle response in teleost fish. The Journal of experimental biology. 1977;66:65–81. doi: 10.1242/jeb.66.1.65. [DOI] [PubMed] [Google Scholar]
- Eaton RC, Emberley DS. How stimulus direction determines the trajectory of the Mauthner-initiated escape response in a teleost fish. The Journal of experimental biology. 1991;161:469–487. doi: 10.1242/jeb.161.1.469. [DOI] [PubMed] [Google Scholar]
- Eaton RC, Lee RK, Foreman MB. The Mauthner cell and other identified neurons of the brainstem escape network of fish. Progress in neurobiology. 2001;63:467–485. doi: 10.1016/s0301-0082(00)00047-2. [DOI] [PubMed] [Google Scholar]
- Eddison M, Belay AT, Sokolowski MB, Heberlein U. A genetic screen for olfactory habituation mutations in Drosophila: analysis of novel foraging alleles and an underlying neural circuit. PLoS ONE. 2012;7:e51684. doi: 10.1371/journal.pone.0051684. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Engel JE, Wu CF. Genetic dissection of functional contributions of specific potassium channel subunits in habituation of an escape circuit in Drosophila. J Neurosci. 1998;18:2254–2267. doi: 10.1523/JNEUROSCI.18-06-02254.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Engel JE, Wu CF. Neurogenetic approaches to habituation and dishabituation in Drosophila. Neurobiol Learn Mem. 2009;92:166–175. doi: 10.1016/j.nlm.2008.08.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Faber DS, Fetcho JR, Korn H. Neuronal networks underlying the escape response in goldfish. General implications for motor control. Annals of the New York Academy of Sciences. 1989;563:11–33. doi: 10.1111/j.1749-6632.1989.tb42187.x. [DOI] [PubMed] [Google Scholar]
- Faber DS, Korn H. Neurobiology of the Mauthner Neuron. Raven Press; 1978. [Google Scholar]
- Fernandez AM, Torres-Aleman I. The many faces of insulin-like peptide signalling in the brain. Nat Rev Neurosci. 2012;13:225–239. doi: 10.1038/nrn3209. [DOI] [PubMed] [Google Scholar]
- Giles AC, Rankin CH. Behavioral and genetic characterization of habituation using Caenorhabditis elegans. Neurobiol Learn Mem. 2009;92:139–146. doi: 10.1016/j.nlm.2008.08.004. [DOI] [PubMed] [Google Scholar]
- Glanzman DL. Habituation in Aplysia: the Cheshire cat of neurobiology. Neurobiol Learn Mem. 2009;92:147–154. doi: 10.1016/j.nlm.2009.03.005. [DOI] [PubMed] [Google Scholar]
- Groves PM, Thompson RF. Habituation: a dual-process theory. Psychol Rev. 1970;77:419–450. doi: 10.1037/h0029810. [DOI] [PubMed] [Google Scholar]
- Gyda M, Wolman M, Lorent K, Granato M. The tumor suppressor gene retinoblastoma-1 is required for retinotectal development and visual function in zebrafish. PLoS genetics. 2012;8:e1003106. doi: 10.1371/journal.pgen.1003106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hackett JT, Faber DS. Relay neurons mediate collateral inhibition of the goldfish Mauthner cell. Brain Res. 1983;264:302–306. doi: 10.1016/0006-8993(83)90829-6. [DOI] [PubMed] [Google Scholar]
- Halberstadt AL, Geyer MA. Habituation and sensitization of acoustic startle: opposite influences of dopamine D1 and D2-family receptors. Neurobiol Learn Mem. 2009;92:243–248. doi: 10.1016/j.nlm.2008.05.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hao le T, Duy PQ, Jontes JD, Wolman M, Granato M, Beattie CE. Temporal requirement for SMN in motoneuron development. Hum Mol Genet. 2013;22:2612–2625. doi: 10.1093/hmg/ddt110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hertz L. Intercellular metabolic compartmentation in the brain: past, present and future. Neurochemistry international. 2004;45:285–296. doi: 10.1016/j.neuint.2003.08.016. [DOI] [PubMed] [Google Scholar]
- Hertz L, Peng L, Dienel GA. Energy metabolism in astrocytes: high rate of oxidative metabolism and spatiotemporal dependence on glycolysis/glycogenolysis. Journal of cerebral blood flow and metabolism : official journal of the International Society of Cerebral Blood Flow and Metabolism. 2007;27:219–249. doi: 10.1038/sj.jcbfm.9600343. [DOI] [PubMed] [Google Scholar]
