Abstract
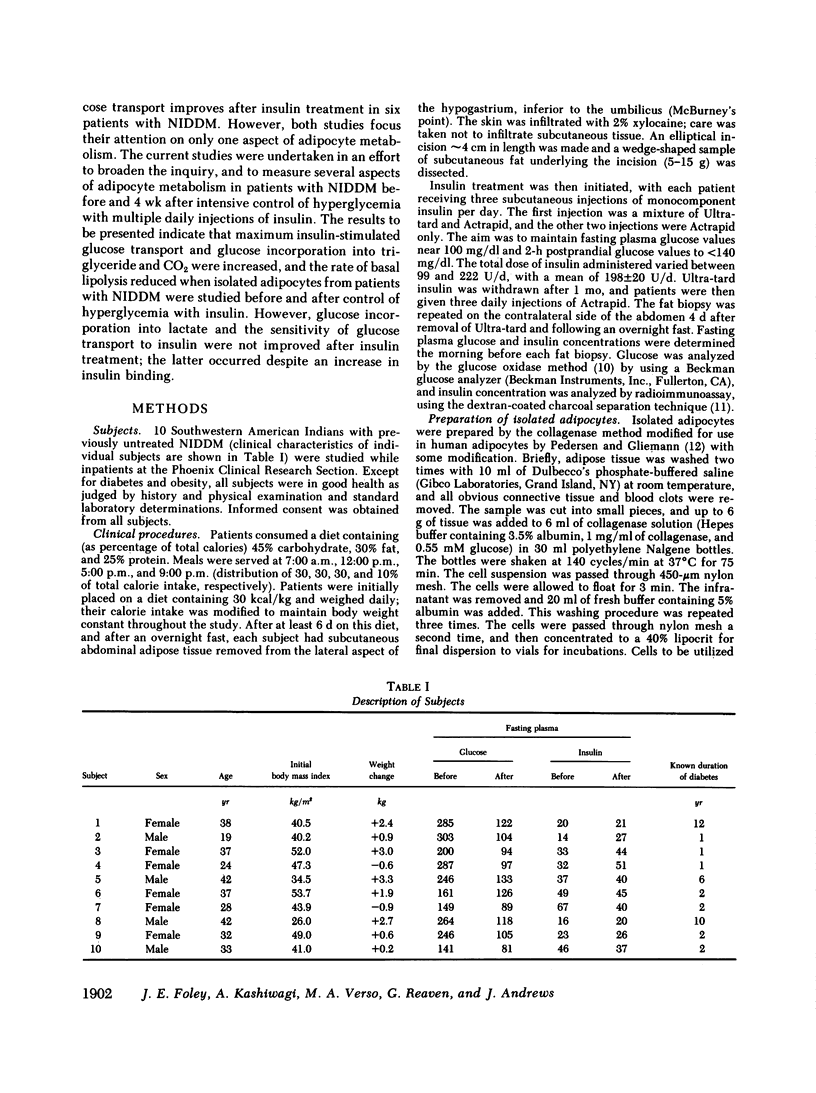
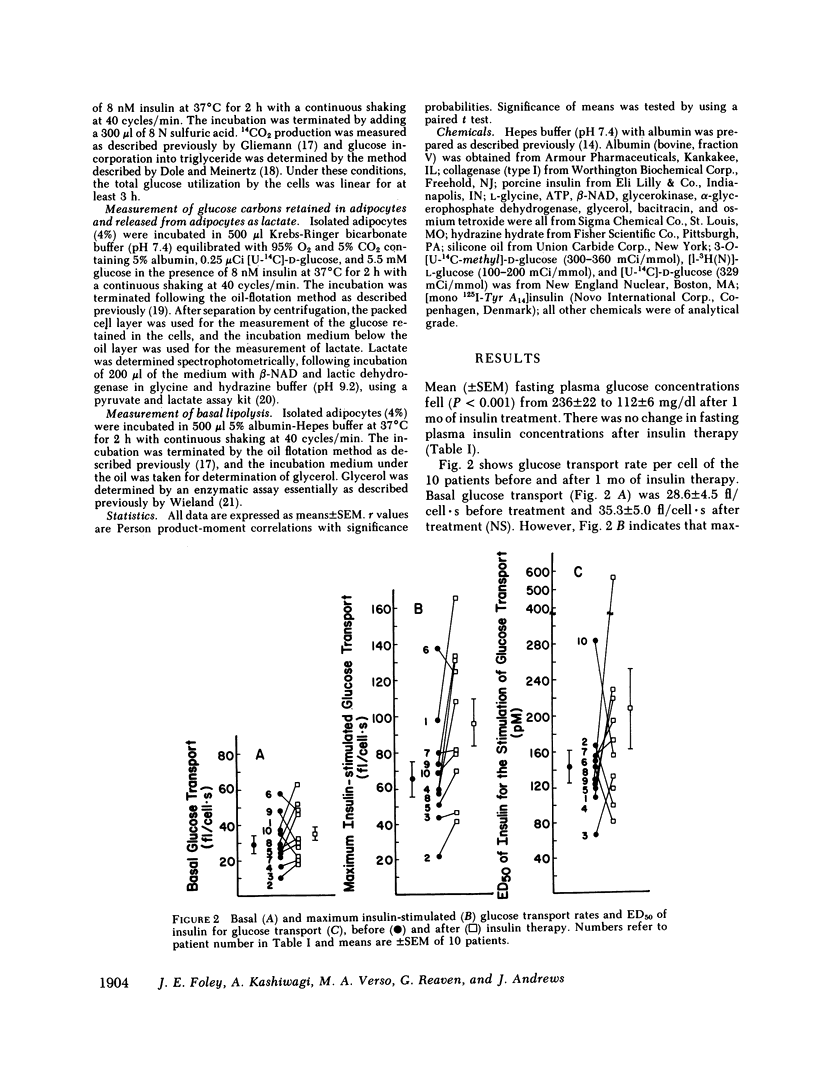
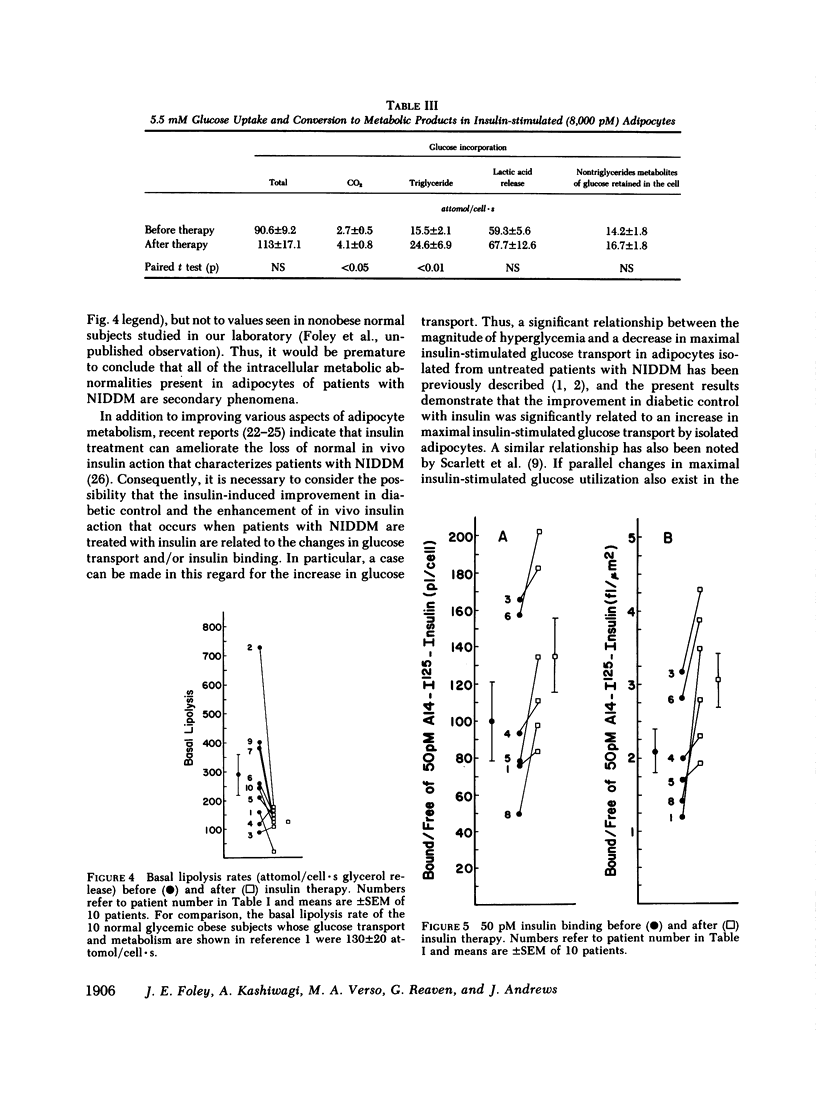
It has been previously reported that maximum insulin-stimulated glucose transport and utilization were both decreased, while basal lipolysis was increased in adipocytes from obese subjects with noninsulin-dependent diabetes mellitus (NIDDM). To determine whether these values can be returned towards those obtained in equally obese subjects with normal glucose tolerance, these measures of adipocyte metabolism were quantified in 10 NIDDM subjects before and after control of hyperglycemia with insulin. The results demonstrate that maximum insulin-stimulated glucose transport (P less than 0.02) and glucose incorporation into triglyceride (P less than 0.01) and CO2 (P less than 0.05) (at 5.5 mM glucose) increased and basal lipolysis decreased (P less than 0.05) after 4 wk of insulin treatment. In contrast, glucose incorporation into lactate and other glycolytic metabolites (at 5.5 mM glucose), and sensitivity of glucose transport to insulin, did not improve with insulin therapy. The latter occurred despite an increase in insulin binding (P less than 0.01). Finally, the improvement in maximal insulin-stimulated glucose transport correlated with the fall in fasting hyperglycemia (r = 0.77, P less than 0.01). These findings demonstrate that several of the abnormalities of carbohydrate and lipid metabolism recently noted to be present in adipocytes from patients with NIDDM can be shown to significantly improve with insulin treatment.