- Hoffman HS, Searle JL. Acoustic and temporal factors in the evocation of startle. The Journal of the Acoustical Society of America. 1968;43:269–282. doi: 10.1121/1.1910776. [DOI] [PubMed] [Google Scholar]
- Ikeda DD, Duan Y, Matsuki M, Kunitomo H, Hutter H, Hedgecock EM, Iino Y. CASY-1, an ortholog of calsyntenins/alcadeins, is essential for learning in Caenorhabditis elegans. Proc Natl Acad Sci U S A. 2008;105:5260–5265. doi: 10.1073/pnas.0711894105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jain RA, Wolman MA, Schmidt LA, Burgess HA, Granato M. Molecular-genetic mapping of zebrafish mutants with variable phenotypic penetrance. PLoS ONE. 2011;6:e26510. doi: 10.1371/journal.pone.0026510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jansiewicz EM, Newschaffer CJ, Denckla MB, Mostofsky SH. Impaired habituation in children with attention deficit hyperactivity disorder. Cogn Behav Neurol. 2004;17:1–8. doi: 10.1097/00146965-200403000-00001. [DOI] [PubMed] [Google Scholar]
- Jitrapakdee S, St Maurice M, Rayment I, Cleland WW, Wallace JC, Attwood PV. Structure, mechanism and regulation of pyruvate carboxylase. Biochem J. 2008;413:369–387. doi: 10.1042/BJ20080709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jo H, Mondal S, Tan D, Nagata E, Takizawa S, Sharma AK, Hou Q, Shanmugasundaram K, Prasad A, Tung JK, et al. Small molecule-induced cytosolic activation of protein kinase Akt rescues ischemia-elicited neuronal death. Proc Natl Acad Sci U S A. 2012;109:10581–10586. doi: 10.1073/pnas.1202810109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kamei H, Ding Y, Kajimura S, Wells M, Chiang P, Duan C. Role of IGF signaling in catch-up growth and accelerated temporal development in zebrafish embryos in response to oxygen availability. Development. 2011;138:777–786. doi: 10.1242/dev.056853. [DOI] [PubMed] [Google Scholar]
- Kimmel CB, Ballard WW, Kimmel SR, Ullmann B, Schilling TF. Stages of embryonic development of the zebrafish. Dev Dyn. 1995;203:253–310. doi: 10.1002/aja.1002030302. [DOI] [PubMed] [Google Scholar]
- Kimmel CB, Patterson J, Kimmel RO. The development and behavioral characteristics of the startle response in the zebra fish. Developmental psychobiology. 1974;7:47–60. doi: 10.1002/dev.420070109. [DOI] [PubMed] [Google Scholar]
- Kjaer-Sorensen K, Engholm DH, Kamei H, Morch MG, Kristensen AO, Zhou J, Conover CA, Duan C, Oxvig C. Pregnancy-associated plasma protein A (PAPP-A) modulates the early developmental rate in zebrafish independently of its proteolytic activity. J Biol Chem. 2013;288:9982–9992. doi: 10.1074/jbc.M112.426304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koch M, Schnitzler HU. The acoustic startle response in rats--circuits mediating evocation, inhibition and potentiation. Behav Brain Res. 1997;89:35–49. doi: 10.1016/s0166-4328(97)02296-1. [DOI] [PubMed] [Google Scholar]
- Koyama M, Kinkhabwala A, Satou C, Higashijima S.-i, Fetcho JR. Mapping a sensory-motor network onto a structural and functional ground plan in the hindbrain. Proceedings of the National Academy of Sciences of the United States of America. 2011;108:1170–1175. doi: 10.1073/pnas.1012189108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- L'Etoile ND, Coburn CM, Eastham J, Kistler A, Gallegos G, Bargmann CI. The cyclic GMP-dependent protein kinase EGL-4 regulates olfactory adaptation in C. elegans. Neuron. 2002;36:1079–1089. doi: 10.1016/s0896-6273(02)01066-8. [DOI] [PubMed] [Google Scholar]
- Lau HL, Timbers TA, Mahmoud R, Rankin CH. Genetic dissection of memory for associative and non-associative learning in Caenorhabditis elegans. Genes Brain Behav. 2012 doi: 10.1111/j.1601-183X.2012.00863.x. [DOI] [PubMed] [Google Scholar]
- Laursen LS, Kjaer-Sorensen K, Andersen MH, Oxvig C. Regulation of insulin-like growth factor (IGF) bioactivity by sequential proteolytic cleavage of IGF binding protein-4 and -5. Mol Endocrinol. 2007;21:1246–1257. doi: 10.1210/me.2006-0522. [DOI] [PubMed] [Google Scholar]
- Laursen LS, Overgaard MT, Soe R, Boldt HB, Sottrup-Jensen L, Giudice LC, Conover CA, Oxvig C. Pregnancy-associated plasma protein-A (PAPP-A) cleaves insulin-like growth factor binding protein (IGFBP)-5 independent of IGF: implications for the mechanism of IGFBP-4 proteolysis by PAPP-A. FEBS Lett. 2001;504:36–40. doi: 10.1016/s0014-5793(01)02760-0. [DOI] [PubMed] [Google Scholar]
- Laursen LS, Overgaard MT, Weyer K, Boldt HB, Ebbesen P, Christiansen M, Sottrup-Jensen L, Giudice LC, Oxvig C. Cell surface targeting of pregnancy-associated plasma protein A proteolytic activity. Reversible adhesion is mediated by two neighboring short consensus repeats. J Biol Chem. 2002;277:47225–47234. doi: 10.1074/jbc.M209155200. [DOI] [PubMed] [Google Scholar]
- Laviola L, Natalicchio A, Giorgino F. The IGF-I signaling pathway. Curr Pharm Des. 2007;13:663–669. doi: 10.2174/138161207780249146. [DOI] [PubMed] [Google Scholar]
- Lawrence JB, Oxvig C, Overgaard MT, Sottrup-Jensen L, Gleich GJ, Hays LG, Yates JR, 3rd, Conover CA. The insulin-like growth factor (IGF)-dependent IGF binding protein-4 protease secreted by human fibroblasts is pregnancy-associated plasma protein-A. Proc Natl Acad Sci U S A. 1999;96:3149–3153. doi: 10.1073/pnas.96.6.3149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li H, Durbin R. Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics. 2009;25:1754–1760. doi: 10.1093/bioinformatics/btp324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin TM, Galbert SP, Kiefer D, Spellacy WN, Gall S. Characterization of four human pregnancy-associated plasma proteins. American journal of obstetrics and gynecology. 1974;118:223–236. doi: 10.1016/0002-9378(74)90553-5. [DOI] [PubMed] [Google Scholar]
- Liou JC, Tsai FZ, Ho SY. Potentiation of quantal secretion by insulin-like growth factor-1 at developing motoneurons in Xenopus cell culture. J Physiol. 2003;553:719–728. doi: 10.1113/jphysiol.2003.050955. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu KS, Fetcho JR. Laser ablations reveal functional relationships of segmental hindbrain neurons in zebrafish. Neuron. 1999;23:325–335. doi: 10.1016/s0896-6273(00)80783-7. [DOI] [PubMed] [Google Scholar]
- Lorent K, Liu KS, Fetcho JR, Granato M. The zebrafish space cadet gene controls axonal pathfinding of neurons that modulate fast turning movements. Development (Cambridge, England) 2001;128:2131–2142. doi: 10.1242/dev.128.11.2131. [DOI] [PubMed] [Google Scholar]
- McKenna A, Hanna M, Banks E, Sivachenko A, Cibulskis K, Kernytsky A, Garimella K, Altshuler D, Gabriel S, Daly M, et al. The Genome Analysis Toolkit: a MapReduce framework for analyzing next-generation DNA sequencing data. Genome research. 2010;20:1297–1303. doi: 10.1101/gr.107524.110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McSweeney FK, Murphy ES, Kowal BP. Regulation of drug taking by sensitization and habituation. Exp Clin Psychopharmacol. 2005;13:163–184. doi: 10.1037/1064-1297.13.3.163. [DOI] [PubMed] [Google Scholar]
- Monnot S, Serre V, Chadefaux-Vekemans B, Aupetit J, Romano S, De Lonlay P, Rival JM, Munnich A, Steffann J, Bonnefont JP. Structural insights on pathogenic effects of novel mutations causing pyruvate carboxylase deficiency. Hum Mutat. 2009;30:734–740. doi: 10.1002/humu.20908. [DOI] [PubMed] [Google Scholar]
- Morrison GE, van der Kooy D. A mutation in the AMPA-type glutamate receptor, glr-1, blocks olfactory associative and nonassociative learning in Caenorhabditis elegans. Behav Neurosci. 2001;115:640–649. doi: 10.1037//0735-7044.115.3.640. [DOI] [PubMed] [Google Scholar]
- Mullins MC, Hammerschmidt M, Haffter P, Nusslein-Volhard C. Large-scale mutagenesis in the zebrafish: in search of genes controlling development in a vertebrate. Curr Biol. 1994;4:189–202. doi: 10.1016/s0960-9822(00)00048-8. [DOI] [PubMed] [Google Scholar]
- Ohno H, Kato S, Naito Y, Kunitomo H, Tomioka M, Iino Y. Role of synaptic phosphatidylinositol 3-kinase in a behavioral learning response in C. elegans. Science. 2014;345:313–317. doi: 10.1126/science.1250709. [DOI] [PubMed] [Google Scholar]