Full text
PDF





Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Arner P., Engfeldt P., Ostman J. Blood glucose control and lipolysis in diabetes mellitus. Acta Med Scand. 1980;208(4):297–299. doi: 10.1111/j.0954-6820.1980.tb01198.x. [DOI] [PubMed] [Google Scholar]
- Björntorp P., Hood B. Studies on adipose tissue from obese patients with or without diabetes mellitus. I. Release of glycerol and free fatty acids. Acta Med Scand. 1966 Feb;179(2):221–227. doi: 10.1111/j.0954-6820.1966.tb05451.x. [DOI] [PubMed] [Google Scholar]
- Bolinder J., Ostman J., Arner P. Postreceptor defects causing insulin resistance in normoinsulinemic non-insulin-dependent diabetes mellitus. Diabetes. 1982 Oct;31(10):911–916. doi: 10.2337/diab.31.10.911. [DOI] [PubMed] [Google Scholar]
- CARLSON L. A., OSTMAN J. IN VITRO STUDIES ON THE GLUCOSE UPTAKE AND FATTY ACID METABOLISM OF HUMAN ADIPOSE TISSUE IN DIABETES MELLITUS. A PRELIMINARY REPORT. Acta Med Scand. 1963 Aug;174:215–218. doi: 10.1111/j.0954-6820.1963.tb07913.x. [DOI] [PubMed] [Google Scholar]
- Ciaraldi T. P., Kolterman O. G., Scarlett J. A., Kao M., Olefsky J. M. Role of glucose transport in the postreceptor defect of non-insulin-dependent diabetes mellitus. Diabetes. 1982 Nov;31(11):1016–1022. doi: 10.2337/diacare.31.11.1016. [DOI] [PubMed] [Google Scholar]
- Cushman S. W., Salans L. B. Determinations of adipose cell size and number in suspensions of isolated rat and human adipose cells. J Lipid Res. 1978 Feb;19(2):269–273. [PubMed] [Google Scholar]
- DOLE V. P., MEINERTZ H. Microdetermination of long-chain fatty acids in plasma and tissues. J Biol Chem. 1960 Sep;235:2595–2599. [PubMed] [Google Scholar]
- Foley J. E., Laursen A. L., Sonne O., Gliemann J. Insulin binding and hexose transport in rat adipocytes. Relation to cell size. Diabetologia. 1980 Sep;19(3):234–241. doi: 10.1007/BF00275275. [DOI] [PubMed] [Google Scholar]
- Ginsberg H., Rayfield E. J. Effect of insulin therapy on insulin resistance in type II diabetic subjects. Evidence for heterogeneity. Diabetes. 1981 Sep;30(9):739–745. doi: 10.2337/diab.30.9.739. [DOI] [PubMed] [Google Scholar]
- Gliemann J. Assay of insulin-like activity by the isolated fat cell method. I. Factors influencing the response to crystalline insulin. Diabetologia. 1967 Aug;3(4):382–388. doi: 10.1007/BF02342631. [DOI] [PubMed] [Google Scholar]
- Gliemann J., Sonne O. Binding and receptor-mediated degradation of insulin in adipocytes. J Biol Chem. 1978 Nov 10;253(21):7857–7863. [PubMed] [Google Scholar]
- Herbert V., Lau K. S., Gottlieb C. W., Bleicher S. J. Coated charcoal immunoassay of insulin. J Clin Endocrinol Metab. 1965 Oct;25(10):1375–1384. doi: 10.1210/jcem-25-10-1375. [DOI] [PubMed] [Google Scholar]
- Hissin P. J., Foley J. E., Wardzala L. J., Karnieli E., Simpson I. A., Salans L. B., Cushman S. W. Mechanism of insulin-resistant glucose transport activity in the enlarged adipose cell of the aged, obese rat. J Clin Invest. 1982 Oct;70(4):780–790. doi: 10.1172/JCI110674. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kashiwagi A., Foley J. E. Opposite effects of a beta-adrenergic agonist and a phosphodiesterase inhibitor on glucose transport in isolated human adipocytes: isoproterenol increases Vmax and IBMX increases Ks. Biochem Biophys Res Commun. 1982 Aug;107(3):1151–1157. doi: 10.1016/0006-291x(82)90642-8. [DOI] [PubMed] [Google Scholar]