- Ohta A, Ujisawa T, Sonoda S, Kuhara A. Light and pheromone-sensing neurons regulates cold habituation through insulin signalling in Caenorhabditis elegans. Nature communications. 2014;5:4412. doi: 10.1038/ncomms5412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peterson SM, Freeman JL. RNA isolation from embryonic zebrafish and cDNA synthesis for gene expression analysis. J Vis Exp. 2009 doi: 10.3791/1470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pierce-Shimomura JT, Chen BL, Mun JJ, Ho R, Sarkis R, McIntire SL. Genetic analysis of crawling and swimming locomotory patterns in C. elegans. Proc Natl Acad Sci U S A. 2008;105:20982–20987. doi: 10.1073/pnas.0810359105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pilz PK, Schnitzler HU. Habituation and sensitization of the acoustic startle response in rats: amplitude, threshold, and latency measures. Neurobiol Learn Mem. 1996;66:67–79. doi: 10.1006/nlme.1996.0044. [DOI] [PubMed] [Google Scholar]
- Poon CS, Young DL. Nonassociative learning as gated neural integrator and differentiator in stimulus-response pathways. Behavioral and brain functions : BBF. 2006;2:29. doi: 10.1186/1744-9081-2-29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rankin CH. Invertebrate learning: what can't a worm learn? Curr Biol. 2004;14:R617–618. doi: 10.1016/j.cub.2004.07.044. [DOI] [PubMed] [Google Scholar]
- Rankin CH, Abrams T, Barry RJ, Bhatnagar S, Clayton DF, Colombo J, Coppola G, Geyer MA, Glanzman DL, Marsland S, et al. Habituation revisited: an updated and revised description of the behavioral characteristics of habituation. Neurobiol Learn Mem. 2009;92:135–138. doi: 10.1016/j.nlm.2008.09.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rankin CH, Beck CD, Chiba CM. Caenorhabditis elegans: a new model system for the study of learning and memory. Behav Brain Res. 1990;37:89–92. doi: 10.1016/0166-4328(90)90074-o. [DOI] [PubMed] [Google Scholar]
- Rankin CH, Wicks SR. Mutations of the caenorhabditis elegans brain-specific inorganic phosphate transporter eat-4 affect habituation of the tap-withdrawal response without affecting the response itself. J Neurosci. 2000;20:4337–4344. doi: 10.1523/JNEUROSCI.20-11-04337.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Riedel G, Platt B, Micheau J. Glutamate receptor function in learning and memory. Behav Brain Res. 2003;140:1–47. doi: 10.1016/s0166-4328(02)00272-3. [DOI] [PubMed] [Google Scholar]
- Roberts AC, Bill BR, Glanzman DL. Learning and memory in zebrafish larvae. Front Neural Circuits. 2013;7:126. doi: 10.3389/fncir.2013.00126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rose JK, Kaun KR, Chen SH, Rankin CH. GLR-1, a non-NMDA glutamate receptor homolog, is critical for long-term memory in Caenorhabditis elegans. J Neurosci. 2003;23:9595–9599. doi: 10.1523/JNEUROSCI.23-29-09595.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rose JK, Rankin CH. Blocking memory reconsolidation reverses memory-associated changes in glutamate receptor expression. J Neurosci. 2006;26:11582–11587. doi: 10.1523/JNEUROSCI.2049-06.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanyal S, Wintle RF, Kindt KS, Nuttley WM, Arvan R, Fitzmaurice P, Bigras E, Merz DC, Hebert TE, van der Kooy D, et al. Dopamine modulates the plasticity of mechanosensory responses in Caenorhabditis elegans. EMBO J. 2004;23:473–482. doi: 10.1038/sj.emboj.7600057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schmid S, Simons NS, Schnitzler HU. Cellular mechanisms of the trigeminally evoked startle response. The European journal of neuroscience. 2003;17:1438–1444. doi: 10.1046/j.1460-9568.2003.02565.x. [DOI] [PubMed] [Google Scholar]
- Schousboe A, Bak LK, Waagepetersen HS. Astrocytic Control of Biosynthesis and Turnover of the Neurotransmitters Glutamate and GABA. Frontiers in endocrinology. 2013;4:102. doi: 10.3389/fendo.2013.00102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scott JW, Zottoli SJ, Beatty NP, Korn H. Origin and function of spiral fibers projecting to the goldfish Mauthner cell. The Journal of comparative neurology. 1994;339:76–90. doi: 10.1002/cne.903390108. [DOI] [PubMed] [Google Scholar]