- Kashiwagi A., Verso M. A., Andrews J., Vasquez B., Reaven G., Foley J. E. In vitro insulin resistance of human adipocytes isolated from subjects with noninsulin-dependent diabetes mellitus. J Clin Invest. 1983 Oct;72(4):1246–1254. doi: 10.1172/JCI111080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lönnroth P., Digirolamo M., Krotkiewski M., Smith U. Insulin binding and responsiveness in fat cells from patients with reduced glucose tolerance and type II diabetes. Diabetes. 1983 Aug;32(8):748–754. doi: 10.2337/diab.32.8.748. [DOI] [PubMed] [Google Scholar]
- Olefsky J. M., Kolterman O. G., Scarlett J. A. Insulin action and resistance in obesity and noninsulin-dependent type II diabetes mellitus. Am J Physiol. 1982 Jul;243(1):E15–E30. doi: 10.1152/ajpendo.1982.243.1.E15. [DOI] [PubMed] [Google Scholar]
- Olefsky J. M., Reaven G. M. Insulin binding in diabetes. Relationships with plasma insulin levels and insulin sensitivity. Diabetes. 1977 Jul;26(7):680–688. doi: 10.2337/diab.26.7.680. [DOI] [PubMed] [Google Scholar]
- Pedersen O., Gliemann J. Hexose transport in human adipocytes: factors influencing the response to insulin and kinetics of methylglucose and glucose transport. Diabetologia. 1981 Jun;20(6):630–635. doi: 10.1007/BF00257432. [DOI] [PubMed] [Google Scholar]
- Pedersen O., Hjøllund E., Beck-Nielsen H., Lindskov H. O., Sonne O., Gliemann J. Insulin receptor binding and receptor-mediated insulin degradation in human adipocytes. Diabetologia. 1981 Jun;20(6):636–641. [PubMed] [Google Scholar]
- Pedersen O., Hjøllund E. Insulin receptor binding to fat and blood cells and insulin action in fat cells from insulin-dependent diabetics. Diabetes. 1982 Aug;31(8 Pt 1):706–715. doi: 10.2337/diab.31.8.706. [DOI] [PubMed] [Google Scholar]
- Reaven G. M. Insulin-independent diabetes mellitus: metabolic characteristics. Metabolism. 1980 May;29(5):445–454. doi: 10.1016/0026-0495(80)90170-5. [DOI] [PubMed] [Google Scholar]
- Roth J., Kahn C. R., Lesniak M. A., Gorden P., De Meyts P., Megyesi K., Neville D. M., Jr, Gavin J. R., 3rd, Soll A. H., Freychet P. Receptors for insulin, NSILA-s, and growth hormone: applications to disease states in man. Recent Prog Horm Res. 1975;31:95–139. doi: 10.1016/b978-0-12-571131-9.50007-4. [DOI] [PubMed] [Google Scholar]
- Scarlett J. A., Gray R. S., Griffin J., Olefsky J. M., Kolterman O. G. Insulin treatment reverses the insulin resistance of type II diabetes mellitus. Diabetes Care. 1982 Jul-Aug;5(4):353–363. doi: 10.2337/diacare.5.4.353. [DOI] [PubMed] [Google Scholar]
- Scarlett J. A., Kolterman O. G., Ciaraldi T. P., Kao M., Olefsky J. M. Insulin treatment reverses the postreceptor defect in adipocyte 3-O-methylglucose transport in type II diabetes mellitus. J Clin Endocrinol Metab. 1983 Jun;56(6):1195–1201. doi: 10.1210/jcem-56-6-1195. [DOI] [PubMed] [Google Scholar]