- Swierczek NA, Giles AC, Rankin CH, Kerr RA. High-throughput behavioral analysis in C. elegans. Nat Methods. 2011;8:592–598. doi: 10.1038/nmeth.1625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thompson RF, Spencer WA. Habituation: a model phenomenon for the study of neuronal substrates of behavior. Psychol Rev. 1966;73:16–43. doi: 10.1037/h0022681. [DOI] [PubMed] [Google Scholar]
- Tomioka M, Adachi T, Suzuki H, Kunitomo H, Schafer WR, Iino Y. The insulin/PI 3-kinase pathway regulates salt chemotaxis learning in Caenorhabditis elegans. Neuron. 2006;51:613–625. doi: 10.1016/j.neuron.2006.07.024. [DOI] [PubMed] [Google Scholar]
- Typlt M, Mirkowski M, Azzopardi E, Ruth P, Pilz PK, Schmid S. Habituation of reflexive and motivated behavior in mice with deficient BK channel function. Frontiers in integrative neuroscience. 2013;7:79. doi: 10.3389/fnint.2013.00079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Venkatasubramanian G, Chittiprol S, Neelakantachar N, Naveen MN, Thirthall J, Gangadhar BN, Shetty KT. Insulin and insulin-like growth factor-1 abnormalities in antipsychotic-naive schizophrenia. The American journal of psychiatry. 2007;164:1557–1560. doi: 10.1176/appi.ajp.2007.07020233. [DOI] [PubMed] [Google Scholar]
- Voz ML, Coppieters W, Manfroid I, Baudhuin A, Von Berg V, Charlier C, Meyer D, Driever W, Martial JA, Peers B. Fast homozygosity mapping and identification of a zebrafish ENU-induced mutation by whole-genome sequencing. PLoS One. 2012;7:e34671. doi: 10.1371/journal.pone.0034671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang YT, Linden DJ. Expression of cerebellar long-term depression requires postsynaptic clathrin-mediated endocytosis. Neuron. 2000;25:635–647. doi: 10.1016/s0896-6273(00)81066-1. [DOI] [PubMed] [Google Scholar]
- Weyer K, Overgaard MT, Laursen LS, Nielsen CG, Schmitz A, Christiansen M, Sottrup-Jensen L, Giudice LC, Oxvig C. Cell surface adhesion of pregnancy-associated plasma protein-A is mediated by four clusters of basic residues located in its third and fourth CCP module. Eur J Biochem. 2004;271:1525–1535. doi: 10.1111/j.1432-1033.2004.04061.x. [DOI] [PubMed] [Google Scholar]
- Whitfield TT, Granato M, van Eeden FJ, Schach U, Brand M, Furutani-Seiki M, Haffter P, Hammerschmidt M, Heisenberg CP, Jiang YJ, et al. Mutations affecting development of the zebrafish inner ear and lateral line. Development. 1996;123:241–254. doi: 10.1242/dev.123.1.241. [DOI] [PubMed] [Google Scholar]
- Williams EJ, Doherty P. Evidence for and against a pivotal role of PI 3-kinase in a neuronal cell survival pathway. Mol Cell Neurosci. 1999;13:272–280. doi: 10.1006/mcne.1999.0750. [DOI] [PubMed] [Google Scholar]
- Wilson DA. Olfaction as a model system for the neurobiology of mammalian short-term habituation. Neurobiol Learn Mem. 2009;92:199–205. doi: 10.1016/j.nlm.2008.07.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wolf FW, Eddison M, Lee S, Cho W, Heberlein U. GSK-3/Shaggy regulates olfactory habituation in Drosophila. Proc Natl Acad Sci U S A. 2007;104:4653–4657. doi: 10.1073/pnas.0700493104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wolman M, Granato M. Behavioral genetics in larval zebrafish-learning from the young. Dev Neurobiol. 2011 doi: 10.1002/dneu.20872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wolman MA, Jain RA, Liss L, Granato M. Chemical modulation of memory formation in larval zebrafish. Proc Natl Acad Sci U S A. 2011;108:15468–15473. doi: 10.1073/pnas.1107156108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xing C, Yin Y, Chang R, Gong X, He X, Xie Z. Effects of insulin-like growth factor 1 on synaptic excitability in cultured rat hippocampal neurons. Experimental neurology. 2007;205:222–229. doi: 10.1016/j.expneurol.2007.01.029. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.